Abstract
Invasive ductal carcinoma of the breast is cancer that originates in the ducts and subsequently invades the fatty tissue of the breast outside the duct. It is the most common form of breast cancer, representing 80% of all breast cancers. Invasive ductal carcinoma has the potential to metastasize via the bloodstream and lymph nodes, making early diagnosis critical. In the case presented, a rare instance of bilateral primary invasive ductal carcinoma is discussed: It was identified by magnetic resonance imaging, and it was visualized, confirmed, and located by sonography; yet, it was undetected by mammography.
Sonography is an important guide for many diagnostic procedures of the breast. 1 It can be used to assist in biopsy procedures, as high-frequency linear transducers are very accurate in guiding needles to a cyst or mass within a breast. It is also a valuable modality to correlate suspicious findings on mammograms or magnetic resonance imaging (MRI). While mammography is the universal screening tool for breast cancer detection, it detects only about 85% of breast cancers. 2 In the case presented, mammography detected a suspicious lesion in the right breast but was unremarkable for left breast, which was later discovered to contain an even larger area of suspicion than the right breast. As described below, ultrasonography successfully located both lesions and was used to assist with bilateral vacuum-assisted biopsies, facilitating a rapid treatment plan and positively influencing the patient’s prognosis.
Case Report
A zero-gravid Caucasian woman early in her seventh decade had her usual annual bilateral digital screening mammogram, with computer-aided detection performed with projections of right- and left-breast mediolateral oblique view (Figure 1) and right- and left-breast craniocaudal view (Figure 2). The patient reported no palpable lumps and no complaints. Her relevant medical history included her current age, no pregnancies, early menses, and late menopause. Findings on mammography were heterogeneously dense breasts, with benign calcifications noted within the right breast. On the mediolateral oblique projection, the right posterior breast at the level of the nipple, just anterior to the pectoralis muscle, showed an asymmetric somewhat spiculated density. No other abnormalities were noted. The patient was informed, and recommendation was made for a diagnostic, spot compression mammogram with an exaggerated craniocaudal of the right breast. This examination showed that the small nodular spiculated density in the right breast persisted in the 12- to 1-o’clock position of zone 2. A sonographic examination showed a solid nodule at the site noted by mammography, measuring less than 