Abstract
Objectives:
Embolization of large portosystemic shunts effectively controls gastric variceal bleeding and prevents hepatic encephalopathy. The significance of dynamic changes in hepatic venous pressure gradient before and after embolization on clinical events and patient outcomes remains unknown.
Methods:
In this retrospective single-center series, 46 patients with gastric variceal bleeding, hepatic encephalopathy, or both undergoing embolization (January 2018 to October 2020) were included, and dynamic changes in portal pressures were analyzed against patient outcomes.
Results:
Males predominated. The most common portosystemic shunt syndrome was the lienorenal shunt. In all, 34 patients underwent embolization for hepatic encephalopathy and 11 for gastric variceal bleeding. The proportion of patients surviving at the end of 12 and 32 months was 86.96 and 54.35%, respectively. The hepatic venous pressure gradient before shunt embolization was 13.4 ± 3.2 and 16.9 ± 3.7 mm Hg after occlusion (
Conclusion:
Close monitoring for the development of infections and optimization of beta-blockers and diuretics after shunt embolization may improve clinical outcomes and help identify patients who will benefit from liver transplantation pending prospective validation.
Introduction
Portosystemic shunt syndrome (PSS) in cirrhosis is a distinct clinical entity in the natural history of the disease. Large portosystemic shunts (PSs) are associated with bleeding from gastric varices (GVB) or spontaneous hepatic encephalopathy (HE). PSs can also lead to recurrent GVB or failure to control bleeding and recurrent (bouts recurring within 6 months or earlier) or persistent HE (persistence of >grade 1), resulting in repeated hospital admissions and compromising the quality of life in affected patients.1,2 The clinical importance of spontaneous PSS (SPSS) in the natural history of cirrhosis was delineated in various studies. Early on, clinical researchers considered SPSS as “decompressing channels” for progressive portal hypertension in patients with advanced cirrhosis. Nonetheless, current evidence demonstrates they are related to worsening liver disease and the severity of portal hypertension. The presence of large shunts on contrast cross-sectional imaging was associated with higher mortality and complications.3,4 Similarly, the total cross-sectional SPSS area, rather than the diameter of the single largest shunt, predicted negative outcomes, including the development of HE or mortality in patients with advanced chronic liver disease, even among those with preserved liver functions. 5 In multiple good-quality replicated studies, the embolization of large PS in cirrhosis was shown to effectively control GVB and prevent and reduce the frequency of overt HE. Furthermore, anecdotal studies have demonstrated that early occlusion of large PS was more beneficial than none or late embolization in cirrhosis with recurrent or refractory HE.6–8 Shunt embolization is generally considered a safe procedure with self-limiting local complications. Nevertheless, some authors have highlighted the potential for worsening portal hypertension and aggravation of the variceal disease. By contrast, some authors have also shown that an increase in portal pressures, as reflected by the elevation of the post-procedure hepatic venous pressure gradient (HVPG), was associated with improved liver function. Tanihata et al. 9 showed that balloon-occluded retrograde transvenous obliteration (BRTO) of gastric varices aggravated esophageal varices. This was related to increased portal pressure systemic gradient >5 mm Hg after shunt embolization. Uehara et al., 10 in their controversial study, showed that an HVPG increase of >20% from baseline was predictive of improved liver function after shunt embolization. Contrary to these findings, Park et al. 11 showed that embolization of large PS resulted in an increase in HVPG with a significant risk of symptomatic esophageal variceal progression. The liver function improved over the 6-month follow-up period.
The limited number of small cohort studies have only looked at HVPG-related changes and portal hypertension complications, specifically variceal disease-related events after shunt embolization. There are no studies in the published literature that assess clinical outcomes in the context of pre- and post-shunt embolization HVPG changes concerning liver (portal hypertension events such as GVB, ascites and HE, shunt recurrence, and hepatocellular carcinoma (HCC)) and non-liver related events (sepsis, short term, and overall mortality) among patients with cirrhosis who undergo shunt embolization for GVB, HE, or both.
