Abstract
Contrast-enhanced ultrasound (CEUS) is a relatively new addition to the diagnostic armamentarium in the United States. The pairing of intravenously injected contrast agents with advanced ultrasound image processing techniques permits improved detection and characterization of different tissue types compared with conventional sonography. The use and efficacy of CEUS is well established in echocardiographic applications in the United States and in many other applications worldwide. Recent approval by the U.S. Food and Drug Administration of an ultrasound contrast agent for use in evaluating adult and pediatric hepatic pathology opens the door to a new era in sonographic diagnosis. An understanding of the underlying physical and technical principles of CEUS is essential for the accurate interpretation of the information obtained from this exciting emerging modality.
Keywords
Background
Contrast imaging, in general, involves the use of an agent to enhance the contrast of structures or fluids in the human body. A variety of contrast agents have been employed in medical imaging over many decades, beginning with the use of air and barium in the early days and evolving into the complex pharmacologic agents currently in use in computed tomography (CT), magnetic resonance, positron emission tomography, and nuclear medicine imaging modalities. Through various physical interactions between the agent and the energy used by the modality creating the image, dissimilarities in tissue types and boundaries are accentuated, which permits better differentiation between them.
Ultrasound Contrast Agents
Ultrasound contrast agents (UCAs) consist of gaseous microbubbles suspended in an aqueous solution that is injected into the human venous vascular system. They are composed of an inert inner gas bubble surrounded by a stabilizing outer shell (Figure 1). Different UCA manufacturers use various gas/shell combinations. As the microbubbles are “blood pool” agents, they remain intravascular at all times and do not permeate into adjacent tissues. As such, they act effectively as red blood cell tracers.
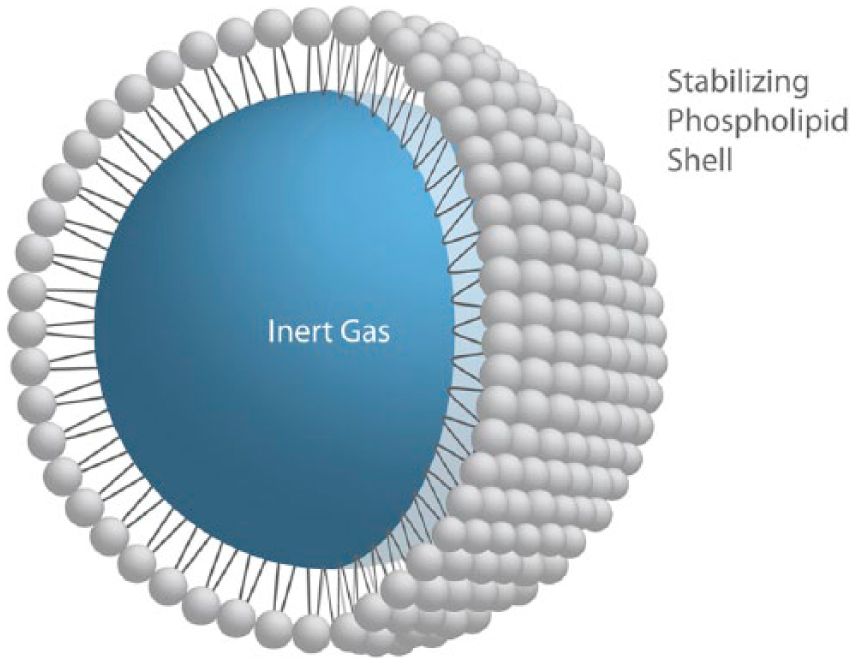
Schematic illustration of a phospholipid-encased ultrasound contrast microbubble.
The great acoustic mismatch between gas and surrounding blood results in dramatic and predictable physical responses when the microbubbles are exposed to an acoustic energy field. Reflection, backscatter and nonlinear harmonic responses create a returning acoustic data set that can be processed in ways that permit the differentiation between bubble-bearing blood and adjacent soft tissue structures. Using the contrast detection methods described below, anatomic B-mode information can be suppressed while the UCA-generated subset of the returning acoustic data set is enhanced, permitting the creation of high-resolution, real-time images of microbubbles moving through the vasculature in the region of interest (ROI).