6 mm in size (Figure 3). Posterior acoustic shadowing was noted for a portion of the nodule, with no significant shadowing noted for the remainder of the nodule. A BI-RADS category 4 was recorded—that is, suspicious abnormality such that biopsy should be considered, per the Breast Imaging–Reporting and Data System.3,4 The patient was informed and a recommendation made for an ultrasound-guided, vacuum-assisted biopsy.
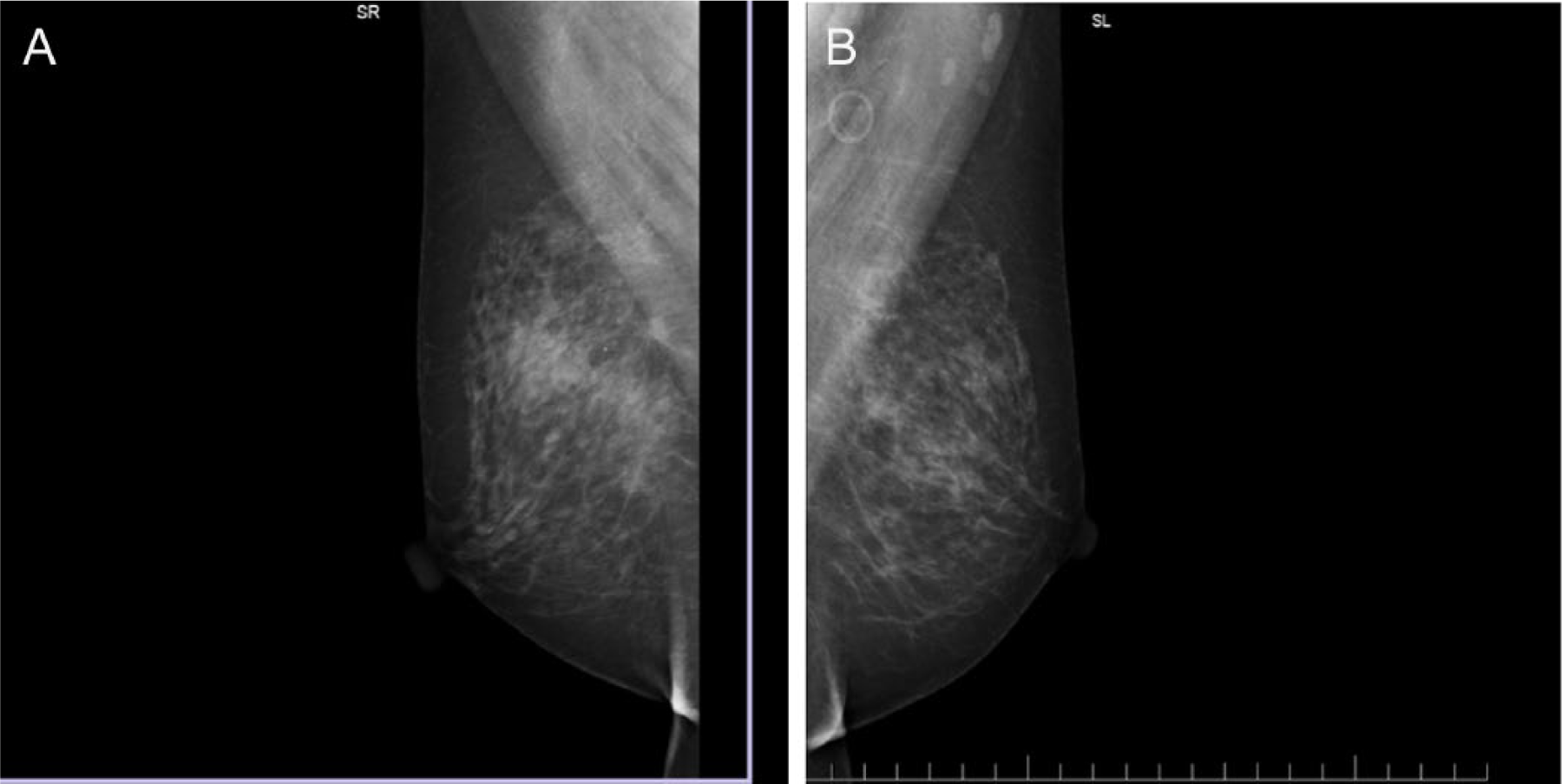
(A) Right and (B) left breast mediolateral oblique screening mammogram images.
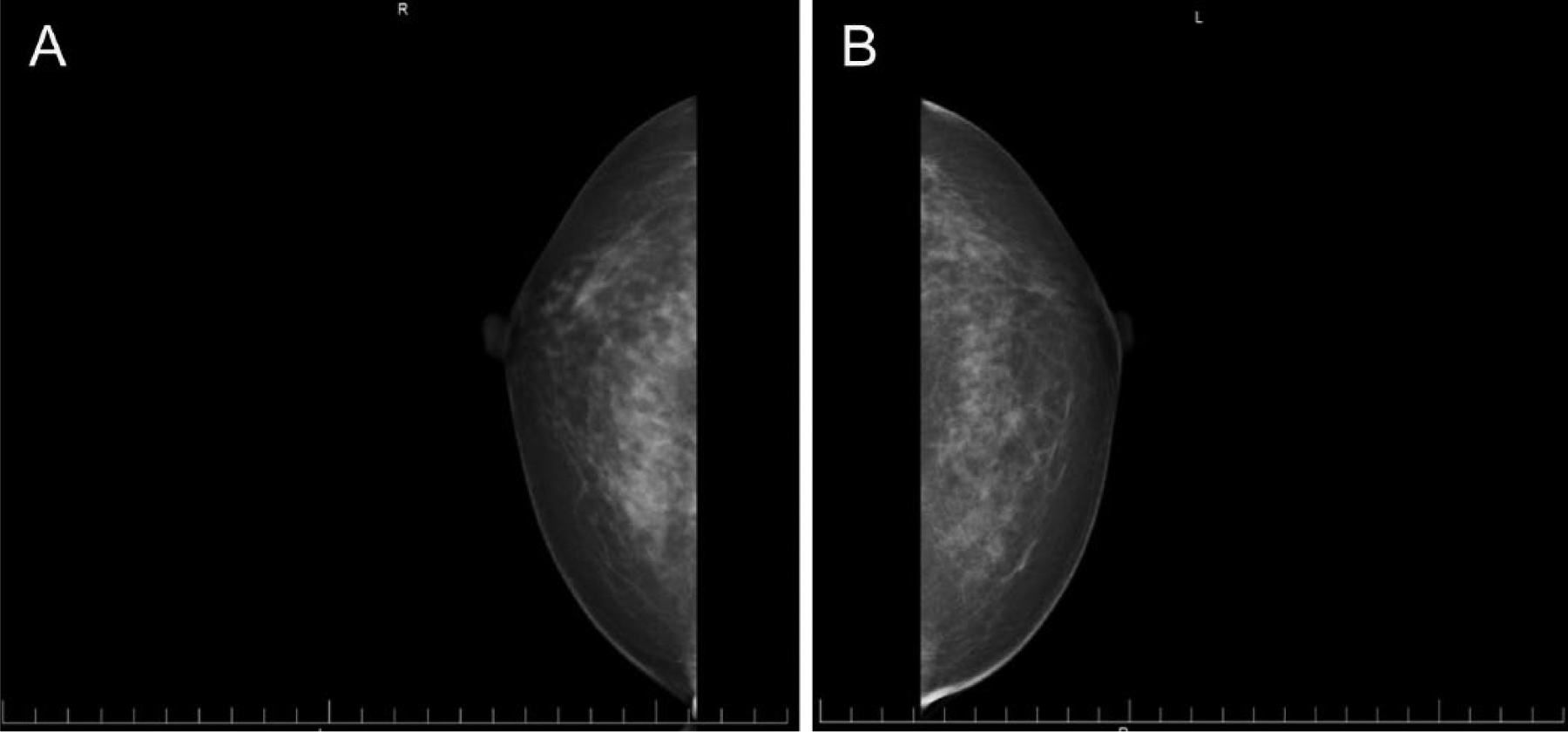
(A) Right and (B) left breast craniocaudal screening mammogram images.
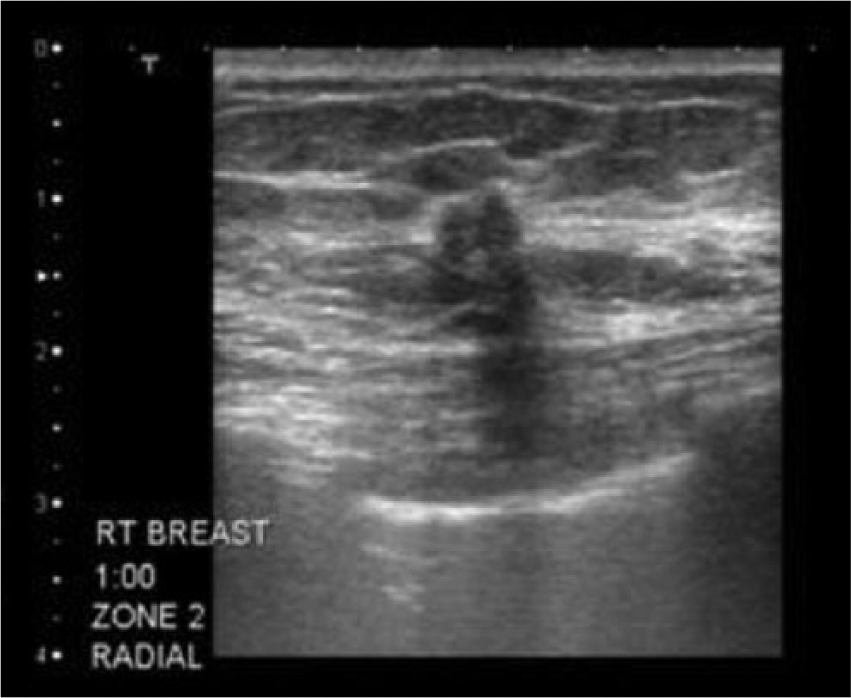
Grayscale image showing a small solid nodule with partial shadowing in the right breast in the 12- to 1-o’clock position of zone 2.
Ten 11-gauge core samples were obtained during the biopsy, removing the majority of the nodule. A metallic clip was deployed at the biopsy site. Pathology results from the right breast biopsy showed a well-differentiated invasive ductal carcinoma occupying 60% of the biopsy tissue. The tissue was given a Nottingham Histologic Score of grade 1: well differentiated with cells that appear most similar to normal and are not growing rapidly.5,6 Surgical consultation was advised, and a right breast lumpectomy with brachytherapy was recommended.
