Abstract
Cor triatriatum is a congenital heart defect often diagnosed in infancy or childhood. This case study presents an adult with metastatic breast cancer, anemia, and an incidental finding of cor triatriatum sinistrum. On the basis of clinical presentation, the patient was treated conservatively and discharged. Sonographic findings of cor triatriatum sinistrum, along with clinical signs, the significance of other imaging modalities, and potential treatments, are presented.
Cor triatriatum is defined as the presence of a fibromuscular membrane within the left or right atrium, giving the appearance of three atria. Cor triatriatum is a rare congenital heart defect often diagnosed in children, which presents with other cardiac anomalies in 80% of cases. 1 The cause of cor triatriatum is not well understood, and several theories exist on the embryologic origin. The treatment for cor triatriatum may be aggressive or conservative and differs according to the severity of symptoms. Overall, in cases of isolated cor triatriatum, the prognosis is excellent. The following case demonstrates the importance of considering a patient’s clinical history when determining the best course of treatment. In addition, a mostly asymptomatic adult with an incidental finding of cor triatriatum sinistrum illustrates a very unusual and interesting case.
Case Presentation
A primary care physician directly admitted a 64-year-old woman with anemia to the hospital. Her medical history was significant for metastatic breast cancer with lesions to the bone and liver following chemotherapy, hydradenitis (chronic inflammation of the apocrine sweat glands), and recurrent anemia with multiple blood transfusions. The last transfusion was one month prior to her hospital admission. She presented with progressive weakness, fatigue, difficulty with ambulation, and shortness of breath upon exertion. The patient denied chest pain.
Upon admission, the patient had a low hemoglobin level and was scheduled to receive another blood transfusion. Physical examination showed bilateral leg swelling and a distended abdomen. Blood pressure was 115/54 mm Hg with a pulse rate of 94 beats per minute. No murmur was detected with auscultation. Laboratory tests showed a negative troponin, elevated brain natriuretic peptide, somewhat elevated prothrombin time, and elevated liver function tests. At that time, vitamin K and Lasix were administered.
Two days later, the patient was marked for a paracentesis with a Zonare Z.One Ultra SP ultrasound system using a 4-MHz probe (Zonare Medical Systems, Mountain View, California). After 1.5 liters of fluid were removed and the blood transfusion was completed, the patient felt significantly better. However, the patient still expressed concerns over her shortness of breath, and the consulting cardiologist ordered an echocardiogram.
A transthoracic echocardiogram was performed with a Philips iE33 ultrasound system using a 5-MHz probe and included a complete 2D, M-mode, and Doppler examination (Philips Ultrasound, Bothell, Washington). Findings from the echocardiogram included normal right and left ventricular size and function, mild enlargement of the right atrium, severe enlargement of the left atrium with an echodense linear membrane noted, normal interatrial septum, normal appearance of the mitral valve with no stenosis or prolapse, mild mitral regurgitation, normal trileaflet appearance and function of the aortic valve, normal appearance and function of the tricuspid valve with mild regurgitation, mild pulmonary hypertension (right ventricular systolic pressure of 31 mm Hg), normal pericardium with no significant pericardial effusion, and normal aortic root size. The pulmonic valve and inferior vena cava were not well visualized. The 2D/M-mode and Doppler measurements are summarized in Tables 1 and 2. Further investigation of the membrane within the left atrium showed there was no obstruction of flow from the pulmonic veins to the mitral valve; therefore, no abnormal pressure gradient was appreciated across the membrane using continuous wave Doppler. The membrane measured approximately 44.1 mm, extending from the interatrial septum to the lateral wall of the left atrium. No fenestration of the membrane was observed throughout the examination (Figures 1–5). The cardiologist was notified of the findings following the echocardiogram, and a diagnosis of cor triatriatum sinistrum was made. A computed tomographic (CT) scan was performed to assess the extent of metastases. The CT demonstrated a membrane within the left atrium as well (Figure 6). Given the patient’s history and medical status at that time, no further imaging or procedures were performed. The patient was subsequently deemed medically stable, discharged, and instructed to follow up with her primary care physician for her anemia and ascites.
2D/M-Mode Measurements From the Patient’s Transthoracic Echocardiogram.
