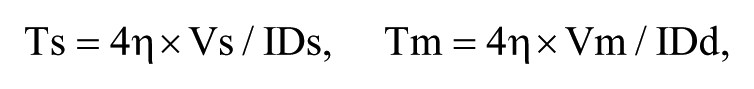Abstract
The aim of this study was to use vascular sonography to evaluate the dynamic forces acting on the wall of the popliteal artery in patients with type 2 diabetes mellitus (T2DM) compared to a control group without diabetes. Forty patients with T2DM and 20 control subjects were enrolled in the study. Patients were divided according to their measured Ankle-Brachial Index (ABI) into two groups, those with evidence of peripheral vascular disease (PVD) based on an ABI < 0.9 (n = 18) and those without peripheral vascular disease (N-PVD, ABI ≥ 0.9, n = 22). The systolic and diastolic internal diameter (IDs, IDd) and the end-diastolic intima media thickness (IMT) of the popliteal artery were measured. Hemodynamic parameters measured were the peak systolic velocity, end-diastolic velocity, and the mean velocity (Vs, Vd, Vm). The peak systolic and mean shear stress (Ts, Tm) were calculated using the Hagen-Poiseuille formula. The shear stress of the popliteal artery was significantly decreased in patients with diabetes in the PVD group compared to the N-PVD group (P < .05). Shear stress also was significantly decreased in both the N-PVD and the PVD patients compared to the control group (P < .05). The shear stress may be a reliable index for the assessment of lower extremity arterial disease status in patients with diabetes mellitus at an early stage when the vascular IMT remains normal.
Introduction
Peripheral vascular disease is very prevalent in patients with type 2 diabetes mellitus (T2DM) and is one of the main causes of lower extremity disability of patients with diabetes mellitus. 1 Significant research has been done on lower extremity atherosclerosis in patients with diabetes mellitus, but there are few reports on early stage vascular lesions in these patients. Studies have shown that the wall shear stress of an artery may reflect the hemodynamic changes associated with atheroscleortic disease and provide an earlier mechanism for diagnosing atherosclerotic changes. 2 Animal research has demonstrated that the high shear stress was agonistic to the atherosclerosis, whereas the low shear stress can lead to the atherosclerosis. 3 The formation and distribution of plaques were mainly due to the state of the low shear stress. 4 The aim of this study is to measure the wall shear stress of lower extremity arteries by using 2D and spectral Doppler ultrasound and assess the dynamic changes of vascular wall in the diabetic patients.
Materials and Methods
Forty patients with T2DM, 23 males and 17 females, were enrolled in the study between October 2008 and September 2009. The patients were 35.7 ± 9.2 years of age (mean ± standard deviation), with an age range of 28 to 50 years. They had had the diagnosis of T2DM for a period ranging from 2 to 10 years. The inclusion criteria were based on the diagnostic criteria distributed by WHO in 1999. 5 Patients were divided into two groups based on their measured Ankle-Brachial Index (ABI): 18 patients with an ABI of < 0.90 were considered to have peripheral vascular disease (PVD), and 22 patients were considered to be without disease based on an ABI ≥ 0.90 (N-PVD). 6 A control group consisted of 20 age- and sex-matched healthy adults (11 male and 9 female) with an age of 34.2 ± 8.3 years and an age range of 25 to 48 years. No control subjects had any diabetes mellitus or evidence of other cardiovascular diseases. There was no hypertension in the control group, with systolic blood pressures ranging from 90 to 140 mmHg and diastolic blood pressures from 60 to 90 mmHg. Likewise, none of the control group had evidence of hyperlipemia (triglyceride, TG: 0.56–1.70 mmol/L; total cholesterol, T-CHol: 1.80–5.17 mmol/L; high density lipoprotein, HDL: 1.04–1.74 mmol/L; low density lipoprotein, LDL: 0.00–3.61 mmol/L). The study was approved by the university Institutional Ethics Committee (Institutional Review Board), and approved informed consent to the protocol was obtained from all patients and control subjects prior to their participation in the study.
Doppler-derived systolic pressures were measured in the arms and at the ankles at rest with the subject supine using the standard method described by Irace et al. 7 ABI was calculated as the higher of the posterior tibial or dorsalis pedis artery systolic pressure divided by the higher of the right or left brachial artery systolic pressure. The lower of the two ABIs was used to stratify the diabetic patients.
