Abstract
Aortic dissection is a rare pathology of the aorta involving separation of the layers of the aortic wall—specifically, the tunica intima and the tunica media. The type of aortic dissection involved is classified by where the separation occurs on the aorta. This case study presents the instrumental significance of performing complete versus limited abdominal sonographic examinations, including abdominal aorta evaluation. Initially, a limited abdominal sonogram was ordered to rule out gallbladder disease. The sonogram was reordered as a complete abdominal study, which led to the diagnosis of aortic dissection. The diagnosis was supported through sonographic features and computed tomography evaluation. The combined use of these two imaging modalities provided an accurate diagnosis and classification regarding the type of aortic dissection. A proper diagnosis and classification facilitated the appropriate treatment related to this type of aortic dissection.
Sonography is a common modality used to image the abdominal aorta, especially to diagnose thrombus, calcification, aneurysm, and dissection of the aortic wall. The aorta is the largest artery in the body, which arises from the outflow tract of the left ventricle of the heart. The aorta arches over the heart and proceeds down through the thoracic and abdominal cavities until it bifurcates at the level of the umbilicus to form the common iliac arteries. The aorta is made up of three layers: the tunica intima (the innermost layer), the tunica media (the middle layer), and the tunica adventitia (the outermost layer). An aortic dissection occurs when a tear develops between the tunica intima and the tunica media. The tear causes these two layers to separate, causing blood flow to course through a “false lumen” created by the resultant intimal flap from the wall separation.
When an aortic dissection occurs, a thrombus may or may not be involved, and blood may or may not be seen flowing through the false lumen when evaluated sonographically. Often, a patient with an aortic dissection will have a sudden onset of chest pain that radiates to the back. Patients are usually men between the ages of 40 and 60 years. 1 Some common associations of an aortic dissection include hypertension, atherosclerotic disease, pregnancy, chest trauma, and inherited diseases such as Marfan syndrome. Marfan is a genetic disorder that mostly affects connective tissue; these patients exhibit such signs as a tall, lanky stature with double joints. Specifically, the individual’s arterial vessels are affected in a way that can cause abnormal dilation, weakened walls, and eventual dissection of the aorta. 1 It is possible for an aortic dissection to rupture into any body cavity; thus, it is considered a medical emergency and is frequently fatal.
Sonography and computed tomography (CT) are imaging modalities used to evaluate aortic dissections. This case illustrates a patient with an aortic dissection and the diagnostic imaging studies utilized to detect and accurately diagnose the pathology. Both sonography and CT proved to be essential in this patient’s diagnosis. Sonography was the instrumental noninvasive study used to detect the presence of the aortic dissection, while CT verified the sonographic findings and helped classify the type of dissection present. This case was initially ordered as a limited abdominal sonogram with special attention to the gallbladder; however, due to the patient’s vague symptoms and no prior imaging studies, the order was changed to a complete abdominal sonogram. The limited abdominal sonogram would have assessed only the patient’s gallbladder and bile ducts, whereas a complete abdominal sonogram offers a more extensive study of the patient’s abdomen—most commonly evaluating the spleen, kidneys, pancreas, liver, gallbladder, bile ducts, abdominal aorta, and iliac arteries.
Case Report
A 57-year-old man with a history of severe back pain, chest pain, shortness of breath, vomiting, diabetes, and a recent heart attack 2 weeks prior presented to the sonography department for a sonographic evaluation of the abdomen, with special attention to the gallbladder. Because the patient’s symptoms were unspecific and he had no recent imaging studies performed, the sonographer obtained permission from the ordering physician to change the order to a complete abdominal sonogram, which was more inclusive. The sonogram was performed with a curvilinear array transducer with a frequency of 4 MHz on a Philips iU22 sonography unit.
