Abstract
Gastrointestinal stromal tumors (GISTs) arise from mesenchymal tissue and represent the majority of gastric tumors. Diagnostic workup of GISTs is usually done through immunohistochemistry and imaging, typically not including sonography. A case is presented of a GIST that was initially identified sonographically with an unknown primary site. Sonography detected and characterized the mass, followed by the use of computed tomography to obtain additional information that led to the diagnosis. The use of both diagnostic imaging modalities allowed for accurate diagnosis and helped to determine the next step in proper treatment of the tumor.
Stomach tumors can be epithelial or mesenchymal in nature, with most duodenal tumors being mesenchymal. 1 Gastrointestinal stromal tumors (GISTs) are a form of mesenchymal tumors that are most commonly found within the gastrointestinal (GI) tract. These tumors typically occur in individuals between the ages of 40 and 70 years, with a mean age of 63 years.1–3 Sonography, fine-needle aspiration, and computed tomography (CT) are all modalities that can be used to detect and properly diagnose GISTs. This case presents a patient with a well-visualized GIST arising from the duodenum and projecting into the abdominal cavity. Sonography provided initial detection of the tumor, while CT confirmed the diagnosis.
Case Report
A 64-year-old Caucasian woman with a history of jaundice, nausea, hemorrhaging, and congestive heart failure presented to the sonography department for an abdominal sonogram. Her laboratory results showed abnormal values, including low potassium levels and increased sodium levels. Sonography was performed using a Philips iU22 ultrasound machine with a 6 MHz curvilinear array transducer (Philips Ultrasound, Bothell, Washington).