Methods
Patients
We included all patients aged 18 years and above with a diagnosis of cirrhosis and portal hypertension from our tertiary liver disease treatment center at Kochi, Kerala, India, who underwent shunt embolization for recurrent or persistent HE, recurrent or failure to control GVB, or both in whom HVPG measurement was performed before and after the procedure at a pre-specified time, based on a strict protocol. Patients who underwent embolization but did not consent to HVPG measurement per protocol were excluded from the current analysis. All included patients completed the required minimum follow-up of 1 year and a maximum of 32 months from the procedure or until death or liver transplantation, whichever came first. Patients who underwent shunt embolization and transjugular intrahepatic portosystemic shunt (TIPS) placement were also excluded from the analysis. Patients with shunt-related HE or variceal bleeding who were already treated for liver cancer or had liver cancer during the diagnosis of shunt syndrome were not considered for inclusion in the analysis. We excluded patients with shunt syndrome from undergoing shunt embolization if they were additionally diagnosed with the following—acute on chronic liver failure, recurrent or refractory ascites requiring paracentesis, uncontrolled sepsis, refractory septic or hypovolemic shock, multiple organ failure, main portal vein trunk thrombosis (malignant or non-malignant) or multiple shunts with high-risk technical challenges, and those willing for liver transplantation.
Statistical analysis
Statistical analysis was performed using MedCalc Statistical Software (Ostend, Belgium). Data are presented as mean and standard deviation or as median and range between brackets. One-way analysis of variance was used to test for differences at baseline between the means of investigational variables of groups.
Primary and secondary outcome measures
The HVPG (in mm Hg) before and after shunt embolization was measured in all patients as per standard and validated protocol.9,10 Elevation in HVPG from baseline and percentage change in HVPG post-shunt embolization were calculated in all included patients. In addition, patients were grouped according to median HVPG pre- and post-shunt embolization, elevation in HVPG, and percentage change in HVPG for comparisons. We aimed to study various clinical events in patients with cirrhosis who underwent shunt embolization procedures in the context of HVPG-related measures. The primary outcome was survival at the end of 1 year after the procedure. The secondary outcome measures included survival at 32 months (beyond 2 years), progression of esophageal varices, infections requiring admission within 100 days and beyond 12 months post-procedure, and incident development of ascites, HE, or HCC post-procedure.
Procedures
All procedures were performed under local anesthesia. After written informed consent was obtained, the right common femoral vein or internal jugular vein was accessed, and a 6F vascular access sheath (Cook, Bloomington, Indiana) was placed. HVPG was then measured using the standard balloon wedge technique. Subsequently, the PS was cannulated using a combination of a 5F angiographic catheter (multipurpose or cobra; Cook) and 0.035″ hydrophilic J-tipped guidewire (Radifocus; Terumo, Japan). The catheter was then replaced with a flexible curved sheath (Flexor Check-Flo Introducer with large valve assembly, Mullin design, and Ansel modification, 7F-14F; Cook) over a 260 cm, 0.035″ Amplatz ultra-stiff guidewire (Cook, Inc). Venography was performed through the sheath to delineate the shunt anatomy and confirm the sheath position. The choice of the occluding modality (BRTO, plug-assisted retrograde transvenous occlusion (PARTO), or coil-assisted retrograde transvenous occlusion (CARTO) with or without glue instillation) was decided by the interventional radiologist performing the procedure based on the shunt anatomy on pre-procedural imaging and standardized techniques.6,7 For balloon-assisted shunt occlusion, a compliant oversized balloon catheter (Equaliser, Boston Scientific Corporation, Natick, MA, USA) was placed into the shunt. After excluding the leak of contrast around the balloon, the sclerosing agent prepared by mixing lipiodol, 3% sodium tetradecyl sulfate (STS; SETROL, Samarth Life Sciences, Mumbai, India), and air in a 1:2:3 ratio was infused to fill the full extent of the shunt. The balloon was kept in place for 4 h to cause effective sclerosis and occlusion of the shunt. For plug-assisted shunt occlusion, an amplatzer vascular plug (AVP-II; St. Jude Medical, Inc., St. Paul, MN, USA or Cera-Vascular plug, Lifetech Scientific, Shenzhen, China; 8–24 mm) with a diameter of 30%–50% more than the narrowest accessible diameter of the shunt was deployed while keeping a 2.7 Fr microcatheter (Progreat, Terumo, Japan) into the shunt distal to the plug. A venogram was then performed through the microcatheter to exclude any leak of contrast and to detect any additional efferent channels that needed to be embolized. Subsequently, a sclerosant mixture comprising 3% STS and contrast (320 mg I/ml; Visipaque, GE Healthcare, Ireland) in a 2:1 ratio along with small gel foam pieces was injected under fluoroscopic visualization till complete filling of the shunt was achieved. The plug was deployed, and the sheath and catheters were removed. For coil-assisted shunt occlusion, a similar procedure as described for the plug was followed, except that oversized detachable coils (Interlock-35 Fibered IDC Occlusion System, Boston Scientific, USA) were used as an occluding agent instead of a plug. The HVPG measurement was repeated the next day, within 24 h, after confirming the obliteration of the PS on contrast-enhanced cross-sectional imaging. The study was performed conforming to the Helsinki Declaration of 1975, as revised in 2000 and 2008, concerning human rights, and the study design and retrospective collection of patient data were approved by the institutional review board of the Liver Institute, Rajagiri Hospital (CoEGIS/TLI/RAJH:83/12/2022). The requirement of obtaining consent from a legally authorized representative (in the case of deceased patients) was waived off by the Institutional Review Board/Ethics Committee. This was due to the retrospective pooled analysis nature of the study design which excluded individualistic scrutiny of existing clinical data.
