Abstract
Transjugular intrahepatic portosystemic shunt, or TIPS, is a procedure used to decompress the portal system resulting from portal hypertension. The technique was inadvertently discovered during a transjugular cholangiography procedure around 1969. Technological advances in the 1980s and 1990s have resulted in more positive outcomes for the TIPS procedure since its inception. There are several indications for performing the procedure, including refractory ascites, variceal bleeding, and portal hypertension. Liver disease can lead to portal hypertension, and few treatments are available; however, with TIPS, many patients obtain favorable results. The goal of placing an intrahepatic portosystemic shunt is to bypass the vascular resistance in the cirrhotic liver by creating a channel between the portal and hepatic veins, thereby reducing portal venous pressure and portal hypertension. Normal and diseased liver function is explained as well as the TIPS procedure process, materials, complications, and long-term outcomes.
History
Interventional radiologist Josef Rösch is recognized as the creator of the transjugular intrahepatic portosystemic shunt (TIPS) procedure. In 1969, while performing transjugular cholangiography, he inadvertently initiated the approach. 1 Transjugular cholangiography is a procedure in which a catheter is introduced into the jugular vein and advanced through the superior vena cava, to the right atrium, and directed into the hepatic vein. Using the catheter, a needle is then introduced through the hepatic vein, into the liver, and passed into the bile ducts. The goal is to administer contrast into the biliary system to evaluate patency. This method was believed to be safer for the patient than puncturing the liver capsule because patients often experienced bleeding, ascites, and obstructive jaundice with the percutaneous liver technique. According to Rösch, 1 the procedure often experienced technical difficulties, and the needle would mistakenly puncture into the portal vein. The idea of shunting the portal system was developed and is currently used to treat portal hypertension, ascites, and variceal hemorrhage. 2
Anatomy and Physiology
Normal liver vascularity includes the portal vein transporting blood into the liver from the bowel along with inflow from the hepatic artery. The hepatic veins drain the liver and transport blood into the inferior vena cava. The portal vein is responsible for supplying about two-thirds of the blood flow volume into the liver. 3 In the diseased liver, blood flow into the liver is often compromised due to fibrotic changes within the liver’s parenchyma. The resistance to the movement of blood into the liver increases, which then increases the portal flow resistance. Over a period of time, the portal circulation is altered, and portal hypertension occurs. The resulting effects are debilitating and progressive. Portal hypertension creates a process where blood is shunted away from the liver. This blood is forced into small, thin vessels connected to veins in the esophagus, stomach, and rectal areas. 4 These thin, dilated vessels are called varices, and they often weaken and hemorrhage. Up to 50% of patients with cirrhosis will eventually form esophageal varices. 5 Hepatorenal syndrome, or HRS, can also occur in patients with cirrhosis and ascites. HRS compromises renal function in addition to liver function and has a poor prognosis. The process that results in HRS begins with vasodilatation of the intestinal arteries in response to portal hypertension and cirrhosis. This vasodilatation dramatically decreases blood volume in the arterial system and triggers vasoconstriction. The effect of vasoconstriction severely impairs kidney function, leading to renal failure.3–6 According to Salerno et al, 7 although there are few studies of TIPS procedures performed on patients with HRS, it has been documented that recurrence of HRS after TIPS is rare, provided there is no shunt malfunction. Moreover, TIPS typically reduced ascites in HRS patients. 7
Indications
The most common indications for TIPS include acute or recurrent variceal bleeding that cannot be medically managed and refractory ascites. These indications make up about 99% of the TIPS procedures performed. Less often, the procedure is performed to treat hepatic hydrothorax, hepatorenal, hepatopulmonary, and Budd-Chiari syndromes. It should be noted that TIPS success rates and clinical outcomes are directly related to the severity of liver disease. 8
Cirrhosis results from damage to the hepatocytes and subsequent scarring and regrowth of the liver parenchyma. The appearance and size of the liver varies based on the underlying causes of the damage to the hepatocytes; these variations can range from a large, greenish liver (due to biliary obstruction) to a shrunken, yellowish liver (due to advanced cirrhosis related to alcohol abuse). A cirrhotic liver feels hard and is nodular in its appearance; the nodules are a result of tissue regrowth and the accompanying fibrosis related to scar tissue formation. Liver fibrosis increases the resistance to blood flowing into the liver and can result in portal hypertension. The increased pressure in the portal system results in blood seeking pathways of lower resistance, which can be found in numerous small anastomoses between the portal and systemic venous systems. These collateral pathways (esophageal, splenorenal, umbilical, and rectal) dilate and begin shunting blood from the higher pressure portal system into the lower pressure venous network. 4 Another significant route of collateralization is via a recanalized paraumbilical vein, which presents sonographically as a tubular structure within the ligamentum teres. 9
