Abstract
The delayed imaging phase of hepatic hemangiomas, during a contrast-enhanced ultrasound (CEUS), was investigated utlizing a recent-generation ultrasound equipment system. From April 2021 to October 2021, ten patients (seven women and three men), with a median age of 52.2 years, were retrospectively reviewed, based on having a known or probable hepatic hemangioma, at least 15–60 mm in diameter. The CEUS examination was conducted using an Aplio i800 (Canon Medical Systems, Otawara, Japan) ultrasound equipment system and a blood pool contrast agent (SonoVue; Bracco, Milan, Italy). The imaging evaluation of the hemangiomas with CEUS was extended beyond 240 seconds from contrast injection. In this study, 80% of hepatic hemangiomas showed washout from 240 to 540 seconds, after contrast bolus injection. Previous studies indicated that hepatic hemangiomas demonstrated a washout in the delayed phase and were considered atypical and rare. However, by using a recent ultrasound equipment system, these lesions may appear more frequently. Since this atypical aspect of hemangioma could lead to confusion, based on a differential diagnosis, with liver malignancy, further studies are needed.
The use of grayscale ultrasonography is widely accepted for the detection of focal hepatic lesions. Hemangiomas are among the most common benign lesions and are frequently encountered as an incidental diagnostic finding. 1 Nevertheless, the diagnostic findings with grayscale ultrasonography can be very nonspecific. 2 Contrast-enhanced ultrasound (CEUS) can aid in the confirmatory diagnosis of a hemangioma, as this imaging technique has a specific enhancement pattern with high diagnostic accuracy.3 –5 The published literature provides few reports of cases of a hepatic hemangioma that presents with a contrast washout, during the venous phase, as this can determine low diagnostic accuracy.6 –8 Indeed, the aforementioned atypical CEUS pattern of constrast enhancement causes difficulties in the differential diagnosis, when considering possible liver metastases or primary liver cancer, especially in those patients with a history of malignancy. However, it is important to underscore that the published cases were investigated with older-generation ultrasound equipment systems. This may explain why the evaluation of the delayed constrast phase resulted in shorter detection, compared to using newer-generation ultrasound equipment systems.6 –9 The objective of this retrospective review was to explore the enhancement pattern of liver hemangiomas, during the delayed phase of contrast imaging, utlizing a newer-generation ultrasound equipment system. The goal was to investigate a hemangioma’s hemodynamic behavior, beyond the imaging time limit, afforded by older-generation ultrasound equipment systems. 9 The specific extended imaging time limit goal was to exceed 240 seconds, after contrast injection.
Case Series
Methodology
From April 2021 to October 2021, ten patients were recruited to join a retrospective study. The inclusion criteria for the study were that the patient must be older than 18 years and have a known or suspected hepatic hemangioma. The only exclusion criterion was a history of malignant tumor or liver disease.
This study was approved by the ethics committee of the host hospital, and the patients provided written informed consent for enrollment into the study.
An Aplio i800 ultrasound equipment system (Canon Medical Systems, Otawara, Japan), with a 2021 date of manufacture, and the contrast harmonic imaging software were used for all the patient examinations.
The mechanical index for the second harmonic imaging was set at 0.07. All examinations were performed with an i8CX1 convex transducer. The sonographic real-time image was displayed on a split imaging display screen. This allowed for the contrast harmonic imaging to be displayed on the left side of the screen, while allowing visualization of the microvascularization, without the fundamental grayscale echoes, on the right side. The electronic calipers were used to identify the lesions on the grayscale side of the screen, which were automatically displayed at same position on the harmonic imaging side of the screen.
A blood pool second-generation contrast agent (SonoVue; Bracco, Milan, Italy) was used for the CEUS.
The contrast agent was prepared according to the manufacturer's recommendations by adding 4.8 mL of saline solution (0.9% sodium chloride) to the vial, which was then vigorously shaken for at least 20 seconds. Each patient had 2.4 mL of SonoVue administered, as an intravenous bolus injection, through a 20-gauge catheter, into their antecubital vein. This was immediately followed by an injection of 10 mL of saline solution.
All CEUS examinations were performed by a single investigator (G.M.) with more than 20 years of experience performing CEUS of the liver. The investigator was aware of the clinical indications for CEUS examination, the patient’s clinical history, and the results of previous imaging tests.