Post-procedure protocol and follow-up
All patients were on optimized beta-blockers (carvedilol or propranolol as per the treating physician’s discretion) before and after shunt embolization. Patients were initiated on a low sodium diet and diuretics if their ascites grade was ⩾2 before or after shunt embolization. Patients who developed symptomatic grade 3 ascites underwent large-volume paracentesis alongside intravenous human albumin infusion cover according to demand. All patients with HE were continued on rifaximin (550 mg twice daily) and oral lactulose (titrated for at least two soft stools per day). Those who developed HE post-shunt embolization were initiated on secondary prophylaxis. None of the patients were on weekly human albumin infusion before or after shunt embolization. All patients underwent upper gastrointestinal endoscopy for variceal disease assessment between 1 and 3 months after the procedure and every 3 and 6 months for the first year and annually thereafter. Contrast computed tomography of the abdomen was performed 24 h after the procedure to assess shunt occlusion and thereafter at 6 months, 1 year, and 2 years. The latest imaging report was utilized to include imaging-based variables in this study. All included patients were followed up for at least 1 year and a maximum of 32 months or death or liver transplantation (both considered a negative outcome), whichever came first.
Results
Patients inclusion
From January 2018 to October 2020, 99 cirrhosis patients with PSS underwent shunt embolization for various indications at our center. Of these, 32 patients underwent shunt embolization with TIPS, and 3 patients treated for prior HCC and diagnosed with HCC during the evaluation of shunt embolization were excluded. Of the remaining 64 patients who underwent shunt embolization only, 9 did not undergo HVPG measurement, 4 had only one HVPG measurement, and 5 had incomplete documentation/follow-up and were excluded from the analysis. Ultimately, 46 patients (

Details of patients included in the study.
Patients characteristics
Males predominated (
Complete and pertinent clinical and investigational details at baseline and follow-up of cirrhotic patients undergoing shunt embolization.