Sonographic images of the popliteal artery in the selected lower extremity were obtained on a Vivid 7 system (GE Healthcare, Milwaukee, WI) using a 7.5-14 MHz linear-array transducer. Imaging was done with the subject’s extremity slightly externally rotated and the knee very slightly flexed to provide a clear, high-quality image of the longitudinal axis of the popliteal artery. Images were measured using the system’s calipers to determine the peak systolic and end-diastolic internal diameters of the popliteal artery (IDs, IDd) and the end-diastolic intima-media thickness (IMT). An IMT ≤ 0.9 mm was considered normal, 0.9 mm < IMT ≤ 1.3 mm was considered a thickened intima, and an IMT > 1.3 mm was considered an atherosclerotic plaque. 8
To measure hemodynamic blood flow parameters, the Doppler sample volume was placed in the center of popliteal artery lumen with a size approximating two-thirds of the vessel lumen diameter. The angle correction cursor was adjusted parallel to the blood vessel walls and the Doppler angle was kept at 60° or less. Peak systolic, end-diastolic, and mean velocities (Vs, Vd, and Vm) of the popliteal artery were measured. Measurements of three successive heartbeats were recorded and averaged using EchoPac 6.3 software (GE Healthcare). Based on these measurements, peak systolic and mean wall shear stress (Ts, Tm) were calculated using the Hagen-Poiseuille formula:9,10
where η is the kinematic blood viscosity.
Blood viscosity was measured with a two-channel MDK-3200 Automatic Blood Rheometer (MDK, Inc., Zhuhai, China). All of the indices were measured on the same day.
Statistical analysis of the data was done using SPSS, V13.0 for Windows (IBM, Armonk, NY). The measurement data were expressed as mean ± standard deviation. Statistical comparisons were performed using the Student-Newman-Keuls test for null hypotheses among the groups. A P-value less than .05 was considered indicative of a significant difference.
Results
Comparisons of the measurements of the popliteal artery systolic and end-diastolic internal diameter, intima media thickness, peak systolic, end-diastolic and mean velocities, and wall shear stress for the N-PVD group, the PVD group, and the control group are shown in Table 1 and displayed graphically in Figures 1 to 6. Popliteal artery IDd and IDs were significantly larger in the N-PVD group compared to those of the control group, but there was no statistically significant difference in IMT between the N-PVD group and control group. IDd, IDs, and IMT were all significantly higher in the PVD group compared with the control group.
Comparison of popliteal artery ID, IMT, Vs, Vd, Vm, Ts, and Tm of each group.
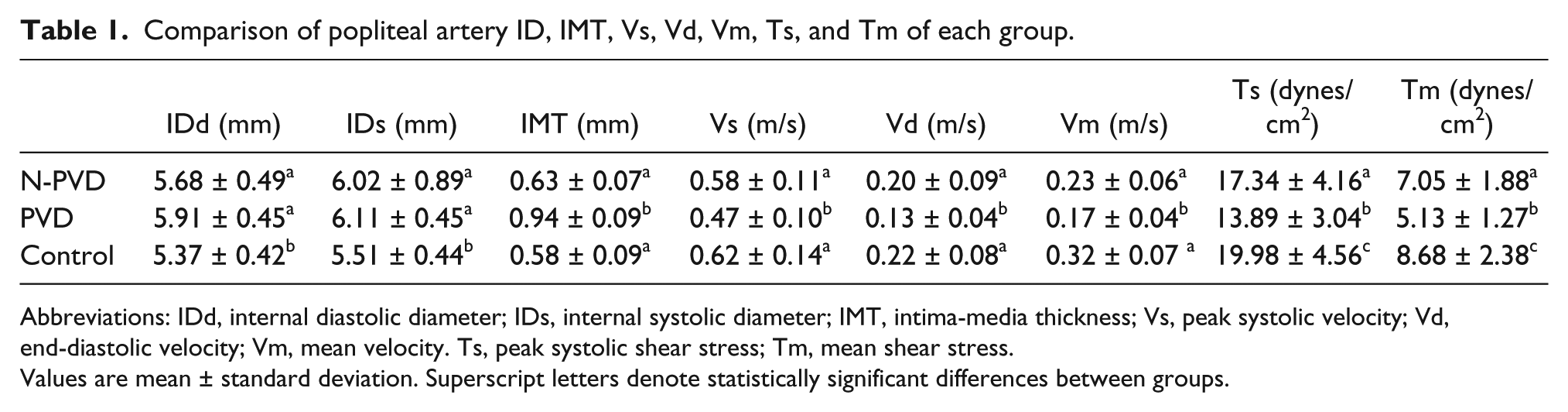
Abbreviations: IDd, internal diastolic diameter; IDs, internal systolic diameter; IMT, intima-media thickness; Vs, peak systolic velocity; Vd, end-diastolic velocity; Vm, mean velocity. Ts, peak systolic shear stress; Tm, mean shear stress.