The abdominal sonogram showed a homogeneous liver, with a hyperechoic lesion seen posteriorly in the left hepatic lobe measuring 1.8 cm in diameter (Figure 1). This lesion was thought to most likely be a hemangioma. Other similar hyperechoic areas of the liver were indicative of hemangiomas, as well as possible lymphoma and/or metastatic disease of the spleen. The portal and hepatic veins appeared patent. The patient had a previous cholecystectomy, and no biliary ductal dilatation was seen. No abnormalities of the pancreas were visualized. Visualized adjacent to the pancreas was a round hypoechoic mass measuring 4.0 × 3.1 cm (Figures 2 and 3). The mass appeared to be associated with the gastric antral wall. The spleen measured 11.95 cm in length, with multiple granulomas noted throughout (Figure 4). Numerous low-density foci were seen throughout the spleen, some of which were partially calcified (Figure 5). Both kidneys were seen to be within normal limits and without any signs of stones or hydronephrosis. The inferior vena cava and abdominal aorta were widely patent, and no aneurysm was seen.
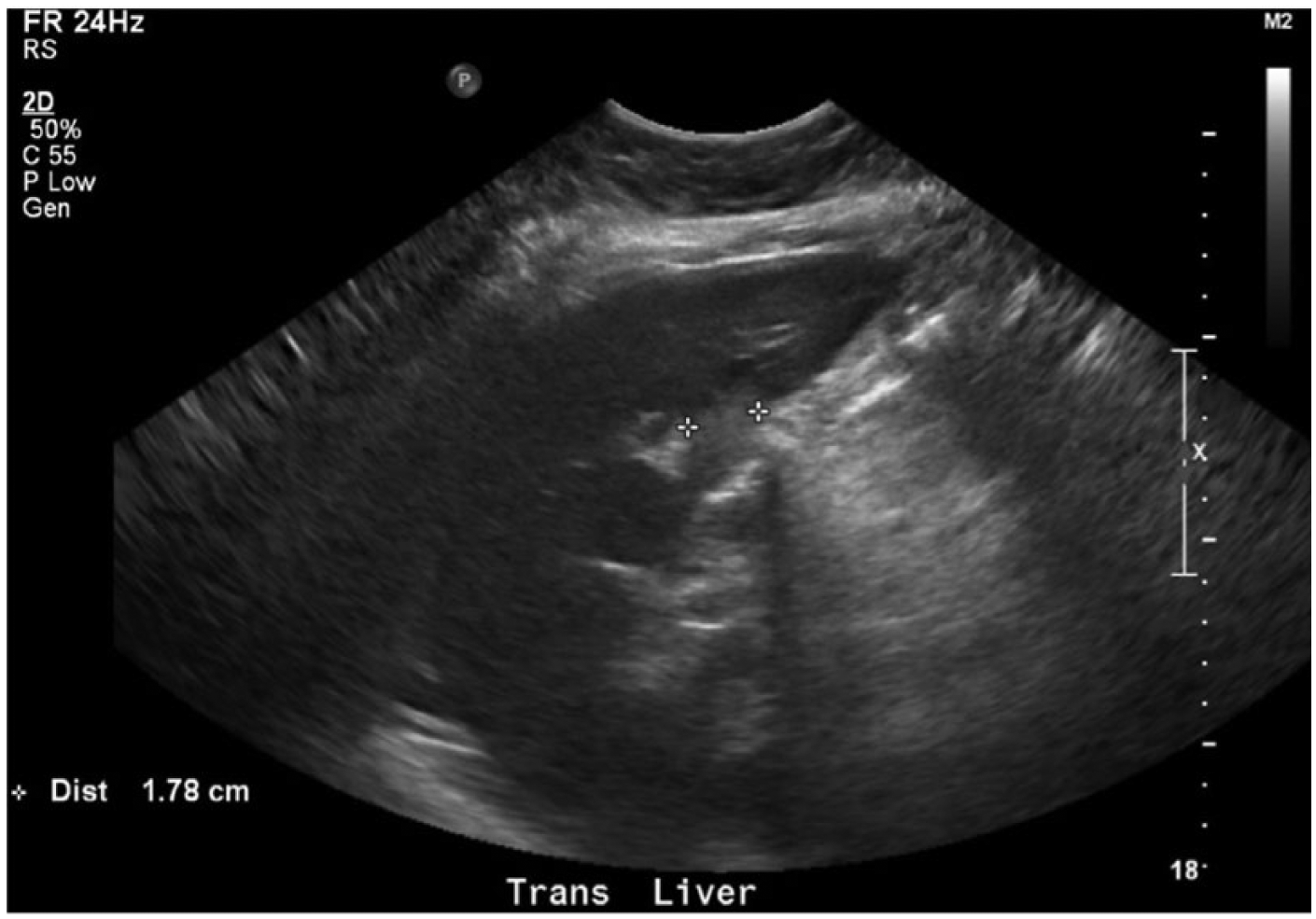
Transverse gray-scale sonogram of the liver showing a 1.8-cm hyperechoic lesion within the left hepatic lobe considered characteristic of a hemangioma.
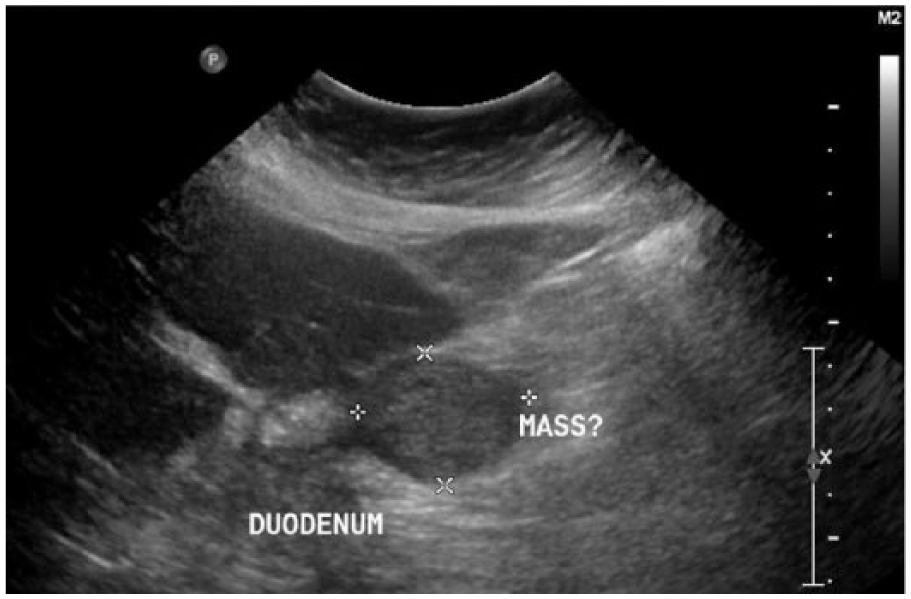
Transverse gray-scale sonogram showing a hypoechoic mass located adjacent to the pancreas and duodenum, measuring 4.0 × 3.1 cm.