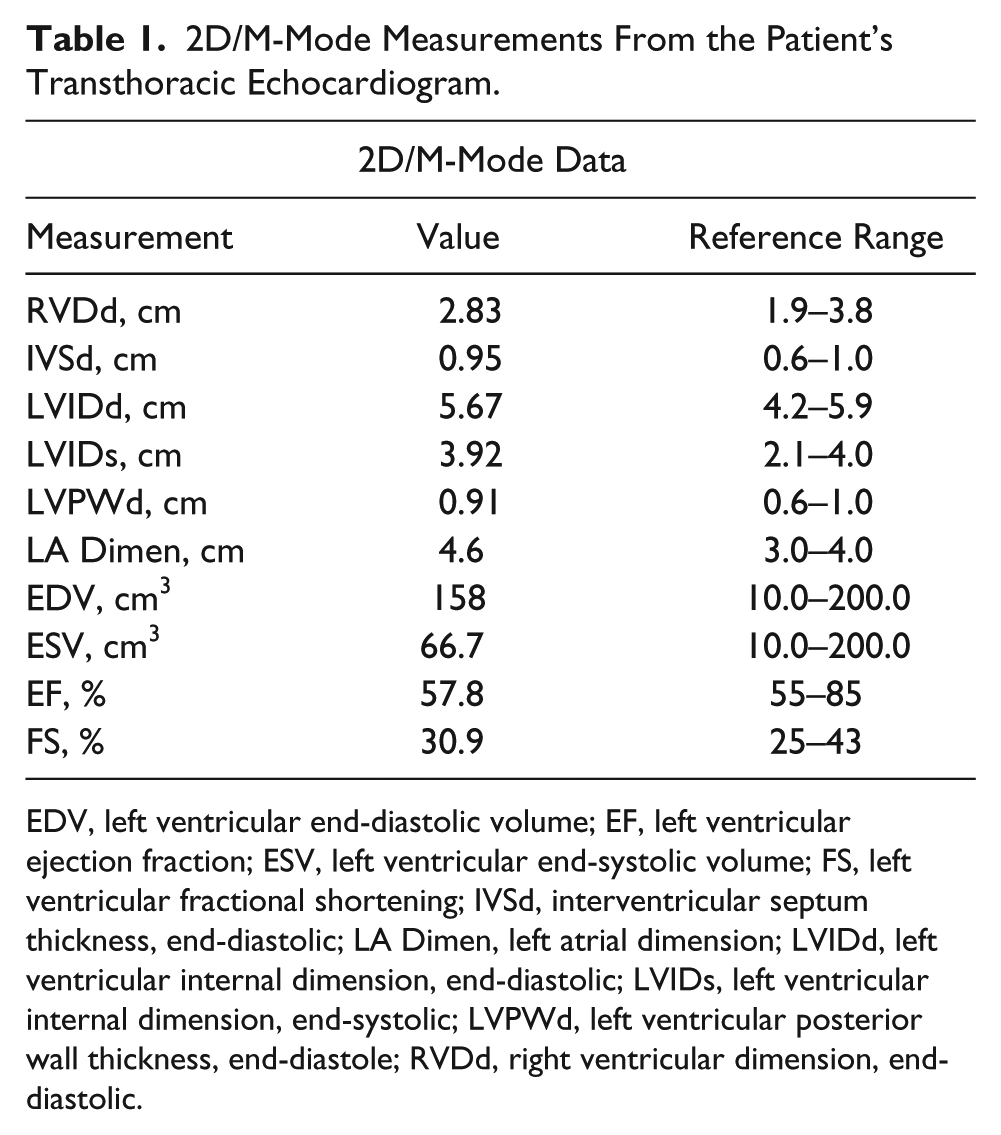
EDV, left ventricular end-diastolic volume; EF, left ventricular ejection fraction; ESV, left ventricular end-systolic volume; FS, left ventricular fractional shortening; IVSd, interventricular septum thickness, end-diastolic; LA Dimen, left atrial dimension; LVIDd, left ventricular internal dimension, end-diastolic; LVIDs, left ventricular internal dimension, end-systolic; LVPWd, left ventricular posterior wall thickness, end-diastole; RVDd, right ventricular dimension, end-diastolic.