Although the initial concern was the gallbladder, there was no sonographic evidence of cholelithiasis or associated pericholecystic fluid. The gallbladder wall measured 3.3 mm, and minimal debris was present within the gallbladder lumen. Sonographic evaluation of the abdominal aorta revealed a linear echogenic line projecting within the infrarenal aortic lumen in both the sagittal and transverse planes (Figures 1 and 2). The echogenic linear structure within the lumen of the aorta appeared to be consistent with an intimal flap associated with aortic dissection. In the transverse plane, color Doppler was applied to the distal aorta, where flow was seen in both the true and false lumen of the vessel (Figure 3). The dissection appeared to involve the infrarenal aorta and extend into the left iliac artery with demonstration of flow within the true lumen (Figures 4 and 5). The aorta at the level of the proximal diaphragm measured 2.8 cm; the proximal midaorta, 2.5 cm; the midaorta, 2.2 cm; the right iliac artery, 0.9 cm; and the left iliac artery, 1.1 cm. The initial diagnosis provided by the radiologist, after receiving the sonographic images, was concern for infrarenal aortic dissection that extended into the left common iliac artery. The radiologist recommended CT of the chest, abdomen, and pelvis for further diagnostic evaluation.
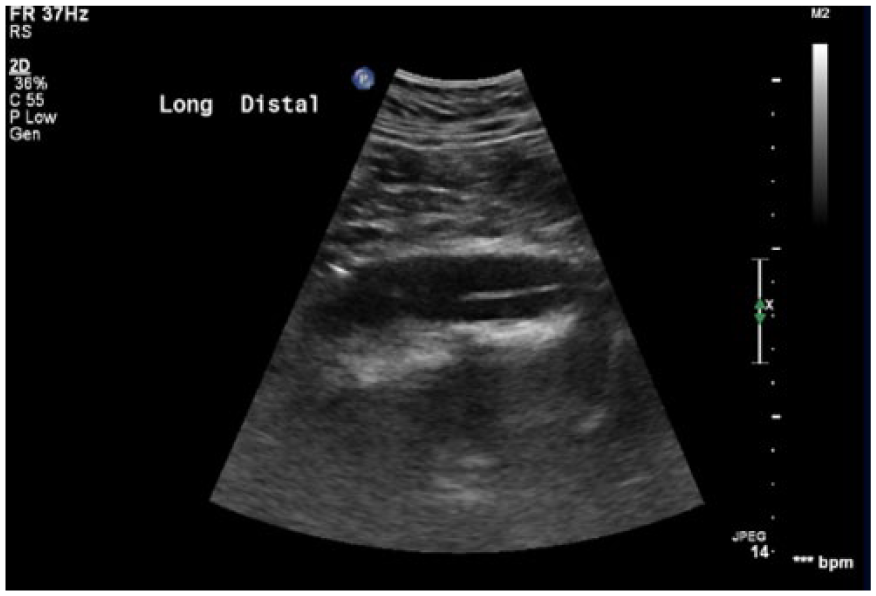
Sonogram demonstrating an echogenic linear structure in the sagittal plane of the lumen in the distal aorta, consistent with aortic dissection.
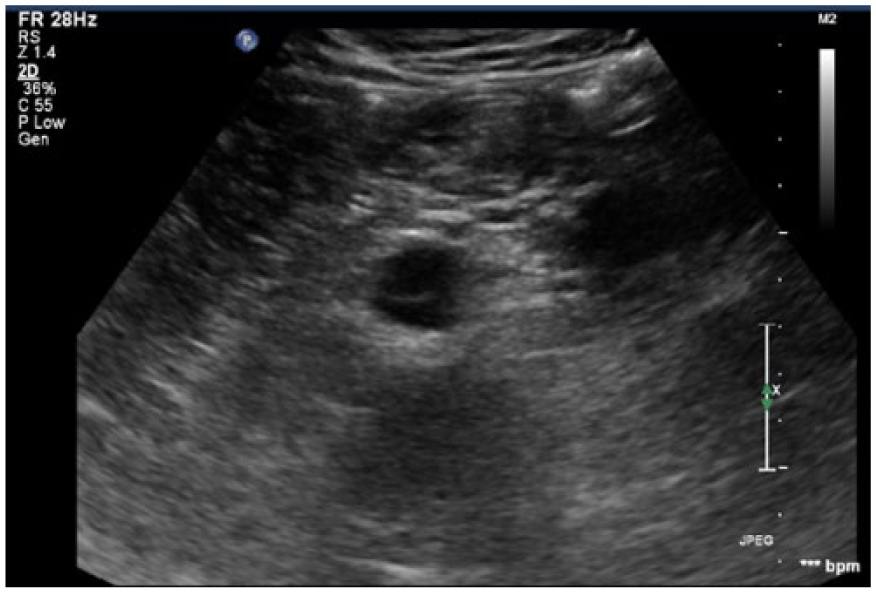
Sonogram demonstrating an echogenic linear structure in the transverse plane within the lumen of the distal aorta, consistent with aortic dissection.
