Abstract
May–Thurner syndrome (MTS), also known as Cockett syndrome or iliac vein compression syndrome, is a condition in which patients develop swelling, deep vein thrombosis (DVT), venous insufficiency, and other symptoms of the left lower extremity due to an anatomic variant in which the right common iliac artery overlies and compresses the left common iliac vein against the lumbar spine. Although it is an uncommonly diagnosed condition, it is estimated to compose up to half of cases of left lower extremity venous disease. Although having some degree of iliac vein compression is considered a normal anatomic variant in an asymptomatic patient, those who experience severe swelling, venous reflux, and DVT often have anatomically abnormal veins with a spur formation. With proper technique and proficiency, transabdominal sonography can be used as a valuable diagnostic tool in the discovery and to facilitate treatment of May–Thurner syndrome. Diagnostic ultrasound also can monitor the development of recurring DVT and identify symptoms of postthrombotic syndrome.
May–Thurner syndrome (MTS) is a rarely diagnosed condition that occurs when the right common iliac artery (RCIA) compresses the left common iliac vein against the lumbar spine (Figure 1). This narrowing of the left common iliac vein (LCIV) can cause severe complications in the left lower extremity (LLE), such as swelling, pain, venous reflux disease, and deep vein thrombosis (DVT). Although it was first described in 1851, there are currently no standardized criteria in diagnosing MTS. Not only is sonography a useful tool in the discovery of DVT, but it also has an ongoing role in patients with venous ulcerations and chronic edema, which can cause lifestyle limitations in patients with postphlebitis syndrome.
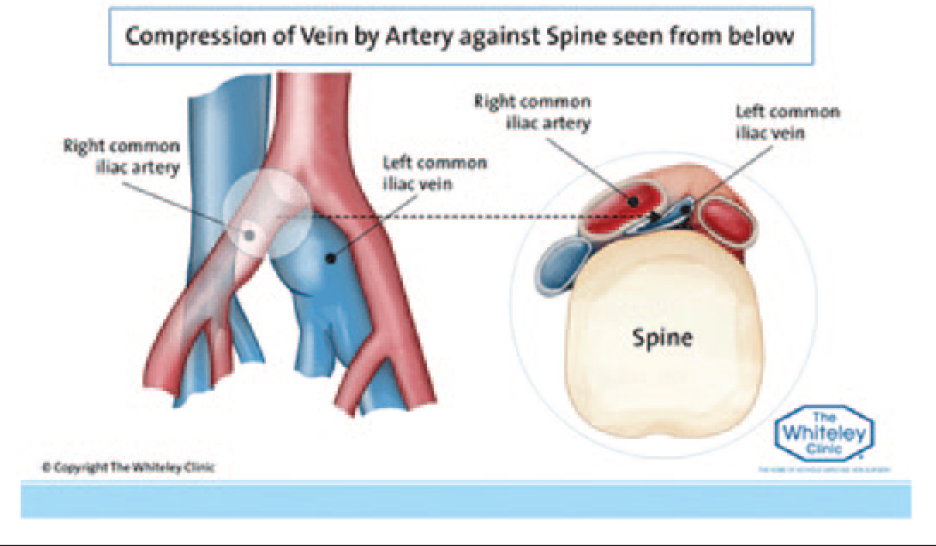
With May–Thurner syndrome, the right common iliac artery compresses the left common iliac vein against the lumbar spine.