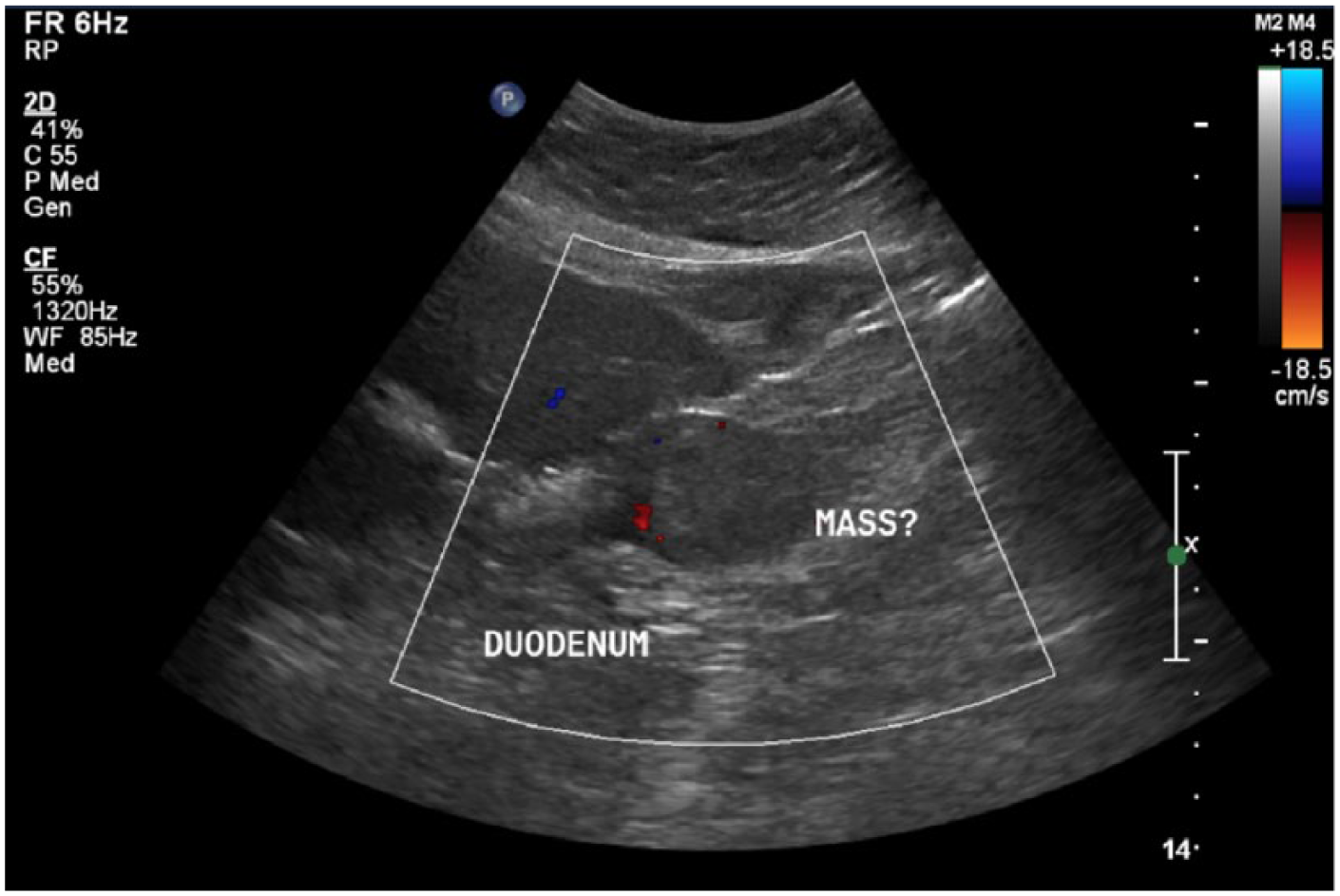
Transverse color Doppler image showing the presence of vascularity within the hypoechoic mass.