Doppler Measurements from the Patient’s Transthoracic Echocardiogram.
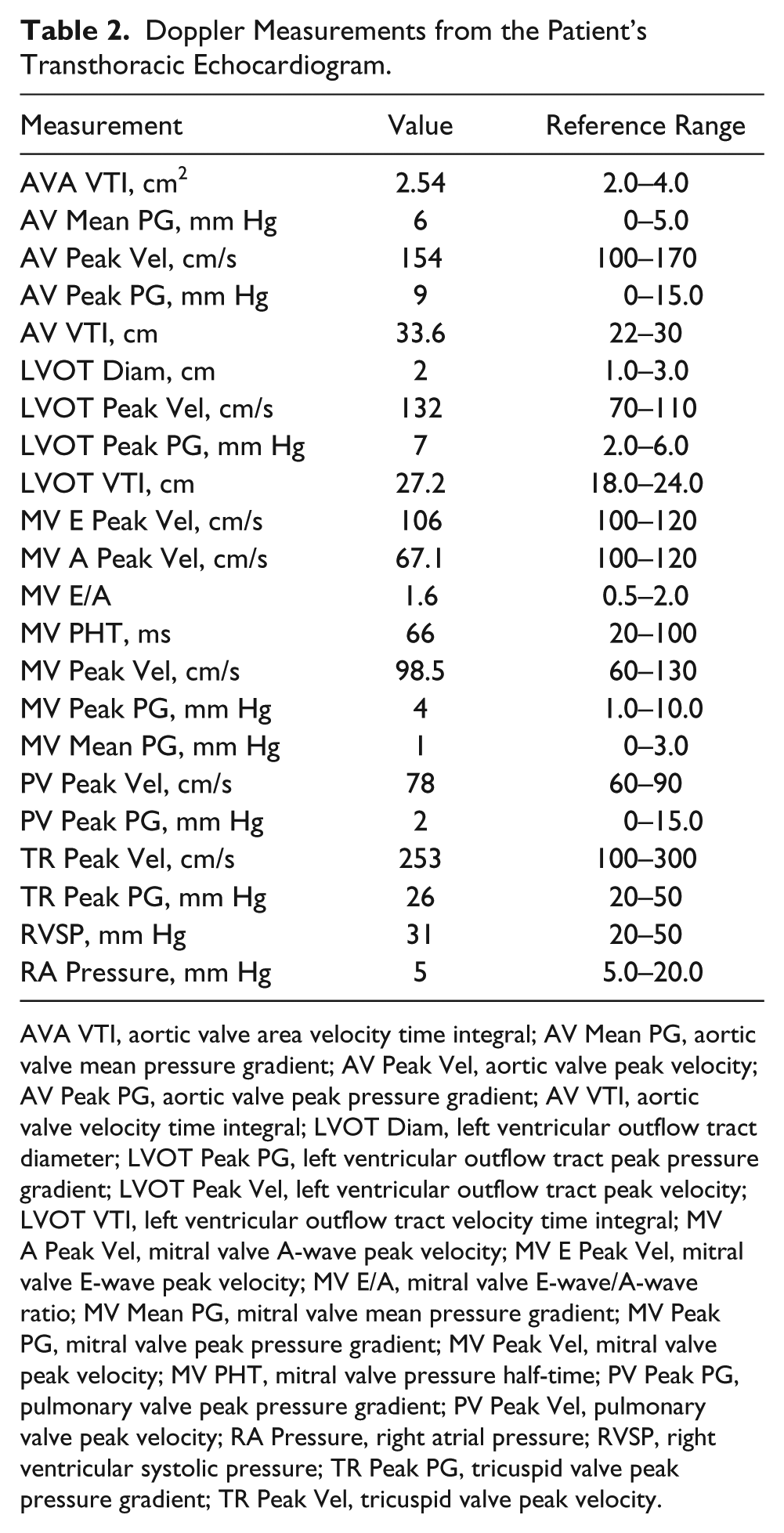
AVA VTI, aortic valve area velocity time integral; AV Mean PG, aortic valve mean pressure gradient; AV Peak Vel, aortic valve peak velocity; AV Peak PG, aortic valve peak pressure gradient; AV VTI, aortic valve velocity time integral; LVOT Diam, left ventricular outflow tract diameter; LVOT Peak PG, left ventricular outflow tract peak pressure gradient; LVOT Peak Vel, left ventricular outflow tract peak velocity; LVOT VTI, left ventricular outflow tract velocity time integral; MV A Peak Vel, mitral valve A-wave peak velocity; MV E Peak Vel, mitral valve E-wave peak velocity; MV E/A, mitral valve E-wave/A-wave ratio; MV Mean PG, mitral valve mean pressure gradient; MV Peak PG, mitral valve peak pressure gradient; MV Peak Vel, mitral valve peak velocity; MV PHT, mitral valve pressure half-time; PV Peak PG, pulmonary valve peak pressure gradient; PV Peak Vel, pulmonary valve peak velocity; RA Pressure, right atrial pressure; RVSP, right ventricular systolic pressure; TR Peak PG, tricuspid valve peak pressure gradient; TR Peak Vel, tricuspid valve peak velocity.