The timer was started at the end of the contrast injection immediately after the saline flushing was completed. Cinematic loops of every CEUS evaluation were saved. The CEUS was conducted with real-time imaging of the lesion throughout the arterial phase, then by intermittent imaging. For intermittent imaging, we carried out real-time scanning of the lesion for 5–10 seconds at 30-second intervals without obtaining additional images to minimize the rupture of the bubbles of SonoVue. 10
The CEUS evaluation was continued beyond 240 seconds, after the bolus of contrast medium, for all patients. This imaging sequence was continued until a hypoechogenicity of the lesion appeared (See Figures 1 and 2) or the contrast enhancement of liver parenchyma adjacent to the lesion became unevenly distributed or unacceptably rarefied. This provided a clear comparison between the lesion and the parenchyma. No reinjection of microbubbles was performed.
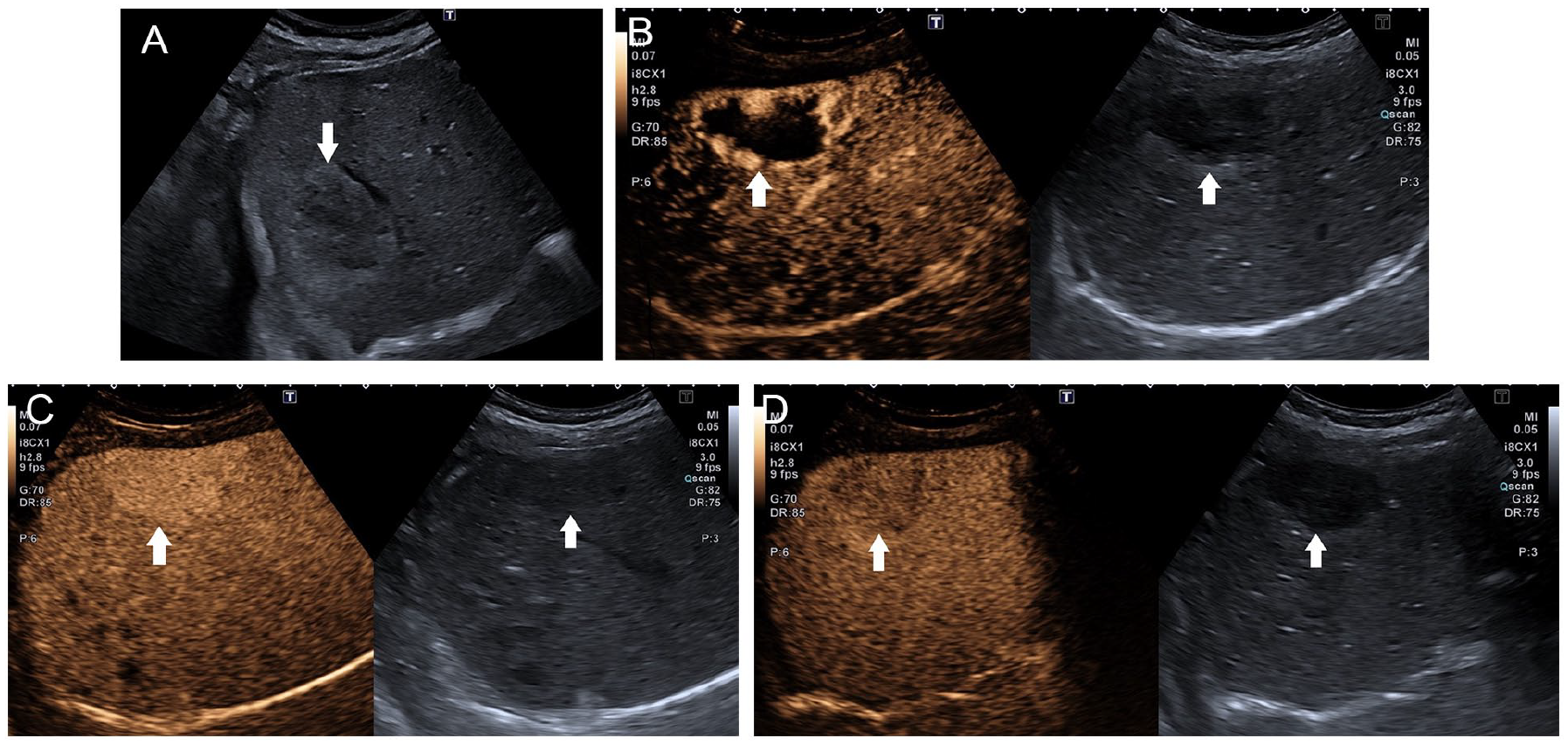
The contrast-enhanced ultrasound (CEUS) examination of a hepatic hemangioma in a patient (See patient #1, in Tables 1 and 2). (A) A grayscale sonogram of an isoechoic lesion with a hyperechoic border of the right hepatic lobe, diameter 60 mm, is provided. The white arrow indicates the edge of the lesion. (B) A CEUS image, during the arterial phase; the white arrows indicate the edge of the lesion. On the left, the peripheral globular enhancement is seen. On the right, the grayscale image is provided for comparison. (C) A CEUS image, during the venous phase (at 120 seconds from contrast injection); the white arrows indicate the edge of the lesion. On the left, a homogeneous hyperenhanced lesion. On the right, the grayscale image is provided for comparison. (D) A CEUS image, during the delayed phase (between 240 and 300 seconds); the white arrows indicate the edge of the lesion. On the left, a hypoechoic lesion, and the right provides a grayscale image for comparison.