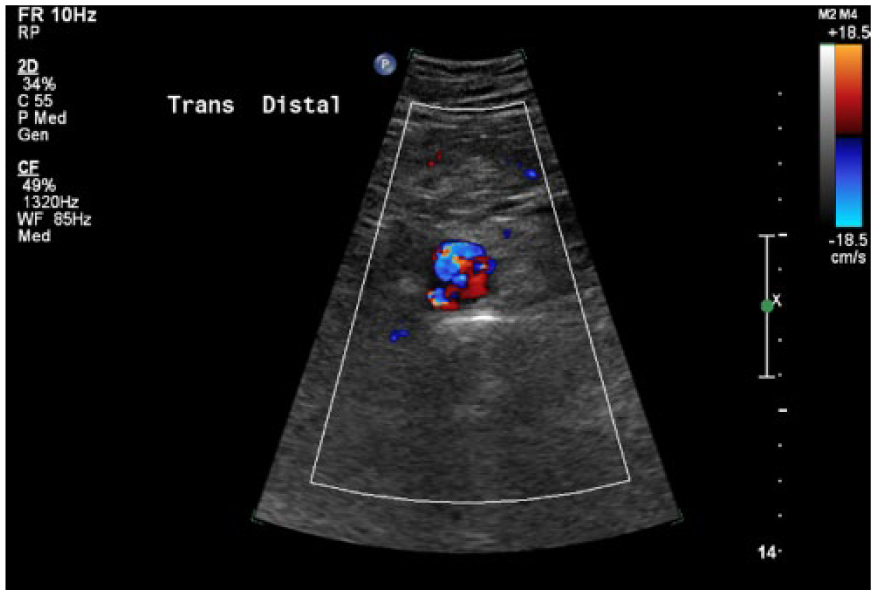
Sonogram with color Doppler demonstrating flow within the true and false lumen of the distal aorta in the transverse plane.
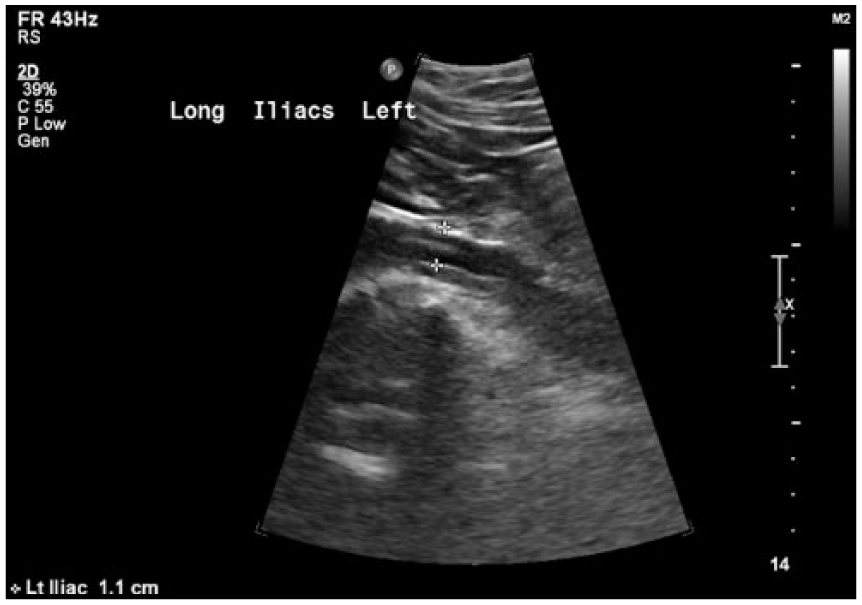
Sonogram demonstrating an echogenic linear structure within the lumen of the left iliac artery, consistent with aortic dissection.