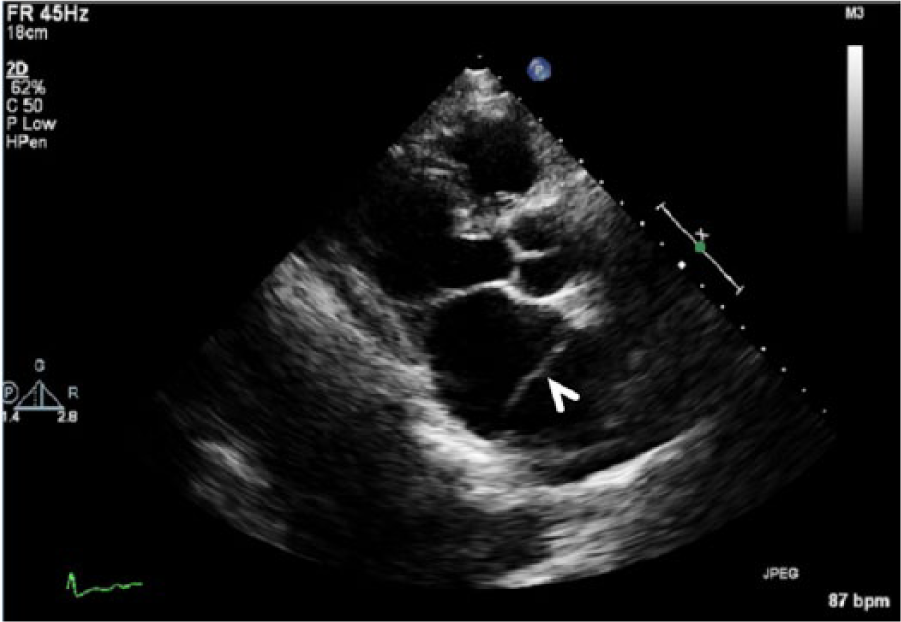
Parasternal long-axis image of the enlarged left atrium, the left and right ventricles, and the left outflow tract. The fibromuscular membrane, indicative of cor triatriatum sinistrum, is seen within the left atrium (arrowhead).