In preparation for the right breast lumpectomy, standard preoperative procedures included bilateral breast MRI with intravenous contrast (Figure 4). Focal areas of interest were evaluated with enhancement activity curves, and correlation was made with the recent mammograms (Figures 1, 2) and the sonogram of the right breast (Figure 3). No enlarged axillary lymph nodes were noted on the right side.
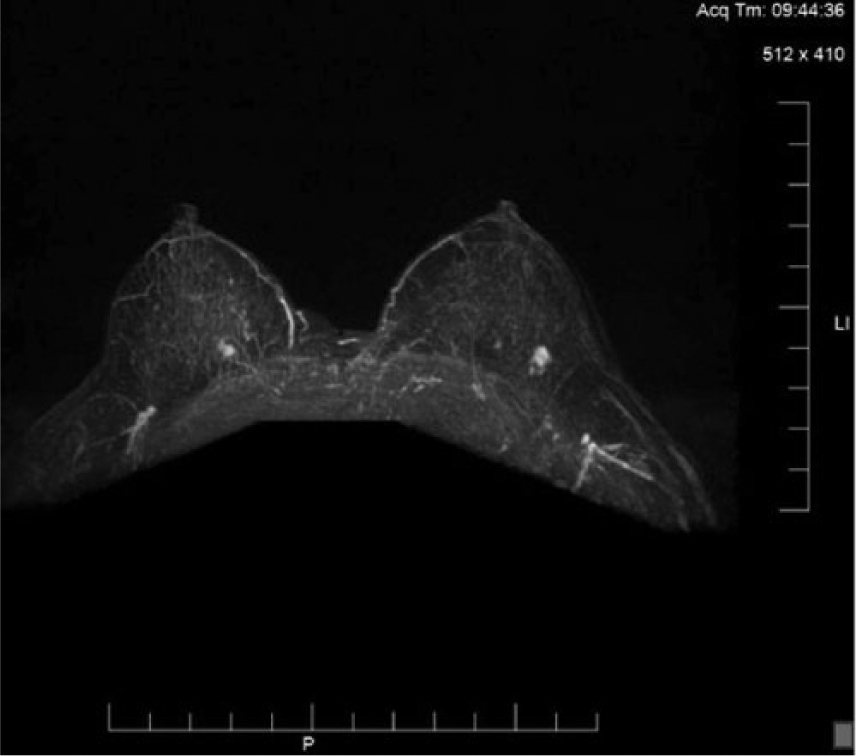
Bilateral breast magnetic resonance image with intravenous contrast demonstrating focal areas of enhancement bilaterally, consistent with the right breast mammogram and bilateral sonographic findings.
In the left breast, MRI with contrast showed a 9-mm focus of intense enhancement with some spiculation in the 3- to 4-o’clock position (Figure 4). Some lymph nodes in the left axilla also showed enhancement but were not enlarged. A BI-RADS category 5 was recorded: highly suggestive of malignancy and requiring biopsy. Because of this new finding in the left breast, there was strong suspicion for contralateral breast malignancy, and an ultrasonogram of the left breast with biopsy was advised.
Real-time sonographic evaluation of each breast was performed. This second sonographic evaluation of the right breast showed the patient’s previous biopsy site marker to be readily seen with no residual suspicious areas. In the left breast, a hypoechoic solid mass in the 4-o’clock position of zone 2 was seen with posterior acoustic shadowing. The mass measured approximately 9 mm with slightly irregular borders and a possible small tail (Figure 5), correlating with the abnormal MRI enhancement. An ultrasound-guided core biopsy was advised, and 5 core samples were obtained with an 18-gauge needle. Pathology results of the left breast samples showed invasive ductal carcinoma, moderately differentiated, occupying the majority of the biopsy tissue, with a Nottingham Histologic Score of grade 2—moderately differentiated with cells that appear somewhat different than normal.