Values are mean ± standard deviation. Superscript letters denote statistically significant differences between groups.
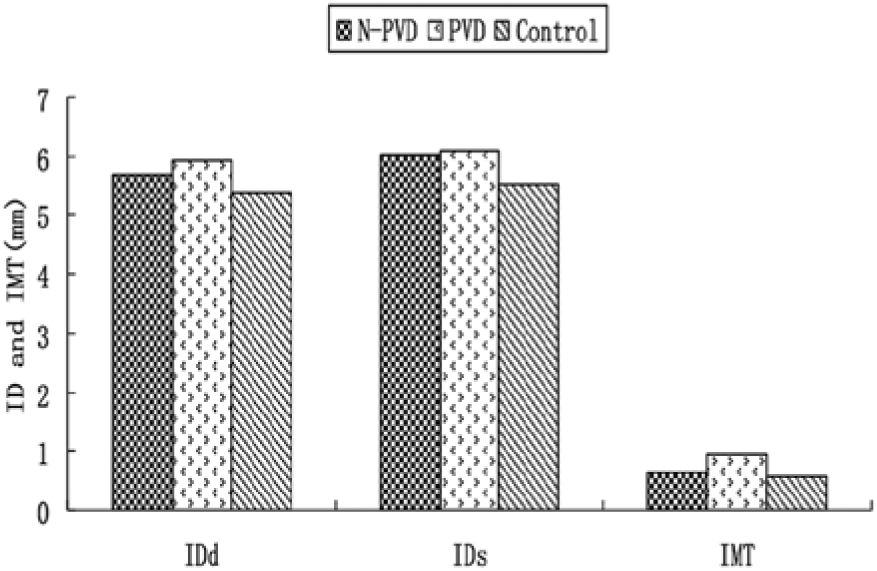
Comparison of popliteal artery inner diameter measurements at peak systole (IDs) and at end-diastole (IDd) and intima media thickness (IMT) between patients with diabetes without peripheral vascular disease (N-PVD), with hemodynamically significant peripheral vascular disease (PVD), and a control group without diabetes with no evidence of peripheral vascular or any other cardiovascular disease.
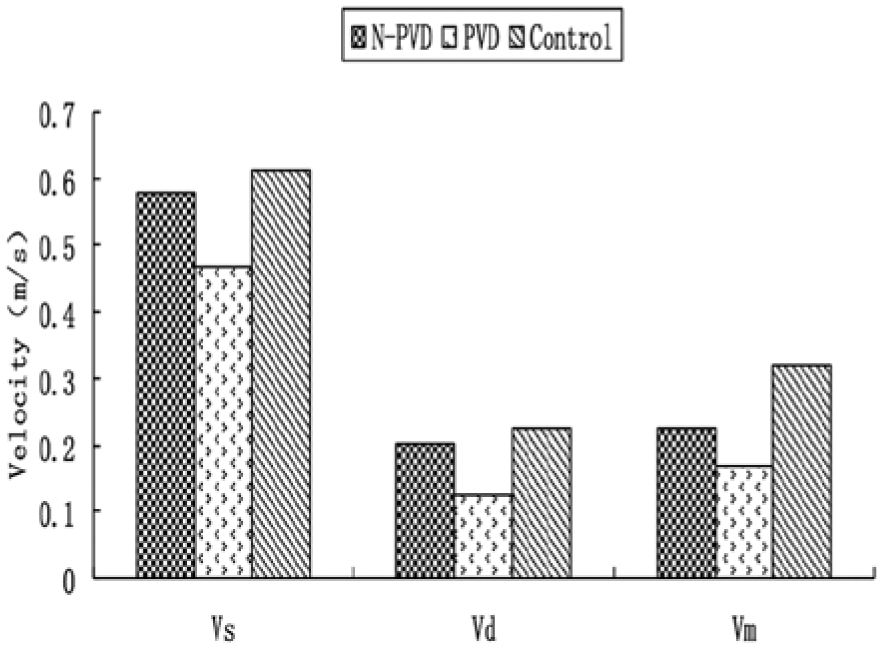
Comparison of popliteal artery peak systolic velocity (Vs), end-diastolic velocity (Vd), and mean velocity (Vm) between the N-PVD, PVD, and control groups. N-PVD patients with diabetes without peripheral vascular disease; PVD, patients with diabetes with hemodynamically significant peripheral vascular disease.