Case Study
A woman late in her fourth decade presented to her vascular physician complaining of LLE varicose veins, achiness, fatigue, and swelling. Three years earlier, she was diagnosed with LLE DVT in an emergency department and was treated with an inferior vena cava (IVC) filter. This was reportedly because she was not a candidate for anticoagulation due to a history of a ruptured stomach ulcer. It is unfortunate that the patient did not follow up with her vascular physician after her initial DVT diagnosis until the LLE symptoms became severe. She did not have any indication of coagulation abnormalities. A bilateral lower extremity venous reflux sonogram and IVC sonogram were ordered. A linear transducer with a center frequency of 9 MHz was used on a Philips iU22 system (Philips Healthcare, Andover, MA) to perform the lower extremity venous reflux sonogram with the patient in reverse Trendelenburg position. The study revealed chronic, nonocclusive DVT in the left superficial femoral vein and acute DVT in the left peroneal vein that was nonocclusive from the distal calf to the middle calf and occlusive from the middle calf to the proximal calf. The common femoral vein (CFV) had continuous flow, suggesting proximal obstruction, and the superficial femoral vein presented as a dual femoral vein system, which may or may not indicate collateral formation in the LLE. She also had severe reflux in the left common femoral vein (6587 ms), femoral vein (6535 ms), and great saphenous vein (2164 ms, measuring 7.2 cm at the saphenous-femoral junction), which implied hemodynamic changes in venous pressure in the left leg and possible damaged valves in the deep system caused by prior DVTs (Figure 2). A curvilinear transducer with a center frequency of 5 MHz was used to perform the IVC sonogram, which revealed extrinsic compression of the LCIV, measuring 2 mm, compared to the right common iliac vein (RCIV), which measured 6 mm (Figure 3). The LCIV had thick, echogenic walls and continuous flow (Figure 4). The study also showed chronic, nonocclusive DVT in the left external iliac vein and numerous venous collaterals throughout the abdomen. Based on the findings, the patient was diagnosed with MTS and placed on baby aspirin to treat the acute DVT. A computed tomography venogram later confirmed the diagnosis. The patient was treated successfully with LCIV stenting and angioplasty of the iliac vein. Two weeks later, a follow-up sonogram showed a patent LCIV stent that measured 8 mm with phasic flow (Figures 5 and 6), confirming the alleviation of extrinsic compression. Upon physical examination, the patient demonstrated symptomatic improvement.
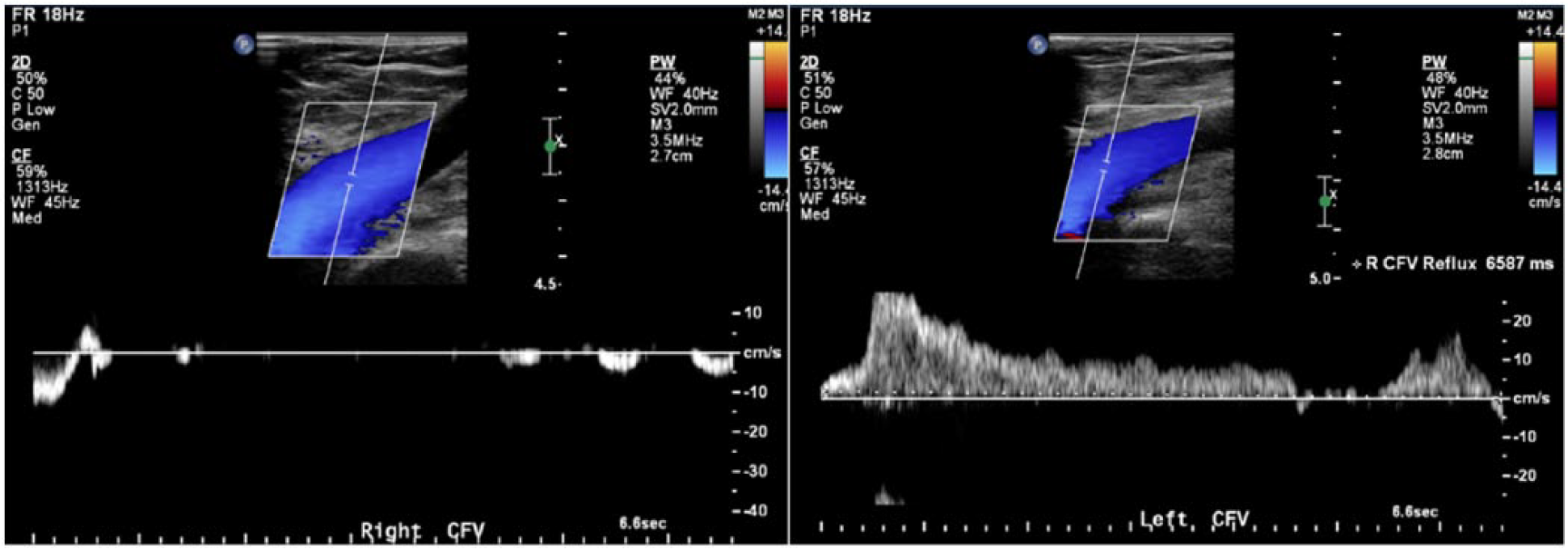
The right common femoral vein (CFV) does not have venous reflux, whereas the left CFV has severe reflux (6587 ms) caused by hemodynamic changes in venous pressure due to extrinsic compression of the left common iliac vein by the right common iliac artery.