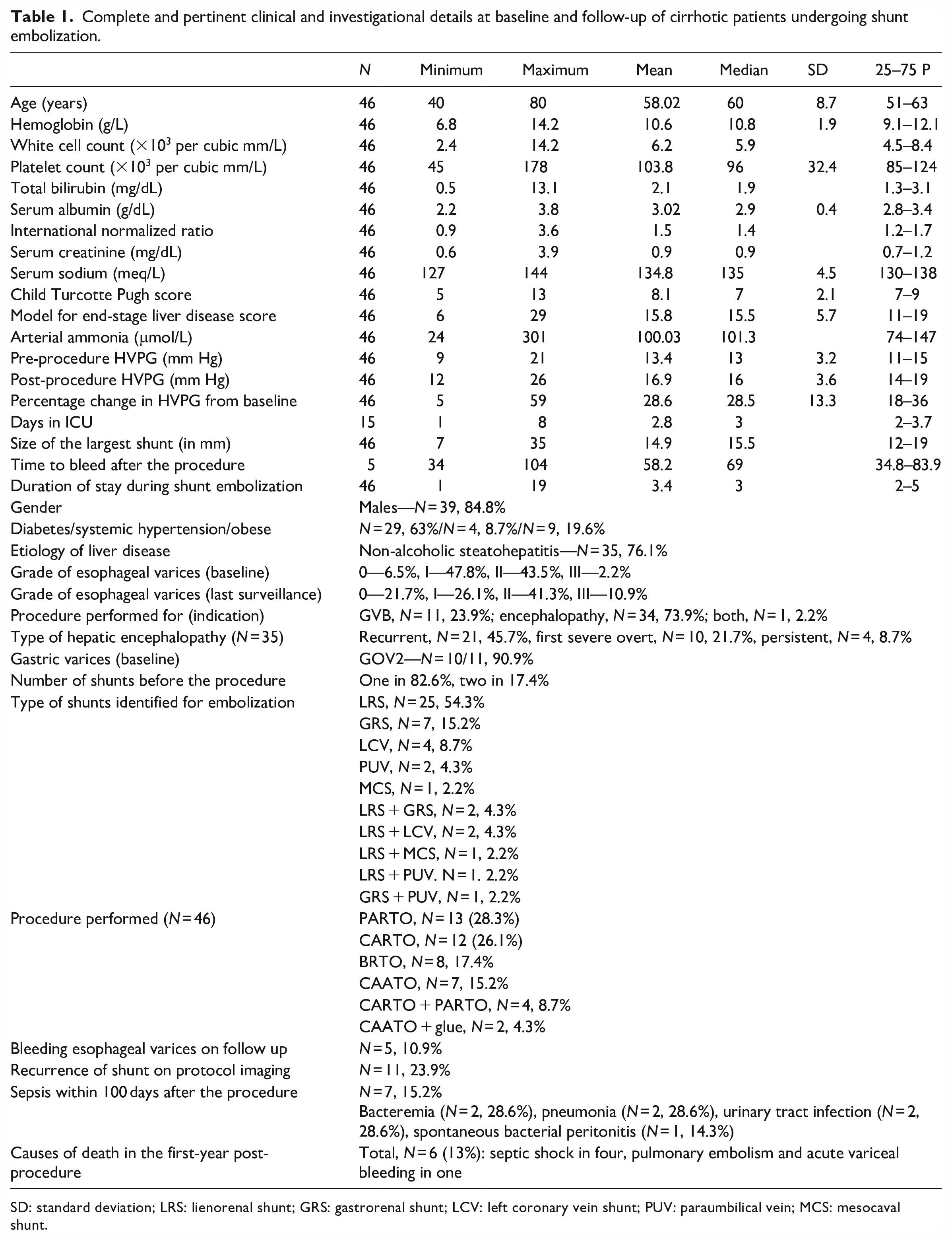
SD: standard deviation; LRS: lienorenal shunt; GRS: gastrorenal shunt; LCV: left coronary vein shunt; PUV: paraumbilical vein; MCS: mesocaval shunt.

Kaplan–Meier analysis shows the proportion of patients surviving at the end of 1 year and long-term follow-up of 32 months after shunt embolization.
Procedure-related characteristics
The PARTO method of shunt embolization was most commonly performed (

The various shunt-embolization methods performed in the patient cohort. Large gastric varices (arrow, a) and post-embolization disappearance of varices (asterisk, b) with the development of ascites (arrow, b); large lienorenal shunt occluded using a plug (arrows, c); balloon-assisted retrograde transvenous occlusion of a large shunt (arrow, d); a large bunch of gastric varices (arrow, e) and associated shunt (asterisk, e) and complete occlusion of shunt and associated obliteration of gastric varices (arrows, f).
The measured HVPG before shunt embolization was 13.4 ± 3.2 mm Hg (median 13, minimum 9, and maximum 21) and 16.9 ± 3.7 mm Hg (16, minimum 12, maximum 26) after occluding the shunt, which was statistically significant (Hodges–Lehmann median difference 3.0,
Portal hypertension-related events
Low-grade (no or grade 1) esophageal varices were notable in 25 patients (
Clinical events and significance related to outcomes
Infections requiring hospital admission within 100 days after shunt embolization were seen in 7 (
Significant HVPG-associated outcomes
The patients were grouped into HVPG ⩽13 mm Hg or >13 mm Hg pre-procedure and HVPG ⩽16 mm Hg or >16 mm Hg post-procedure, based on median value cutoffs. The HVPG >16 mm Hg after shunt embolization was significantly associated with the development of ascites between months 1 and 6 post-procedure (relative risk 1.6 (1.1–2.3),

The significance of pre- and post-procedure portal pressures measured as the HVPG on various clinical, investigational, and patient outcomes.