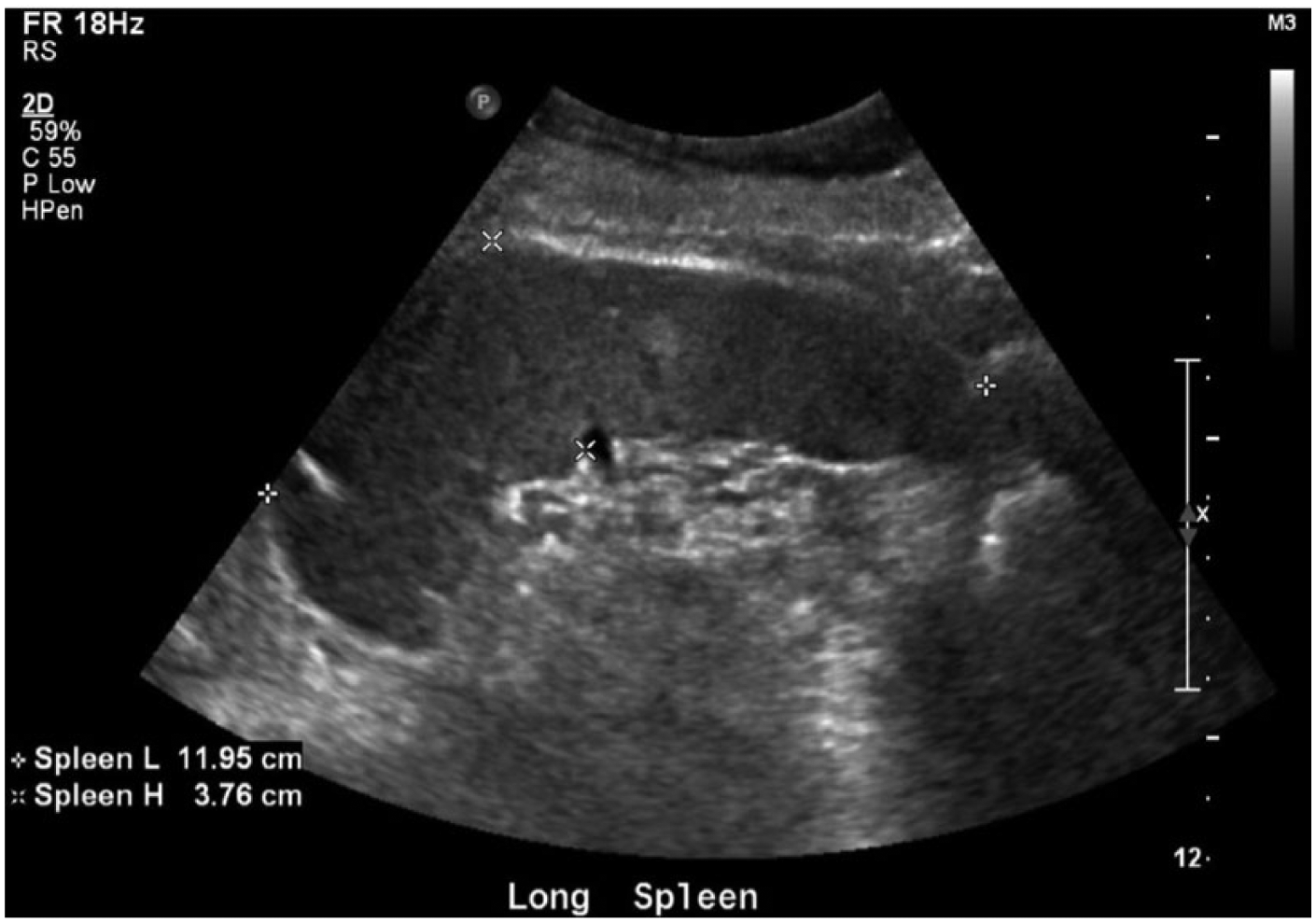
Longitudinal gray-scale sonogram of the spleen showing a splenic length of 11.95 cm and a region of hyperechogenicity.