A significant advantage of UCAs over other types of contrast agents, particularly those used in CT and magnetic resonance imaging, is that they are rarely, if ever, associated with either immediate or delayed clinical complications. 1 Iodine-based intravenous contrast agents, frequently used in CT scanning, have an infrequent but well documented association with potentially serious postinjection sequelae. 2 Immediate anaphylactic reactions to the intravenous injection of iodine-based CT contrast agents range from mild allergic response such as itching, hives, and vomiting to potentially life-threatening anaphylactic shock. Anaphylactic shock, which is typically characterized by severe cutaneous, respiratory, cardiovascular, and gastrointestinal symptoms, may occur within minutes of administration of as little as 1 cc of contrast agent.3,4 Delayed reactions to intravenous iodine administration are related to their nephrotoxic effects, as they are primarily excreted by the kidneys and may include acute renal injury and contrast-induced nephropathy. 5 Again, although uncommon, the risk of both immediate and delayed serious clinical sequelae does exist.
Gadolinium-based contrast agents used in magnetic resonance imaging are also infrequently but definitely associated with both proximate nephrotoxic and delayed neurotoxic effects. Gadolinium in its isolated state is a highly toxic heavy metal. Although this toxicity is virtually obviated by binding it to other molecules for use in human imaging studies, it has nevertheless been shown to be associated with nephrogenic systemic fibrosis and gadolinium body storage. The former, which has been essentially eliminated by adherence to radiologic usage guidelines, is of particular concern in patients with existing chronic renal disease as the contrast agent is excreted by the kidneys. The latter has been observed in both brain and bone, many years after initial administration. 6 The long-term clinical ramifications of gadolinium body storage, if any, have yet to be determined. 7
Given that both the gaseous core of the UCA microbubble and its encasing shell are composed of inert, nonallergenic materials, there is virtually no risk of anaphylactoid reactions immediately after injection. The microbubbles eventually degrade as they pass through capillary beds. The inert gas becomes suspended in plasma and is ultimately cleared from the bloodstream via the lungs. The degraded encasing shell material is engulfed by macrophages in the reticuloendothelial system in the liver and spleen. As a result, UCAs are not nephrotoxic and have not shown any delayed or long-term adverse effects associated with their use.8,9
Vascular Phases
Ultrasound contrast agent microbubbles remain in the blood pool for a limited period of time. They are transported from the venous injection site to the heart and ultimately to the systemic circulation. Enhancement patterns reflect various vascular phases from early arterial “wash-in” to late venous “wash-out.” In general, as UCA-bearing blood flows into a tissue bed during the early arterial phase, the microbubble-generated acoustic response initially causes the feeding arteries to “light up.” As blood passes from feeding vessels and into the arterioles and capillary bed, the degree of diffuse tissue enhancement increases until it reaches a maximum level. In the case of the liver, enhancement continues during the portal venous phase as well (Figure 2). Subsequently, non-bubble-bearing blood flows into the tissue and enhancement begins to diminish (wash-out) until the sonographic appearance returns to its normal nonenhanced level in the late venous phase. As enhancement variations reflect perfusion patterns into and out of a tissue bed, contrast-enhanced ultrasound (CEUS) is a valuable tool in differentiating normal tissue from abnormal adjacent tissue and is ideally suited for characterization of focal lesions in the liver and other solid parenchymal organs.