Discussion
Spontaneous large PSS are associated with HE (spontaneous, recurrent, and persistent) and lower transplant-free survival in patients with cirrhosis—independent of liver function, in the presence of lower MELD scores, and independent of beta-blocker treatment. 3 The embolization of symptomatic large PSS early on in the disease course was associated with improved clinical outcomes in cirrhosis patients regarding better control of bleeding from gastric varices and preventing HE recurrence. 13 Only a few studies on shunt embolization have analyzed clinical and investigational variables associated with outcomes. The MELD score >11–15 and CTP above 11 were associated with negative outcomes, while liver stiffness values below 21.6 kPa before shunt embolization correlated with better survival and lesser adverse events. Similarly, shunt embolization performed early in the disease course was associated with improved transplant-free survival. 14
In the current study, we demonstrate novel findings concerning the association of portal pressures before and after shunt embolization and follow-up clinical outcomes in the short and long term in a large series of patients from a single center. Ours is the largest series on shunt occlusion with serial HVPG measurements, which included various upgraded techniques of shunt embolization. In our study cohort, the 1-year survival was approximately 87%, and nearly 55% of patients survived beyond 2.5 years. The development of infections (sepsis) within 100 days and beyond the first year after embolization was associated with the risk of dying at the end of 12 and 32 months, respectively. The presence of infections requiring hospital admission was also the only independent factor that predicted death in the long term in cirrhotic patients undergoing shunt embolization. The elevation of HVPG by >4 mm Hg from baseline and an absolute increase to >16 mm Hg immediately post-procedure significantly predicted the development of early and late-onset ascites, respectively. An absolute increase in HVPG above 16 mm Hg post-shunt embolization was also associated with a trend toward an increased incidence of primary liver cancer. Nonetheless, the percentage change in HVPG from the baseline did not affect any clinical outcome.
Tanihata and colleagues demonstrated that HVPG elevation >5 mm Hg post-shunt embolization (using only the BRTO technique) significantly aggravated esophageal varices. The presence of varices and higher grades of varices (grade 2 > grade 1) at baseline were the other important factors that predicted the worsening of the variceal disease. But most importantly, this significant aggravation was notable in CTP class B patients. 9 In our study, the proportion of patients having aggravation of variceal disease post-procedure was evident, and 1 in 10 patients in the whole cohort bled on follow-up. This was probably due to two important aspects in our patients—one, the beta-blocker optimization that was homogenously performed before and after shunt embolization, and two, better techniques of shunt embolization (such as PARTO, CARTO with or without glue use) which ensured complete occlusion of the variceal complex and its tributaries, thereby preventing significant clinical event even after aggravated variceal disease.
In the study by Uehara et al., the authors found that an increase in HVPG ⩾20% from baseline improved liver functions. The patients did not have significant HE or bleeding post-shunt embolization using the BRTO technique. However, this study had contradictory findings that were not clearly explained. 10 As per current literature, HVPG is associated with an increased risk of variceal bleeding, ascites, infections, HE, liver disease progression, liver disease severity, and the development of HCC.14,15 Nonetheless, robust data on an increased risk for HCC development in the context of shunt embolization remain unclear. In the Uehara study, the authors showcase contradictory findings in their patients concerning HVPG and could have unintentionally failed to control for multiple confounders that affected the study conclusions. In our study, in line with the current literature, we found that HVPG elevation by >4 mm Hg from baseline and absolute increase to above 16 mm Hg was associated with a higher incidence of ascites. A large multicenter retrospective study from Korea showed that only CTP class C was associated with rebleeding after shunt embolization, strengthening the argument that HVPG elevation post-procedure was associated with aggravated portal hypertension events and not amelioration in the severity of the liver disease. 16
Similarly, in the study by Park and colleagues, shunt embolization for GVB using the PARTO technique was associated with 100% technical success and worsening of varices in 53% of patients. 11 The HVPG was significantly associated with esophageal variceal aggravation. Details on optimized beta-blockers and other confounding factors that could have led to variceal disease aggravation and associated events were not discussed. The improvement in liver function after the PARTO procedure lasted only the first 6 months. It was not significantly associated with portal pressures before and after the procedure—showcasing that other core events (such as control of etiological factors) may have been at play post-procedure that improved liver disease severity. 11 In our patient cohort, intermediate and long-term outcomes were determined by the development of infections post-shunt embolization. A previous study on shunt embolization in HE demonstrated that a CTP score above 11 was associated with higher mortality in cirrhotics undergoing shunt occlusion. Nonetheless, in our study, statistical significance was probably not reached, given the inclusion of cirrhosis patients with a lesser degree of disease severity. 17