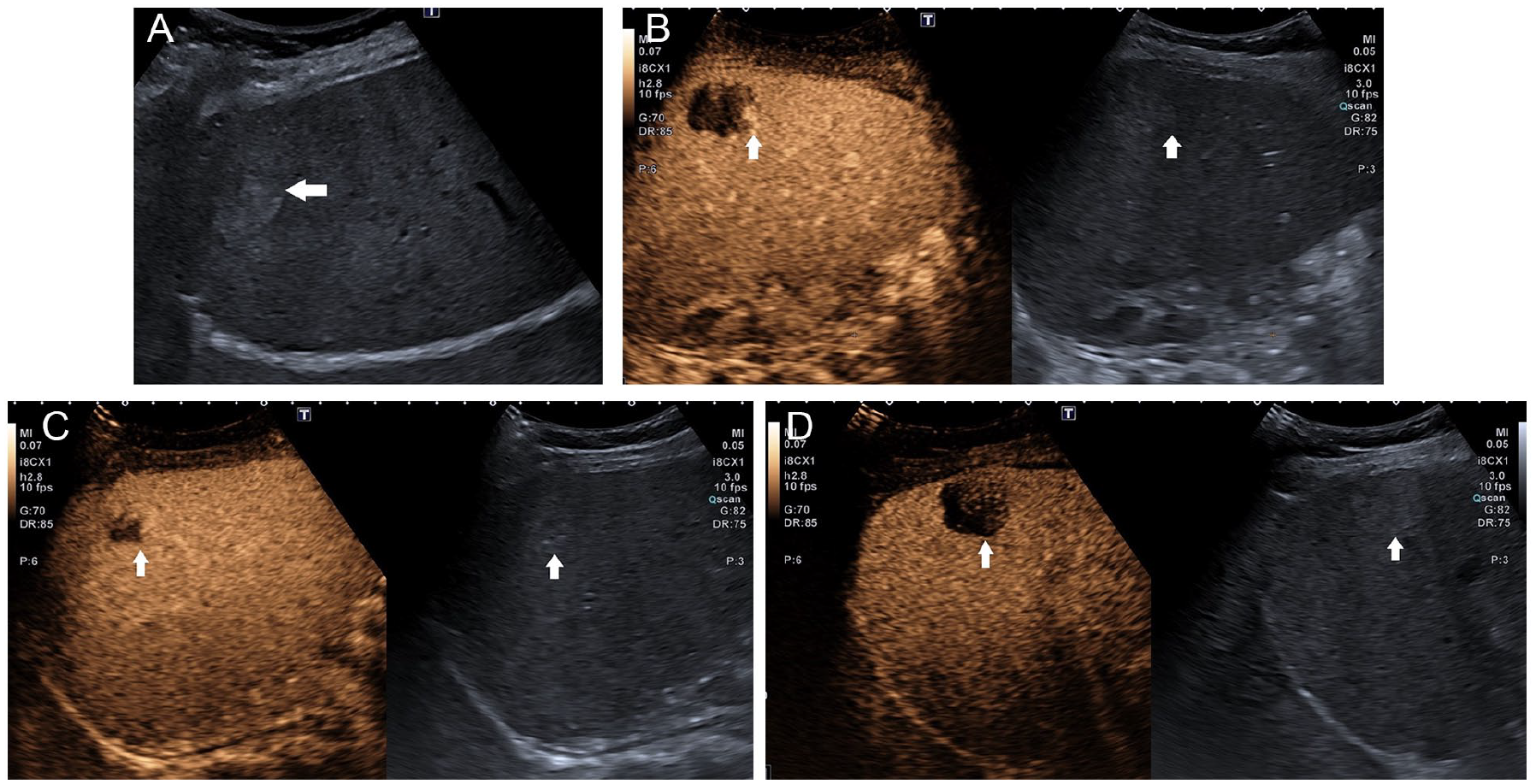
The contrast-enhanced ultrasound (CEUS) examination of a hepatic hemangioma (See patient #3, in Tables 1 and 2). (A) A grayscale sonography image demonstrating a hyperechoic lesion, within the right hepatic lobe, with a measured diameter of 27 mm. The white arrow indicates the edge of the suspected lesion. (B) A CEUS image is provided, during the arterial phase; the white arrows indicate the edge of the hepatic lesion. On the left, the peripheral globular enhancement is demonstrated, and on the right, the grayscale sonogram is provided for comparison. (C) A CEUS image during the venous phase (at 120 seconds after contrast injection); the white arrows indicate the edge of the hepatic lesion. On the left, a isoechoic appearing lesion with an intralesional hypoechoic area. On the right, the grayscale sonogram is provided for comparison. (D) A CEUS image during the delayed phase is provided (between 240 and 300 seconds); the white arrows indicate the edge of the hepatic lesion. On the left, a hypoechoic appearing lesion, and on the right, a grayscale sonogram is shown for comparison.
The Descriptive Data on Each Patient, the Sonographic Appearance of the Liver Nodule, and the Criteria for Certain or Suspected Hepatic Hemangioma, Before the Contrast-Enhanced Ultrasound Examination.