Liver cirrhosis can create ascites, determined as free fluid within the abdominal cavity that can rapidly increase in volume, causing discomfort, a distended abdomen, and shortness of breath. “Ascites is commonly seen as a major complication of cirrhosis and is associated with renal failure, poor quality of life, increased risk of infections, and an overall poor long-term prognosis.” 8 Ascites is primarily triggered by vasodilatation of the splanchnic arteries. Portal hypertension initiates the release of nitric oxide, which is a vasodilator. As the vasodilator acts, the arterial pressure and blood volume decrease dramatically, triggering the release of vasoconstrictors that will raise the arterial pressure again. The vasoconstriction results in sodium and fluid retention. Excessive fluid is retained in the abdominal cavity, which is referred to as ascites. 8
Variceal hemorrhages occurring in an acute and or recurrent manner led to the initiation of the TIPS procedure. 10 Variceal hemorrhage, which can be life threatening, is common in patients with cirrhosis, especially in severe cases. Up to 33% of patients with cirrhosis will experience some episode of variceal bleeding. Recurrent hemorrhage becomes particularly likely after an initial event, occurring up to 70% of the time. When alternative treatments such as pharmacologic and endoscopic procedures fail to stop variceal bleeding, the TIPS procedure has shown to be effective in treating uncontrolled bleeding in 90% of cases. 11 In a random clinical trial (RCT) performed on 75 patients with liver cirrhosis and variceal bleeding, TIPS was used early in acute hemorrhage for 45 patients and was found to be highly successful at treating the uncontrolled bleeding. 12 “The results of the current surveillance study confirm the ones of the original RCT, clearly showing that the early use of polytetrafluoroethylene (PTFE) covered TIPS markedly and significantly reduces failures to control bleeding, re-bleeding, and improves survival of high-risk cirrhotic patients, admitted for acute variceal bleeding.” 12
According to Grace and Minor 11 , the 12th leading causes of mortality in the United States are chronic liver disease and cirrhosis. There are several different methods to identify cirrhosis, portal hypertension, ascites, and varices. To obtain a definitive diagnosis of cirrhosis, the most reliable procedure is a liver biopsy, which has an accurate rate of 83% to 95%. 13 However, the procedure is invasive and has several risks, including but not limited to pain, bleeding, intrahepatic hematoma, pneumothorax, hemothorax, hemobilia, and pancreatitis. Liver functions can be assessed with routine hematologic testing. Imaging studies including sonography, computerized tomography, and magnetic resonance imaging can reveal information about the liver’s size, texture, density, and vascularity. Sonography is optimal in determining the presence of ascites. Portal hypertension can be assessed with duplex imaging, but there is limited reliability and reproducibility when using peak velocities and resistive indices to quantify portal hypertension. 14 For example, Haag et al 14 reported a sensitivity and specificity of 85% to 95% when using various sonographic parameters to assess portal hypertension. In addition, their research indicated that using sonography to quantify the degree of portal hypertension wasn’t feasible.
Procedure
The TIPS procedure has evolved and matured considerably since the early days of its inception. 1988 marked the first TIPS process in Freiburg, Germany, when seven patients were treated, but the procedure was far from ideal in that two of the patients died soon after the shunts were placed. The TIPS method for treating portal hypertension was still in its infancy and had numerous technical imperfections. For example, early procedures were known to take up to eight hours to complete. Although the method had a promising future, more clinical research was needed to establish a more consistent technique and better outcomes. 15 During the last years of the 20th century, additional research and clinical trials were completed. Post-TIPS complications fostered newer protocols and materials. Early procedures utilized Teflon dilators and plastic tubing for the shunts. When balloon angioplasty was developed in the mid to late 20th century, it became the dilator of choice. 16 Stent material was not adequate in the early procedures to maintain patency, and many stents either thrombosed or stenosed. In the 1980s, expandable metallic stents were developed that greatly improved their reliability. Today, stent grafts, also known as covered stents, have replaced the metallic stents with even more positive long-term results. 17 These stents are lined with PTFE, which is expanded polytetrafluoroethylene and fluorinated ethylene propylene. 18 According to Keller et al, 17 Gore is a manufacturer of PTFE-covered stents that are widely used during TIPS procedures. The endoprosthesis is the tube (stent) that is permanently implanted in the liver and remains in place to redirect blood flow through the organ. It looks like a tube made of “chain linked fence” with white material covering most of the metal tube’s inner surface. 18 Figure 1 demonstrates the GORE® VIATORR® TIPS Endoprosthesis.

GORE® VIATORR® TIPS Endoprosthesis. Figure copyrighted 2013 by W. L. Gore & Associates in Phoenix, Arizona. Used with permission.
The materials surrounding the stent are chemically inert fluoropolymers and have properties that minimize the invasion of bile and mucin and also lessen the growth of tissue into the stent, both of which can lead to stent failure. The design of the stent offers improved strength to decrease the deformation caused by tissue growth around the stent and also provides adequate anchoring within the liver. A segment of the stent is uncovered and placed in the portal vein to allow for perfusion through the stent to the distal portions of the hepatic vein. 18 Ferral et al 19 state that the GORE® VIATORR® TIPS Endoprosthesis is the first dedicated stent-graft system specifically designed for the TIPS application. It was designed to improve the long-term patency of TIPS and received approval by the Food and Drug Administration for the TIPS application in 2004.