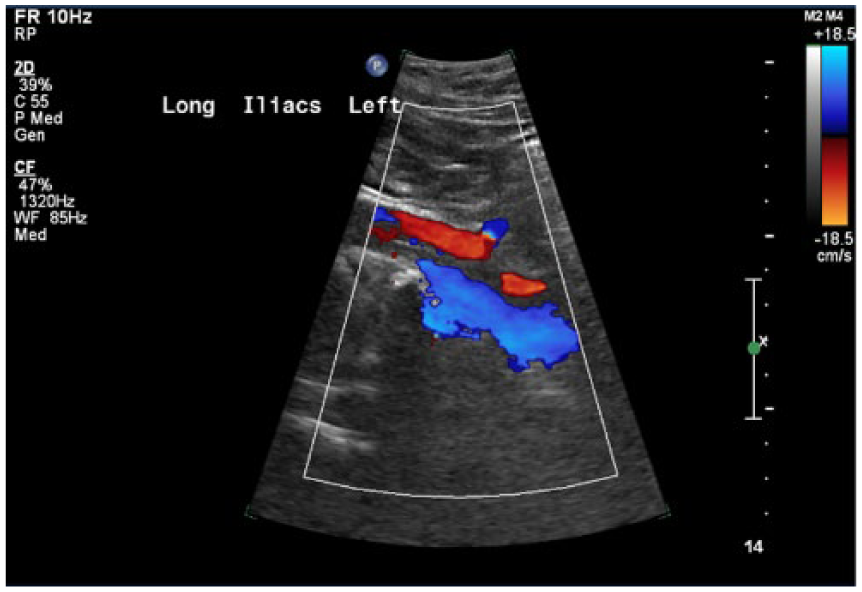
Sonogram of the left iliac artery with color Doppler, demonstrating flow within the true lumen of the dissection.
An axial CT angiogram (CTA) of the chest, abdomen, and pelvis was then performed; 75 mL of Omnipaque contrast at 3 mL/s was administered intravenously. The CTA results were comparable to the sonographic findings in that the aortic measurements were similar; however, CTA demonstrated that the aortic dissection actually extended from the aortic arch (Figure 6) all the way down the abdominal aorta and into the left common iliac artery, external iliac artery, and the internal iliac artery. With the dissection extending into the left internal iliac artery, there was subsequent luminal narrowing by 60%. With the dissection extending into the left external iliac artery, there was complete occlusion of a portion of the distal left external iliac artery (Figure 7). The dissection within the left external iliac artery also extended into the left common femoral artery and the left superficial femoral artery. The dissection also appeared to extend into the celiac artery, with narrowing of the proximal celiac artery by approximately 50% (Figure 8). It was noted that the dissection was also possibly extending into the gastric artery, off the celiac trunk, with resultant narrowing of the vessel lumen by 80%. Both the superior mesenteric artery and the inferior mesenteric artery arose from the true lumen of the aorta. No frank rupture, leak of the aorta, pulmonary embolus, or pericardial effusion were visualized on CTA images. On the basis of the sonographic and CTA findings, the patient was strictly instructed to go to the hospital for further evaluation and care.
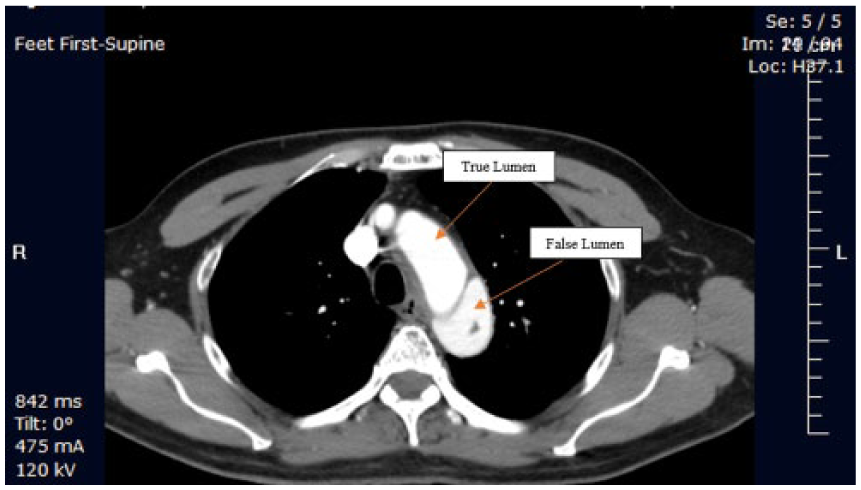
Computed tomography angiogram demonstrating both the true and false lumens of aortic arch, beginning just distal to the left subclavian artery but not extending into the great vessels, consistent with aortic dissection of the descending aorta.