Abbreviations: CT, computed tomogram; F, female; M, male; MRI, magnetic resonance imaging; US, ultrasound; US, grayscale ultrasonography.
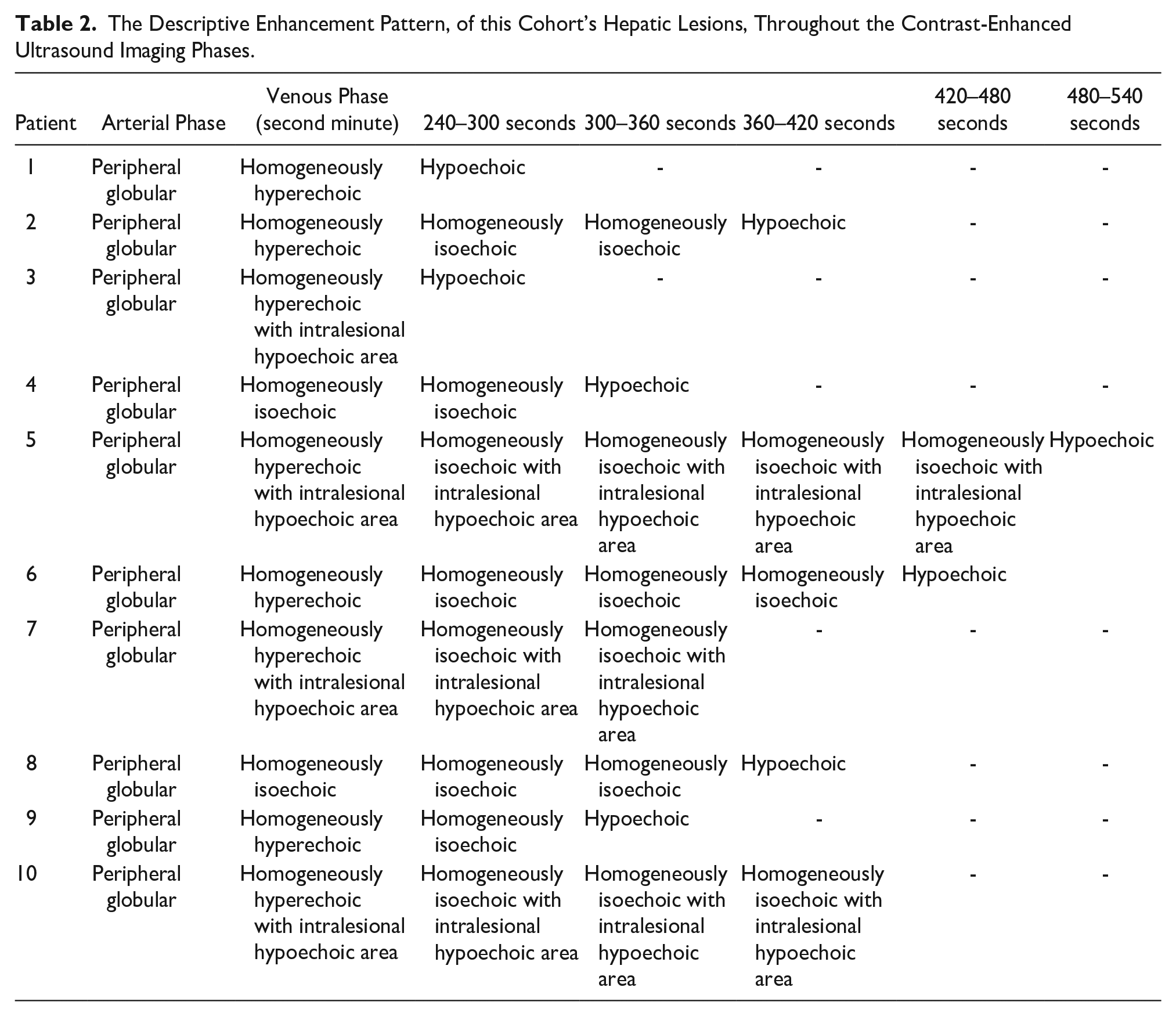
The Descriptive Enhancement Pattern, of this Cohort’s Hepatic Lesions, Throughout the Contrast-Enhanced Ultrasound Imaging Phases.
Results
There were seven female and three male patients recruited, ages 28–70 years (median age 52.2 years) (See Table 1). On previous grayscale imaging, one patient had three hepatic lesions, six patients had two hepatic lesions, and three patients had one hepatic lesion. All the lesions were diagnosed as hemangiomas. For this particular study, only the patient’s largest hepatic lesion was evaluated (See Table 1). The venous contrast transit phase showed an evenly enhanced liver parenchyma in all patients beyond 240 seconds from the contrast injection, and the evaluation of the focal lesions was still achievable. To wit, the CEUS evaluation was performed from 240 to 300 seconds from the intravenous injection in two patients, from 300 to 360 seconds with three patients, from 360 to 420 seconds in three patients, from 420 to 480 seconds in a single patient, and from 480 to 540 seconds in a single patient (See Table 2). The CEUS evaluation had to be concluded in two patients (See Table 2 for patients #7 and #10) because the liver parenchyma, adjacent to the lesions, demonstrated an uneven enhancement and rarefied at 300 and 360 seconds. In these