Abstract
Aortic dissection is an emergent condition requiring rapid diagnosis and immediate treatment. Patients with aortic dissection typically present with complaints of a sudden onset of severe abdominal and back pain. Patients with a suspected dissection may undergo a variety of imaging tests such as computed tomography angiography, sonography, and/or magnetic resonance angiography. The role of these diagnostic imaging modalities for aortic dissection is examined in detail in this literature review. These imaging modalities are compared and contrasted by reviewing their advantages, disadvantages, and overall effectiveness for aortic dissection.
Aortic dissection is a serious life-threatening condition and is one of the most common potentially deadly conditions of the aorta.1–5 In the United States, nearly 10,000 people are affected by this condition each year.3,6 Aortic dissection is a tear in the aorta caused by a weakened wall in which the intima, or inner wall, separates from the media (middle wall layer).3,7–12 The separation in the wall causes the formation of two separate lumens, a true lumen and a false lumen, with blood flow penetrating the intima into the false lumen.5,8,9,11 Once the intima is penetrated, the dissection may propagate toward the iliac arteries (an antegrade dissection) or back toward the heart (a retrograde dissection). In either case, blood may pool in the false lumen, constricting the true lumen and causing a low volume of blood flow to other parts of the body, creating a fatal compromise of blood flow to essential organs at or downstream from the constriction.3,8,9 The serious complications secondary to an aortic dissection may include stroke, heart attack, aortic insufficiency, congestive heart failure, cardiac tamponade, aortic rupture, renal and/or mesenteric ischemia, and hemorrhage.5,7,9,10,13–15
Dissection typically occurs in older patients.1,7,13,16 Nearly 75% of patients are between the ages of 40 and 70, with the majority being 50 to 65 years old.1,3,6,7,9 Men are more likely to have an aortic dissection compared with women and blacks more commonly than whites.3,4,7,13,16–19 The cause of aortic dissection is unknown, but several risk factors precipitate its occurrence in individuals. 7 Some of the most common risk factors are chronic hypertension, atherosclerosis, and a blunt-force trauma to the chest or abdomen.3,7,9,16,17,20 Other risk factors may include a bicuspid aortic valve, prior heart surgery, narrowing of the aorta, connective tissue disorders, or genetic disorders such as Marfan syndrome.1–3,7,9,13,16,20
There are two classification systems for aortic dissections. The Stanford classification divides dissections into type A and type B based on their location.2,3,5,9,10,15,17 Type A dissections originate in the ascending aorta and may extend through the aortic arch to involve the descending aorta as well; type B dissections occur distal to the brachiocephalic (innominate) artery.8,14,17,21 The DeBakey classification system helps distinguish dissections requiring surgical intervention from those not needing surgery.1,2,5,8,22–24 Type I dissections originate in the ascending aorta but propagate to involve both the ascending and descending aorta; these constitute approximately 60% of cases, tend to occur in younger patients, and are the most lethal of all dissection types. Type II dissections are the least common, making up approximately 10% to 15% of cases, and only involve the ascending aorta; type III dissections constitute approximately 25% to 30% of cases and involve only the descending aorta.1,8,22 Stanford Type A dissections include both the DeBakey type I and type II dissections, while Stanford type B includes the DeBakey type III dissections.