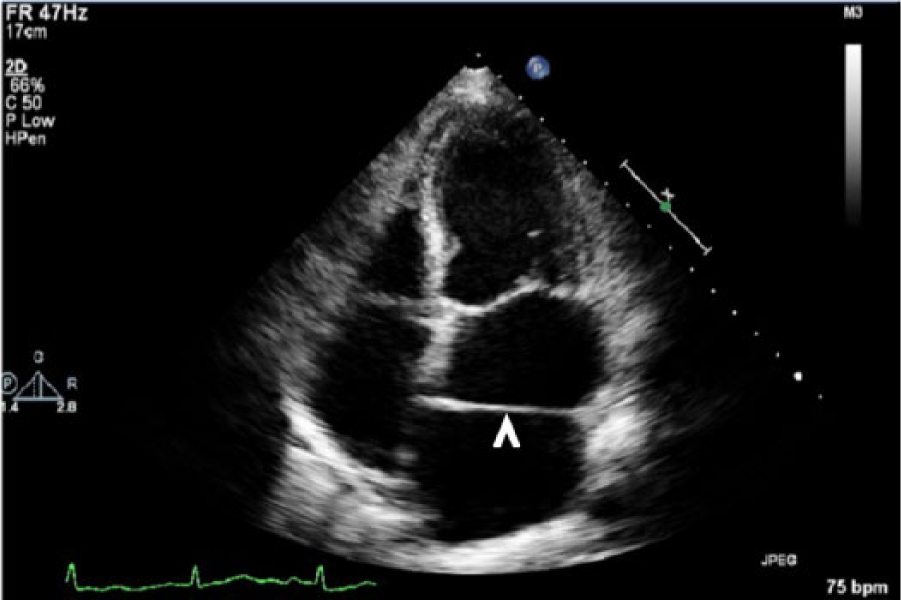
Apical four-chamber view of the heart showing the echogenic fibromuscular membrane characteristic of cor triatriatum sinistrum in the left atrium (arrowhead).
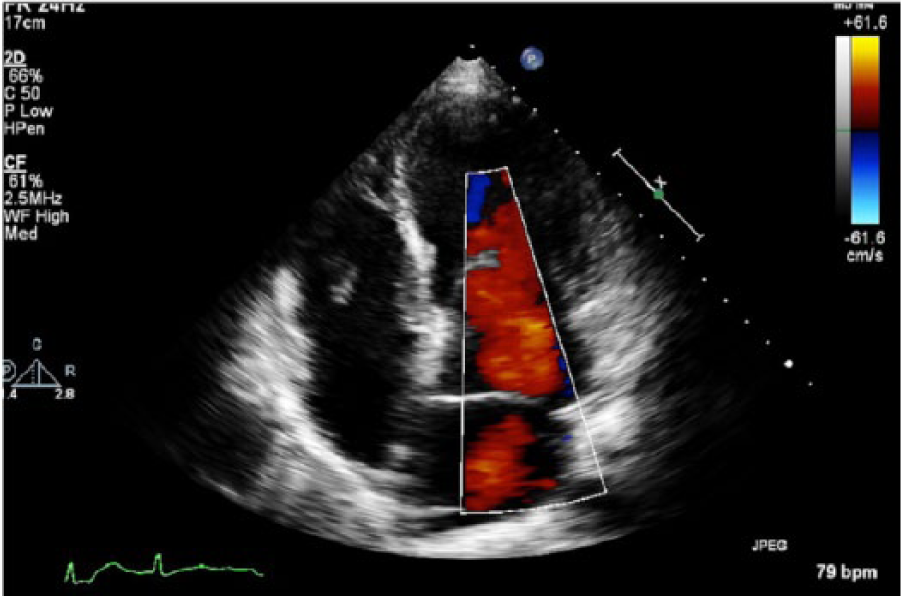
Apical four-chamber view of the heart during ventricular filling with color Doppler imaging. No obstruction to flow is seen.
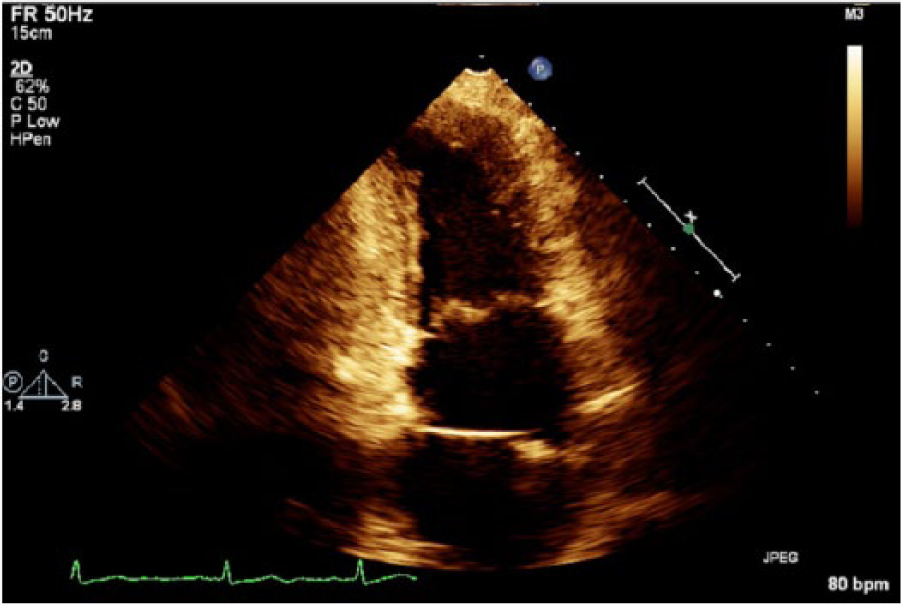
Apical two-chamber view of the enlarged left atrium and the left ventricle showing the echogenic membrane of cor triatriatum sinistrum.