cases, the lesions in both patients demonstrated isoechogenicity, and the lesions showed an intralesional hypoechoic area. In all the other patients, the CEUS evaluation was concluded from 240 to 540 seconds, after contrast injection, when the lesions demonstrated hypoechogenicity, with respect to the adjacent liver parenchyma (See Table 2 and Figure 1). All 10 hemangiomas showed the typical CEUS pattern of peripheral globular arterial enhancement. In this case series, hemangiomas demonstrated an intralesional hypoechoic area during all the dynamic phases (See Table 2 for patients #3, 5, 7, and 10 and Figure 2). In eight patients, the lesion appeared hypoechoic, during the delayed contrast transit phase. Between 240 and 540 seconds, after contrast bolus injection, the lesions appeared hypoechoic in comparison to the adjacent liver parenchyma, which maintained an even distribution of contrast. In particular, the areas of the lesions that demonstrated contrast enhancement in the early phase appeared hypoechoic in the delayed contrast transit phase (See Table 2 and Figures 1 and 2). The hypoechogenicity of the lesions appeared after the intravenous injection of SonoVue in two patients from 240 to 300 seconds, in two patients from 300 to 360 seconds, in two patients from 360 to 420 seconds, in a patient from 420 to 480 seconds, and in a patient from 480 to 540 seconds (See Table 2).
