Abstract
Biopsy of the transplanted kidney is known to cause intraparenchymal renal vascular injury. The most common vascular complications include arteriovenous fistula (AVF) and pseudoaneurysm (PA). We report a case of a patient who underwent cadaveric renal transplantation and developed an AVF and a PA after an on-table wedge biopsy. The PA was missed during the standard Doppler sonographic examination because of its small size but was unmasked with the use of contrast-enhanced ultrasound (CEUS). Fourteen days after the first CEUS, the procedure was repeated and showed complete thrombosis of the PA.
Introduction
Baseline biopsy of a transplanted kidney has become a routine tool to provide insight into preexisting diseases relevant for comparative analyses after transplantation.1–5 However, renal transplant biopsy using the core needle technique is known to cause intraparenchymal vascular injury.6–12 The most common complications include arteriovenous fistula (AVF) and pseudoaneurysm (PA).10,12 We report a case of an AVF and a PA in a kidney transplant recipient after an on-table wedge biopsy. This case highlights the importance of contrast-enhanced ultrasonography (CEUS) for diagnosis.
Case Report
A 39-year-old man with end-stage renal failure underwent cadaveric renal transplantation from a 21-year-old male donor. An on-table wedge transplant biopsy was performed. Post-transplant recovery was complicated by delayed graft function requiring dialysis. Abdominal color Doppler ultrasonography (CDUS) done at day 5 post transplant showed an AVF. Since we began to perform CEUS in our facility as clinical research, the hospital ethics committee approved the study according to local legal requirements, and informed consent for contrast use was obtained from the patient. Two days later, a CEUS using a Sonovue (Bracco Int; Milan, Italy) bolus of 2.4 ml injected through a 20-gauge intravenous cannula, followed by a 10 ml saline flush, showed in addition a PA measuring 10.6 × 8.5 mm in the lower lobe (Figure 1, arrow) whose outer border was 6.3 mm deep in relation to the kidney surface. Two days later, the patient had an episode of hypotension due to gastrointestinal hemorrhage, presumed secondary to the high dosage of corticosteroids used in his immunosuppressive regimen, which rapidly improved after blood transfusion. Fourteen days after the first CEUS, the procedure was repeated in the same manner and showed complete spontaneous thrombosis of the PA (Figure 2, arrow). The patient reported no side effects during either contrast procedure.
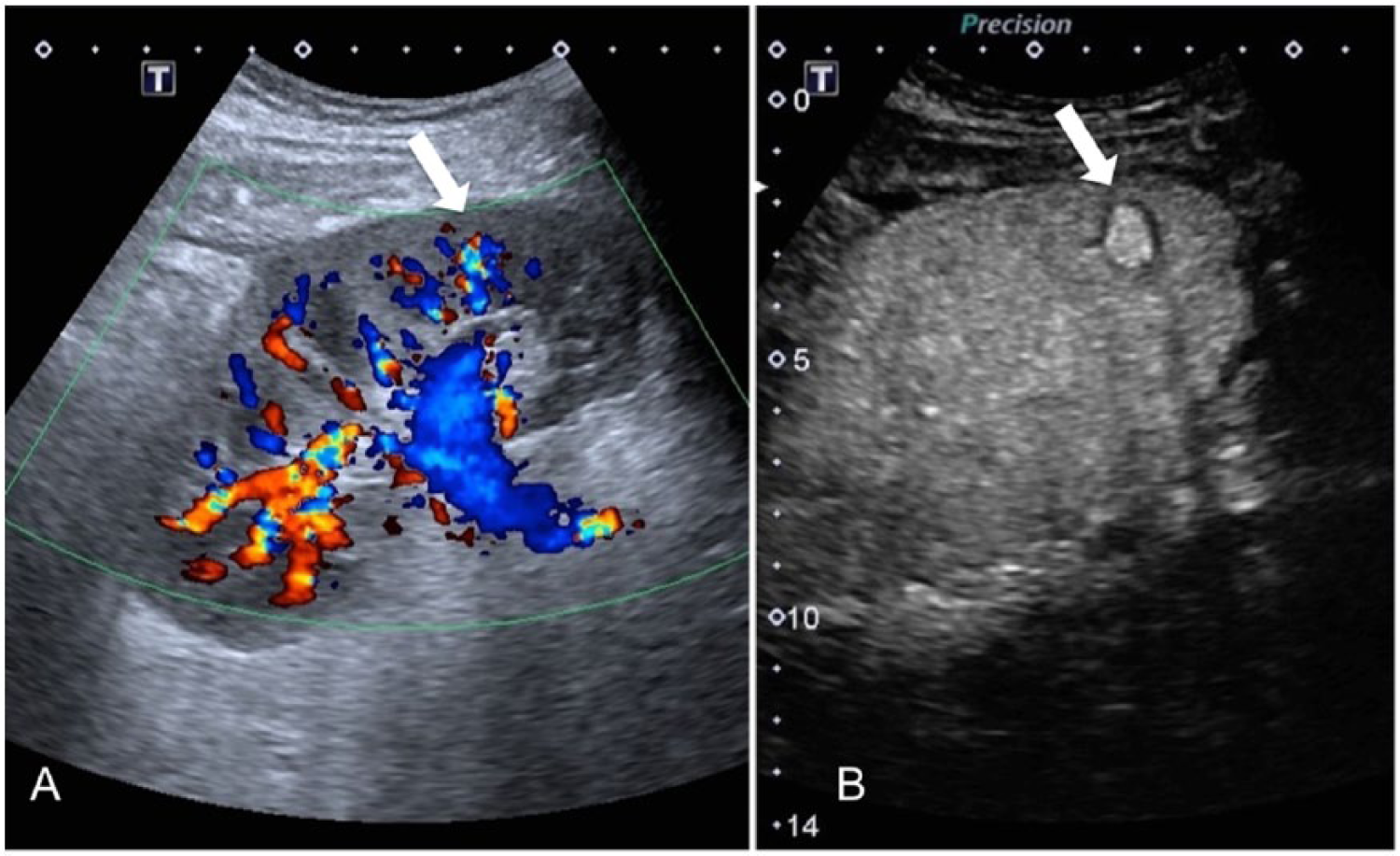
(A) Contrast-enhanced color Doppler image of the transplant kidney showing a small region of flow in the lower pole (arrow) in the cortical region. (B) Contrast-enhanced gray-scale image of the lower pole of the transplant kidney clearly showing the site and size of the small pseudoaneurysm (arrow).