Patients with aortic dissection may present with various displays of signs and symptoms that can imitate other medical conditions. 1 Common symptoms are chest pain that may mimic a heart attack or stroke-like symptoms. 9 Patients may have abdominal or back pain, fainting, dizziness, syncope, heavy sweating with clammy skin, weakness, shortness of breath, or a rapid, weak pulse.7,17,18 Most patients experience an acute onset of symptoms. Many have described pain as being “tearing” or “ripping” in nature.3,4,5,7,10,17,19,23 Clinical signs of a dissection may include low blood sugar, blood pressure differences between arms, heart attack or stroke signs, signs of shock, and an unusual “murmur-like” sound of the aorta. 7
Diagnostic Imaging
Because of its potential for fatal complications, aortic dissection requires rapid diagnosis and immediate treatment. Several imaging techniques may help to diagnose this condition. These include computed tomography/computed tomographic angiography (CT/CTA), sonography, and magnetic resonance imaging/angiography (MRI/MRA). Each modality has its own set of specific findings (Table 1) and its own set of relative advantages and disadvantages (Table 2). CTA is typically the imaging modality of choice when an aortic dissection is suspected because of its high degree of accuracy and its ability to provide a rapid diagnosis. Since MRA is more time-consuming, it is usually used for following up the condition. Sonography can also play a vital role in diagnosis. Sonography may not always be the modality of choice, but in some patient populations, it is able to demonstrate the pathology well. Sonography may also be the first imaging test ordered, especially for patients presenting with generalized abdominal pain. As the first line of imaging, sonography must be fast and accurate in the diagnosis of this condition.
Imaging Signs of Aortic Dissection.

CTA, computed tomographic angiography; MRA, magnetic resonance angiography.
Comparison and Contrast of Imaging Modalities for Aortic Dissection.
CTA, computed tomographic angiography; MRA, magnetic resonance angiography.
Computed Tomographic Angiography
Imaging of the aorta obviously plays a vital role in rapidly and accurately diagnosing dissection, and CT is typically the imaging modality of choice when aortic dissection is suspected.1,2,6,9,11,12,18,19,25 Although aortic dissections may be evident on a noncontrast CT examination, they are best illustrated with contrast-enhanced scans.9,20 CTA is very accurate with close to 100% sensitivity and specificity,2,19 has the advantage of being easily accessible in most medical settings,5,18 can be programmed to provide a rapid acquisition of images allowing for a quick diagnosis,1,6 and usually is able to demonstrate well the extent of the dissection.5,25 CTA may be contraindicated in some patients, though, such as those allergic to iodine contrast and patients with renal insufficiency. CTA may also occasionally exhibit false positives, showing a pseudo-dissection when there is streaking of contrast caused by motion artifact. Other conditions also may mimic a dissection such as mural thrombus, intramural hematoma, or a penetrating atherosclerotic ulceration. 3 The classic signs for dissection to look for on CTA include the visualization of a double lumen and an intimal flap of the aorta (Figure 1).5,20 Other signs seen by CTA that may help distinguish the true and false lumens are the “beak sign” and the “cobweb sign.”2,20 The beak sign can be seen when there is hematoma present within the distal portion of the false lumen.2,20 The cobweb sign is sometimes present when thin, string-like filaments of the media layer are seen in the false lumen.2,20
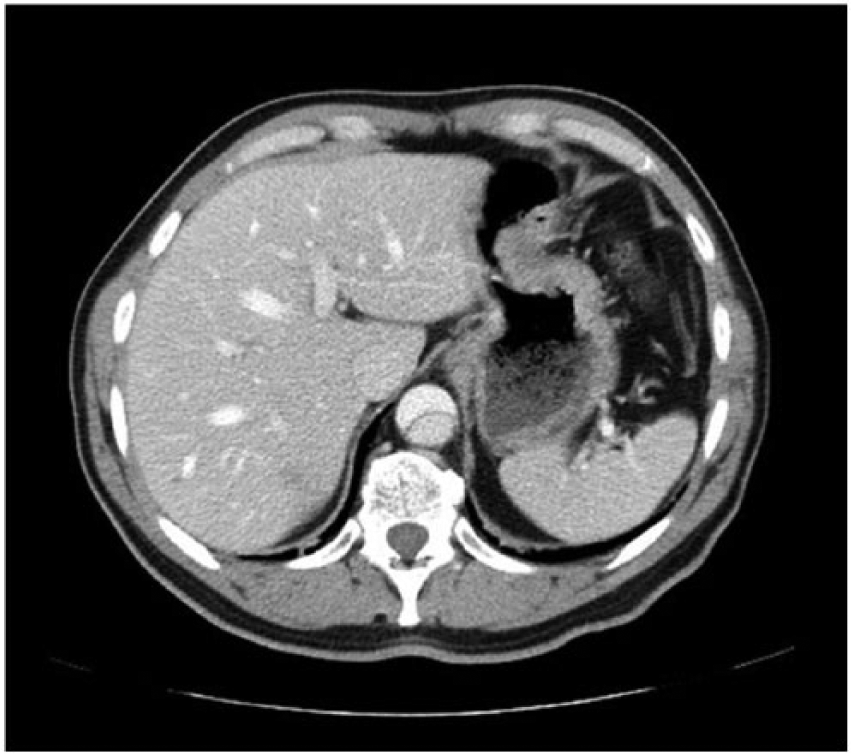
Computed tomographic angiographic axial image of a Stanford type B aortic dissection extending antegrade with involvement of the abdominal aorta (with permission from radiopaedia.org).