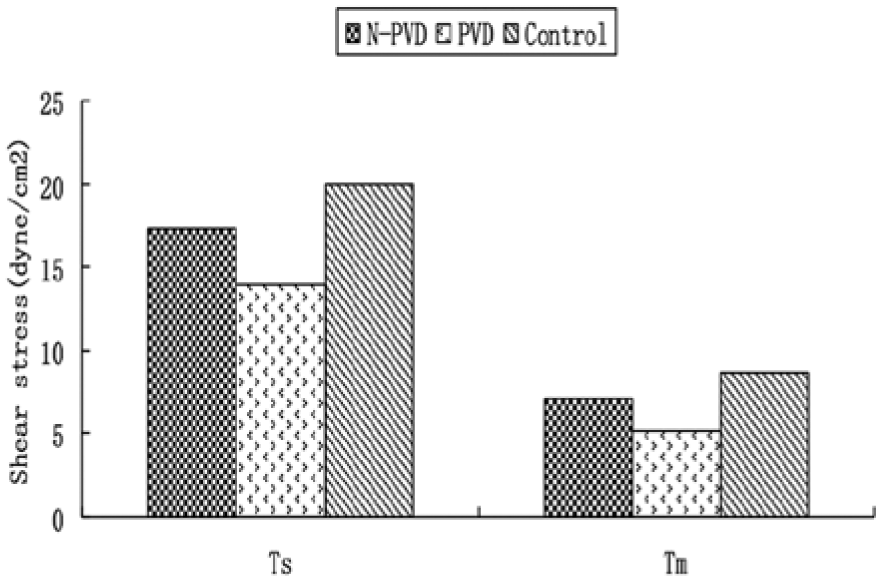
Comparison of popliteal artery peak systolic wall shear stress (Ts) and mean wall shear stress (Tm) between the N-PVD, PVD, and control groups. N-PVD patients with diabetes without peripheral vascular disease; PVD, patients with diabetes with hemodynamically significant peripheral vascular disease.
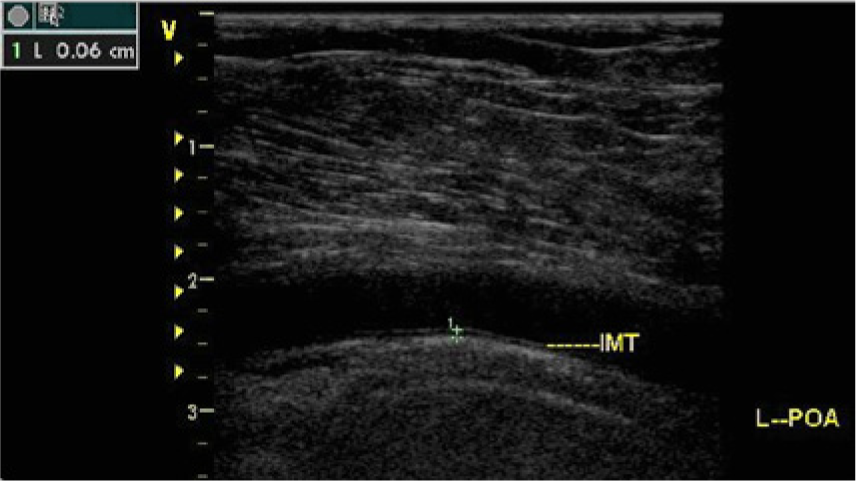
Sagittal B-mode image of the popliteal artery in a control group subject showing the measurement of the intima media thickness (IMT). (L-POA: Left popliteal artery).