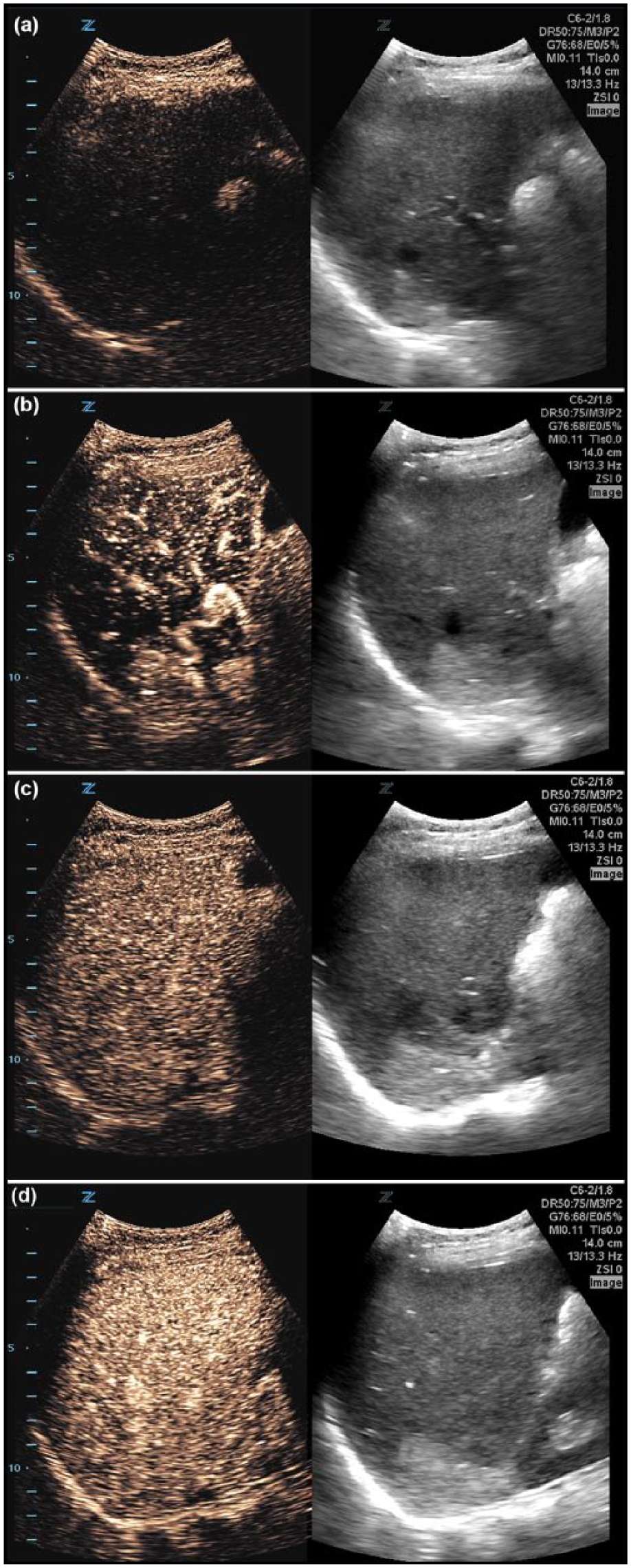
Contrast-enhanced appearance over vascular phases in a normal liver. (a) Preinjection. (b) Early arterial phase with enhancement of feeding arterial vasculature. (c) Arteriolar/capillary phase. (d) Portal venous phase.