A patient who is being considered for the TIPS must undergo testing to qualify to have the procedure performed; severe liver disease is a direct contraindication. Pre-procedure imaging studies are necessary to determine anatomical structures, verify portal system patency, and establish the portal and hepatic vein relationships. Cirrhosis can create diffuse parenchymal differences in the liver and initiate the need for an alternative form of vessel identification and selection. Computerized tomography, magnetic resonance imaging, and sonography are often used to define the liver’s anatomy. When an excessive amount of ascites is found pre-procedure, it will usually be drained prior to TIPS insertion. 17
A TIPS procedure is performed in an interventional radiology suite or operating room. Some physicians prefer to use general anesthesia and intubation while others choose to use conscious sedation. Prior to the procedure, the physician will provide details about the steps used throughout the procedure, explain the risks, provide expectations, give post-procedure instructions, and obtain consent from the patient. A nurse will obtain pre-procedure vital signs, and an intravenous line will be placed for medication administration. Frequently, antibiotics are administered pre-procedure to reduce the risk of infection. Sterile technique is utilized throughout the entire process. The patient will be prepped and draped in a standard manner for a minimally invasive procedure. Skin cleansing varies among facilities, but a standard sterilizing solution is often used. Sterile surgical drapes are used over the entire body length to accommodate the use of long guidewires while maintaining a sterile field. 17
Using sonography, the right jugular vein is visualized. The right jugular is favored over the left because of the more direct route through the vena cava, but the left can be used if needed. The ultrasound transducer is covered with a sterile cover, and sterile gel is used directly on the skin. The physician will manipulate the transducer in one hand while using the other to cannulate the vein. The skin will be anesthetized using a typical solution such as 1% Lidocaine. Access will then be obtained via the jugular vein using a micro-puncture needle. 20 A small guidewire will be inserted and the needle removed. A catheter and longer guidewire are placed and advanced through the superior vena cava, through the right atrium into the inferior vena cava, and into the right hepatic vein. At this point, hepatic pressures are recorded. After pressures are taken, the catheter is removed, and a needle/puncture assembly designed for the liver is advanced over the wire and directed through the hepatic vein, passing through the liver, making a channel, and then puncturing the right portal vein. A special needle is used to enter the portal vein through the liver parenchyma. It is advanced along the guidewire and inserted into the liver tissue between the hepatic and portal veins. 21 Figure 2 illustrates the generic placement of the shunt between the portal vein and a hepatic vein.
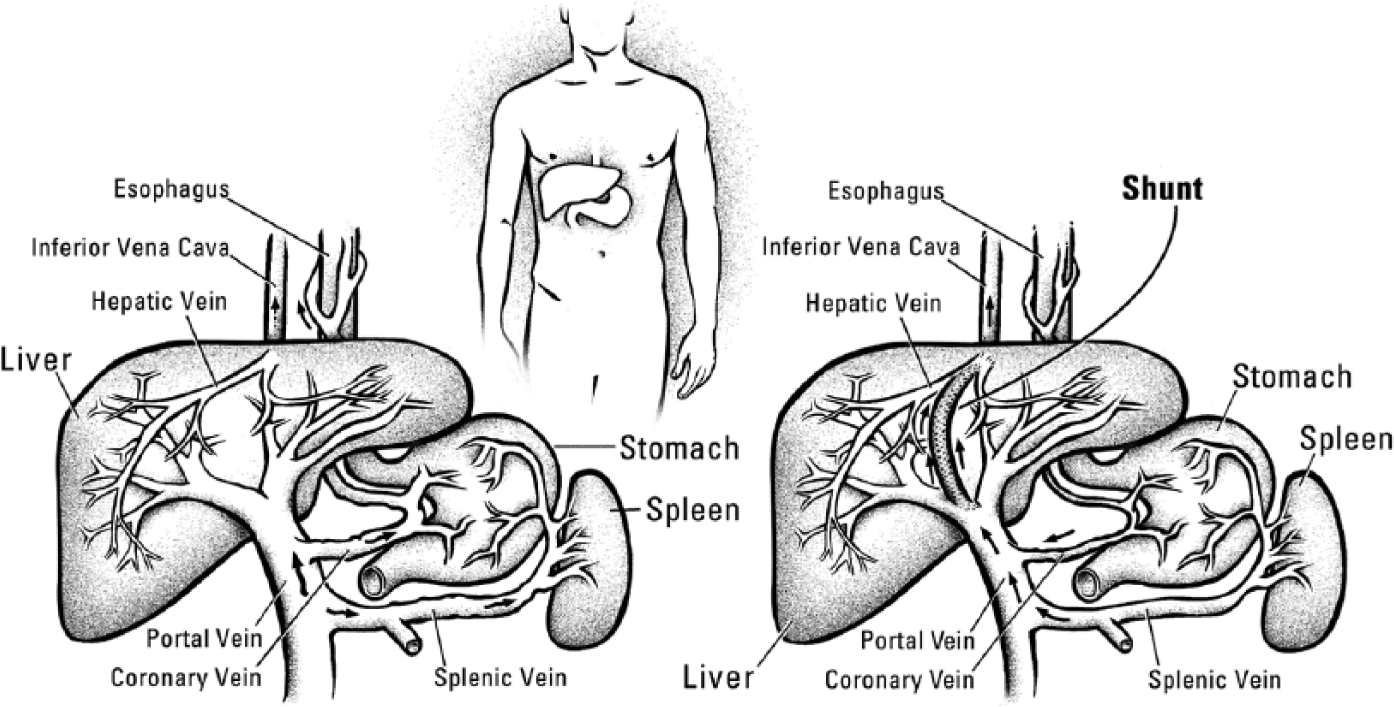
Illustration of the placement of the transjugular intrahepatic portosystemic shunt (TIPS) endoprosthesis between the portal vein and a hepatic vein. Illustration copyrighted 2012 by University of Washington Medical Center, Seattle, Washington. Used with permission.