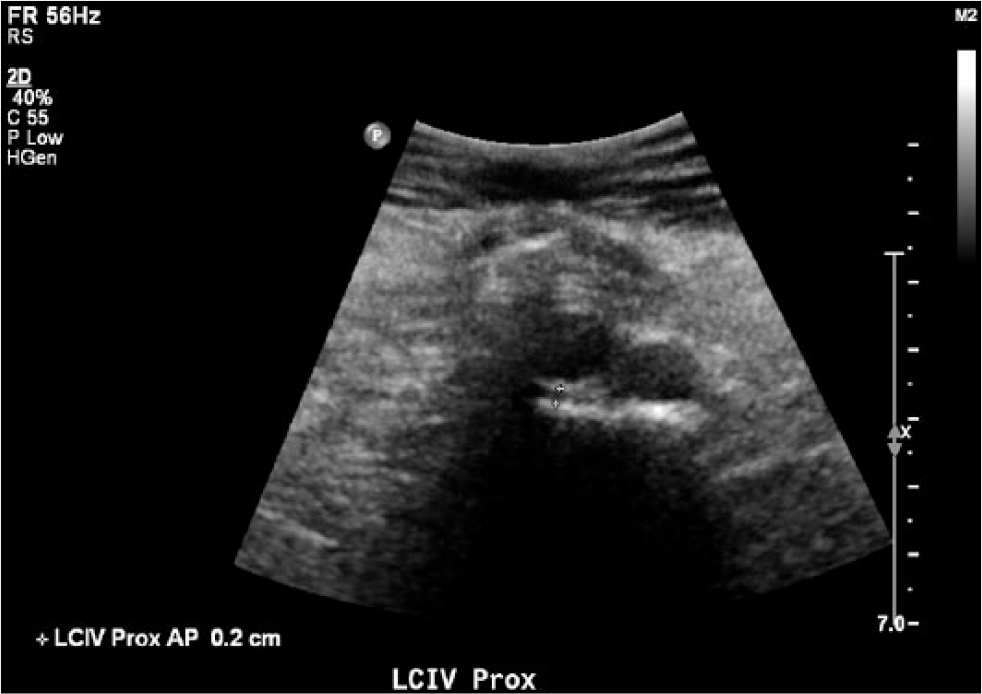
The left common iliac vein has a small caliper of 2 mm with thick, echogenic walls.
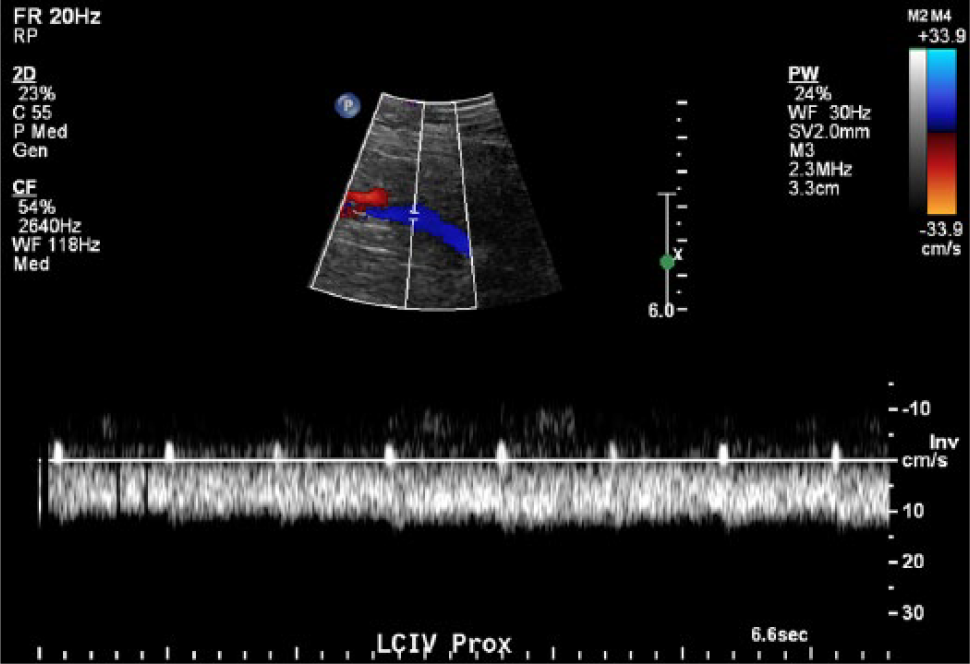
Continuous flow is noted in the left common iliac vein, distal to the site of compression.