Sonography
Since CTA is better able to demonstrate the extent of the dissection in an emergent situation, sonography may not always be the imaging modality of choice. It is important to recognize key sonographic findings, though, since for patients with atypical symptoms of dissection such as generalized abdominal pain, sonography may be the initial imaging test. 10 The classic sonographic appearance of an aortic dissection is the presence of an intimal flap that appears as a linear, hyperechoic structure separating the aorta into true and false lumens (Figures 2 and 3). 10 With color Doppler imaging and spectral Doppler, the two lumens will exhibit different hemodynamic characteristics and flow patterns (Figures 4 and 5). Sonographic evaluation has the advantage of being easily accessible, and it is a rapid imaging technique much like CTA without the need for ionizing radiation.10,11 Disadvantages of the modality are that it may be limited by patient habitus and bowel gas. Abdominal ultrasound may also be limited in showing the extent of the disease, especially if the dissection involves the ascending aorta, aortic arch, or small vessel branches. Transesophageal echocardiography (TEE) is now becoming more widely used in the diagnosis of aortic dissection involving the thoracic aorta, particularly the ascending aorta, with some limitations in its ability to visualize completely the aortic arch. TEE is reported to have greater than 95% sensitivity and specificity for this disease. 26 Another potential disadvantage of sonography is that imaging is highly dependent on the sonographer’s expertise and skill level.
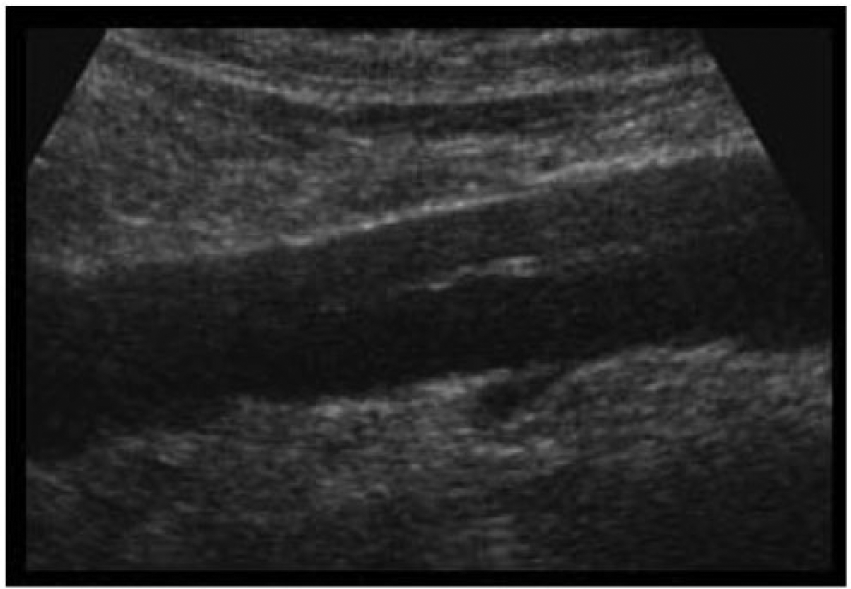
Longitudinal gray-scale image of the abdominal aorta showing a hyperechoic intimal flap separating the aorta into a true lumen and a false lumen characteristic of a dissection (with permission from ultrasoundcases.info).
