Abstract
The presence of a goiter in the fetus is an indication of maternal thyroid dysfunction. It carries an increased morbidity and mortality for the fetus and into the neonatal period. Maternal thyroid disease must be monitored closely and medications adjusted throughout pregnancy to prevent adverse outcomes. In females with poorly controlled thyroid disease, sonography is useful in the evaluation of the fetus at risk for goiter.
Introduction
Thyroid dysfunction is a common condition in adults and can be difficult to control during pregnancy. Normal pregnancy-related hormones affect the thyroid gland and signal it to increase production of thyroid hormones.1,2 The fetal brain is dependent on maternal thyroid hormones for normal development, and maternal thyroid dysfunction can cause serious complications for both the mother and fetus.1,3,4 Thyroid function should be closely monitored throughout pregnancy in mothers with thyroid disease and medications adjusted as necessary. In cases of thyroid disease that are difficult to control, sonography may be used to evaluate for fetal complications such as goiter. The presence of a fetal goiter is very rare and indicates abnormal thyroid function in the fetus. Treatment of fetal goiter depends on the type of thyroid dysfunction present. Sonography plays an important role in determining whether the goiter is due to an underactive fetal thyroid or due to maternal antibodies from Graves’ disease. 5 Neonates born to mothers with severe thyroid disease are also at risk for thyroid disease in the first few months of life. 6
Case Report
A 38-year-old pregnant woman, G4P3003, presented to an outside clinic for initial prenatal care in the first trimester. The patient had noticed increased neck swelling during the preceding 4 months. In addition to neck fullness, she complained of insomnia, tremulousness, anxiety, weight loss, rapid heart rate, rapid respiratory rate, proptosis, fatigue, nausea, and general pruritus. At the time of initial presentation, laboratory evaluation of her thyroid function revealed significant hyperthyroidism with thyroid stimulating hormone (TSH) <0.01 mIU/L (normal laboratory reference values, 0.50–5.0 mIU/L) and free thyroxine (T4) of 5.8 ng/dL (normal, 0.8–1.8 ng/dL). She was started on anti-thyroid drug therapy (ATD) and referred to our clinic at 15 weeks gestation for close management during pregnancy.
At 15 weeks gestation, her laboratory values were improved but still abnormal, with free T4 at 4.29 ng/dL. She was also found to have positive TSH Receptor Antibodies (TRAB) >40 IU/L (normal, <1.75 IU/L) and thyroid stimulating immunoglobulin (TSI) >500% (normal, <122%). Her anti-thyroid medication dose was increased secondary to a diagnosis of Graves’ disease and the high levels of antibodies. Her symptoms improved with the exception of occasional palpitations, upper extremity tremors, and some memory difficulty. A sonographic examination was performed at 20 weeks gestation using a Philips iU-22 ultrasound system (Philips Medical, Bothell, Washington) with a C5-1 MHz transducer. The examination showed normal fetal growth and anatomy and no evidence of fetal thyroid disease. Given the severity of her Graves’ disease and persistently high levels of antibodies, her medications were adjusted again. The patient’s thyroid hormones began to trend downward and at 26 weeks gestation her free T4 was in the lower range of normal at 1.0 ng/dL. Her antibodies began to decrease to levels that were measureable but were still high. She remained on ATD, but the dosage was continuously lowered as her disease improved with increasing gestational age.
Serial growth sonograms at 23, 26, and 32 weeks gestation showed normal growth and no evidence of thyroid disease in the fetus. At 34 weeks gestation, the patient’s laboratory values showed a rapid decrease in free T4 to 0.53 ng/dL, consistent with hypothyroidism. An additional sonogram was ordered due to concern for fetal hypothyroidism. The sonogram showed bilateral hypoechoic masses in the fetal neck anterior and lateral to the trachea (Figure 1 and Figure 2). The masses were symmetric in size and each thyroid lobe measured 3.8 × 2.3 × 1.4 cm. The circumference of the entire goiter was 11.1 cm, which is in the 95th percentile for the fetal thyroid. Color Doppler imaging showed that the entire thyroid gland was hyperemic (Figure 3 and Figure 4). The diffuse hyperemia was unusual and did not indicate which type of thyroid dysfunction the fetus had. No other abnormalities were identified.