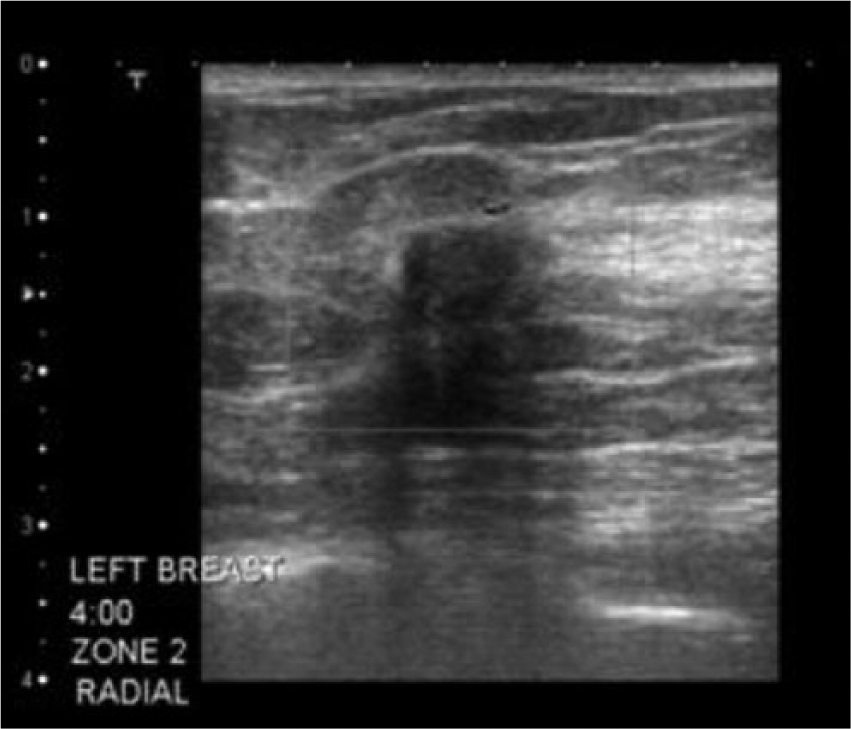
Grayscale image of the left breast showing a 9-mm hypoechoic solid mass in the 4-o’clock position of zone 2, with slightly irregular borders and posterior acoustic shadowing correlating with the magnetic resonance image of the left breast.
Because of the new diagnosis—a change from right to bilateral breast cancer and a higher Nottingham grade for the newly found lesion on the left breast—the patient’s previously scheduled right breast lumpectomy with brachytherapy was canceled. Further consultation with the patient was done to explore her options, which now included bilateral lumpectomies with sentinel lymph node biopsies with the possibility of axillary lymph node dissection to include mastectomy. The patient chose to proceed with bilateral mastectomies with no immediate reconstruction. Bilateral sentinel lymph nodes were removed, and histopathology was negative for malignancy.
Discussion
According to the Physicians Insurers Association of America, delay in diagnosis of breast cancer is the most common reason for which physicians are sued for malpractice. 3 The rendering of a diagnosis based on mammography that improperly fails to indicate a sufficient suspicion for malignancy is considered a false-negative mammogram. 7 The American Cancer Society stated in 2013 that overall, screening mammograms miss about one in five breast cancers; the main cause of false-negative results is high breast density. 8 Theoretically, these false negatives should occur more often among younger women than older women because as a woman ages, her breasts usually become more fatty (less dense), and false-negative results become less likely.9-11 Other possible causes for missed breast cancers include poor positioning or technique, perception error, incorrect interpretation of a suspicious finding, subtle features of malignancy, and slow growth of a lesion. 12
Sonography is proving to be an important imaging modality to complement mammography in an attempt to leave no nodule undetected. Grayscale is the most common form of sonography used in breast imaging. Newer technologies—such as harmonic imaging, tissue Doppler imaging, 3-dimensional sonography, and elasticity imaging—have been used to enhance conventional grayscale imaging, particularly in younger patients more likely to have dense breast tissue. 13 Breast sonographic imaging can differentiate solid from cystic masses, suspicious from benign lesions, and abnormal from normal lymph nodes. 13
Sonographic images with mixed features cannot exclude the possibility of malignancy. Suspicious pathology seen in real time can be sampled with sonographically guided breast biopsies, a valuable determinant in treatment planning and patient long-term health. Biopsies are indicated for nodules that are classified on sonography as either malignant or indeterminate, even after a patient may have had a MRI evaluation, as benign lesions continue to be common false-positive findings on MRI. 14
The technical advances in sonographic imaging of the past 30 years have allowed radiologists to differentiate benign from malignant solid breast masses with a high level of accuracy,15,16 thereby simultaneously influencing the recommendation for biopsy as needed and avoiding unnecessary procedures for patients. To decrease the number of biopsies for benign lesions, sonography should be exploited for accurate interpretation of a lesion’s characteristics of suspicion.4,17 Attention to detail in this area renders the most accurate diagnosis and patient care.