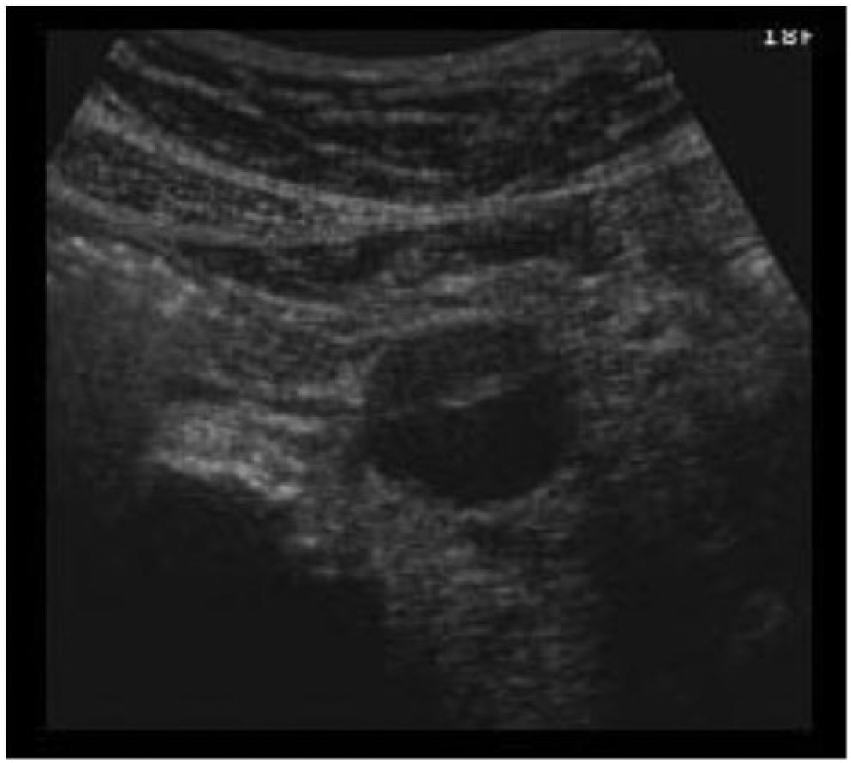
Transverse gray-scale image of the abdominal aorta showing a hyperechoic intimal flap characteristic of a dissection (with permission from ultrasoundcases.info).
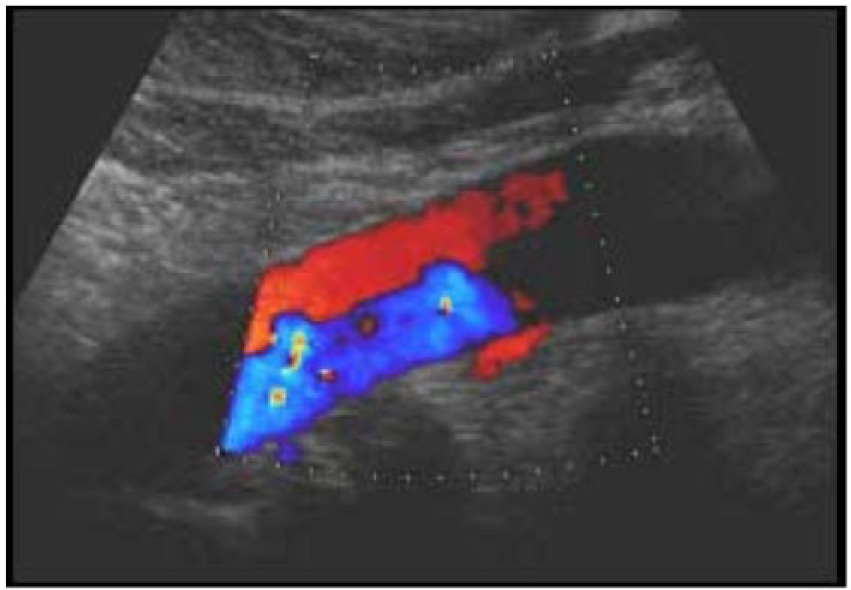
Longitudinal color Doppler image of the abdominal aorta showing two distinct lumens with different hemodynamic characteristics, one lumen with antegrade flow and one with retrograde flow (with permission from ultrasoundcases.info).