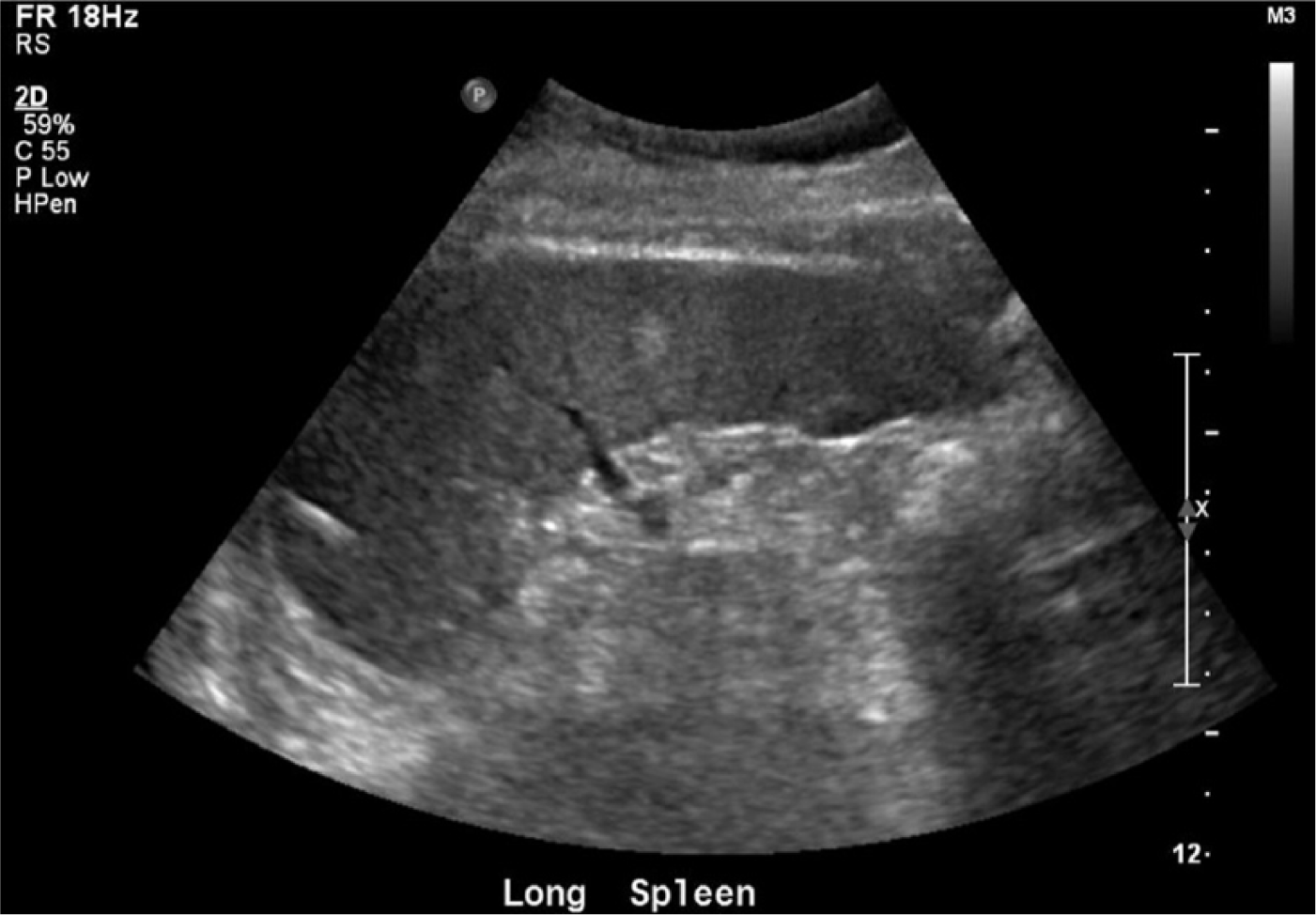
Longitudinal gray-scale sonogram of the spleen showing similar multiple areas of echogenicity throughout the spleen.
Previous CT of the chest that was completed 5 days prior to the sonogram showed a suspicious area near the gastric antrum (Figure 6); however, the area was visualized in only the last few images. Based on the sonographic findings, which suggested a potentially malignant mass, abdominal CT with contrast was performed for further evaluation, which demonstrated clear lung bases and a 3.4-cm mass arising from the gastric antrum/proximal duodenum (Figures 7 and 8), corresponding with the sonographic findings. Unfortunately, no further follow-up was available for this patient.
