Abstract
The purpose of the Rex shunt is to restore normal blood flow to a hemodynamically compromised liver that is otherwise normal. It has proven to be an effective treatment for children with extrahepatic portal vein thrombosis as in the case presented. The shunt allows blood from the superior mesenteric vein to bypass the obstructed extrahepatic portal vein and enter the liver through the still patent left portal vein. The operation is successful in more than 90% of patients. The name “Rex shunt” is derived from the location in the liver in which the shunt is placed, the area between segments three and four of the left lobe of the liver.
Introduction
The purpose of the Rex shunt is to restore normal blood flow to a hemodynamically compromised liver that is otherwise normal. The role of the sonographer in overall patient management in such cases is an important one. Preoperative sonography is used to confirm cavernous transformation of the main portal vein and to demonstrate a still patent left portal vein. Postoperative sonography allows direct evaluation and comparison with prior examinations to detect any change in the status of the shunt. An understanding of the anatomy of the surgery performed and the Doppler characteristics of the resulting hemodynamic changes is vital for postoperative evaluation because the portal venous flow will have a new route and will now enter the liver above the typical location of the porta hepatis.
Case Report
A 2-year-old boy was referred to pediatric surgery with a diagnosis of portal hypertension, extrahepatic portal vein thrombosis (EHPVT), splenomegaly, and hematemesis. Both computed tomography (CT) and sonographic imaging confirmed cavernous transformation of the portal vein and marked splenomegaly with a patent left portal vein (Figures 1 and 2). After further extensive workup, it was determined that the patient would likely benefit from a Rex shunt, from the superior mesenteric vein to the left portal vein, to attempt to restore normal blood flow to the liver and normal pressures to the mesenteric venous system.
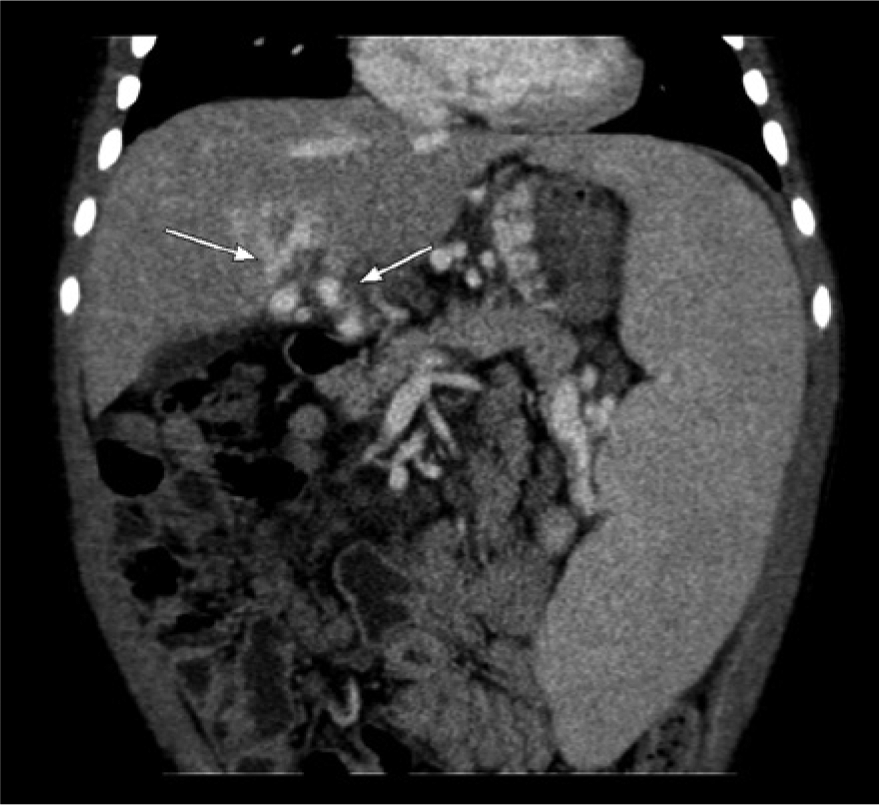
Computed tomographic scan of the patient with extrahepatic portal vein thrombosis shows cavernous transformation of the portal vein (arrows). Also note the markedly enlarged spleen.