It is stated that the most demanding part of the TIPS procedure is detecting and establishing access into the portal vein. 1 Upon entry into the portal system, the portal pressure is measured. This pressure is referred to as the portosystemic pressure gradient or PSG; normal PSG is less than 6 mmHg. “Portal hypertension is defined when the PSG is greater than 6 mmHg; however, bleeding complications of portal hypertension usually occur only when the PSG exceeds 10 mmHg.” 17 After pressures are measured, the new channel between the hepatic vein and portal vein is dilated using a balloon catheter device. 22 Once adequate tract dilation is achieved, the stent or endograft is deployed. The endograft should entirely cover the tract from the hepatic vein to the portal vein to decrease the possibility of stent stenosis due to fibrous tissue growth. With the stent in place, portovenography is performed to ensure proper placement and confirm patency of the stent. Contrast is injected through the catheter into the stent and should be visualized radiographically as it flows through the portal vein into the hepatic vein and IVC. Usually this evaluation is performed under real-time fluoroscopy. Once more, pressure measurements are recorded in the portal vein. According to Moran et al, 22 the PSG post-procedure should be much less than the prior measurement and should be 12 mmHg at the most. Variceal bleeding risk is lower when the portal pressure is at or below this level. Optimally, the pressure would be in the 8 to 12 mmHg range to decrease the chances for ascites accumulation. 22 Different practices have different guidelines on what portal pressure is optimal, with some in agreement that the post-procedure pressures should be 6 to 10 mmHg. 17 After the pressure is obtained and acceptable pressures are established, the remaining guidewires and catheter are removed, leaving the endograft in place. In some cases, varices that are still demonstrated on venography will be embolized before the procedure is terminated. Monitoring of the patient is performed post-procedure and typically includes vital signs, hematocrit surveillance, and urine output for 24 hours. Most patients do not require critical care following TIPS procedures. 15 Duplex imaging is performed before the patient leaves the hospital, and the portal and hepatic blood flow velocities are measured as well as visualization of the stent’s position.1,15–17,20–22 Figure 3 is a sonogram showing the position of the shunt within the liver. Figure 4 is a color Doppler image showing blood flow through the shunt, and Figure 5 is a spectral Doppler measurement of the blood flow velocity.
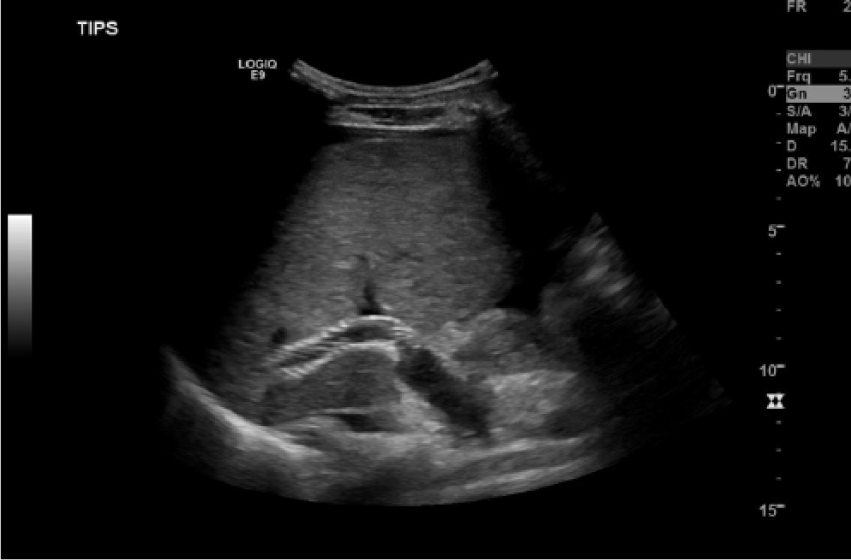
B-Mode image demonstrating the location of the transjugular intrahepatic portosystemic shunt (TIPS) endoprosthesis in the main portal vein. Note the characteristic echogenicity and sonographic appearance of the shunt.