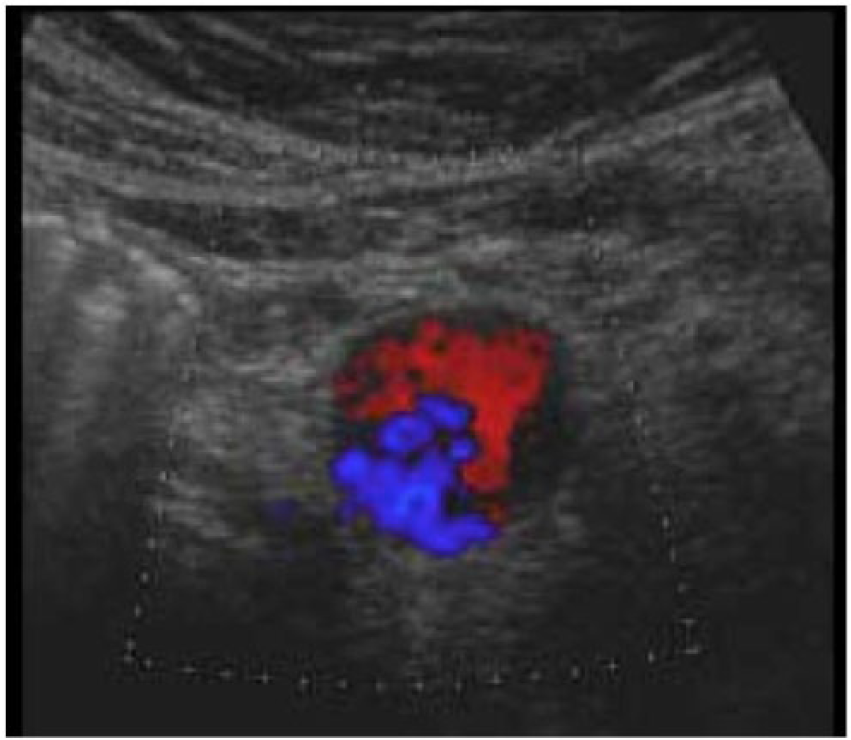
Transverse color Doppler image of the abdominal aorta showing two distinct lumens with different hemodynamic characteristics (with permission from ultrasoundcases.info).
Magnetic Resonance Angiography
MRA may also play a key role in imaging the patient with aortic dissection. As imaging technology developed in the 1990s, MRA was considered at the forefront and was the best imaging technique for displaying aortic dissection. 25 Advantages of this technique are its high degree of accuracy and its lack of ionizing radiation. 18 Like CTA, MRA has also been found to have close to 100% sensitivity and specificity.18,19 Although contrast MRA is excellent at demonstrating aortic dissection, it is rarely used now for emergent cases because it is not as widely available as CTA in that setting. 10 It also is rarely employed due to the time-consuming nature of the examination, which contradicts the need for rapid diagnosis,1,6,9 and it is not as safe an