A retrospective review of shunt-embolized patients from the Royal Free Hospital, London, by Privitera et al., revealed that patients who did not improve post-procedure did not do so due to sepsis, 20% developed severe ascites, and approximately 8% developed variceal bleeding. The authors did not find an association between pre-procedural MELD score and outcome, suggesting that a MELD score of >11 may not be an optimal method for stratification, and a substantial proportion of patients undergoing shunt embolization developed portal hypertension-related complications. These findings are in line with our study. Privitera et al. also called for more detailed patient assessment through portal pressure measurement to better define a subgroup at a lower risk of complications. In our study, we analyzed clinical outcomes based on HVPG measurement before and after shunt embolization. 18 We found that absolute increase in HVPG (⩾16 mm Hg), the pertinent elevation of HVPG from baseline (>4 mm Hg), and patients with infections post-procedure were the subgroup who were at risk of negative outcomes, including the development of portal hypertension complications, HCC, and lower transplant-free survival.
Our study has several strengths as well as limitations. This is the largest single-center study that analyzed clinical outcomes in cirrhosis patients undergoing shunt embolization for HE or GVB based on pre- and post-procedure portal pressures. We identified a subgroup of patients developing negative outcomes concerning the baseline and post-procedure HVPG. Based on our findings, pre- and post-shunt embolization assessment could help us identify patients who require close monitoring for the development of infections, assess and control risk factors for infections, optimization of beta-blockers and diuretics to improve control of inadvertent portal hypertension complications, and close surveillance for the development of HCC. Ultimately, in patients with any such risk factors or the development of complications related to those, early liver transplantation can be offered for improved survival (Figure 5, summary infographics).

Summary infographics of the study.
Our retrospective single-center study requires validation from well-designed prospective studies. The realistic influence of beta-blocker therapy and its targeted optimization require further studies with the inclusion of a control group. Stringent well-defined inclusion criteria can help us identify clinical and investigational variables associated with improved liver functions post-shunt occlusion. Due to the retrospective, cross-sectional study design, a sample size calculation was not performed. Finally, the role of different methods of shunt embolization on outcomes needs clarification.
Conclusion
Baseline and post-procedure HVPG and associated dynamic changes in portal pressures in cirrhosis patients undergoing shunt embolization for HE or GVB dictate intermediate and long-term clinical outcomes. The HVPG elevation by >4 mm Hg from baseline and absolute increase in HVPG to ⩾16 mm Hg after the procedure were associated with clinically significant portal hypertension events. Sepsis after shunt embolization was an independent risk factor for post-procedure death in the long term.
Footnotes
Author contributions
Sasidharan Rajesh has been involved in conceptualization, methodology, data curation, writing (review and editing), validation; Cyriac Abby Philips has been involved in conceptualization, visualization, methodology, writing (original draft and review), validation; Rizwan Ahamed has been involved in writing (review and editing), validation, supervision; Shobhit Singh has been involved in data curation, writing (review and editing), validation; Jinsha K Abduljaleel has been involved in writing (review and editing), validation, supervision; Ajit Tharakan has been involved in writing (review and editing), validation, supervision; Philip Augustine has been involved in writing (review and editing), validation, and supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the Institutional Review Board of the Liver Institute, Rajagiri Hospital (CoEGIS/TLI/RAJH:83/12/2022). All authors confirm that they are accountable for all aspects of the work (if applied, including full data access, the integrity of the data, and the accuracy of the data analysis) in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The requirement of obtaining consent from a legally authorized representative (in the case of deceased patients) was waived off by the Institutional Review Board/Ethics Committee.
Trial registration
Not applicable as this is not a prospective randomized trial.