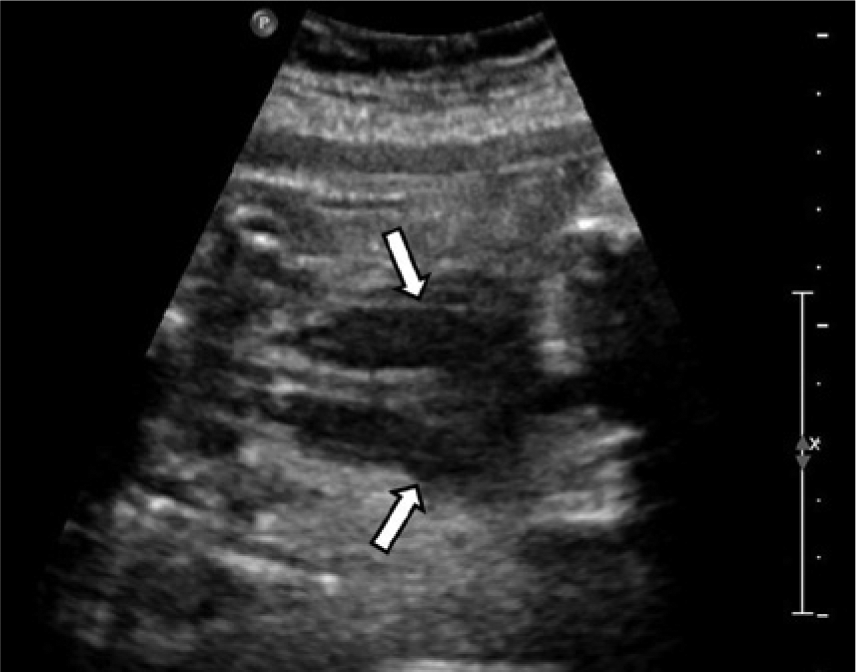
Coronal image of the fetal neck showing bilateral hypoechoic masses (arrows).
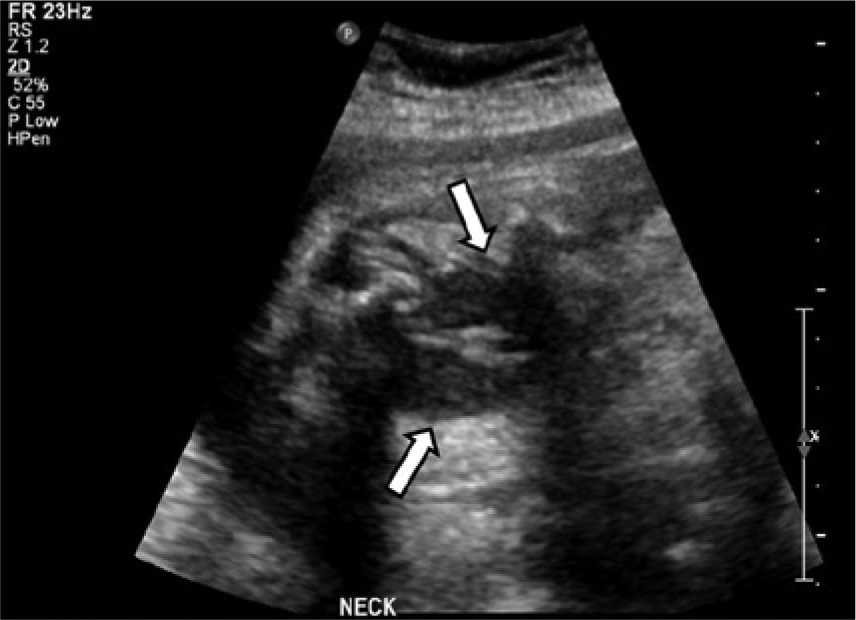
Transverse view of the fetal neck showing bilateral hypoechoic masses (arrows) anterior and lateral to the trachea.