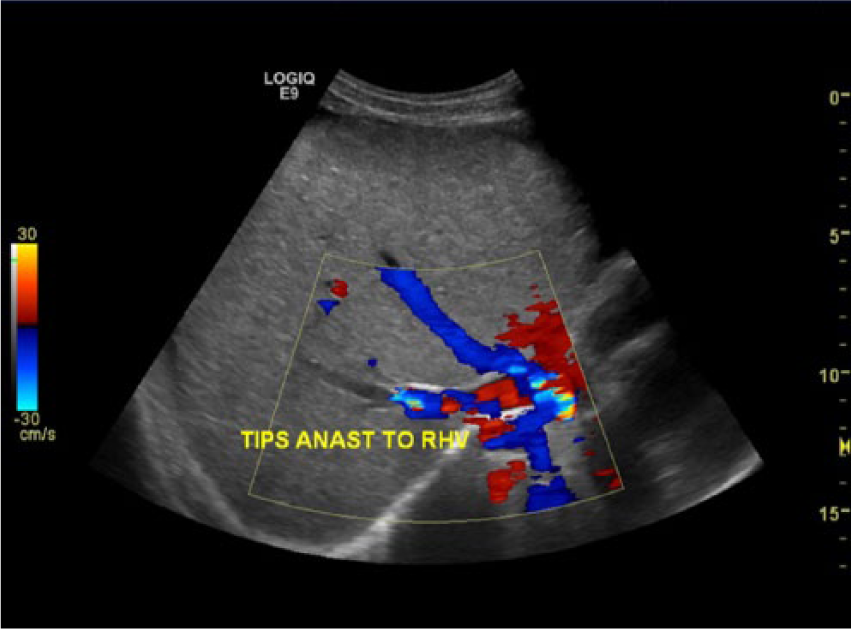
Color flow image demonstrating blood flow within the shunt (marked with yellow arrow) and into the right hepatic vein.
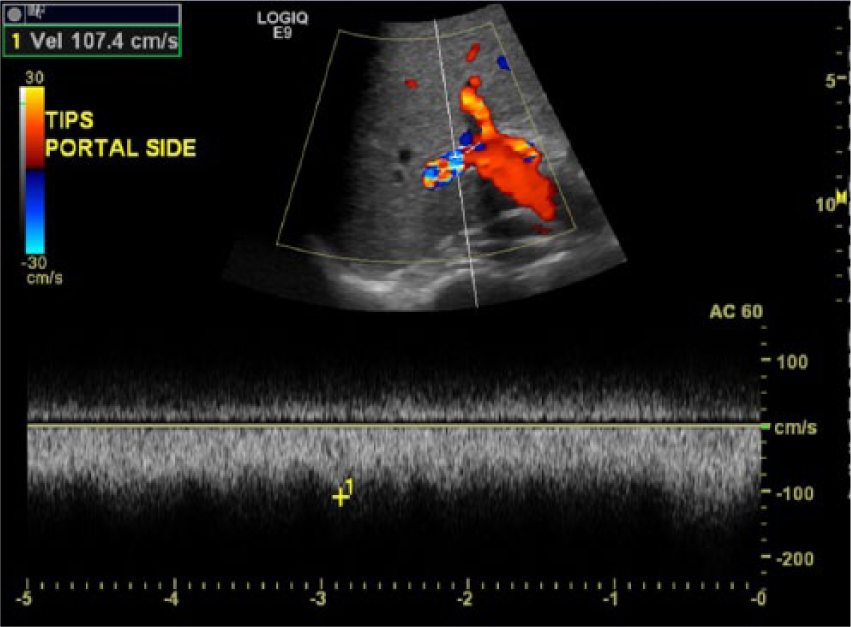
Spectral Doppler measurement of the flow velocity within the shunt.