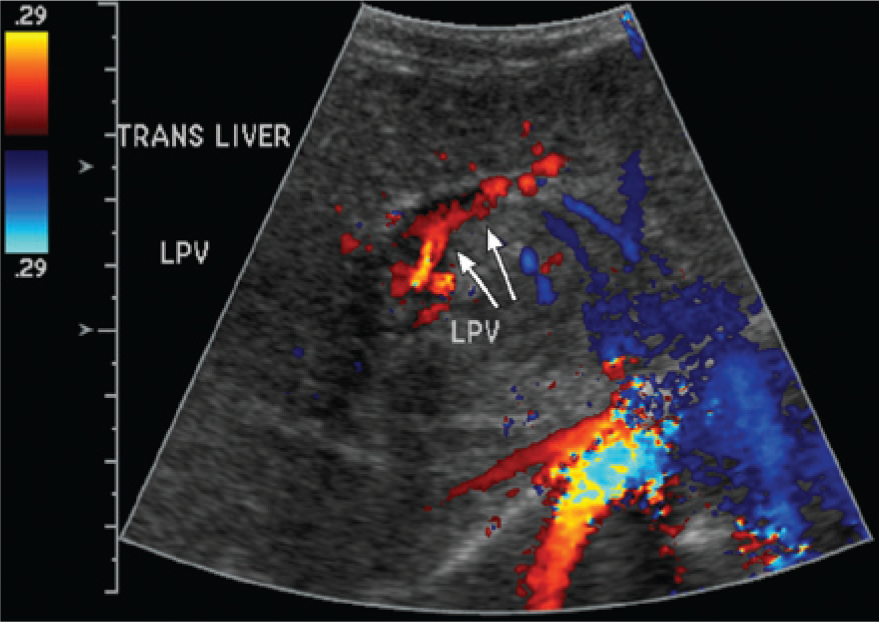
Preoperative color Doppler imaging demonstrates flow in the still patent left portal vein.
The morning following surgery, a portable abdominal Doppler sonogram was ordered by the pediatric intensive care unit to assess the patency of the shunt. Of primary importance was confirmation of the presence and velocity of blood flow throughout the shunt in the immediate postoperative period to determine the anatomic and hemodynamic success of the procedure.
Multiple longitudinal and transverse gray-scale and Doppler images were taken using a GE LOGIQ E9 (GE Ultrasound, Milwaukee, WI) with both a linear 9LD and curvilinear C1-5D transducer, with the patient in a supine position. The liver was noted to be normal in size and contour, but with slightly heterogeneous echogenicity. Doppler and gray-scale interrogation at the liver hilum showed multiple small torturous vessels consistent with cavernous transformation/collateral vessels of the portal vein (Figures 3 and 4). Normal direction of flow was confirmed in the splenic vessels, hepatic veins, and hepatic artery. The spleen remained markedly enlarged for the patient’s age, measuring 14.7 cm (normal for age is 7.6 cm). 1
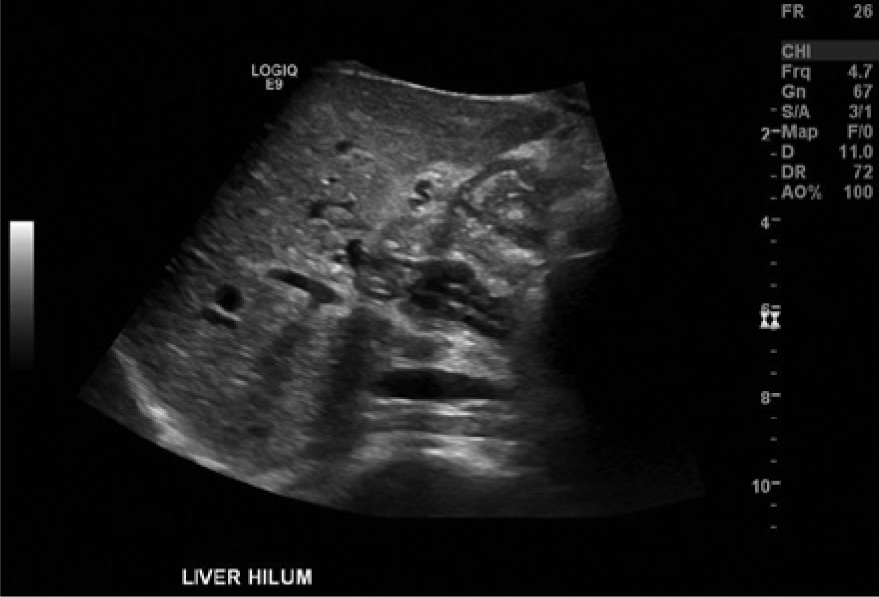
Gray-scale imaging at the hilum of the liver shows multiple collateral vessels consistent with cavernous transformation of the portal vein.