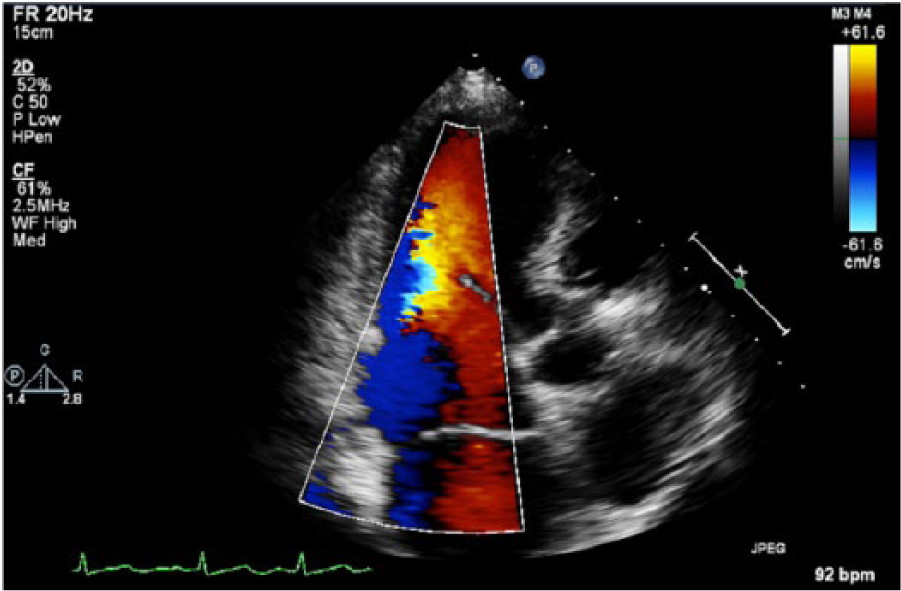
Apical three-chamber view of the heart using color Doppler imaging, again showing no obstruction to flow.
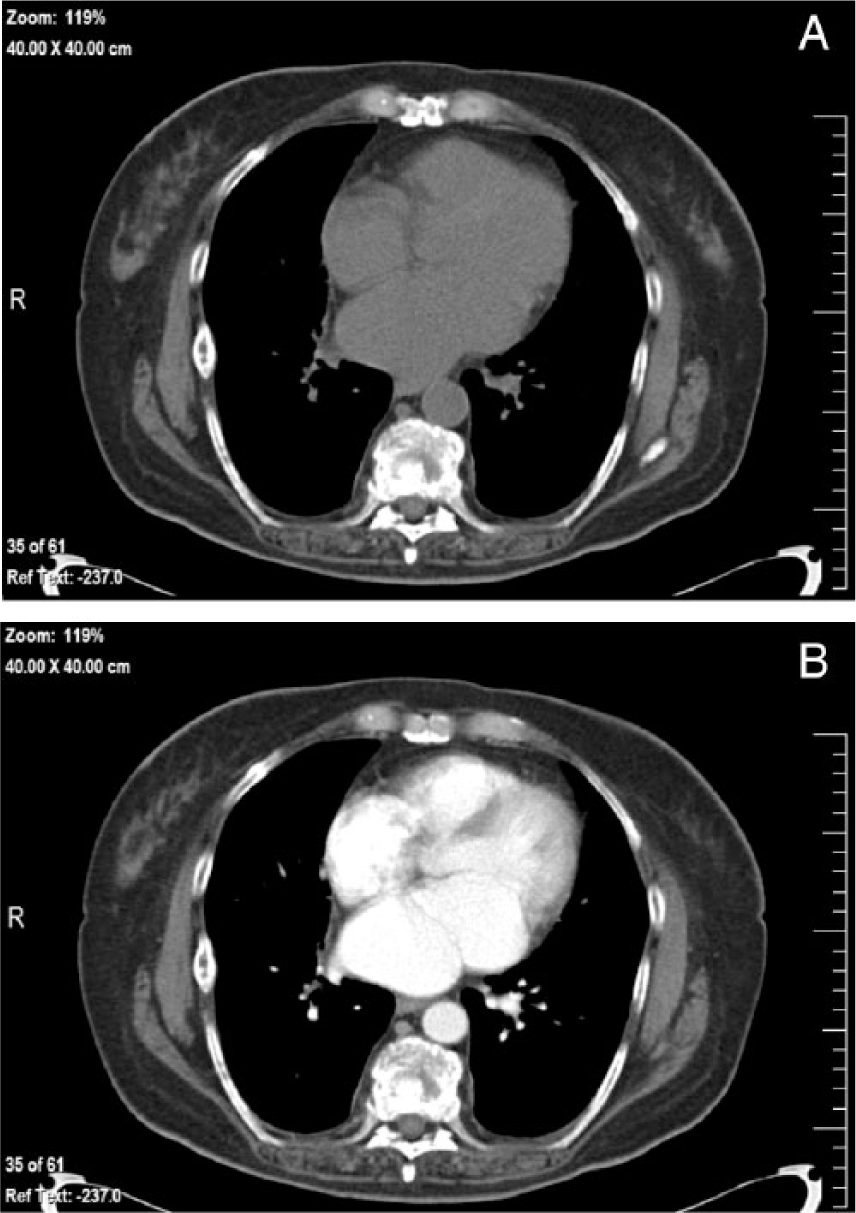
Chest computed tomogram at the level of the left atrium without (A) and with (B) intravenous contrast. The left atrial membrane can be clearly seen in the contrast image (B).
Discussion
A congenital heart defect occurs when the structure of the heart is altered in the early stages of embryonic development. Congenital heart defects are the most common type of birth defect and can involve the walls, valves, arteries, and/or veins of the heart. 2 Severe defects are often found during pregnancy or soon after birth; however, some defects may cause few symptoms and are not diagnosed until later in life.
Cor triatriatum is a rare congenital heart defect usually diagnosed in childhood. 1 Occasionally, some cases remain asymptomatic until adulthood or are found incidentally, as with this case. The reported incidence of cor triatriatum is 0.1% to 0.4%. 1 There is no information to suggest an increase or a decrease in incidence based on sex. In addition, cor triatriatum cannot be linked to any particular genetic disorder; however, the anomaly is associated with other congenital heart defects. The most common defects seen with cor triatriatum are ostium secundum atrial septal defect and anomalous pulmonary vein return. 1 Other defects associated with cor triatriatum are summarized in Table 3. The patient discussed in this case has no known cardiac anomalies other than the recently discovered cor triatriatum sinistrum.
Congenital Heart Defects Associated With Cor Triatriatum.

Boldface items represent the most common congenital heart defects.
Cor triatriatum was first described in 1868 as an abnormal septum located within an atrium, giving the appearance of three atria. 1 The anomaly can be divided into two main groups: cor triatriatum sinistrum (sinister) and cor triatriatum dextrum (dexter). Cor triatriatum dexter occurs in the right atrium and is extremely rare. Conversely, cor triatriatum sinistrum involves the left atrium, as seen with this case. The true left atrium is located distally/inferiorly, connects with the atrioventricular valve, and contains the atrial appendage and true interatrial septum. The accessory atrium receives blood from the pulmonary veins. 1 Several theories about the embryologic process of this anomaly exist. The most widely accepted theory is called “malincorporation,” which is based on the belief that the common pulmonary vein did not join the left atrium in its ordinary fashion. 1 Other hypotheses include malseptation and the entrapment theory, which speculates that part of the embryonic sinus venosus entraps the common pulmonary vein and prevents integration into the left atrium.