Variations in liver anatomy necessitate considerable flexibility from the physician since approximately 60% of patients have differences in hepatic vein anatomy. In addition, due to these normal variants, the devices used for the procedure will differ based on which vessels are used to create the shunt between the portal and hepatic veins. At times, the middle hepatic vein may be used if the right cannot be accessed, and on occasion, the left hepatic vein is used. Imaging modalities including sonography, fluoroscopy, and venography are often used to assure appropriate placement of the catheter and the stent. 17 “The best method, in the opinion of many, for selecting the hepatic vein, with the added benefit of real time guidance during venous puncture, is sonography.” 16 Meticulous care should be used when selecting hepatic veins for TIPS procedures because about one-third of patients with hepatic vein variation present with two or three right hepatic branches, which can be of very small caliber. Similarly, the portal system may have deviations from normal as well. The right portal vein is known to have multiple variations and must be fully assessed prior to TIPS procedures. On occasion, the left portal branch may be utilized. 16
Complications
Post-procedure complications do occur but fortunately at a much lower percentage than in the early days of TIPS procedures. These risks for the TIPS method are reported at a 4% likelihood for minor complications and 3% for major complications, but more importantly, only a 1.7% procedural mortality rate was found in a large retrospective study. 21 One of the major complications encountered is stenosis, which is often caused by fibrous tissue growth into the endograft that subsequently compromises the lumen. The evolution of the PTFE-covered stents has reduced the number of stenoses that occur, but it is still a problem. “Their introduction [PTFE stents] led to marked decrease in shunt dysfunction (15% vs. 44%), and a lower rate of clinical relapse (10% vs. 29%).” 15 Stent stenosis can lead to failure that in turn can cause recurrent portal hypertension and/or variceal bleeding. 21
Stent thrombosis or occlusion can occur in approximately 10% to 15% of patients, which necessitates steps to remove the thrombosis by angiographic techniques or dilatation of the thrombosed section by balloon catheter.22,23 Surgical removal of the thrombus may be attempted, but in some cases, liver transplant is the only treatment. 22 When stent thrombosis occurs, it is typically soon after the procedure has been completed. It is thought that bile may leak into the stent and initiate the development of a thrombus. Using anticoagulants is contraindicated in patients with variceal bleeding, but according to Boyer, 24 even in patients who have been given Heparin (an anticoagulant), the chances for stent thrombus remain the same as for patients who do not receive blood thinners. Thrombus in the stent has the potential to dislodge and migrate to the lungs and cause pulmonary embolism. 24
Stent retraction into the liver parenchyma is another complication that can occur, but this may be resolved by accessing the stent and dilating it or adding additional stent material to the existing one already in place. The stent should be placed 1 to 2 cm into the portal and hepatic veins to allow for the retraction that typically occurs. However, caution must be used to ensure the stent doesn’t extend too far into the vessels, which can result in the shunt collapsing. 23 According to Rössle, 15 the newer endografts, such as the Viatorr, are specifically designed to facilitate positioning and rarely migrate from the original deployed position.
Saxon et al 25 explains that many of the early stent malfunctions were a result of technical errors. The more experienced physician will produce fewer mistakes that might lead to complications. Another problem that can occur is perforation of the liver capsule. Perforation with hemorrhage has been described, but evidence gathered by Rössle 15 states that when sonography is utilized during the liver puncture process, the risk of complications due to capsule damage is drastically reduced. Even in cases of perforation, intraperitoneal bleeding risk is low, occurring in 1% to 2% of cases. 15
Hepatic encephalopathy (HE) is an additional complication associated with TIPS. Patients with an increased stent diameter, older than 62 years of age, and with advanced liver disease are at an increased risk of developing this complication. 26 Hepatic encephalopathy can cause neurologic disturbances, irritability, imbalance, behavioral changes, sleep pattern disruptions, and tremors. Hepatic encephalopathy occurs due to the shunting of ammonia and neurotoxins, which are normally removed by the liver, directly into the bloodstream; this generally occurs within the first three months after TIPS placement. 27 Hepatic encephalopathy can often be treated successfully with lactulose and antibiotics. In addition to HE, the process of portal shunting increases blood flow into the venous circulation, possibly initiating heart failure and pulmonary hypertension, especially in patients who have a history of heart failure. 27