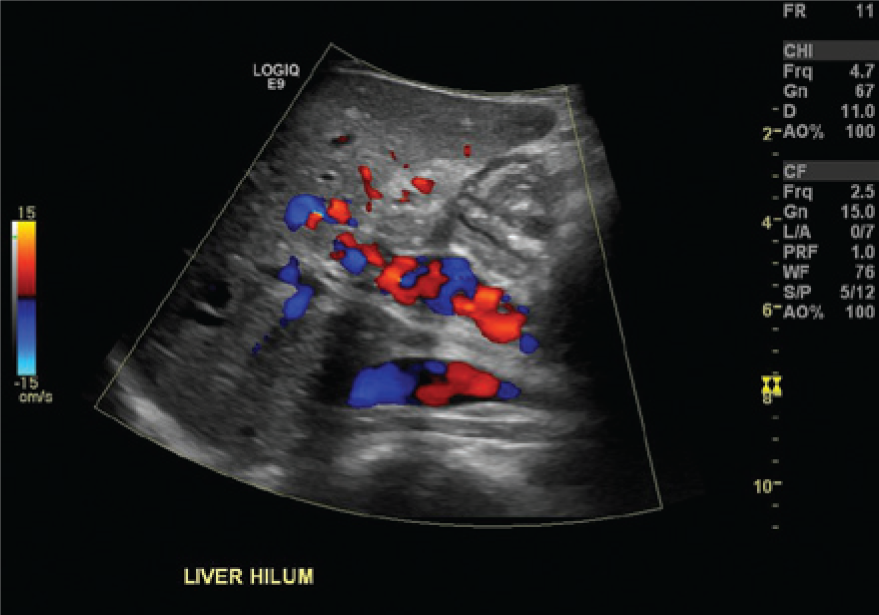
Color Doppler imaging at the hilum of the liver confirms the finding of cavernous transformation of the portal vein.
The Rex shunt was visualized within the Rex recess (left portal branch) of the liver (Figures 5 and 6). Beginning in an oblique plane near the left lobe of the liver, images of the shunt were acquired by first locating the left portal vein where the flow direction has been purposely reversed to deliver blood to the intrahepatic portal system. Next, by tracing the left portal vein slightly superior and medial, a surgical anastomosis near the edge of the liver could be identified. Finally, while continuing to scan out of the liver, the extrahepatic portion of the shunt could be followed as distally as could be appreciated. Diameter measurements were obtained in five locations: near the connection to the superior mesenteric vein, the mid portion of the shunt, near the connection to the left portal vein, at the left portal vein anastomosis, and in the intrahepatic left portal vein. Hepatopedal flow was demonstrated throughout the shunt (Figure 7), and flow within the left portal vein was now appropriately reversed (Figure 8). A distinct narrowing along with an accelerated velocity of 174.9 cm/s was noted at the anastomosis (Figure 9), while velocities within the mid portion of the shunt and in the left portal vein were noted to be 28.6 cm/s and 124.7 cm/s, respectively. Despite the narrowing at the hepatic anastomosis, sonography confirmed that the shunt was patent and that blood flow was moving in the intended direction(s).
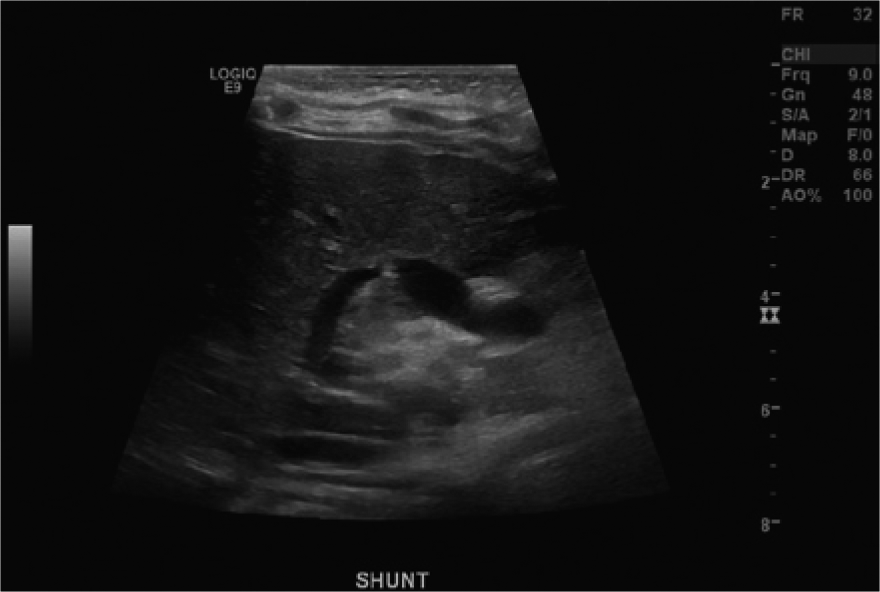
Gray-scale imaging of the Rex shunt. Note the area of narrowing at the anastomosis to the left portal vein at the center of the image.