Bubble Behavior
The unique interaction between an incident acoustic energy field and UCA microbubbles is the basic physical foundation of CEUS. First, as there is a significant impedance mismatch between the gas-filled bubbles and the encompassing blood plasma, linear reflection occurs much as it does in static soft tissue—the underpinning phenomenon of B-mode imaging. Second, as the bubble size is smaller than the wavelength of the acoustic beam, typically 3 to 4 μm, oscillations are produced as the bubble is compressed and rarefied by the passing acoustic wavefront. The subwavelength size of the bubbles results in nonlinear backscatter; the disparate compression/expansion response produces harmonic frequencies at twice the incident fundamental frequency. All three of these types of acoustic data are used in creating CEUS images.10,11
An important consideration in understanding bubble behavior in CEUS is that these phenomena occur in different proportions at different incident acoustic pressures. At lower pressures, linear reflection and backscatter (B-mode type) is the dominant component of the returning acoustic data set. At increased pressures, nonlinear backscatter and harmonic responses dominate; at even higher pressures, bubble destruction occurs. The practical tradeoff is to use transmit energies, as expressed by the mechanical index (MI), that will penetrate to the ROI without destroying the bubble density required to obtain adequate diagnostic information. For this reason, contemporary CEUS-enabled preset parameters use
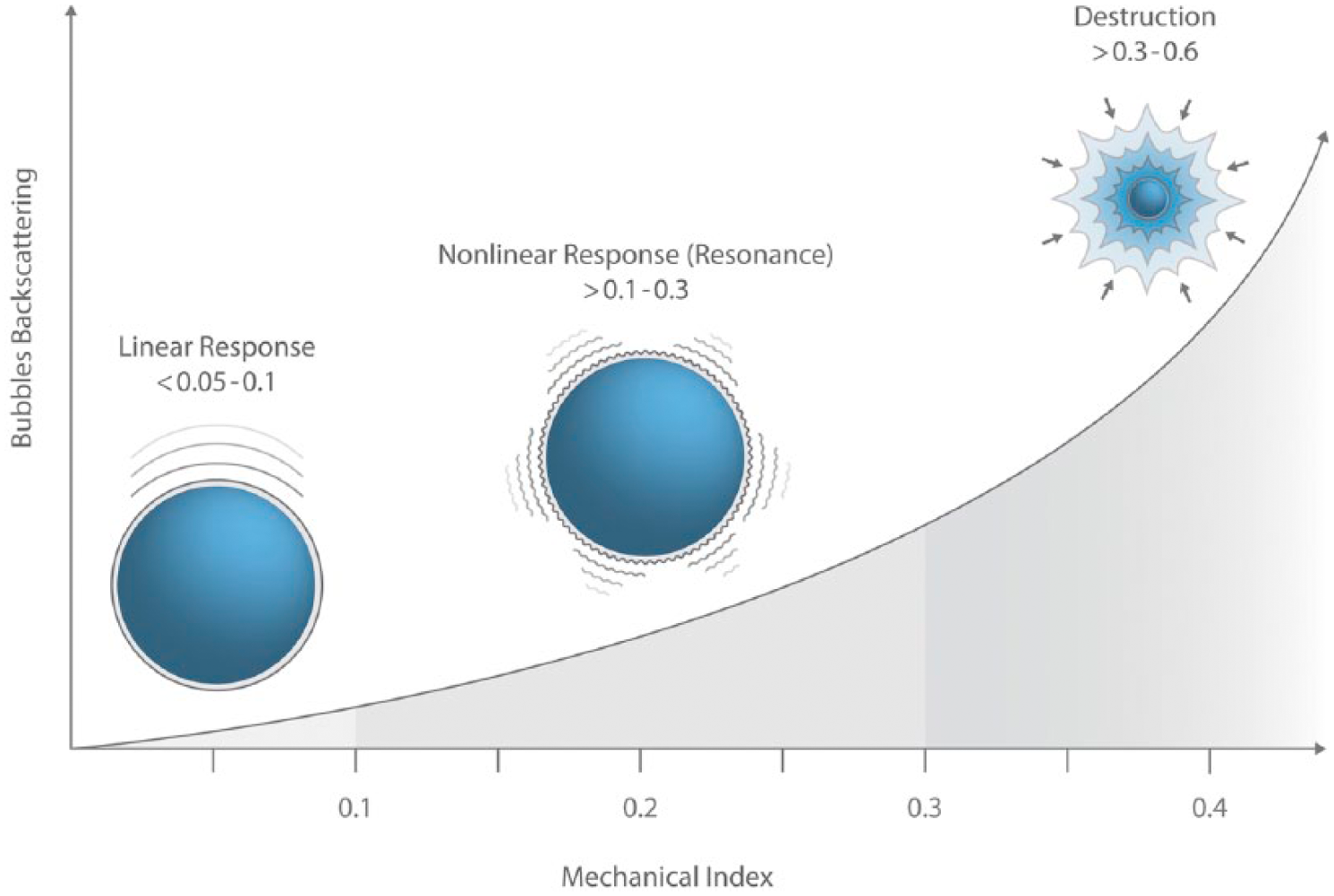
Schematic representation of microbubble response to increasing levels of acoustic power (mechanical index).