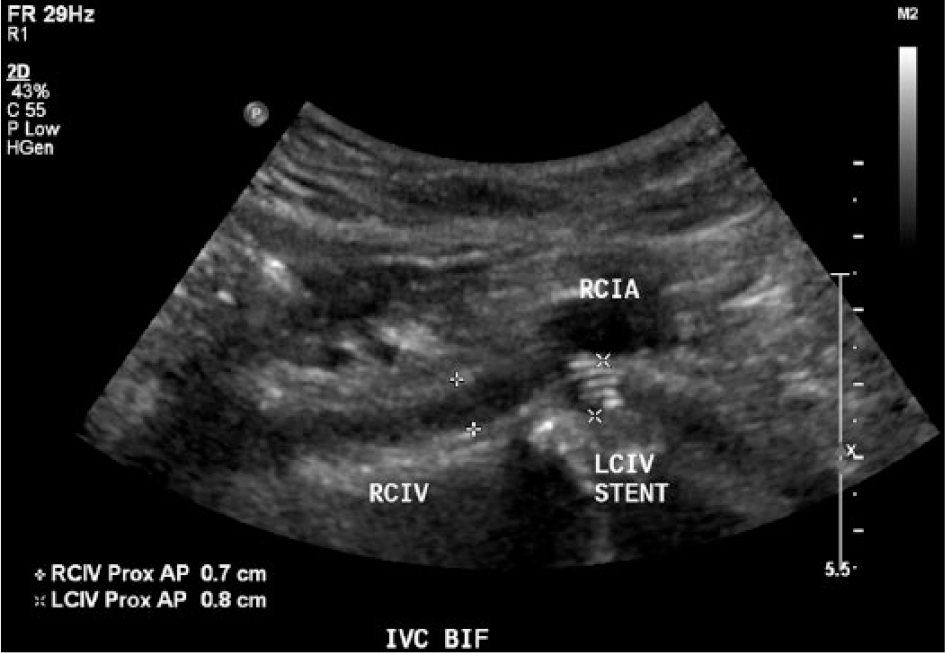
After stent placement, the left common iliac vein (LCIV) measures 8 mm. RCIA, right common iliac artery; RCIV, right common iliac vein.
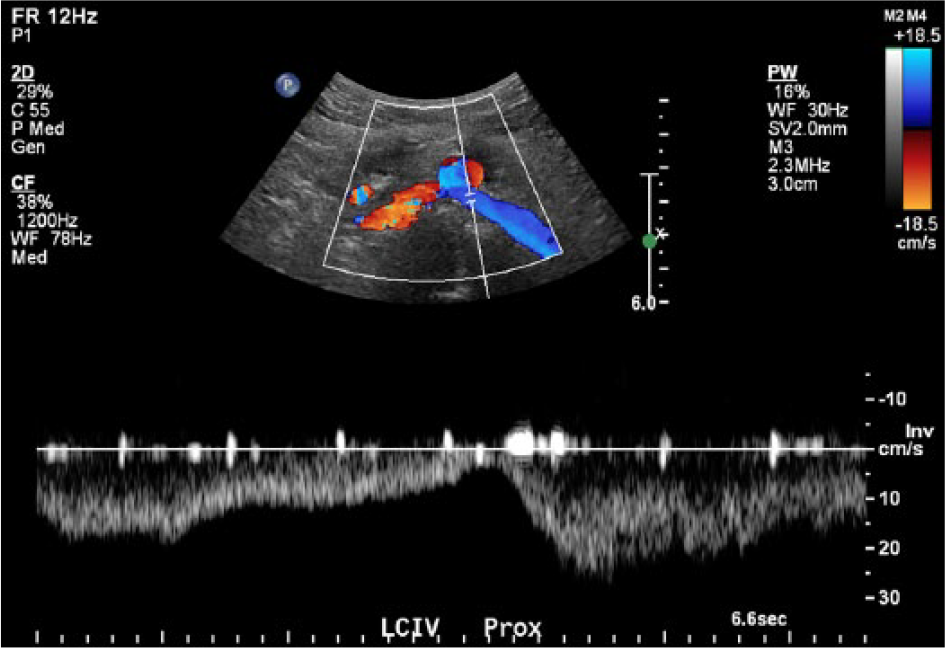
After stent placement, the left common iliac vein has normal phasic flow.
