Abstract
Sonographic follow-up of surgical or other invasive procedures may lead to incidental findings, and knowledge of a patient’s treatment history is an important factor in properly interpreting these results. The treatment regimen for a patient with deep vein thrombosis and its sequelae can include a combination of anticoagulation, thrombolytic therapy, compression stocking, venous bypass, and stenting of the veins. This case presents an incidental finding during a stress echo examination of embolization into the right ventricle of a previously placed iliofemoral venous stent that required surgical removal. It provides an example of the reasons that sonographers need to understand patient history as well as examination protocols to properly evaluate the sonographic images and to anticipate that there may be unexpected findings with every case.
Keywords
Introduction
Following a laboratory’s protocol in any modality of sonography is crucial to understanding and answering the question as to why the physician ordered the test.1,2 Typical protocols are set up so that disease can be readily ruled in or ruled out. In the echocardiographic case presented, it is made clear that paying special attention to all of the cardiac structures, some of which may be outside of the normal protocol implemented by the sonographer, can be beneficial to the patient’s health. Incidental findings of an urgent nature may be detected that can prompt unexpected immediate intervention. The case report below demonstrates how a sonographer’s role in the routine examination for any modality can move beyond the conventional protocol to incorporate significant incidental findings and affect patient care.
Case Report
A 57-year-old male with a history of left ankle and foot swelling presented to his primary care physician and had a venous duplex sonography examination. The examination showed significant valvular incompetence in the left common femoral vein, femoral vein, and greater saphenous vein. Compression stockings were ordered and used in an attempt to control the swelling. Despite appropriate use of stockings, swelling in the ankle and foot persisted and remained a significant lifestyle issue for the patient. Referral to a vascular surgeon was made to explore interventional management of the patient’s peripheral symptoms. At that time, venous insufficiency testing using duplex sonography showed patent deep and superficial veins in the left lower extremity; evaluation of the proximal left common iliac vein showed marked compression between the proximal right iliac artery and the pelvic rim. The patient was admitted to the hospital for endovascular intervention. Iliofemoral ascending venography was performed to confirm stenosis of the left common iliac vein. Stenting of the vein was performed using intravascular ultrasound guidance, and poststenting, the patient was placed on warfarin anticoagulation for 4 weeks.
On the first poststenting day, the patient developed premature ventricular contractions and chest tightness. Cardiology consultation was obtained, and the ectopy was deemed to be benign and possibly secondary to a noncardiac cause such as caffeine overload, lack of sleep, or stress. The patient was considered stable and was discharged home. During the patient’s next follow-up visit with his cardiologist, it was noted that the patient had had a coronary artery calcium score of 28 (50th percentile) in 2010, and a stress echo examination was ordered to assess coronary reserve. Standard resting parasternal long axis, parasternal short axis, apical four-chamber, and apical two-chamber views were obtained. While obtaining the apical four-chamber view, an echogenic foreign structure was visualized below the tricuspid valve near the chordae tendinae (Figure 1). The stress echo imaging protocol was halted and further evaluation focused on the right heart was carried out immediately. Spectral and color Doppler interrogation of the tricuspid valve showed a significant amount of tricuspid regurgitation (Figure 2).