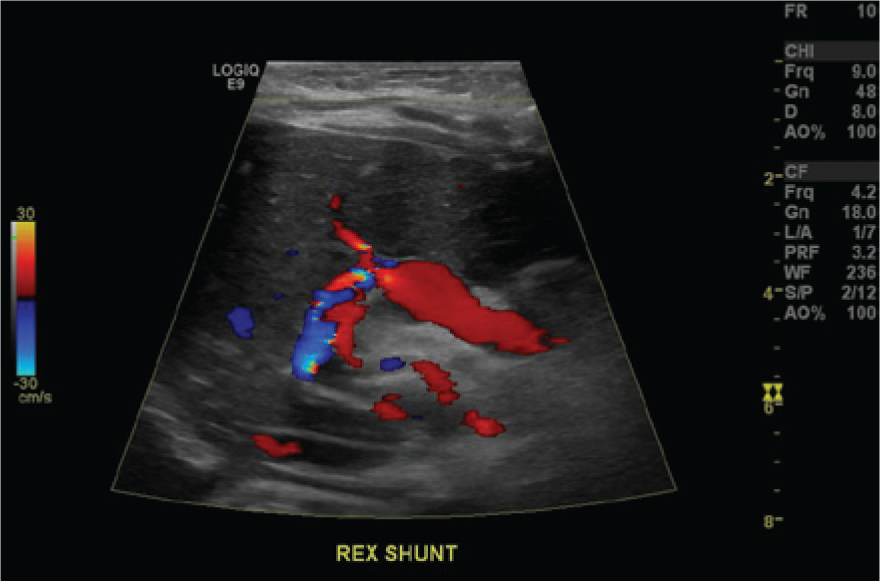
Corresponding color Doppler image of the patent Rex shunt shows disturbed flows at the anastomosis to the left portal vein.
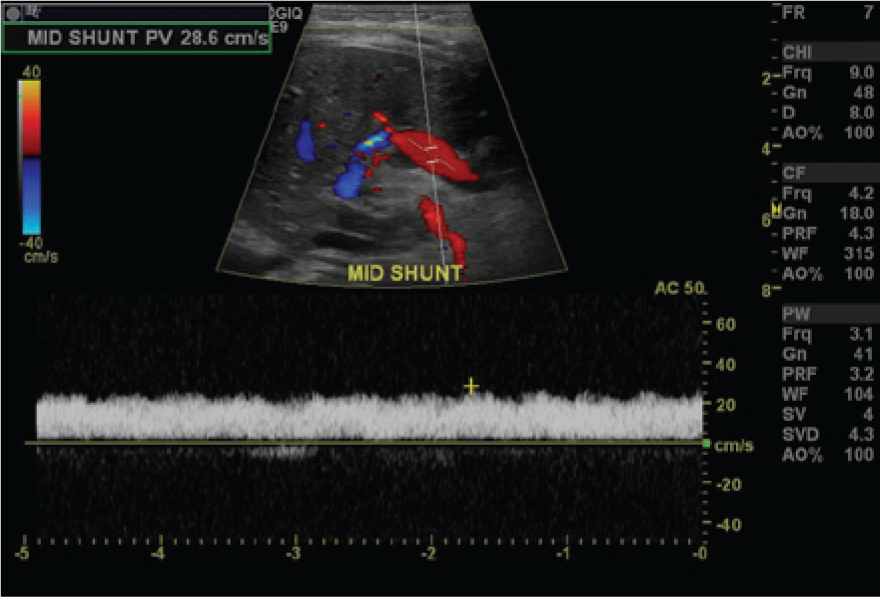
Color Doppler image and spectral Doppler flow signals taken from the mid portion of the Rex shunt show hepatopedal flow.