Discussion
Anatomic evidence of MTS was first noted in 1851 by Virchow when he observed that iliofemoral DVT was 5 times more likely to occur in the left leg than in the right leg due to compression of the left iliac vein between the right iliac artery and fifth lumbar vertebrae. 1 In 1908, McMurrich examined 107 cadavers and noticed that 29.9% had obstruction in the left iliac vein that was responsible for the increased incidence of LLE DVT. He believed that this obstruction was congenital in origin. 2 However, it was realized that these obstructions were acquired rather than congenital in 1943 when Ehrich and Krumbhaar discovered that these endovenous lesions were made of elastin and collagen and increased with age. Their research revealed that on 127 autopsied newborns and children, only 4.7% had endovenous anomalies, compared to 33% of autopsied adults. 3 It was not until 1957 that May and Thurner developed a comprehensive understanding of the anatomic variants of MTS. They discovered that 22% of 430 cadavers exhibited lesions described as “spurs” in the LCIV and were predicated to arise from the chronic compression of the LCIV by the RCIA. The combination of chronic extrinsic compression along with pulsatile vibratory pressure from the overlying iliac artery causes repetitive microtrauma and, therefore, endothelial injury to the vein. 4 This endothelial damage leads to the deposition of elastin and collagen, which leads to intraluminal webs, channels, and spurs over time. These spurs can have several morphologic features, which cause a predisposition to DVT. 5 May and Thurner classified these spurs into three categories based on location and size: central, lateral, or resulting in partial obliteration. Central spurs occur on the anteroposterior plane and split the vessel lumen in two. Lateral spurs occur along the sides of the LCIV. Partial obliteration results in the lumen being covered in a reticulation of spurs and results in decreased venous flow. 2 This decreased venous flow can thus lead to thrombosis in the LCIV and/or LLE. In 1965, Cockett et al. 6 expounded on May and Thurner’s research when they determined that patients with LCIV spurs could remain asymptomatic for a period of time due to the development of venous collaterals. However, spur formation is an irreversible process, and early diagnosis of MTS is imperative to prevent further complications. 2
Iliac vein compression is a frequent anatomic variant and can be seen in up to 25% of asymptomatic healthy individuals. 5 This compression becomes clinically significant only if it causes hemodynamic changes in venous flow or venous pressure, evident by venous reflux and/or the presence of varices, or if it leads to DVT. 5 Although MTS is an uncommonly diagnosed condition, it has been said to be responsible for one-half to two-thirds of cases with left-side iliac femoral DVT. 7 It is believed that compression from the right common iliac artery (RCIA) leads to thrombosis in the LCIV either by trapping small emboli from thrombus in the distal veins or by de novo thrombus formation at the site of compression. 8 The compression of the LCIV causes the vein to have a narrow pathway, incidentally trapping the emboli formed from the low flow of venous stasis. In fact, MTS reduces the risk of pulmonary embolism since the embolus would have difficulty passing through the narrow pathway. 2 Trapping of emboli from distal thrombosis at the site of compression would explain the occurrence of divergent, noncontiguous thrombosis in the calf and iliofemoral veins, such as in the example of the aforementioned case study. The formation of de novo thrombus at the site of compression is thought to be related to the turbulence and fibrosis of the venous wall at this site. 8 However, MTS does not always produce DVT. When there is external compression of the LCIV producing web-like intrinsic intraluminal lesions but not causing a thrombosis, it is termed nonthrombotic iliac vein lesion. This is a so-called permissive lesion, which is a pathology that remains silent until additional insult or pathology is superimposed. 9 Triggering factors such as trauma, cellulitis, distal thrombosis, and decreased mobility could thus cause more severe and hindering symptoms.