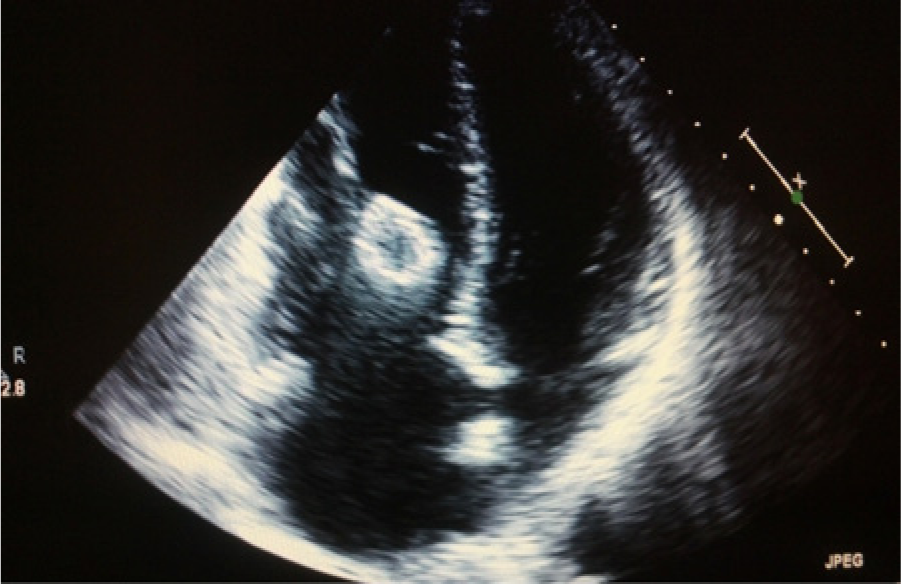
Resting stress echo apical four-chamber view of the heart showing the iliofemoral stent within the right ventricle just below the site of the tricuspid valve.
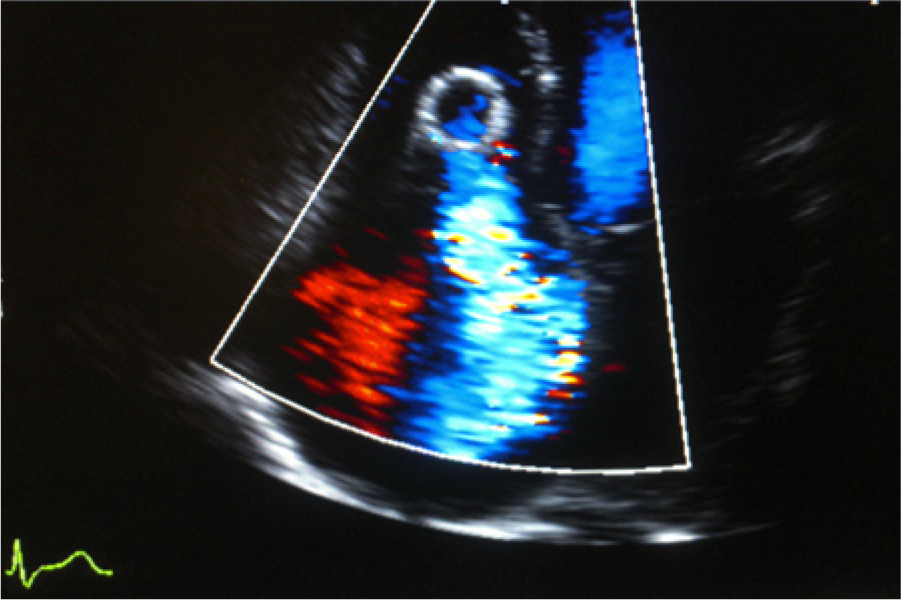
Color Doppler image of the right heart showing significant tricuspid valve regurgitation caused by tangling of the stent within the chordae tendinae of the valve.
Upon review of the patient’s history, the structure was felt to be the peripheral venous stent that had migrated to the right ventricle and entangled itself within the chordae tendinae. The attending physician referred the patient to the hospital for admission. After admission, a bedside echocardiogram was performed to analyze the anatomic positioning of the stent. The dimensions of the stent were measured to be 3.5 cm by 2 cm, and it was noted to be positioned horizontally below the tricuspid valve in the sub-tricuspid valvular apparatus, with one end facing the right ventricular outflow tract. In the cardiac catheterization laboratory, percutaneous catheter-directed attempts to retrieve the stent were unsuccessful. A cardiothoracic surgeon was consulted and open heart surgery was scheduled promptly.