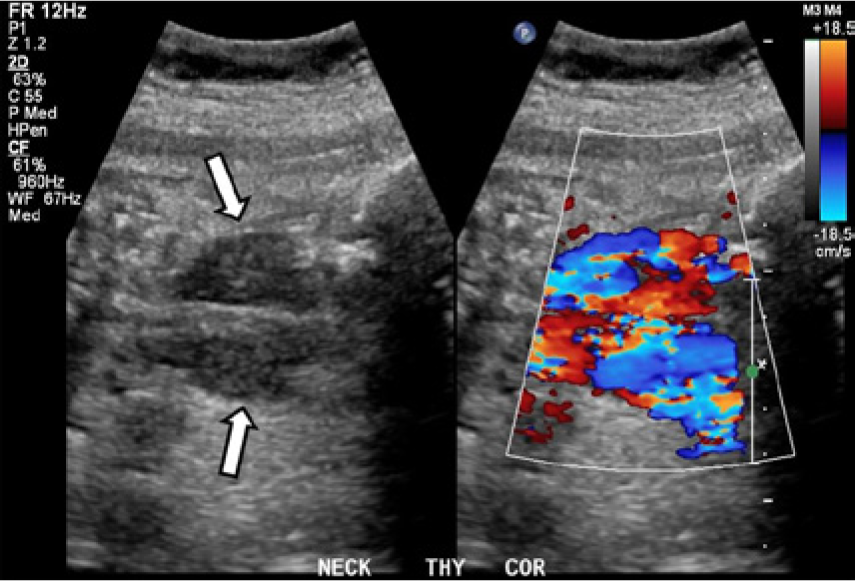
Coronal image of the fetal neck showing the thyroid gland in grayscale on the left (arrows) and with color Doppler imaging on the right. The entire gland can be seen to be diffusely hyperemic; differentiation of hyperthyroidism (typically central vascularity) versus hypothyroidism (typically peripheral vascularity) was not possible in this case.
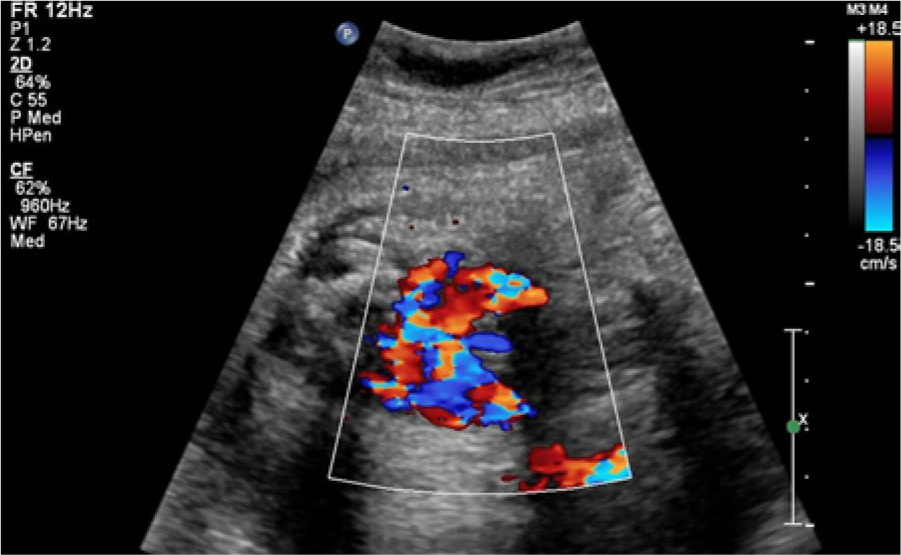
Transverse image of the fetal neck showing the bilateral hyperemic masses with color Doppler imaging, again demonstrating the diffuse hyperemia present.