Although breast MRI is recommended for screening women at high risk for breast cancer, its role in evaluating the extent of disease in women with recently diagnosed cancer is not universally accepted. 14 The large number of biopsies performed for benign abnormalities because of patient fear, physician uncertainty, or standard protocols has long been recognized as a problem.17,18 Excessive biopsies can have adverse effects on society, increasing the costs of screening projects and health care. 17 Grayscale sonographic imaging has improved significantly, consequently improving diagnostic accuracy where mammography may prove to be limited. Sonographic imaging is so dependable at differentiating simple cysts from solid masses that unnecessary biopsies can be avoided based on techniques for predicting malignancy. 15
In most circumstances, the decision to biopsy should not be based solely on the radiographic report from mammography, and radiologists should review films directly with the sonographer to ensure that the anomaly is real. Attention should be given to these specific signs of malignancy that sonography details, such as hypoechoic lesions with ill-defined nodular borders, “taller than wide” shape, spiculated margins, posterior acoustic shadowing, irregularly branching neovascularity, and microcalcifications, as well as heterogeneous interior echoes and a hyperechoic halo.15,19 Consistent predictions of malignancy can be made with these guidelines as an accurate method of distinguishing cancer from fibroadenomas and from benign tumors, which display such sonographic characteristics as a smooth and well-circumscribed lesion, with the echogenicity being hyperechoic, isoechoic, or mildly hypoechoic; thin echogenic capsule; ellipsoid shape with the maximum diameter being in the transverse plane; and three or fewer gentle lobulations. 15 In a revision of their study, Costantini et al. noted that echographic signs such as hypoechogenicity and indistinct margins are often less predictive of malignancy compared with an irregular shape; spiculated, angular, or microlobulated margins; a hyperechoic halo; and posterior acoustic shadowing. 17
Biopsies are indicated for nodules that are classified by sonography as either malignant or indeterminate, having appearances that differ greatly yet maintaining the typical patterns common to malignancies. In a landmark study by Stavros et al., of 424 lesions classified as benign by means of ultrasound, only 2 were found to be malignant at biopsy, resulting in a negative predictive value of 99.5%. 20
Cancers that spread into nearby tissue are said to be invasive. 1 Invasive ductal carcinoma invades fatty tissue of the breast and has the potential to metastasize to other parts of the body, as carried through the bloodstream or lymphatic system. It is imperative that the cancer be detected before it spreads to other organs. Early detection and treatment, which includes lumpectomy with brachytherapy or complete mastectomy, are the only options to ensure that the patient has received the most aggressive treatment for survival. In the case presented, the patient’s cancer was detected before it metastasized, yet the implications of bilateral primary breast cancer suggest an even higher chance for invasion to spread if not diagnosed and properly treated. While the current standard screening method of mammography for pre- and postmenopausal women was able to detect this patient’s right breast nodule, limitations in its ability to penetrate dense breast tissue, perhaps uncharacteristic for this patient’s age group, cannot be ignored. Berg recently demonstrated that incident annual supplemental screening ultrasound in intermediate- and high-risk women with mammographically dense breast tissue enabled detection of an additional 3.7 cancers per 1000 women screened. 21
Conclusion
Sonography is very reliable at characterizing breast masses, as well as revealing nodules otherwise not detected on mammogram. With sonographic breast imaging, decisions can be made regarding the need for tissue biopsy, eliminating the need for painful, costly, unnecessary procedures for patients while also providing diagnostic results to guide the best treatment approach. Sonographic imaging proves to be a practice to better support breast cancer screening, staging, and treatment. Sonography, in the case presented, was the resource that physicians employed for detection, confirmation of mass characteristics, specific location, and guidance of patient treatment.
Footnotes
Acknowledgements
The author would like to thank and recognize the hardworking sonographers, mammographers, radiologists Dennis C. Luetkemeyer, MD, and Mitchell Godbee, MD, along with the entire imaging staff at the clinical site where this patient’s case was studied.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