To help understand the nebulous concept of low MI settings used in CEUS imaging, it is helpful to put it in context with the normal MI levels used in general sonography studies. Mechanical index is a U.S. Food and Drug Administration (FDA)-mandated on-screen indicator of the relative potential for the induction of nonthermal bioeffects by acoustic energy, that is, cavitation and acoustic streaming. It is not an indicator that a bioeffect will actually occur; rather, it provides a single piece of information on the relative probability of a biological event ensuing. It is mandated as an output display data point, along with a tissue index (TI), so that the sonographer can set system controls
Bubble destruction can, however, be used to clinical advantage. Some manufacturers provide the operator with the ability to transmit a single frame at a much higher MI to intentionally clear out the microbubbles in the ROI. This is useful when there is residual contrast in the image and an additional injection of UCA is required. Initiating a
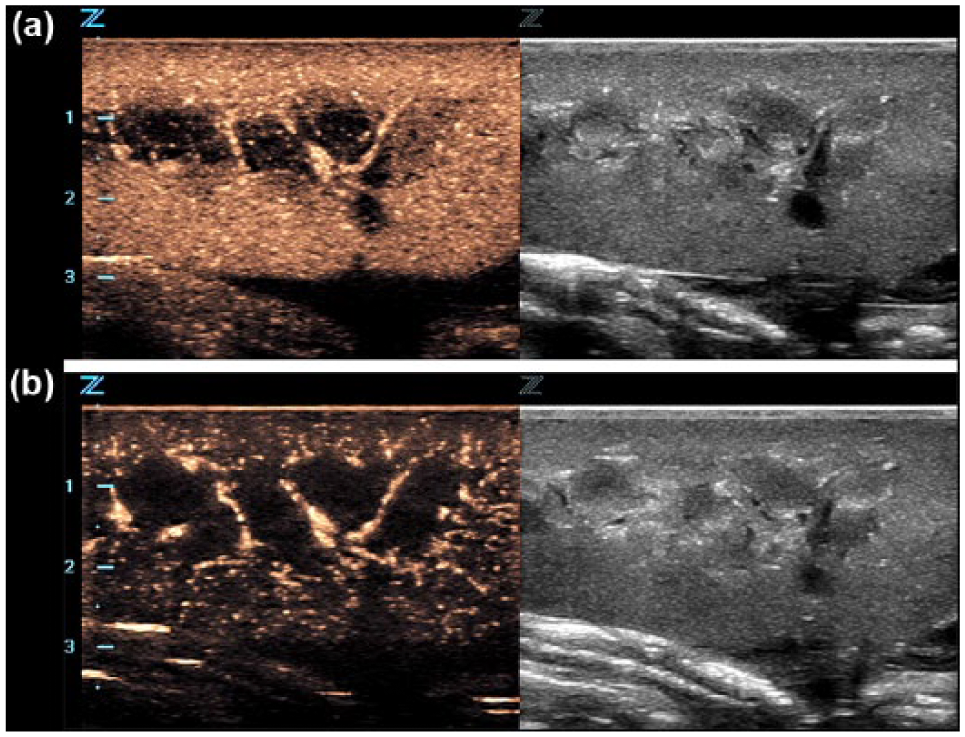
Use of a higher mechanical index flash frame to cancel contrast information in a contrast-enhanced ultrasound image. (a) Preflash frame. (b) Postflash frame.
Another bioeffect consideration when performing CEUS is the increased probability of nonthermal events occurring because of the presence of microbubbles in the acoustic field. Although no clinical sequelae related to these events have been reported in the medical literature, that they can and do occur has been clearly demonstrated. In fact, emerging therapeutic methods have been developed to take advantage of the predictable effects that bubble oscillations, cavitation, and microstreaming have on cell walls. These physical phenomena alter cell wall permeability and permit targeted delivery of pharmacologic and genetic agents attached to the microbubbles directly into cells through the altered membrane (sonoporation). In these methods, CEUS plays both a diagnostic and therapeutic role in patient management.13–15 A full discussion of these exciting new ultrasound applications is beyond the scope of this article.