Along with these major problems, there are numerous minor complications that are related to TIPS procedures; however, the small risks are often considered worth taking for the overall benefits that can be obtained with portosystemic shunting.15,21–23,28
Post-Procedure Monitoring
Careful follow-up and consistent monitoring are essential to ensure proper function of the portosystemic stent. While portal venography is excellent at demonstrating stent patency, Doppler imaging allows visualization of blood flow within the stent, portal system, and hepatic venous system without the need for radiation exposure and contrast injections. According to Boyer, 24 TIPS dysfunction occurs in about 80% of treated patients within the first year. He suggests using Doppler imaging to assess the stent within 24 hours of placement. A recent review of the TIPS procedure by Rössle 15 reveals that when anticoagulants are used with uncovered stents, the shunt dysfunction rate drops from 44% to 15%. Rössle 15 also mentions that at the time of his review of the literature (2013), no clinical studies had been performed on the efficacy of anticoagulants and covered shunts. Regular follow-up Doppler studies should be performed every three months to ensure patency of the stent and assess for recurrent portal hypertension. However, because of the improved long-term patency of covered stents, Carr et al 29 suggest that regular assessment of the shunt with sonography isn’t necessary. If symptoms of portal hypertension persist after TIPS or increase, Freedman et al 26 suggest immediate Doppler studies. If abnormalities are discovered on the sonogram, it is advised that angiography of the TIPS should be conducted with portal pressure measurements. Additionally, Boyer 24 states that TIPS patients should be evaluated with angiography at one-year post-procedure to confirm portal decompression and shunt patency. In cases of stent stenosis, dilatation should be performed to maintain normal portal pressures. Comprehensive follow-up of post-procedure TIPS is necessary for the life of the patient or until liver transplant has occurred. 24
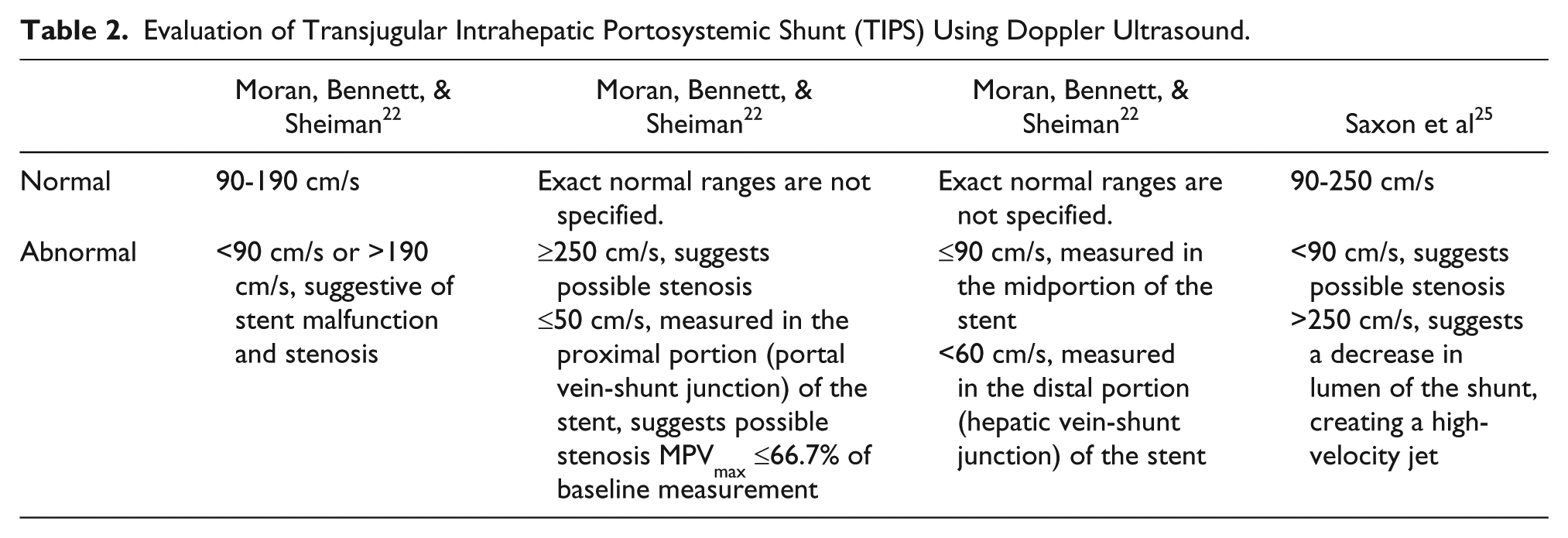
Several researchers have evaluated the usefulness of Doppler evaluation of a TIPS shunt, and the results have been encouraging. 25 Protocols pertaining to the Doppler examination may vary between facilities. Typical protocols include visualization of the stent within the portal vein, color and spectral Doppler interrogation of the stent, hepatic veins, portal vein and its branches, the IVC, and the hepatic artery. Saxon et al 25 uses the following Doppler parameters when evaluating a shunt: normal velocities within the stent may range from 90 to 250 cm/s, velocities under 90 cm/s suggest stent stenosis of about 50%, and velocities <50 to 60 cm/s suggest stenosis is likely and failure may occur. Moreover, if the velocities are over 250 cm/s, it can imply a stenosis where the lumen of the shunt is significantly decreased and a “jet” is created, as in a high-grade arterial stenosis. 25 Evidence reported by Fidelman et al 30 implies that the sensitivity and specificity for detecting stent malfunction is greater than 90% when Doppler velocities are below 50 cm/s or above 250 cm/s within the stent.
To adequately image a stent within the liver, a scanning system with the ability to penetrate the diseased liver should be used. Manipulating the transducer to obtain an optimal Doppler angle is important when performing color Doppler and spectral Doppler analysis of the blood flow through the shunt. Subxiphoid, subcostal, and intercostal views are all potentially used to align the beam with the vessel of interest. Figure 3 shows the characteristic sonographic appearance of the stent material, which is easily seen using two-dimensional (2D) gray-scale sonography. Stents covered with a fabric, such as polytetrafluorethylene, can exhibit shadowing that can persist for several days until the gas causing the shadowing is absorbed. 9 Once the gas is absorbed and the shadowing dissipates, the stent should sonographically appear as a conventional stent. Color Doppler as well as spectral Doppler should be used to interrogate the stent and supporting vessels. Information regarding the normal and abnormal appearance of the TIPS stent by use of 2D sonography and color Doppler can be found in Table 1.
2D and Color Doppler Ultrasound Evaluation of Transjugular Intrahepatic Portosystemic Shunt (TIPS).