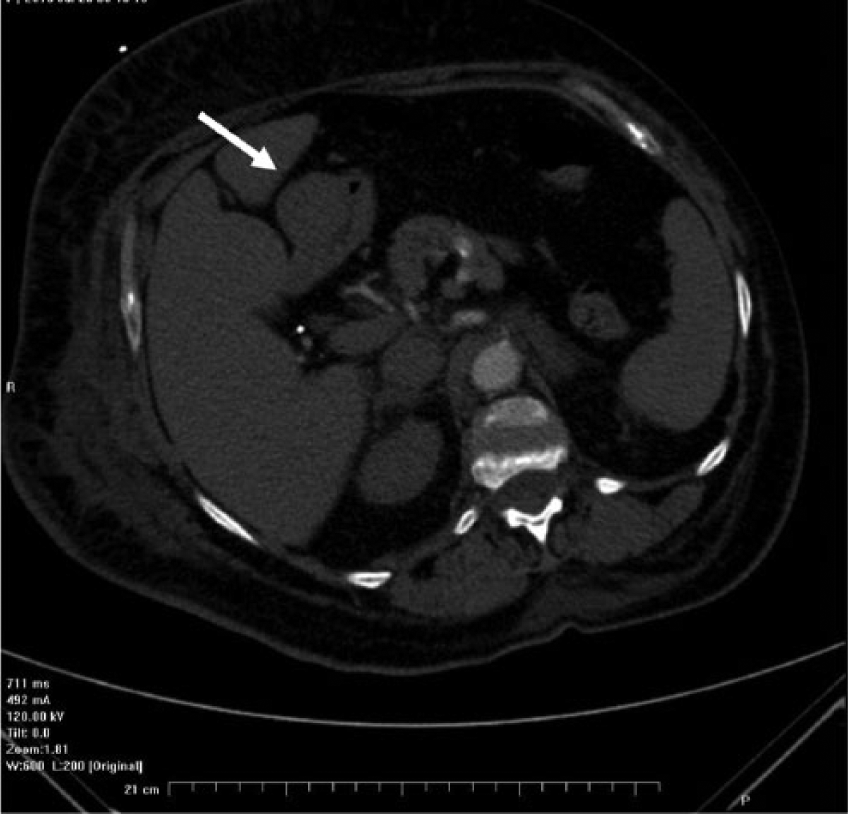
Initial computed tomogram without contrast showing the presence of a questionable mass (arrow) located near the gastric antrum.
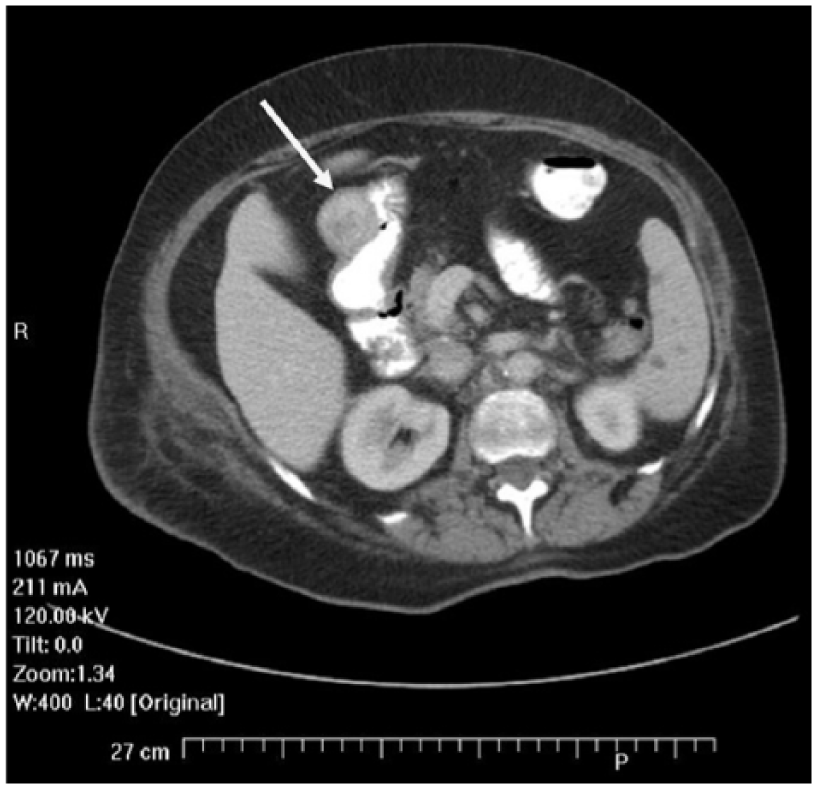
Secondary computed tomogram with contrast clearly depicting the mass (arrow) located at the gastric antrum.