Due to the persistently elevated thyroid antibodies and the development of fetal goiter, the fetus was considered at high risk for either fetal hypothyroidism or Graves’ disease. The patient was counseled regarding her options and elected to undergo a percutaneous umbilical blood sampling (PUBS) procedure to definitively diagnose the fetal thyroid function in order to initiate in utero treatment with levothyroxine (Synthroid, AbbVie Inc, Chicago, Illinois) if the fetus was found to be hypothyroid. Upon completion of the PUBS, a slow stream of blood was noted arising from the site of the cordocentesis. The fetus’ heart rate dropped from 90 beats per minute to 20 beats per minute and an emergent cesarean section was performed.
A female infant was born at 34.6 weeks gestation. There was initial concern for hypothyroidism secondary to the initial maternal treatment with Propylthiouracil (PTU) for Graves’ disease, so the infant was given a brief course of 12.5 mcg of levothyroxine. This was discontinued on day 5 of life. The infant later developed neonatal Graves’ disease with elevated thyrotropin receptor antibodies and thyroid stimulating immunoglobulin. The infant’s laboratory values showed a TSH of 0.19 mIU/L, down from 0.5 mIU/L, a T4 of 2.43 ng/dL, down from 2.9 ng/dL, a TSI of 361%, and a TRAB of 18.05 IU/L. She remained largely asymptomatic throughout hospitalization. The infant required close follow-up care with endocrinology and her pediatrician for management of her neonatal Graves’ disease. After 3 months she was doing well and was off all medications.
Discussion
Maintaining a normal thyroid function during pregnancy is essential for fetal neurologic development.7,8 During pregnancy, thyroid physiology changes considerably with advancing gestational age and is affected by human chorionic gonadotropin (hCG) and estrogen. Therefore, it is important to use gestational age–specific reference values when testing thyroid function.1,2 Two indicators of thyroid function are the levels of TSH and T4. The thyroid gland secretes thyroxine into the blood stream; the pituitary gland functions to detect how much T4 is in the blood and secretes TSH to stimulate the thyroid to produce more or less T4, depending on how much is present. A low level of TSH occurs when the thyroid gland is overactive and secreting too much T4, a condition known as hyperthyroidism. A high level of TSH occurs when there is an underactive thyroid gland and too little T4 circulating in the blood, thus indicating an underactive thyroid or hypothyroidism.
During pregnancy, hCG stimulates the TSH receptors in the thyroid gland, which causes an increase in T4 and a decrease in TSH levels. Compared to a non-pregnant state, levels of T4 tend to be 1.5 times higher during gestation. 1 The rise in T4 is greatest in the first trimester due to the high concentration of hCG present. T4 concentration decreases in the second and third trimesters, although it consistently remains higher than in the non-pregnant state.1–3 If maternal hyperthyroidism is present prior to pregnancy, it will be exacerbated during pregnancy, especially in the first trimester. The incidence of hyperthyroidism in pregnancy is approximately 2 out of 1000, with greater than 90% of cases being autoimmune in nature.1,3,4 Graves’ disease is the most common form of autoimmune hyperthyroidism. It is characterized by the presence of thyroid antibodies that stimulate the thyroid gland and cause increased secretion of T4, which in turn suppresses TSH. The two common antibodies that are tested for with Graves’ disease are thyrotropin receptor antibodies and thyroid stimulating immunoglobulin.1,3,9
The fetus relies solely on the maternal contribution of thyroid hormones through the placenta until the 17th week of gestation when the fetal thyroid begins functioning. After this point, if maternal antibodies are present, they are able to cross the placenta and affect the fetal thyroid.3,9 Antibody levels naturally decrease during pregnancy, which can lead to remission of Graves’ disease.4,6 The maternal contribution of thyroid hormones is around 30% at term, and the maternal component is not fully cleared from the neonate until the second to the fourth month of life.6,9