examination for a critically and acutely ill patient in the emergency department. Since MRA is excellent at demonstrating dissection, though, it is commonly used for routine follow-up and management of the condition.5,9 Dissection is visible on MRA with the visualization of an intimal flap, which appears as a linear structure (Figures 6 and 7). 27 Contrast enhancement helps to distinguish the true and false lumens. 26 The true lumen is seen void of signal, whereas the false lumen is seen with high signal intensities representing disturbed flow.27,28
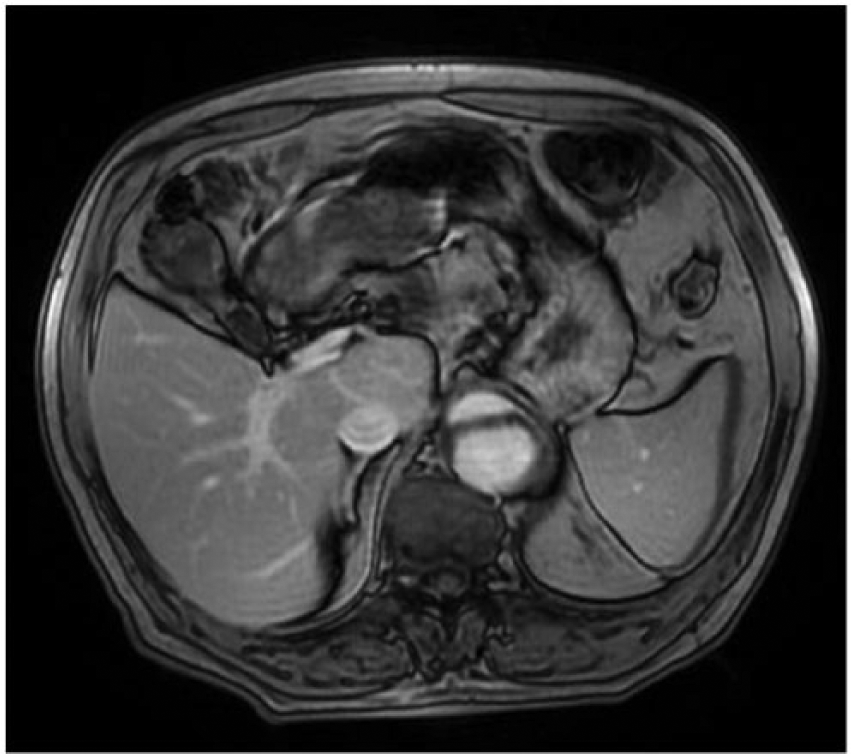
Magnetic resonance angiographic axial image of a Stanford type B aortic dissection extending antegrade with involvement of the abdominal aorta (with permission from radiopaedia.org).
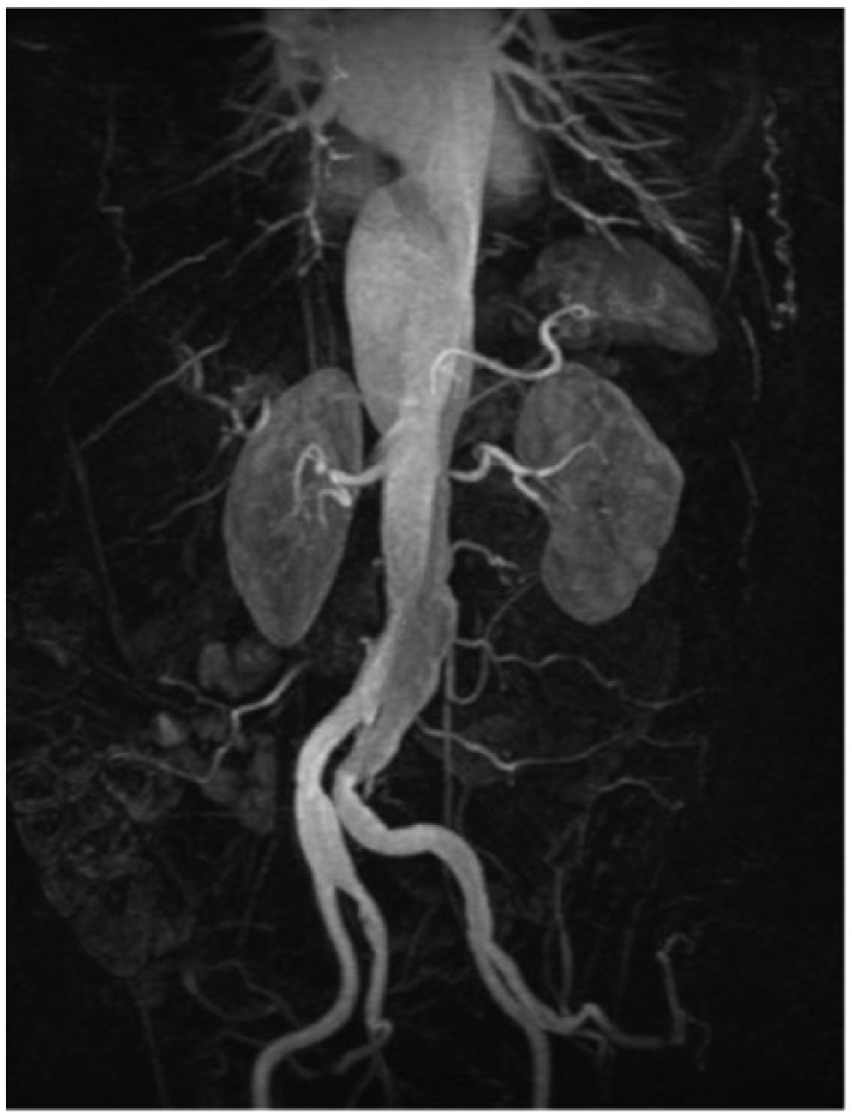
Magnetic resonance angiographic longitudinal image of a Stanford type B aortic dissection extending antegrade into the iliac arteries (with permission from radiopaedia.org).