Following pericardiotomy, a robotic-assisted procedure was performed to help break down the stent and remove it from the heart. The tricuspid valve was inspected and it was determined that the stent was causing septal leaflet compression of the valve as well as prominent inclusion of the subvalvular apparatus. The stent was also tangled within the chordae tendinae, putting the valve at further risk for mechanical damage for chordal rupture, leading to severe tricuspid regurgitation. In addition, a thrombus was visualized in the right ventricular outflow tract where the stent was being pushed by hemodynamic forces. A 4-0 expanded polytetrafluoroethylene (Gore-Tex®; WL Gore, Newark, Delaware) neochord was attached, tested, and secured into position. There were no complications during the operation and a postoperative echocardiogram showed only mild tricuspid regurgitation with normal biventricular function. Postoperative recovery was uneventful, and after 4 weeks of cardiac rehabilitation, the patient had increased his strength and endurance.
At that time, a standard Bruce treadmill protocol showed that he was able to exercise at a metabolic equivalent (MET) level of 5.5 for 40 to 50 minutes with no cardiovascular symptoms. (METs are a physiologic measure of energy expenditure during physical activity, where 1 MET represents the resting metabolic state [3.5 milliliters of oxygen per kilogram of body weight per minute]. An individual exercising at 10 METs is exercising at a vigorous intensity.) A follow-up echocardiogram performed 3 months postoperatively confirmed interval removal of the vascular stent from the right ventricle without residual thrombus (Figure 3). Inspection of the tricuspid valve showed restricted mobility of the septal leaflet with abnormal leaflet coaptation due to tricuspid valve repair. Doppler interrogation of the repaired tricuspid valve demonstrated a mean pressure gradient of 1 mmHg and moderate residual tricuspid regurgitation was seen.
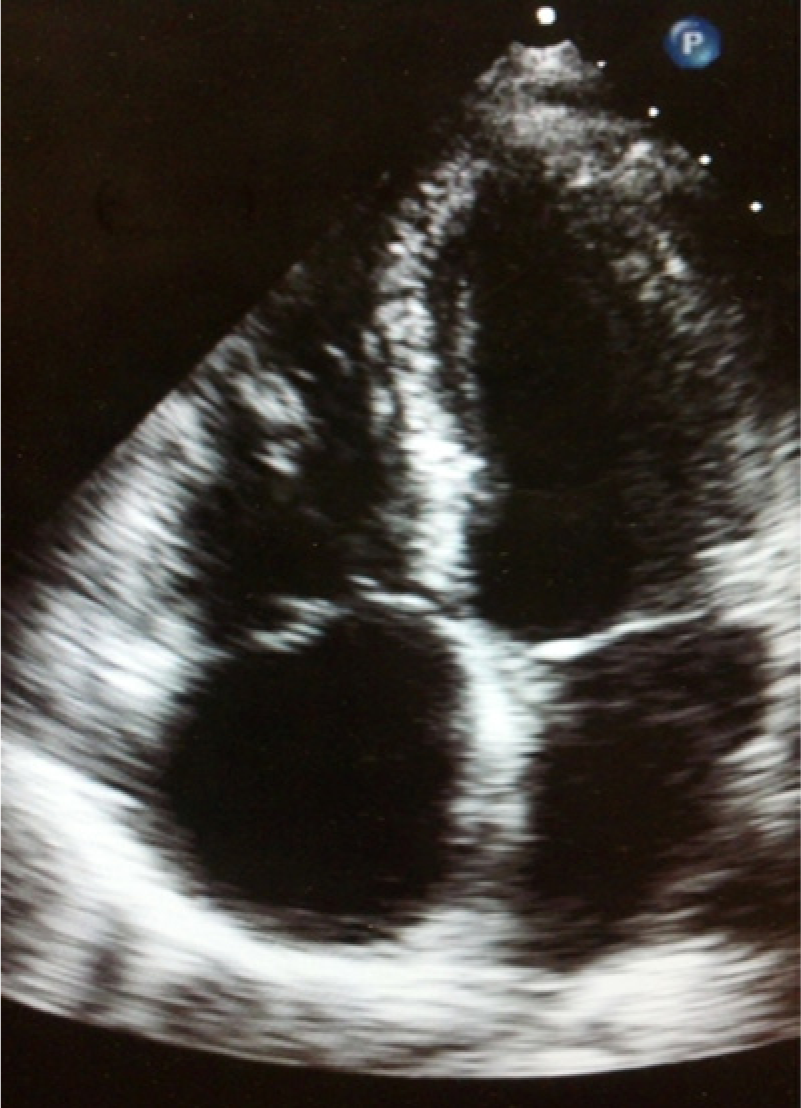
Apical four-chamber view of the heart following surgical retrieval of the stent from the right ventricle.