In 1949, Loeffler further classified cor triatriatum sinistrum (CTS) according to the number and size of orifices within the septum. 3 Group 1 has no fenestrations within the fibromuscular septum; therefore, the left atrium must communicate with the right side of the heart. Group 2 has a few small openings, which may result in a high degree of obstruction. 3 In group 3, the true atrium and the accessory atrium communicate without obstruction through a large, single fenestration. 1 Groups 1 and 2 are often diagnosed in infancy due to their clinical significance and the symptoms with which they present; they are also associated with an increased mortality rate. 1 Of the three classifications, group 3 may be asymptomatic and is mostly found in adults due to the heart’s ability to function without obstruction. Unfortunately, since further imaging was not performed, there is no way to definitively determine which type of CTS the patient has in this case study. Group 3 is most likely demonstrated in this case due to the lack of obstruction in the left atrium and the incidental discovery in adulthood.
According to the literature, it is very rare that a patient with CTS presents in adulthood with no symptoms or other cardiac anomalies.1,3,4 The symptoms of CTS directly reflect the size of the communication between the true and accessory atria. The most common symptoms include dyspnea, orthopnea, and hemoptysis. 5 These symptoms are consistent with obstruction at the level of the left atrium, which means CTS may be misdiagnosed as mitral valve stenosis.1,3 In adulthood, CTS symptoms are initiated by calcification of the orifice or the development of atrial fibrillation.1,4,6,7 Upon auscultation, typically a loud P2 sound, created by closure of the pulmonary valve at the end of ventricular systole/beginning of diastole, can be heard. Absence of an opening snap or a loud S1 sound, created by closure of the mitral and tricuspid valves at the beginning of ventricular systole, can differentiate between CTS and mitral stenosis. 1
CTS is most frequently diagnosed by echocardiography; however, several other diagnostic imaging modalities exist. 5 CT, magnetic resonance imaging (MRI), and cardiac angiography are all acceptable alternatives when diagnosing CTS. Transesophageal echocardiography (TEE) is often used to characterize the anatomy of the membrane. 5 With the addition of 3D imaging, the physician has the ability to assess the membrane from multiple planes, which enhances diagnostic capabilities. 8 Electrocardiograms (EKGs) will most likely be normal in the presence of CTS. In the past, cardiac catheterization was the suggested technique for diagnosis; however, it is rarely used for diagnostic purposes given the minimally invasive choices now available. 9 For the patient presented, EKGs were normal and no murmurs were heard on auscultation. Had the cardiologist performed a 2D/3D TEE, there would have been more definitive information about the type of CTS present; however, the lack of any significant benefit of a specific diagnosis in this basically asymptomatic patient did not warrant this procedure.
The treatment for CTS varies based on the patient’s symptoms. In the case of an incidental finding in an asymptomatic patient, no medical treatment is required. When the patient complains of exertional dyspnea, diuretics and preload reduction are the suggested medical treatment. 1 Surgical treatment is usually indicated as well. In symptomatic children and adults, surgical resection of the membrane is performed and the overall surgical outcome is excellent.1,4,5
Ultimately, the patient discussed in this case was treated conservatively due to her history of metastatic breast cancer, anemia, and lack of previous symptoms. It is difficult to determine if the existing CTS and mild pulmonary hypertension caused the new onset of exertional dyspnea or if it was caused by other exacerbating factors. Given there were no other cardiac anomalies and the patient was otherwise asymptomatic, she was discharged with a prescription for 40 mg of furosemide daily. Furosemide is a diuretic that can be used for fluid retention, high blood pressure, and heart failure. 10 The patient was instructed to follow up with the cardiologist if symptoms worsened.
Conclusion
Correlating clinical presentations and imaging modalities is imperative for an accurate diagnosis of cor triatriatum. When cor triatriatum is discovered, it is essential to evaluate the presence of other cardiac anomalies and the size of fenestrations within the membrane. Transthoracic echocardiograms and 2D/3D TEEs are valuable tools in the anatomic and hemodynamic assessment of these membranes. As with this patient, surgical intervention is not always indicated. It is important to continue patient monitoring to ensure symptoms resolve or do not worsen with conservative treatment.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