May–Thurner syndrome is more prevalent in women than in men and typically presents in the second to fourth decade of life, especially after a recent surgery, periods of immobility, or pregnancy. 10 A study done at the Northwestern University Feinberg School of Medicine revealed that LCIV compression affects 41.2% (± 3.1%) of women, compared to 27.0% (± 3.0%) of men. 10 This was thought to be because a woman’s pelvis has more of an accentuation of the lumbar lordosis that pushes the lower lumbar vertebrae forward, thus compressing the overlying LCIV against the RCIA. 8 These changes are further accentuated during pregnancy, hence the reason that pregnancy can be a triggering factor that causes venous complications. Patients usually complain of LLE pain and swelling in the acute phases of MTS, whereas varicose veins, skin discoloration, venous claudication, and ulceration around the ankle due to venous stasis are evident in the chronic stages.5,6 Those who have recurring LLE DVT should also be evaluated for MTS.
The gold standard for diagnosing MTS is left common femoral or iliac venography. This confirms the presence of venous obstruction, assesses hemodynamic significance, and allows treatment to be offered in the same setting. 5 Computed tomography venography and magnetic resonance venography can also be used to evaluate the iliac veins. These newer, minimally invasive modalities can detect occult pelvic masses and other causes of extrinsic venous compression as well as provide important information about venous architecture and the extent of thrombus. 11 Although sonography has its limitations, such as overlying bowel gas and a large body habitus, it can also be used as a valuable tool in diagnosing MTS. Most publications state that sonography is useful in detecting a DVT but may be limited in detecting iliac vein compression or spurs. Experienced vascular sonographers who use a proper technique can provide high-quality diagnostic evidence of this syndrome. A lack of respiratory variations and absence of response to Valsalva maneuver in the common femoral vein is a sign of proximal compression or obstruction and should be investigated proximally with transabdominal sonography. 5 It is also prudent to evaluate the contralateral CFV Doppler waveform to determine whether there is a difference in phasicity and respiratory status between the two limbs.
Furthermore, when symptoms of swelling, DVT, and/or venous insufficiency are limited only to the LLE, MTS should be suspected. When evaluating MTS with transabdominal sonography, it is important to measure the LCIV at the exact site of RCIA compression. This is done best in transverse to the abdomen at the bifurcation of the common iliac arteries. Due to the anatomic layout of the abdominal vasculature, even when transverse, the LCIV can be seen longitudinally, traversing underneath the RCIA (Figure 7). Three things are identified sonographically when MTS is present: (1) the LCIV has a small caliper; (2) the LCIV has thickened echogenic walls; and (3) the LCIV has continuous flow. A contralateral comparison of the Doppler waveforms within the CFV/iliac system is strongly suggested by the Intersocietal Accreditation Commision standards (Table 1). In severe cases of MTS and in cases of iliac DVT, as seen in the presented case study, continuous flow can be identified in the ipsilateral CFV. However, this is not evident in all cases of MTS, and CFV phasicity should not discourage sonographers from investigating further if iliac compression is suspected.
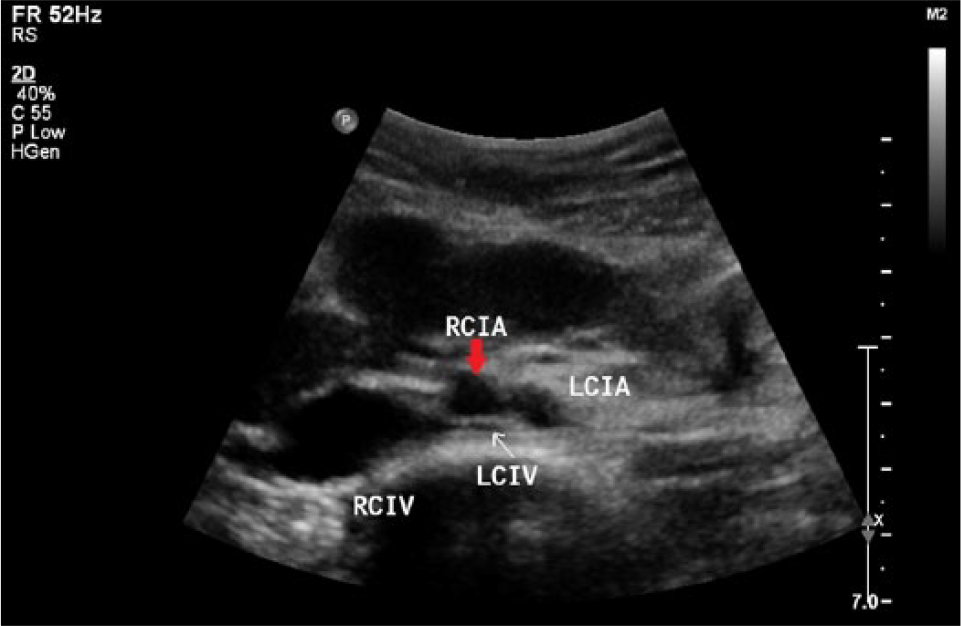
When transverse on the abdomen, the right and left common iliac veins (RCIV and LCIV, respectively) will appear longitudinal. The LCIV (white arrow) can be seen traversing under the right common iliac artery (RCIA) (red arrow). This is an example of extrinsic compression evident by thick, echogenic walls and small caliper of the LCIV. LCIA, left common iliac artery.