Careful examination of the liver vasculature is imperative to verify proper stent function. Velocity parameters may vary from patient to patient and will change from immediately post-procedure velocities to those obtained during long-term follow-up. Universal Doppler criteria have not been established as of now, although some physicians and facilities have developed standards from their trials and are currently using them in their institutions. Table 2 demonstrates specified Doppler values for TIPS evaluation derived from multiple research studies. While there is variation in protocols at facilities and in the data collected, the information from Table 2 can be used to aid in assessment of TIPS function/dysfunction. A more comprehensive table of Doppler regarding TIPS dysfunction has been developed by Moran et al. 22
Evaluation of Transjugular Intrahepatic Portosystemic Shunt (TIPS) Using Doppler Ultrasound.
Hepatic arterial flow may increase due to the shunting of blood from the liver by the stent and be more prominent than normal. 10 Research from Owen and Gaba 31 using Doppler imaging and venography along with clinical symptoms to compare findings conveyed less than optimal results in concordance between these two diagnostic imaging modalities. The study hoped to find an acceptable correlation with abnormal or normal Doppler imaging and venography. The results were not sufficient to determine that Doppler imaging alone was an adequate tool to consistently determine stent malfunction. Future objectives are for noninvasive Doppler testing to be sensitive and reproducible enough to detect early stent malfunction even before symptoms occur. 31 Boyer 24 reports that Doppler imaging has sensitivity and specificity rates from 70% to 100% in cases of stent stenosis. A review of the diagnostic criteria for Doppler evaluation of TIPS by Darcy 32 revealed similar results with a sensitivity of 86% and a specificity of 54%. Sonographic diagnosis of a shunt malfunction requires a highly skilled sonographer and an experienced radiologist and must be conducted with consistent parameters to obtain dependable results. Most of the trials recommend follow-up studies, including some or all of the following: venography, Doppler imaging combined with gray-scale sonography, angiography, magnetic resonance imaging, and computerized tomography.
Contraindications
Contraindications to TIPS procedures are sometimes objective and vary between facilities and physicians. Advanced age is a potential contraindication as well as patients who have had previous hepatic encepha-lopathy. Severe pulmonary hypertension with a mean pressure gradient above 45 mmHg, heart failure, severe liver failure, and polycystic liver disease are absolute contraindications. 27 Overall, it has been proven that discriminatory patient selection for TIPS is paramount to a successful outcome. 8
Discussion
Mortality rates related to TIPS procedures are approximately 2%. 10 According to Middleton et al, 10 TIPS placement is successful in about 90% of patients but does hold a complication rate of 10% to 16%. Research has shown that placement of TIPS as a secondary treatment for variceal bleeding has produced encouraging results. In 13 RCTs that included over 900 patients, recurrent variceal bleeding rates were 9% to 40%, which is superior to endoscopic treatments that have recurrent bleeding rates of 20% to 60%. 27 This success rate has resulted in increased utilization of TIPS procedures in the treatment of variceal bleeding. Additionally, the same clinical trials indicate that ascites reduction after shunting ranges from 38% to 84%, which is favorable to large volume paracentesis that reported rates of 0% to 43%. 27 “Compared with large-volume paracentesis, TIPS improved transplant-free survival and the incidence of recurrent ascites in cirrhotic patients with refractory ascites.” 27 There is evidence of an increase in the development of HE in patients who undergo TIPS, whereas endoscopic treatment does not show an increase. Furthermore, HE is twice as likely to occur when paracentesis is performed. 30
Presently, TIPS is often used as a temporary solution for patients who are waiting on a liver transplant. Research from Owen et al 1 suggests that patients who undergo TIPS prior to liver transplant do not have increased survival rates prior to or following a transplant. The key is that TIPS intervention does have good results in controlling variceal bleeding and ascites formation while patients are awaiting transplant. Interestingly, patients who do have TIPS placed may present some technical problems to the surgeon while undergoing liver transplant surgery.1,10,27,30
Conclusion
While TIPS procedures are still maturing and advancing, many significant accomplishments have been made. Without a doubt, the more experienced physician tends to have better outcomes. Precise patient selection is key, as reported by many clinical trials. Routine monitoring and defined follow-up are necessary. Technically, the procedure is improving, and newer materials and methods are surfacing to facilitate the process. As with most medical therapies, there is a great need for continual improvements and research. Once used only as a secondary treatment or for emergent rescue for critical cases, TIPS procedures are improving, possibly becoming a first line of treatment for portal hypertension, variceal bleeding, and refractory ascites. The techniques and methods for TIPS are in a constant state of improvement as well as the protocols for follow-up and maintenance. As new approaches are implemented, clinical trials must still be utilized to analyze the best protocols and measure outcomes, mortality, and morbidity.
Footnotes
Acknowledgements
The authors thank TaShayla L. Withers, RDMS, RVT, for her assistance in obtaining the clinical images used in this literature review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