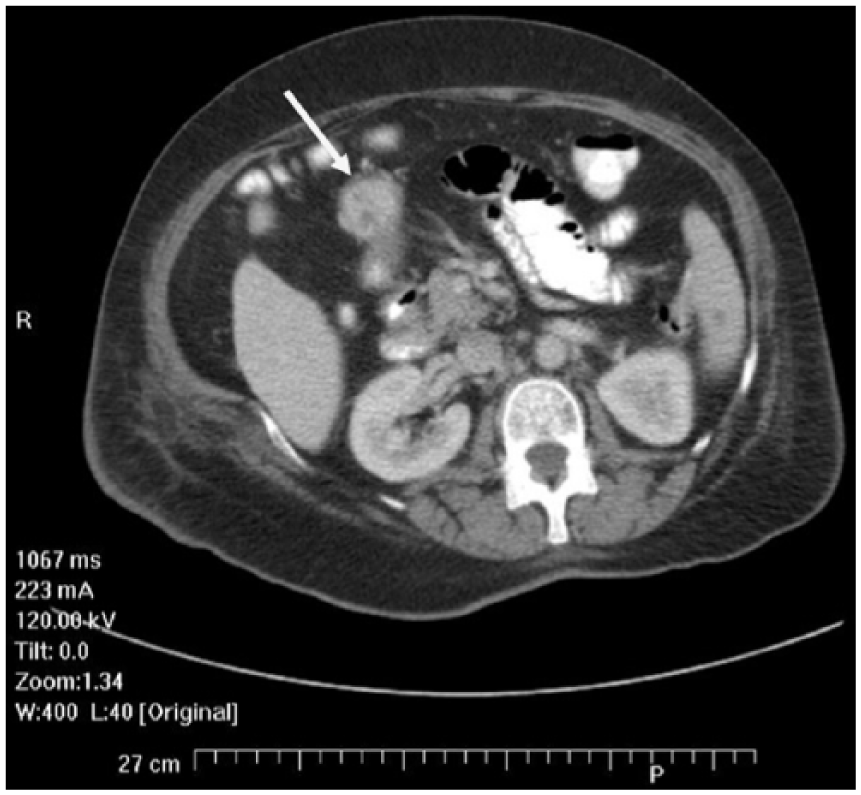
Additional image from the secondary computed tomogram study taken more caudad showing the presence of the mass (arrow) located at the gastric antrum.
Discussion
Stomach tumors can be epithelial or mesenchymal in nature, with most duodenal tumors being mesenchymal. 1 Tumors of the GI tract can include polyps, leiomyomas, lipomas, adenomas, as well as other vascular and neurogenic neoplasms. GISTs are a form of mesenchymal tumors that are most commonly found within the GI tract. In 90% to 95% of cases, these tumors are transmembrane tyrosine kinase (KIT, CD117)–expressing and signaling tumors.3,4 They were originally thought to arise from cells found within the GI tract called interstitial cells of Cajal. 2 More recently, it was discovered that they arise from multipotential mesenchymal stem cells. 3 Mutations are associated with a familial and sporadic likelihood that these tumors will develop; however, most are not inherited. GISTs are diagnosed in 10 to 20 million people per year; these tumors are most often of a benign and slow-growing nature, and any further treatment is completed on a case-by-case basis.1–3 Fifty to sixty percent of GISTs are found within the stomach, whereas 30% to 40% are found within the small intestine. GISTs make up <1% of all malignancies associated with the GI tract and are said to affect men slightly more so than women.5,6 The annual occurrence rate of malignant tumors in the United States ranges between 3000 and 4000 cases a year. 1 These tumors typically occur in individuals between the ages of 40 and 70 years, with a mean age of 63 years.