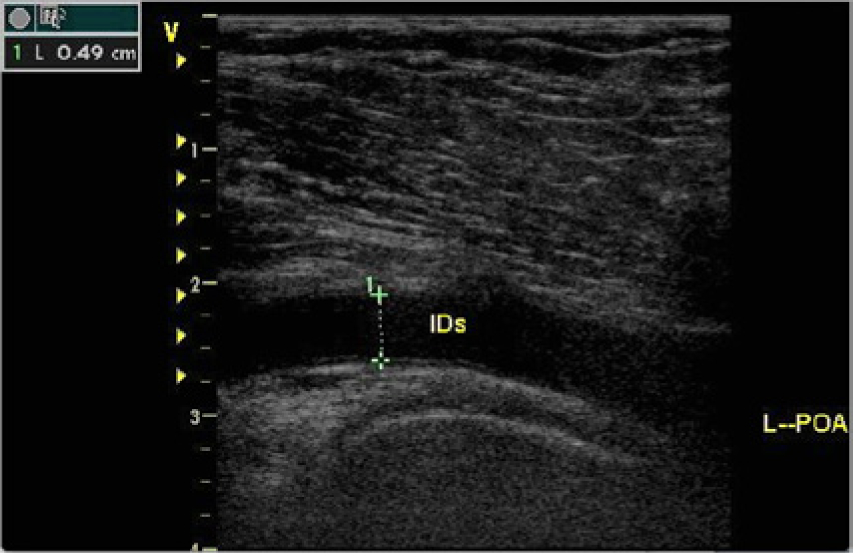
Sagittal B-mode image of the popliteal artery in a patient in the N-PVD group showing the measurement of the inner diameter at peak systole (IDs). N-PVD patients with diabetes without peripheral vascular disease; L-POA, left popliteal artery.
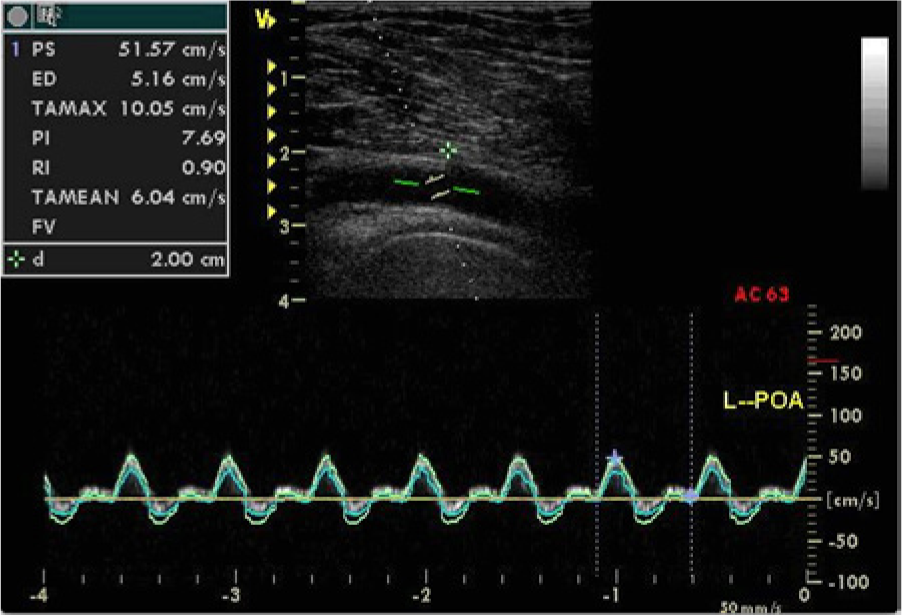
Spectral Doppler display showing the flow velocity in the popliteal artery of a patient in the PVD group with measurement of peak systolic (PS) and mean (TAMEAN) velocities. PVD, patients with diabetes with hemodynamically significant peripheral vascular disease; L-POA: left popliteal artery.