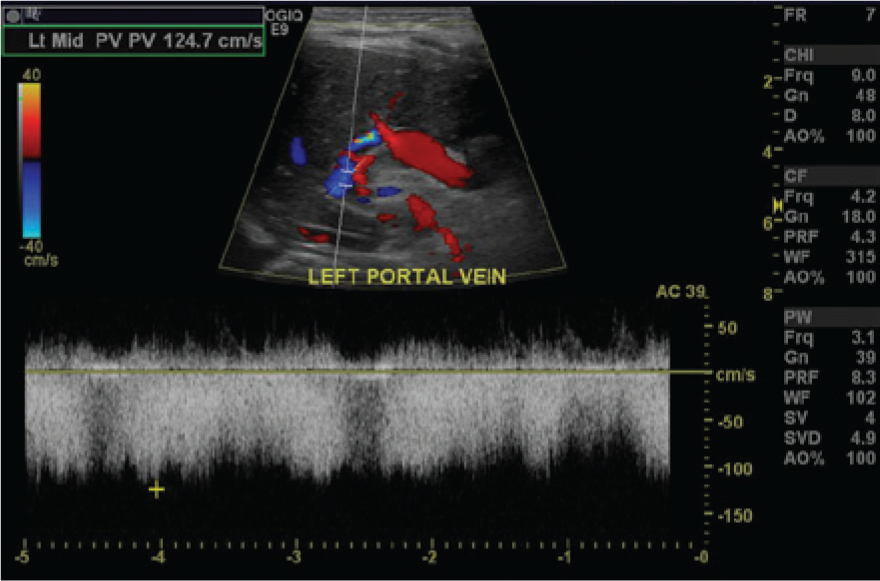
Color Doppler image and spectral Doppler flow signals show reversed flow in the left portal vein delivering blood from the Rex shunt into the liver.
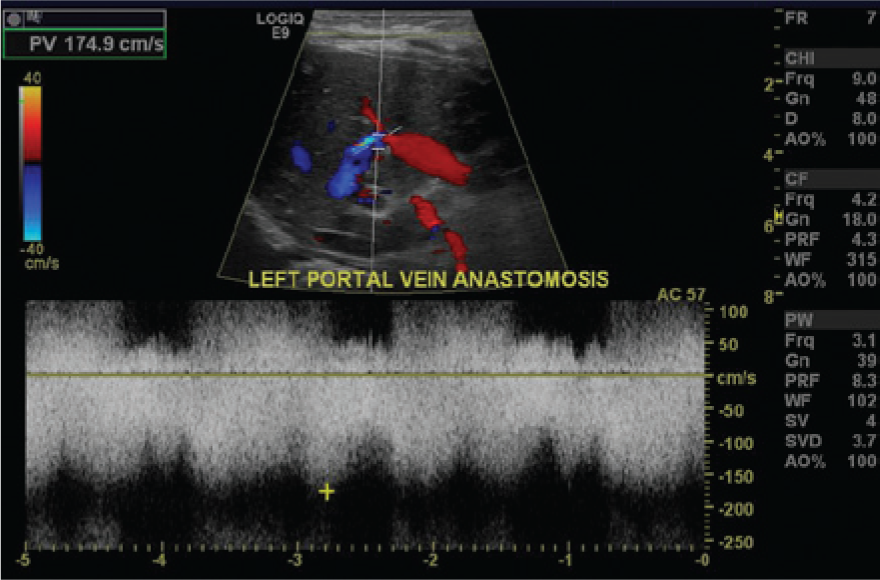
Spectral Doppler flow signal at the anastomosis of the Rex shunt to the left portal vein shows significantly increased velocities and associated flow disturbances characteristic of anastomotic narrowing.