Sonographic Signs of May–Thurner Syndrome.

CFV, common femoral vein.
Some compression of the LCIV by the RCIA is considered normal and results in the diameter of the LCIV measuring smaller than the RCIV. It is unfortunate that there currently is no normal measurement for the smallest diameter of the LCIV. A study by Stanford University School of Medicine discovered that patients with DVT had a mean minimum LCIV diameter of 4.0 mm, compared to 6.5 mm for patients without a DVT. The odds of LLE DVT increased by a factor of 1.68 for each millimeter decrease in the LCIV diameter. 12 It is also suggested that if the LCIV measures < 50% of the RCIV diameter, MTS can be suspected. Another factor noted with MTS is thickened, echogenic walls of the LCIV at the site of compression. This marked thickening of the venous wall likely represents the spur, which consists of intimal hyperplasia, fibrous tissue, and a possible organized thrombus. However, the most important factor in diagnosing MTS with sonography is with Doppler analysis. A normal waveform in the iliac veins is phasic with respiratory variations. When there is severe compression of the iliac vein, the waveform becomes continuous distal to the site of compression. When these three factors are identified, MTS should be considered and further evaluated with computed tomography venography or magnetic resonance venography.
May–Thurner syndrome should be treated only when it is symptomatic. In the presence of both iliac compression and superficial or deep venous reflux, the course of treatment should be to correct the obstructive component first. 13 Endovenous stenting of the LCIV is the method of choice in treating MTS. This allows relief of the extrinsic compression and expands the narrowed portion of the iliac vein. This form of treatment has low morbidity, no mortality, a long-term high patency rate, and a low rate of in-stent restenosis. 13 It would be advised to record a Doppler waveform prior to the iliac stent procedure demonstrating continuous flow to compare the poststent Doppler waveform confirming procedural success.
Anticoagulation with warfarin is initiated in patients who present with acute DVT and MTS and is continued for a specified duration. 10 Interventional treatment of outflow obstruction in the form of angioplasty and stenting has been shown to relieve symptoms and improve quality of life, whether or not an adjunct procedure to control superficial reflux is performed.14–16 However, when there is evidence of significant superficial reflux, obliterating the great saphenous vein with endovenous radiofrequency ablation or endovenous laser therapy is recommended to further alleviate symptoms. As May and Thurner discovered, spur formation is an irreversible process; therefore, early detection and treatment are imperative to prevent further complications. 2 Studies have shown that early thrombus removal and decreasing the incidence of recurrent thrombotic events can reduce the likelihood of developing postthrombotic syndrome. 17
Conclusion
May–Thurner syndrome is a condition that can cause severe swelling, pain, venous insufficiency, and DVT in the LLE due to compression of the LCIV by the RCIA. Although numerous articles state that sonography cannot detect iliac compression or spur formation, transabdominal sonography should be considered a very beneficial tool in diagnosing MTS. Recognizing the common symptoms and evaluating the LCIV for the three signs of MTS to include a contralateral CFV waveform for comparison (Table 1) can accelerate early treatment and help reduce the development of recurrent DVT and postthrombotic syndrome.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