There were no statistically significant differences in popliteal artery Vs, Vd, and Vm between the N-PVD group and control group. Vs, Vd, and Vm were significantly decreased in the PVD group compared to the control group and the N-PVD group. The peak systolic and mean shear stress were significantly decreased in both the N-PVD group and the PVD group compared to the control group. Additionally, Ts and Tm were significantly diminished in the PVD group compared to the N-PVD group.
Discussion
Lower extremity atherosclerotic disease is one of the principal peripheral complications of diabetes mellitus, and it frequently leads to intermittent claudication with rapid progression to critical limb ischemia, gangrene, and significant disability. 11 Early diagnosis is important for successful treatment and improving quality of life.12,13 At least a part of the atherogenesis mechanism is related to lipid retention, oxidation, and modification, which contribute to chronic inflammation at susceptible sites on the walls of all the major conduit arteries.14,15 Initial fatty streaks evolve into fibrous plaques, some of which develop into lesions that are unstable and vulnerable to rupture, causing stenosis or outright thrombosis. 16
An increased arterial IMT can be considered an early manifestation of atherosclerosis. Intima-media thickness provides a measure of vessel morphologic information that reflects the changes of the arterial structure. 17 In conjunction with arterial wall structural changes, there are stress forces on the wall. Circumferential stress, or hoop stress, is a function of the vessel wall thickness and blood pressure. Shear stress is the friction force per unit area exerted created by blood viscosity on the surface of the vascular intima and endothelial cell layer.18,19 The shear stress can be calculated using the Hagen-Poiseuille formula once the vessel lumen diameter and flow velocity are known; the shear stress is directly proportional to the blood viscosity and velocity and inversely proportional to the blood vessel internal diameter.10,20,21 In addition to some of the risk factors associated with diabetes mellitus such as hyperglycemia and hyperlipemia, which may lead to endothelial damage of the vessel wall, the shear stress acting on the vessel wall while it is undergoing atherosclerotic changes and remodeling may have some role in disease progression. 22
This study was designed to investigate quantitatively wall shear stress and the dynamic forces on the popliteal artery in patients with relatively early stage diabetic mellitus using vascular sonography. The Ankle-Brachial Index, a reliable general indicator of systemic atherosclerotic disease and specifically of lower extremity arterial obstructive disease,23,24 was used to group patients with diabetes into those with peripheral vascular disease and those without PVD. The data showed that IDd, IDs, and IMT were significantly higher and Vs, Vd, Vm, Ts, and Tm were significantly lower in the PVD group than in control group without evidence of any diabetes or cardiovascular disease. The diminished shear stress in the PVD group can be attributed both to the increased lumen diameter, possibly a result of vasodilatory remodeling in response to the atherosclerotic disease, 9 and the reduced velocities in the popliteal artery, which may be a result of more distal atherosclerotic disease in the lower leg limiting overall flow and runoff.
Similarly, IDd and IDs were significantly higher and Vm, Ts, and Tm were significantly lower in the N-PVD group compared to the control group. However, there were no significant differences in Vd, Vs, or IMT between these two groups. The calculated shear stress in the popliteal artery in the N-PVD group was significantly lower than the control group, primarily a result of the increases seen in the internal diameter of the vessel. In the N-PVD group, patients with diabetes mellitus but no evidence of hemodyanmically significant atherosclerotic disease and with IMT values comparable to the control group, it may be that the shear stress provides an earlier indicator of the vessel injuries that occur secondary to diabetes.
In comparing the two groups with diabetes, IDd, IDs, and IMT in the PVD group were significantly higher than those in the N-PVD group, and Vs, Vd, Vm, Ts, and Tm in the PVD group were significantly lower than those in the N-PVD group. It could be hypothesized that the thickened IMT, widened ID, and decreased velocity in the PVD group, combining to give a lower shear stress, represent greater degrees of change than the N-PVD group secondary to the progressive nature of diabetic induced atherosclerosis.
Conclusion
Shear stress may be a sensitive measure of peripheral vascular disease and provide an early indicator of changes, even when the IMT and blood flow velocities are normal. The shear stress can be readily calculated using easily acquired hemodynamic and anatomic data from vascular sonography, and it may offer useful information about the vascular status and prognosis of diabetic patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.