The appearance of these tumors will vary, ranging in size from 0.5 to 44.0 cm. 1 Normally, they appear as an exophytic growth resembling a mass attached to the stomach or small intestine, projecting out into the abdominal cavity. Sonographically, they appear as well-circumscribed masses displaying mixed gray tones, and they usually contain a pseudo-capsule.2,3 Hemorrhage, cystic degeneration, and necrosis may also be seen within the tumor. Clinically, the presentation of these tumors will vary, with 70% of patients being symptomatic and 20% asymptomatic; the remaining 10% go undetected until an autopsy is performed.
Tumor location in the fundus or gastroesophageal junction of the stomach has been documented as being unfavorable toward treatment, whereas tumors in the antrum of the stomach have been documented as a favorable location for successful treatment.5,6 Some patients will remain completely asymptomatic with the presence of a GIST; however, common signs and symptoms may include pain or abdominal swelling, nausea, vomiting, and loss of appetite, as well as weight loss. Hemorrhaging can sometimes occur, which in turn will lower the red blood cell count of the individual, as well as lead to weakness and fatigue. Hemorrhaging within the intestinal tract may lead to tarry-appearing stool, whereas bleeding within the throat may lead to hematemesis.
Sonography, fine-needle aspiration, and CT are all modalities that can be used to detect and properly diagnose GISTs.5–7 Diagnostic evaluation of this tumor can prove to be difficult through sonography alone; therefore, incorporating other imaging modalities is beneficial in making the diagnosis and evaluating the tumor components and origination. This case presented a patient with a well-visualized GIST arising from the duodenum and projecting into the abdominal cavity. Sonography provided the initial detection of the tumor, while CT confirmed the diagnosis. The well-circumscribed mass visualized adjacent to the pancreatic head warranted further investigation. CT confirmed the diagnosis of a gastric antrum tumor—specifically, a GIST. The lack of available follow-up leaves the presence of metastatic disease to the spleen and the precise origin of the malignancy unknown.
Surgical removal is the only known curative therapy for treating malignant GISTs. Stomach lesions can be treated endoscopically with snare resection, laser ablation, or mucosal resection.8,9 Other options include open procedures involving local and wedge resections or gastrectomy. Newer treatment methods are completed laparoscopically, providing a shorter period of recovery, better immune response, and an earlier discharge. The median survival rate of a recurrent GIST after resection was only 15 months prior to the introduction of imatinib. 10 Imatinib mesylate is a targeted therapy against cancer cells—specifically, an inhibitor of signal transduction (i.e., a protein tyrosine kinase inhibitor). This drug essentially repairs the inactive conformation of the KIT gene.10,11 Treatment with this drug therapy has become the standard protocol for dealing with solid masses of the GI tract.
Despite this success, GISTs are not always completely resolved with targeted therapy. If there is any progression in the tumor within 6 months of treatment, 50% of these patients will relapse after 2 years.10,11 In terms of secondary resistance, progression of the tumor can appear as an increase in the density of the tumor as opposed to an increase in the actual size. When visualized with CT, it can also appear as a hyperdense lesion within a hypodense lesion. 3 The prognosis of a low-risk GIST after a complete resection is promising, with a 5-year survival rate up to 30%.2,9 The most common time frame that a GIST will return is within 3 and 5 years of resection; however, this recurrence is strongly correlated to the size, location, and mitotic rate of the tumor. However, GISTs can be very unpredictable, so it is imperative that long-term follow-up be implemented for each patient, regardless of whether the tumor was originally malignant or benign. 12 In a study by Dematteo et al. in 2000 involving 200 patients with GISTs, more than half had gastric GISTs. 13 Ninety-three of those patients had primary GISTs and opted for resection, with a median survival rate of 54%. In a study by Fujimoto et al. of 140 patients with GISTs, of the 129 that had “curative” treatment, the survival rates were 88% to 93%. 14
Conclusion
GISTs are among the most common mesenchymal tumors of the GI system. 2 Complete surgical resection of a GIST remains the primary treatment option; however, these are all considered on a case-by-case basis. The present case shows that sonography can play an important role as an imaging modality to detect an abnormality outright and assist in proper diagnosis. Prompt detection and diagnosis are critical to provide the patient with proper treatment.
Footnotes
Acknowledgements
The authors thank Alli Egart, RDMS, RVT, Susan Urban, RMDS, RVT, Kimberly Grandstaff, RDMS, Melissa Hirner, RDMS, Ella Rakhlenko, RDMS, RVT, and Janet Baker for their assistance, leadership, and guidance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