Discussion
EHPVT and Portal Hypertension
The majority of pediatric Rex shunt recipients are completely healthy prior to the sudden onset of symptoms. 2 More than 50% of children with EHPVT will present with hematemesis from gastroesophageal varices or splenomegaly of unknown origin. 2 Their liver function tests and liver histology are typically unremarkable, and the architecture of the portal vein is preserved.2–4
Extrahepatic portal vein thrombosis is typically diagnosed between the ages of 2 and 4 years. 5 Very often, EHPVT in children is idiopathic. 3 There are several conditions, however, that will decrease portal vein flow velocity and lead to stasis. These conditions include umbilical vein catheterization, dehydration, shock, sepsis, chemotherapy, malignancy, hereditary thrombophiliac disorders, liver transplantation, and splenectomy.1–3
Once the EHPVT reaches a chronic state, small periportal collateral veins begin to form. 1 These small channels, which function to increase flow to the liver, are called “cavernous transformation of the portal vein.” 1 Extrahepatic portal vein thrombosis will ultimately cause extrahepatic portal hypertension and result in these cavernous transformations. 3 Almost all patients with portal hypertension also will develop dilated submucosal veins (varices) involving the esophagus, stomach, or spleen, and most of these patients will present with episodes of variceal bleeding and hematemesis.2–3 The bleeding commonly will originate in the lower esophagus and cardia of the stomach and will begin when there is an erosion of the thin variceal walls in combination with the increased venous pressure. 2
The Rex Shunt
According to current surgical guidelines, any child with EHPVT should be evaluated for a Rex shunt.3–4 In order to be an appropriate candidate, however, several parameters must be met. The patient must have an absence of intrinsic liver disease, a patent left portal vein measuring 2 mm or larger in diameter, and patent mesenteric and jugular veins.3–5 The purpose of the Rex shunt surgery is to restore normal blood flow to an otherwise normal liver. 5 Typically, the jugular vein is harvested from the neck and used as a conduit to anastomose the superior mesenteric vein to the left portal vein, bypassing the blocked portal vein. 3 Hemodynamically, this will cause reversal of flow in the left portal vein, bringing blood into the liver. The Rex recess (left portal vein system) is an ideal location for the placement of the shunt because rarely will it be involved with cavernous transformations and collaterals. 3
After placement of the shunt, symptoms of portal hypertension usually will resolve quickly.4–5 Flow will increase to the liver, branches of the portal system will begin to expand, the size and complexity of the cavernous transformations will decrease, and the overall liver volume will increase to 1.3 to 1.7 times the baseline. 3 Additionally, splenomegaly will decrease.7–9
Alternative treatments for EHPVT include distal splenorenal shunts, mesocaval shunts, sclerotherapy, variceal banding, serial endoscopic procedures to control varices, and splenectomy.6–10 These options will relieve only the symptoms of portal hypertension and do not have any lasting hemodynamic or metabolic benefits. 7 In comparison, the Rex shunt will decrease splenomegaly and ascites, restore overall normal portal flow, normalize liver function, and improve platelet count, liver atrophy, encephalopathy, and any associated cholestasis.7,9
On occasion, stenosis of the shunt either within the conduit or at an anastomosis is possible and will require intervention such as surgical revision, balloon venoplasty, and stent placement. 10 This is particularly true if the patient experiences any recurrence of symptoms such as variceal bleeding, a decrease in platelet count, and an enlarging spleen. The current CT criterion for stenosis for which intervention should be considered is a focal narrowing in the shunt measuring less than 3 mm in diameter. 10 Sonographic findings of stenosis will demonstrate a visible narrowing at the stenotic site with increased velocities and a jet by Doppler interrogation, similar to that of the case study. An advantage of sonography is the ability to evaluate the shunt in the very early postoperative period and closely follow with serial examinations any suspected sites of narrowing.
Conclusion
Sonography plays an important role in the pre- and postsurgical evaluation of patients who are candidates for and receive a Rex shunt. It has the advantages of being portable, cost effective, and suitable for serial evaluations, and it can provide an early indication if there is any concern for a narrowing or stenosis of the shunt that might lead to thrombosis and failure. Additionally, compared to CT, sonography uses no ionizing radiation and when performed properly can yield comparable results. Sonographers do need to be familiar with the location of the Rex shunt and have a general understanding of the surgical procedure as well as the expected hemodynamic changes. Documentation must be made of gray-scale measurements and blood velocities not only in the shunt but also at the anastomoses and within the now-reversed left portal vein. Close attention should be paid to any narrowing at the anastomosis and for any increase in velocity along the course of the shunt. Typically, patients will return to the department for serial sonographic examinations immediately following surgery, at 2 weeks, 1 month, 3 months, 6 months, and 1 year, and at least annually thereafter. Consistency in scanning techniques is imperative as the gray-scale diameter measurements and velocities throughout the shunt are always compared to prior imaging studies.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