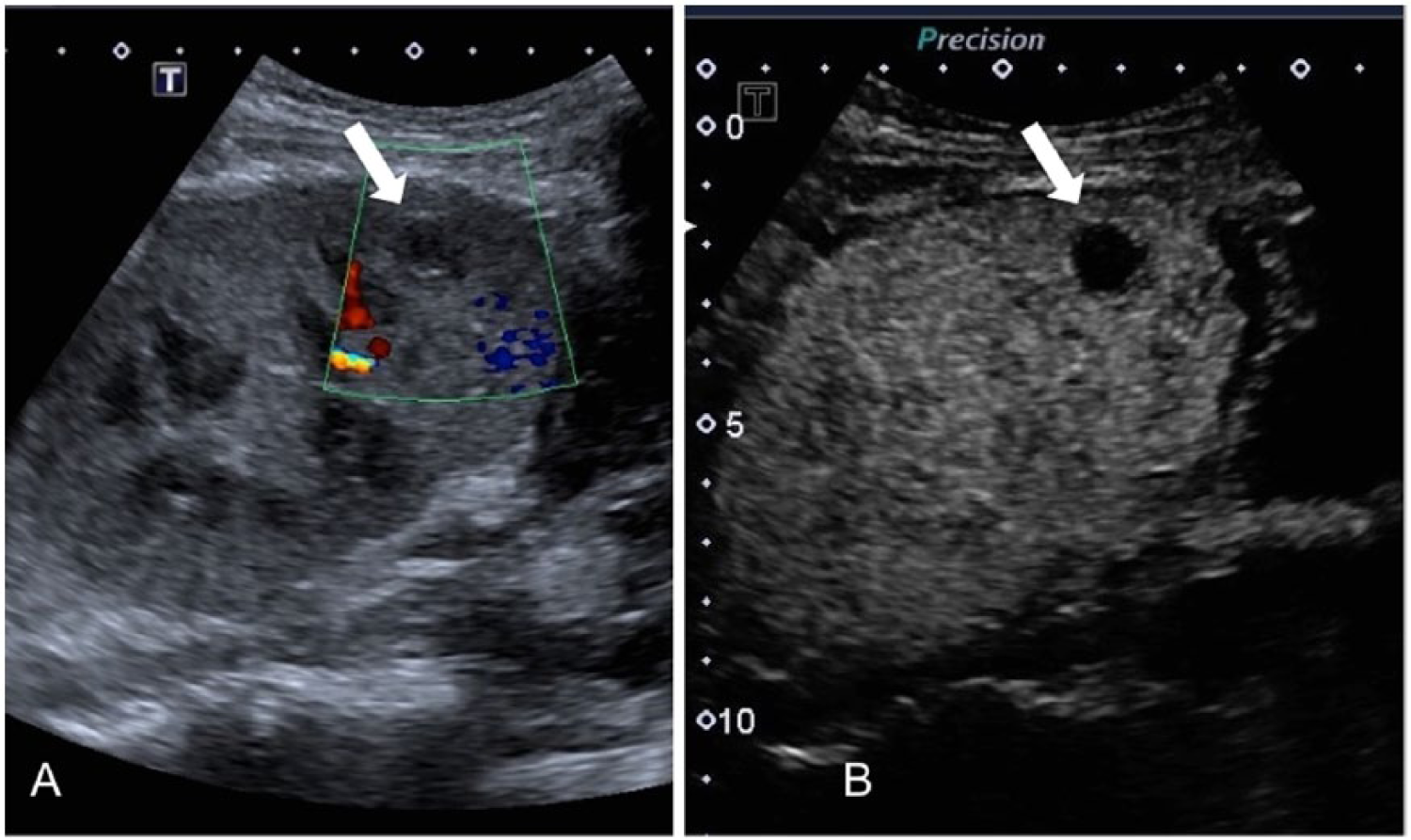
(A) Follow-up contrast-enhanced color Doppler image of the site of the previously seen pseudoaneurysm (arrow) with no evidence of flow within the sac. (B) Follow-up contrast-enhanced gray-scale image of the site of the previously seen pseudoaneurysm (arrow) with no evidence of flow within the sac, indicative of complete spontaneous thrombosis.