Maternal thyroid dysfunction during pregnancy can cause severe complications for both the mother and fetus. Hyperthyroidism can lead to preeclampsia, hypertension, heart failure, or thyroid storm, while hypothyroidism can lead to preeclampsia and postpartum hemorrhage in the mother. 1 Women with either hypertension or hypotension are also at an increased risk for miscarriage. Fetuses of affected mothers are at risk for intrauterine growth restriction (IUGR), prematurity, cardiomegaly, decreased brain development, and fetal death.3,9 The fetal brain relies on maternal thyroxine for proper development. 8 Abnormalities in maternal thyroid status can cause neurologic deficiencies including lower IQ, lethargy, motor disabilities, and intellectual development disorders.1,8 Therefore it is necessary to treat maternal thyroid disease during pregnancy to prevent serious adverse outcomes. Careful monitoring of maternal thyroid hormone levels and antibody levels throughout pregnancy is required and medications are often adjusted throughout gestation as hormone levels change. 7 Anti-thyroid drugs are used to treat maternal Graves’ disease but must be monitored closely to prevent overtreatment and conversion to hypothyroidism in either the mother or the fetus.1,3
Sonography plays an important role in the care of pregnancy complicated by maternal thyroid disease, especially in cases that respond poorly to treatment. Serial sonograms should be performed to evaluate fetal growth and look for evidence of fetal goiter. The development of fetal goiter is very rare, with an incidence as low as 1 in 50 000 live births. A fetal goiter is diagnosed when the thyroid circumference is greater than the 95th percentile for a given gestational age. 5 If a fetal goiter is identified, it is necessary to determine whether the cause is due to fetal hypothyroidism from overtreatment of maternal hyperthyroidism with anti-thyroid drug therapy, or whether it is a result of maternal antibodies crossing the placenta leading to fetal hyperthyroidism. 5
Sonography may be helpful in distinguishing between the two. Hyperthyroid fetal goiters tend to have central vascularity visualized by color Doppler, while hypothyroid goiters are more likely to have peripheral vascularization.3,5 A fetus with hyperthyroidism also may have IUGR, oligohydramnios, tachycardia, and evidence of early bone maturation. 4 Bone maturation can be evaluated in the third trimester by identification of the distal femoral epiphysis. The distal femoral ossification center should not be evident before 28 weeks gestation and is usually just a small echogenic dot at 32 weeks. After 35 weeks gestation it should be easily identifiable. 5 A fetus with hypothyroid goiter may show evidence of delayed bone maturation in the third trimester.
Other nonspecific sonographic findings in a fetus with goiter include polyhydramnios due to compression of the trachea by a large goiter and a hyperextended neck position. 5 In cases where the cause of fetal goiter cannot be determined, percutaneous umbilical blood sampling may be indicated. By sampling fetal blood from the umbilical cord, fetal thyroid status can be definitively diagnosed and proper treatment can ensue.4,9 In cases of hyperthyroid goiter, increased doses of ATD can be helpful in decreasing the size of the goiter and improving fetal thyroid function. In cases of fetal hypothyroidism, maternal ATD therapy may be decreased or stopped and in utero injections of levothyroxine into the amniotic fluid may be given to the fetus to improve thyroid function and reduce the size of the goiter. 3
Neonates born to mothers with positive antibodies should be evaluated for neonatal autoimmune hyperthyroidism. Although only 2% of neonates develop Graves’ disease, those with the disease have increased morbidity and mortality rates. The mortality rate in neonatal Graves’ has been reported as high as 25% and can be attributed to tachycardia and cardiac insufficiency. 6 Neonates may also be at risk for respiratory complications from compression of the trachea by a large goiter. The advanced bone maturation seen in fetal hyperthyroidism can also affect the infant if antibodies persist and can lead to early fusion of the sutures of the skull, a condition known as craniosynostosis. 8 The neonate usually eliminates the maternal antibodies from circulation by 2 to 4 months of age and symptoms resolve as the infant becomes euthyroid. 6
Conclusion
Maternal thyroid dysfunction is a common problem in pregnancy and it can be exacerbated by increased levels of estrogen and hCG during pregnancy. Clinicians should use pregnancy-based reference levels when evaluating thyroid function over the course of gestation. Uncontrolled or poorly controlled maternal thyroid disease can have deleterious effects on both the mother and the fetus. Careful monitoring of maternal thyroid function should be performed throughout gestation, and sonography should be utilized to evaluate fetal growth and well-being and to look for evidence of fetal goiter, especially in cases of severe thyroid disease.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