Discussion
One of the cornerstones of hepatic CEUS is that a hypoechoic lesion in the delayed phase should be considered malignant, that is, if the lesion is hyperenhanced in the arterial phase.3,5 In fact, for the typical CEUS enhancement pattern of a liver hemangioma, a completely isoenhanced or hyperenhanced lesion in the venous phase is expected.3,5 Nevertheless, it is known that hemangiomas may also present at CEUS with an intralesional nonenhancing area, provided that it is present in all the phases of contrast transit and not as a result of washout.3,5
With the contrast technology available at the beginning of CEUS era, disappearance of bubbles from the liver parenchyma was expected approximately within 240–360 seconds after the contrast injection.3,5
In the past, some authors described an atypical behavior of liver hemangiomas with CEUS as the venous washout of arterial hyperenhanced peripheral areas within 360 seconds from contrast injection.
Therefore, the number of lesions with such a dynamic behavior was reported as lower than the number of lesions presenting the classical CEUS, and they were defined as “atypical.”6,7 The older-generation ultrasound equipment systems were incapable to obtain an evenly enough enhanced liver parenchyma beyond 240–360 seconds after the intravenous injection of SonoVue to permit a comparison with focal lesions.3,5,9 Instead, using the Aplio i800 ultrasound equipment system (manufactured in 2021), as well as a i8CX1 convex probe and a mechanical index of 0.07, the liver parenchyma demonstrated an even enhancement much longer. Additionally, this study provided an elevated number of lesions with the aforementioned atypical aspect of liver hemangioma with CEUS.
In this cohort, 80% of liver hemangiomas had a diameter ranging from 15 to 60 mm, which resulted in a hypoechoic apperance, with respect to the adjacent liver parenchyma, in the delayed venous phase (as noted beyond 240 seconds after contrast injection). Moreover, in this study cohort, none of the hepatic lesions were attributable to thrombosed or sclerosing hemangiomas as all the lesions had at least a partially homogeneously hyperechoic appearance, in the arterial and early venous phases. Therefore, the hemodynamic behavior beyond 240 seconds found in this study represents a true washout of the lesions as the contrast medium is a pure blood pool agent. Nevertheless, late washout, as shown in this cohort, remains a poorly understood phenomenon. A more thorough evaluation of CEUS of delayed-phase liver hemangiomas, a frequently encountered lesion, should be performed. Indeed, this study may underscore the potential impact of newer-generation ultrasound equipment systems in terms of their diagnostic accuracy in differentiating between benign and malignant lesions in the delayed phase. All the hemangiomas in this cohort presented a typical arterial enhancement pattern, which seems to remain the only reliable criterion for diagnosis.
Limitations
This cohort study has several limitations. The primary limtiation is the study design, a retrospective case series; therefore, these results can not be generalized beyond the study cohort. Additionally, this cohort had a small number of lesions evaluated, and this was completed by only one investigator. Furthermore, all the liver lesions had a diameter greater than 15 mm; even smaller lesions should be investigated. An additional concern was the diagnosis of a hemangioma was not established on histology. In fact, we considered all the study lesions were assumed as angiomas, even in the absence of a histological or pathological examination, as all the patients had no history of malignant neoplasm or liver disease. For those who had previously only undergone grayscale sonography, this diagnostic step helped to confirm the stability of the lesions, which was deemed as enough time to diagnose the lesion as benign (the shortest observation time was 2 years). For those in whom a focal liver lesion was detected for the first time, behavior on CEUS was that of a typical angioma up to 300 seconds from the intravenous injection of the contrast media. Finally, the other patients had previously undergone a computed tomogram or a magnetic resonance imaging examination, with contrast media, which was diagnostic for angioma.
Conclusion
All hemangiomas in this patient case series presented with a peripheral pattern of arterial enhancement. Future clinical research should also investigate hemangiomas with a homogeneous pattern of arterial hyperenhancement. An additional concern with this study was the use of a single newer-generation ultrasound equipment system. The replication of these results with CEUS should be more extensively evaluated by using a variety of newer-generation ultrasound equipment systems to confirm these findings.
Footnotes
Acknowledgements
The authors thank Livia Davolio, Nicolina Solita, and Silvia Angerame, the nurses that work with them at their ultrasound unit; without their precious daily practice, this study could not have been done. They also thank Jacqueline M. Costa for the English language editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval was not sought for the present study because This study was approved by the ethics committee of the host hospital.
Informed Consent
Informed consent was not sought for the present study because all case data was de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