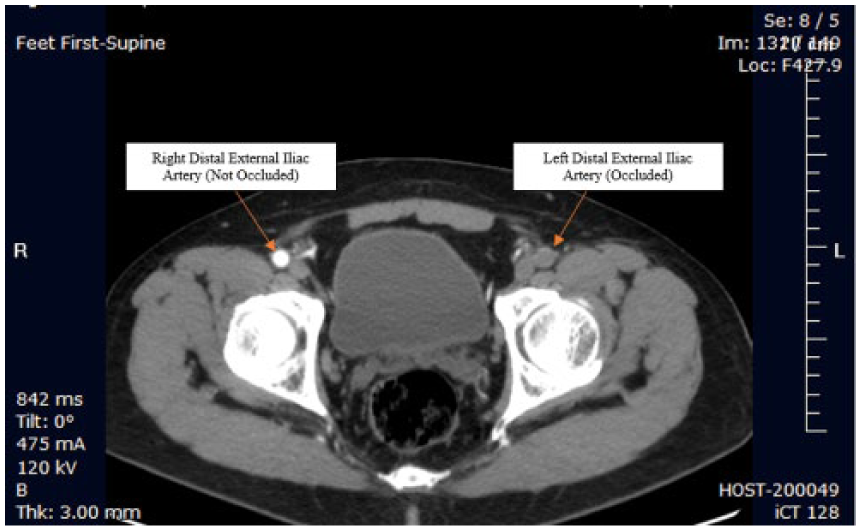
Computed tomography angiogram demonstrating complete occlusion of the distal left external iliac artery, consistent with aortic dissection.
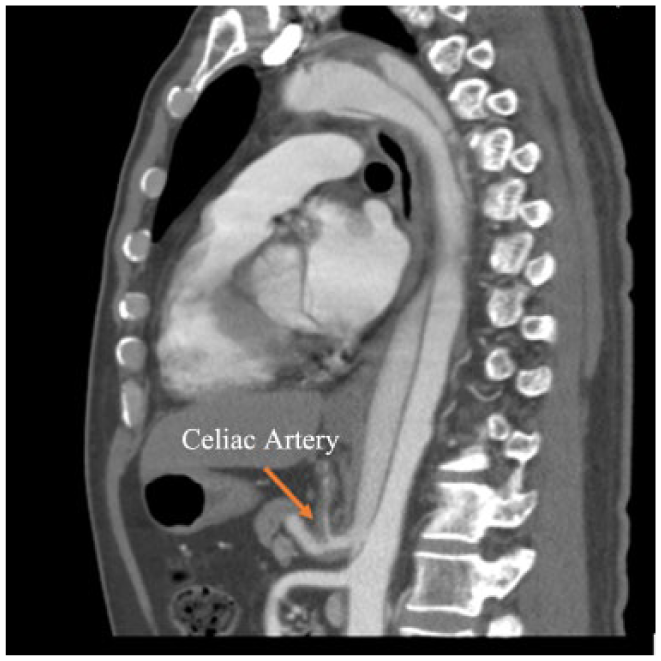
Computed tomography angiogram demonstrating extensions of the aortic dissection into the celiac artery with narrowing of the artery.
Discussion
Although the etiology of aortic dissection is not entirely clear, it is presumed that a dissection results from a tear of the intimal lining of the aorta due to damage or weakening of the intimal wall. However, not all aortic dissections can be assumed to develop in this same manner. A rupture of the network of small blood vessels that supplies blood to the walls of larger vessels, known as the vasa vasorum, can also initiate a dissection. 2 Aortic dissections are strongly associated with hypertension and are related to other underlying causes—notably, cystic medial necrosis of the vessel, Marfan syndrome, pregnancy, aortic valve disease, congenital cardiac anomalies, Cushing syndrome, pheochromocytoma, and catheter-induced needle wounds. 2 The risk of death from an aortic dissection is greatest in patients ≥65 years, males, and those who smoke >100 cigarettes in a lifetime. 3
Symptoms such as intense chest pain, abdominal, and lower back pain, as well as arm or leg pain may occur with aortic dissection. Patients may also experience vomiting, paralysis, transient blindness, coma, confusion, syncope, headache, and dyspnea, and extremity pulses may be absent. 2 It is important to note that the signs and symptoms of aortic dissection often mimic other pathologies. For example, diseases of the gallbladder share some of the most common symptoms of aortic dissection, such as chest, back, and abdominal pain, as well as vomiting, which may result in similar clinical presentations. In this particular case, it was advantageous that the sonographer was aware of these overlapping symptoms and was able to get the initial order changed from a limited to a complete abdominal sonogram. This clinical situation underscores the potential to miss significant pathology when limited sonography examinations are performed. Given that some sonographers are working under increased pressure to perform more examinations in limited time, laboratories have embraced the performance of limited sonograms at the discretion of the ordering physician. The sonography laboratory highlighted in this case study performs limited sonographic studies only when a recent imaging study is available for comparison. This protocol is performed in order to identify pathology that would otherwise not be detected.