Ultrasound Contrast Detection Methods
There are several engineering methods for ciphering out the received linear echo data that are the foundation of B-mode anatomic imaging from the nonlinear backscatter and harmonic data that are used for the creation of a contrast image.
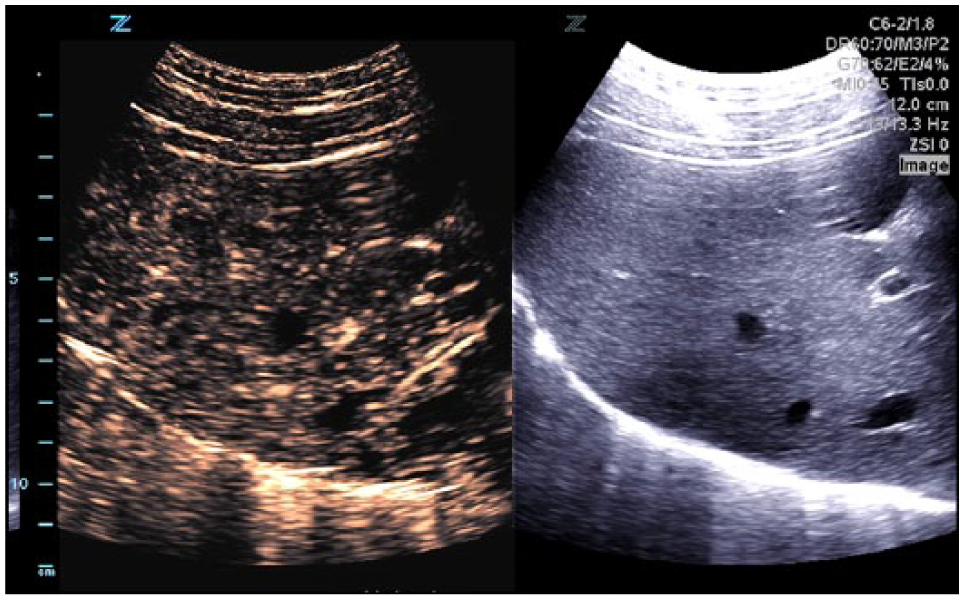
Typical contrast-enhanced ultrasound display. Tissue suppressed contrast image (left) and reference B-mode image (right).
Pulse inversion, the most common tissue suppression technique, involves the transmission of two acoustic waves into tissue. The first pulse is transmitted as usual. A second pulse is transmitted after a short delay and is an inverted replica of the first. In a linear medium, the response of the second wave, then, is also an inverted response of the first wave. This permits the ability to cancel out all linear responses from the tissue, leaving only the nonlinear backscatter and harmonics that are present in the received acoustic data sets. These residual data are used to create the contrast image 16 (Figure 6).
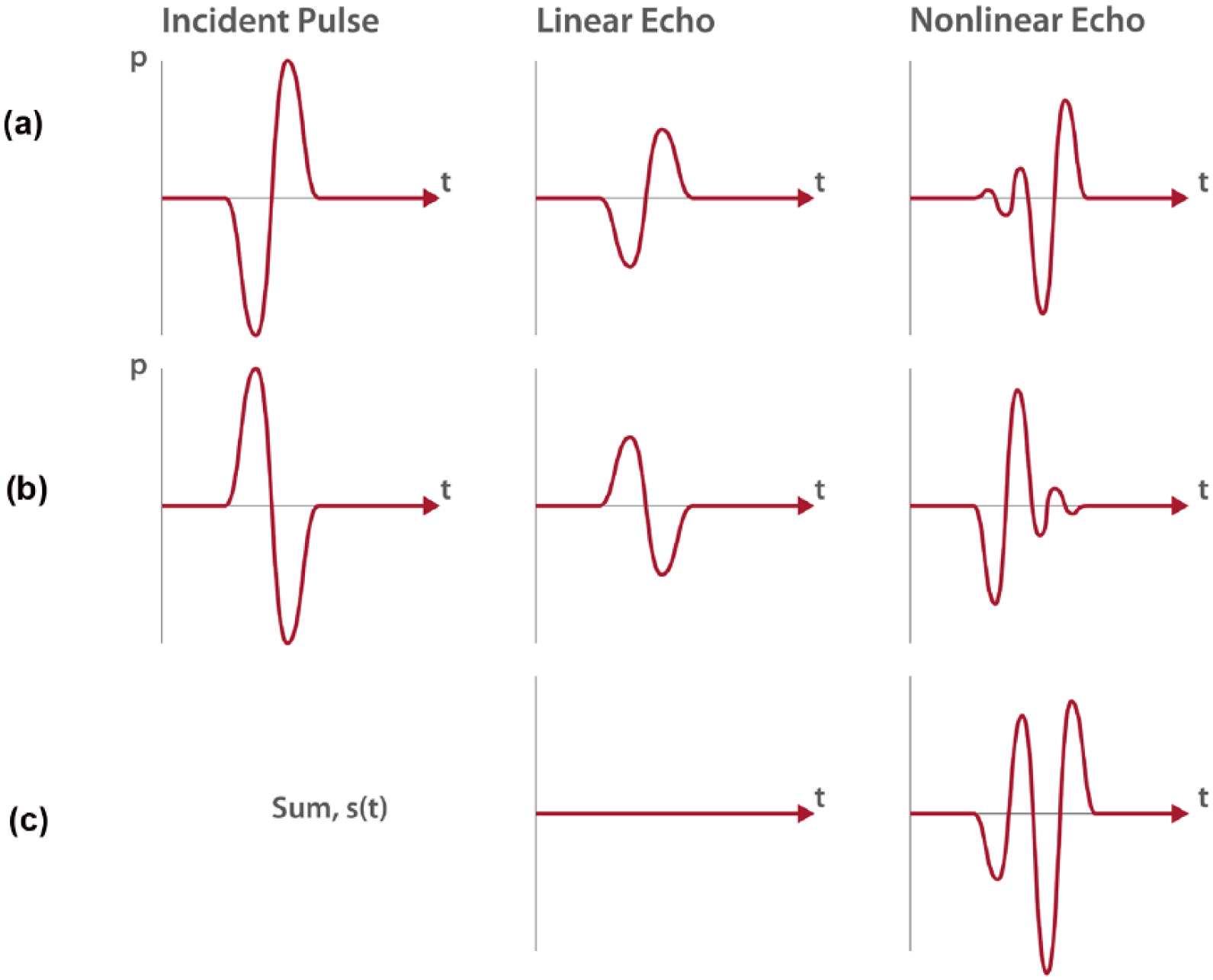
Schematic illustration of pulse inversion technique of displaying contrast-enhanced ultrasound (CEUS) information. (a) First pulse transmitted as usual. (b) Second pulse, an inverted replica of the first pulse, is transmitted after a short delay. (c) Linear responses from tissue are cancelled out, leaving only nonlinear backscatter and harmonics used to create the CEUS image.
CEUS Performance Parameters
Performance evaluation for CEUS imaging includes parameters related to the equipment as well as bubble-specific criteria. Imaging equipment criteria are similar to those associated with B-mode and color Doppler imaging; bubble-specific criteria are related to the biochemistry of the UCA and the injection methods employed.
Bubble Sensitivity
Sensitivity has several meanings in evaluating the performance of a CEUS-enabled ultrasound imaging system. All of them relate to the ability of the system to detect, differentiate, and display bubble-generated components of the returning acoustic data set versus stationary, tissue-generated data.
The first specific definition of CEUS sensitivity refers to the ability of a system to detect a small quantity of bubbles and, in most instances, this is dose dependent. More sensitive devices require a lower dose of UCA for satisfactory imaging, which translates into fewer injections required per diagnosis and more economic use of each vial of agent. Second, sensitivity is the ability to detect individual bubbles and is intimately tied in with spatial resolution. Finally, sensitivity is the ability to detect small differences in local contrast agent concentration, which reflects the dynamic range of contrast performance. This parameter is similar to contrast resolution in B-mode imaging.