Discussion
Baseline biopsies, also called on-table or “zero-hour” biopsies, are procedures done while the patient is still in the operating room and constitute a reference point for future grafts’ histological assessments.1–5 However, it is a procedure with certain risks, with AVF and PA being the most common complications.6,11,12 These 2 complications may even develop concomitantly, 13 as seen in the case report presented.
An unusual finding in this case was the occurrence of an AVF and PA secondary to a wedge technique biopsy, which is done using a scalpel instead of a core needle. A wedge biopsy is generally considered limited to the subcapsular-cortical area of the kidney, with the recommended depth of no more than 5 mm, though depths from 1.8 to 15.0 mm have been reported.1,4,7,14,15 A deeper sample, normally obtained by needle core, is more likely to include arcuate arteries than the wedge technique.4,7 However, in practice, the size of a wedge biopsy is operator dependent and therefore highly variable.7,14 For the case presented, it is reasonable to assume that the site of the arterial wall lesion is near the center of the pseudoaneurysm, which was noted to be at a depth of 11.6 cm. That depth is about the same as that of the corticomedullary border where the arcuate vessels run; therefore, the cutting edge could have damaged arcuate vessels during the biopsy.
Although it has been stated that there is a lower incidence of post-biopsy complications using a wedge in comparison to the core needle technique, studies about this issue are lacking as most comparative reports look at different core needle techniques.6,8,9,11,12 No report on the complication rate of wedge biopsy was found in published studies.4,7 In three reports on post-biopsy pseudoaneurysm, the biopsy was done by means of the core needle technique.13,16,17 Two studies reported an AVF but no PA secondary to an on-table biopsy; however, in one of them, the biopsy was performed with a needle, and the other did not state the biopsy technique used.5,18
The second finding in this case worth commenting on is that the PA was unmasked only with contrast-enhanced ultrasonography. This is the first case of biopsy-related renal transplant PA that was diagnosed in our facility and coincidentally found only after instituting the use of CEUS. After finding the PA by CEUS, a second CDUS was done to ascertain that the prior lack of visualization was not caused by poor examination. Even with the knowledge of the location of the PA, color Doppler ultrasonography showed no significant characteristic Doppler signals, with no mosaic or to-and-fro yin-yang sign present, which could be interpreted as a PA without this prior knowledge.
Post-biopsy renal transplant pseudoaneurysms have been seen using conventional CDUS. Unlike our case, however, the PA size was equal to or greater than 2 cm. 6 Thus, the discrepancy may be explained by the fact that in our case the PA was smaller (approximately 1 cm) and the blood flow velocity within it probably too low to produce a mosaic or classic yin-yang sign. This result is consistent with the findings of a previous study that evaluated a total of 2824 biopsies with Doppler and found 235 AVFs but no PA, 19 while a reported study that used angiography to investigate 29 haematuria cases following renal procedures found more PAs (19) than AVFs (5). 6 We hypothesize that PAs were likely missed by Doppler evaluation in the former study because of their small size. In one published report, CDUS failed to show a PA that was seen later by arteriography; unfortunately, the size of the PA was not given. 13
Contrast-enhanced ultrasonography clearly provided excellent visualization of the morphology of the PA before and after thrombosis. We have found the use of contrast to be an easy and safe procedure, with no contrast-related complications. 20 In the case presented, certainly it provided increased sensitivity to a small vascular lesion not detected by conventional color Doppler sonography.
Another finding in our case was the spontaneous resolution of the PA. We could not determine whether the resolution of the PA was secondary to the patient’s episode of hypotension caused by the gastric hemorrhage or not; however, hypotension is a well-known cause of arteriovenous fistulae thrombosis. 21 It has also been well documented that post-biopsy AVFs and PAs resolve spontaneously in the great majority of cases. 22 It should be noted, however, that in some cases a PA may expand to an extent that requires intervention, so follow-up of these lesions is important clinically.17,23
Conclusion
To the best of our knowledge, this is the first case in which the appearance and spontaneous resolution of a post-biopsy renal transplant pseudoaneurysm was detected and followed by contrast-enhanced ultrasonography. Based on this case report and the literature, we feel confident that CEUS is an easy and safe procedure that is superior to CDUS for small vascular focal lesions. However, with so few reported cases in the literature on complications due to PA, the strict criteria to recommend surveillance of PA with CEUS remain to be delineated.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for the research, authorship, and/or publication of this article received in part via Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ). Bracco (Milan, Italy) provided the contrast agent (Sonovue) free of charge.