An aortic dissection is classified by the location of the dissection. In this case, the dissection extended from the aortic arch down to the left iliac artery, thus extending nearly the entire length of the descending thoracic and abdominal aorta. The radiologist concluded that the dissection began just distal to the left subclavian artery but did not extend into the great vessels; therefore, this dissection was isolated to the descending aorta, starting from the aortic arch. The two classifications of aortic dissection are described by the DeBakey and Stanford classification systems. The DeBakey classification consists of three types. Type I involves the entire aorta, ascending and descending; this accounts for 50% of all aortic dissections. 4 Type II involves only the ascending aorta and is associated with Marfan syndrome. 2 Type III involves only the descending aorta and possesses the danger of cutting off supply to the renal arteries. 1 Type III is the least common type of aortic dissection. 4 Because types I and II have the possibility of extending into the pericardium, they are considered the most dangerous. 2 The Stanford classification consists of two types: type A involves the ascending aorta and may or may not involve the descending aorta; type B dissections involve only the descending aorta. 1 Based on this information, the dissection in this case was classified as Stanford type B and DeBakey type III.
Treatment for aortic dissections may include surgery or medications. Medications help lessen the chance of the aortic dissection progressing and may also be used to prepare a patient for surgery. Although surgery and medications could be used for both type A and type B dissections, the different types usually require varied treatment options. Stanford type A dissections (DeBakey I and II) often require surgical intervention, especially if there is involvement of pathology with the aortic valve and aortic root. 5 A type B (DeBakey type III) dissection is more commonly treated conservatively, with medical management, provided there are no other life-threatening complications.3,6,7 Since the patient in this case had a Stanford type B, DeBakey type III dissection and no apparent life-threatening complications, medical management was decided as the appropriate treatment option.
This case study is an informative example of how sonography is a valuable, cost-effective, and efficient imaging modality in the detection of aortic dissection. It also demonstrates the importance of physicians not diagnosing patients solely on their clinical signs and symptoms but referring suspected patients for a complete sonographic examination to obtain a differential diagnosis.
A compatible case study 8 was reviewed of a 72-year-old woman who presented to the emergency department with chest pain radiating to the neck and back. An emergency transthoracic echocardiogram and bedside abdominal sonogram were utilized to confirm an aortic dissection. The type of dissection in this case was Stanford type A, which extended into the left common carotid artery. Due to an early accurate diagnosis, the patient was able to receive emergent surgical consultation, additional imaging evaluation to confirm the diagnosis, and an intervention to avert further complications. 8 Although this compatible case features a different type of aortic dissection (Stanford type A), it provides an excellent example of how an emergent complete sonogram can be used to accurately diagnose aortic dissection and provide prompt treatment in cases where patient symptoms mimic other pathologies.
Conclusion
Sonography and CTA both played an essential role in this case study to diagnose and accurately classify an aortic dissection. Sonography acted as an initial noninvasive imaging modality that allowed for dynamic detection and evaluation of the aortic dissection. CTA is rated by the American College of Radiology as the most appropriate diagnostic test for suspected aortic dissection. 9 Never-theless, combining the CTA examination with the screening results of the complete abdominal sonogram confirmed the diagnosis and gave even further insight into the extent and origin of the aortic dissection. The synergy of both imaging examinations was formative in confirming the presence and type of aortic dissection. Dual imaging also was pivotal in recommending the best treatment option for the patient.
Footnotes
Acknowledgements
Samantha Simpson thanks Fran Richey, RDMS, Lara Oidtman, RDMS, Sarah Stockman, RDMS, Kimberly Perkins, RDMS, RVT, and LeeAnn Rippey, RDMS, for their assistance, leadership, and guidance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