Bubble Specificity
Bubble specificity is the ability to differentiate bubbles from adjacent soft tissue on the CEUS image itself.
Whereas bubble sensitivity is the ability of the imaging system to detect bubble-generated image information, specificity refers to the ability to differentiate between bubbles and soft tissue on the visual display itself. It also refers to the ability to differentiate between areas of UCA concentration within a particular ROI or identified lesion. Adequate bubble specificity is essential in differentiating hyper- or hypoenhancing lesions within a solid parenchymal organ and in commenting on UCA concentration in discrete areas within the ROI over the various vascular phases.
Tissue Suppression
As discussed above, tissue suppression is the ability to eliminate B-mode anatomic information from the contrast image. This is a critical CEUS performance parameter as it permits the differentiation of normal from pathologic tissues. Strong reflectors such as the diaphragm, large vessel walls, and gas-filled structures can break through and appear in the contrast images. Separate gain controls for both reference and contrast images can provide the operator with the ability to correct for much of this breakthrough and improve the quality and accuracy of the contrast image. 17
Spatial Resolution
As in traditional B-mode ultrasound imaging, spatial resolution refers to the ability of an imaging system to distinguish between point sources of acoustic reflection or backscatter. In CEUS, it also refers to the ability to display bubble information with optimal detail as the microbubble UCA passes from feeding vessels into the arterioles and capillary bed.
Temporal Resolution
Temporal resolution is the ability of an ultrasound system to differentiate between the presence of bubbles within a given sample of tissue at different points in time. It is determined by the frame rate and line density of each frame. There is a trade-off in CEUS imaging between faster frame rates and bubble longevity (described below). High frame rates require more beam transmit sequences, which introduce more acoustic energy, which increases the probability of bubble destruction. The goal in designing CEUS-enabled platforms is to minimize bubble destruction while maintaining frame rates that permit accurate assessment of enhancement patterns over the various vascular phases. In CEUS, good temporal resolution is imperative as it permits an accurate display of wash-in and wash-out times as well as the ability to differentiate between trapped and mobile bubbles over the various vascular phases.
Image Uniformity
Image uniformity refers to the ability to maintain consistent spatial and contrast resolution throughout the contrast-enhanced ROI. Similar to B-mode imaging, it is a function of transducer frequency, which determines penetration depth, and an ultrasound system’s ability to maintain lateral resolution at all depths.
Bubble Longevity
It is important that UCA microbubbles remain stable for the duration of the diagnostic examination. Longevity is a function of both bubble chemistry and the equipment used. Early generation free gas bubbles were large and easily collapsed by the acoustic energy field. Subsequent iterations of microbubble UCAs saw a uniform reduction in size and better longevity due to the improved chemistry of the shell.18,19 Bubble longevity is also a function of the acoustic power used as reflected by the MI.
Low MI scanning techniques have been almost universally adopted by ultrasound equipment manufacturers and clinical users, ensuring the stability of the agent through all vascular phases of the examination. The mantra of bubble longevity in CEUS imaging is “to see without destroying.”
Conclusion
Contrast-enhanced ultrasound offers an additional level of diagnostic information to standard sonographic examinations. Although this modality has been in use for decades outside the United States in a wide array of clinical applications, recent FDA approval for use of an ultrasound contrast agent in evaluating both adult and pediatric liver pathology presents the opportunity for a new era in sonographic diagnosis. As CEUS enters the mainstream sonography armamentarium, an understanding of the physical principles underpinning this technique and a command of equipment performance and control parameters become necessary so that optimum quality diagnostic information can be obtained. Ultrasound contrast agent composition and UCA microbubble behavior exhibited when exposed to an acoustic energy field are also important considerations when performing CEUS studies, minimizing potential adverse biological phenomena while still ensuring the production of a quality diagnostic examination.
Footnotes
Acknowledgements
The author would like to thank Dr. Glen McLaughlin and Mr. Rob Steins for review of the technical content of this article and Mr. Kendall Dea for the original technical illustrations presented herein.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