Differential Diagnosis
There may be many different differential diagnoses to consider, since patients typically present with acute chest, abdominal, and/or back pain. 9 Even though nearly 10,000 people in the United States each year are affected by aortic dissection, it is a rare condition compared with other acute abdominal pathologies. When patients present with acute and generalized abdominal or back pain, physicians may suspect the more common abdominal pathologies such as gallbladder or renal diseases and conditions, typically the reasons that sonography may likely be the first type of imaging a patient receives. It is important for sonographers to carefully evaluate the aorta when a patient presents with an acute onset of these symptoms. By doing so, the sonographer will be able to rule out or pinpoint involvement of the abdominal aorta in a dissection quickly so that the patient can get the proper treatment in a timely manner. Since aortic dissection can have similar signs and symptoms as intramural thrombosis, intramural hematoma, or penetrating aortic ulcer, it may sometimes be mistaken clinically for these acute aortic conditions.3,20 For patients experiencing chest pain with an aortic dissection, the physician may suspect other pathology with the differential diagnosis, including myocardial infarction or a pulmonary embolism.2,7
Treatment
Treatment of aortic dissections is dependent on the severity and type. Most dissections are type A, which requires surgically invention.13,17,20 Type B or DeBakey type III descending aortic dissections typically are medically managed without surgical invention.5,7,13 Treatment is aimed at stabilizing the dissection and effectively managing hypertension using beta-blockers or calcium channel blockers.7,9,13,24 Patients are very closely followed by routinely checking their blood pressure and performing serial imaging studies. 24 A baseline CTA or MRA study should be obtained prior to a patient’s release from the hospital, with follow-up studies done at 3 months and then at 6-month intervals for a 2-year period. If the dissection remains stable after 2 years, it is recommended that patients have a yearly imaging examination thereafter. 9
Prognosis
Aortic dissection has a high mortality rate, which is why rapid diagnosis and treatment is so important. The mortality rate for patients with type A who undergo surgical invention can be as high as 30%.1,4,13 Patients who are unable to undergo surgery and must be medically managed have even higher mortality rates, approaching 60%.4,13 Patients with type B dissections who are medically managed have a 10% mortality rate, and those who undergo surgery have a 30% mortality rate.3,13
Conclusion
Imaging plays a critical role in accurately and rapidly diagnosing aortic dissection. Since this is a potentially fatal condition, a fast diagnosis is required to improve the patient’s outcome and chances of survival. CTA, sonography, and MRA may all play a role in the diagnosis and follow-up imaging of dissections. Each modality has its advantages and disadvantages. For this particular condition, CTA is typically the best imaging modality to use for rapid diagnosis. MRA is excellent at following up a known dissecting aorta. Although sonography is not typically used because of the effectiveness of CTA, in the emergent setting, it may be the initial test performed. This is especially true for patients with atypical or generalized symptoms and is the reason sonographers must possess adequate knowledge of the condition and know the key signs to look for to make a positive diagnosis.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