Discussion
In the venous system, normal flow hemodynamics carry blood from the peripheral circulation back to the right side of the heart. Pathologic states such as deep vein thrombosis (DVT) can lead to damaged venous valves and alter these hemodynamic patterns such that blood flow may be obstructed and/or reversed, causing peripheral swelling and edema secondary to blood pooling in the extremities. 3 Conversely, persistent stasis and edema involving damaged venous valves secondary to other etiologies are risk factors for the formation of DVT.4,5 Patients with chronic DVT can develop persistent edema of the extremities, chronic leg pain, venous stasis ulcers, and secondary varicose veins.6,7 Typically, the treatment regimen for a patient with acute DVT involves anticoagulation and possible thrombolytic therapy. 8 When chronic venous disease develops, the treatment options expand to include compression stocking, bypass, endovascular thrombolysis, and possible stenting of the obstructed veins.9,10
Often, the symptoms of chronic edema and leg pain are the result of a previous thrombotic event, but this is not always so. In the case presented, venography ruled out any thrombotic manifestation but showed that the vessel anatomy was consistent with May-Thurner syndrome.11,12 This anatomic arrangement of the proximal right common iliac artery crossing over the proximal left iliac vein causes compression of the vein between the artery and the pelvic rim just posterior to the vein. Pain and swelling are typically experienced in the left leg in significant cases due to the chronic compression of the vein. When sonographic assessment of the left lower extremity shows that there is no infrainguinal thrombosis causing the edema and pain, the iliac vein itself should be evaluated to rule out this syndrome.13,14 Current management of symptomatic May-Thurner syndrome is frequently percutaneous venous dilation and stenting.15-18 Migration of these stents further proximally into the heart is similar in nature to the more commonly reported migration of an inferior vena cava filter or an upper extremity venous stent placed in dialysis access patients; it is a rare event but can be a life-threatening one.19-22
Stress echocardiography is routinely ordered to detect significant obstructive coronary artery disease, typically manifested by new regional wall motion abnormalities postexercise secondary to the myocardial ischemia induced by increasing the heart rate and myocardial oxygen demand. With hemodynamically significant coronary artery stenosis, the amount of oxygen demand due to an increase in exercise cannot be met adequately. As with any echocardiogram, however, it is important to take note of all the cardiac structures, even those outside the attention of the immediate protocol. 23
In the case presented, the awareness of the sonographer at the very outset of the examination in recognizing the rare event of a migrated stent in the right ventricle, before beginning the stress echo protocol, prevented further damage to the heart and complications to the patient. The sonographer immediately stopped the stress echo protocol and began to interrogate the right side of the patient’s heart based on the finding of the echogenic foreign body. The resultant resting echocardiographic images showed the stent adjacent to the chordae tendinae of the tricuspid valve with significant regurgitation. If the stent had been overlooked and the patient had exercised vigorously during the stress echo examination, the increased flow within the heart would have put the structural integrity of the tricuspid and pulmonic valves at an even greater risk, and ultimately it could have resulted in further embolization to the lungs with potentially grave consequences.
Conclusion
Anticipating the possibility of the unexpected is the ideal mindset when beginning any sonographic examination. Echocardiographers will come across incidental findings that may not be discovered with other cardiac testing and will ultimately change the future care of the patient. If gone undetected, the iliofemoral stent in the patient presented could have created irreparable damage to the patient’s heart and lungs. The risk of putting the patient’s life in danger would have greatly increased if the stent had gone unnoticed and not been investigated further and the stress echo had simply been done following routine protocol. Full understanding of the anatomy and documentation of high-quality images is the standard of care that our patients expect from sonography professionals. The ability to recognize the clinical importance of incidental findings that could potentially affect a patient’s wellbeing is another hallmark of the professional and is a motivational factor that allows sonographers to take pride in their work.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
