Abstract
Background:
Fluid overload—a critical risk in pediatric/neonatal care—is often driven by excessive fluid during medication dilution, increasing complications like acute kidney injury(AKI), prolonged ventilation, and mortality.
Methods:
A 2024 cross-sectional study across 14 pediatric units (PICUs, NICUs, surgical/general wards) evaluated fluid administration practices and nurses’ knowledge. Observations included 150 patients and 200 nurses administering 8 medications. Data on fluid volumes, infusion durations, compatibility, and storage compliance were collected via checklist and questionnaires.
Results:
Nurses demonstrated moderate overall knowledge (mean score: 68.3 ± 10.4), with unit-level variability. Performance scores were highest in PICUs (76.0 ± 9.7) and lowest in surgical wards (62.86 ± 12.7). Key issues included excessive dilution (vancomycin, ampicillin, ceftriaxone) and storage noncompliance (eg, 40% in cardiac NICUs), posing risks to vulnerable patients.
Conclusion:
Standardized dilution guidelines, enhanced education on drug compatibility/storage, and real-time monitoring tools are critical to mitigating fluid overload risks and improving pediatric outcomes.
Keywords
Introduction
Maintaining fluid balance is critical in pediatric and neonatal care, where even minor errors in intravenous (IV) fluid administration can disrupt homeostasis and precipitate life-threatening complications such as fluid overload.1,2 Hospitalized children who cannot receive oral fluids depend on IV therapy—including maintenance fluids, resuscitation boluses, total parenteral nutrition (TPN), and diluents used in medication preparation—to meet metabolic and therapeutic needs.3,4 Although resuscitation fluids are essential for sustaining intravascular volume, diluent volumes used during medication preparation—a frequently overlooked source—can account for up to 30% of daily fluid intake in critically ill children.5,6 Nurses, as frontline clinicians responsible for preparing and administering IV medications, play a pivotal role in managing this risk. Their decisions regarding diluent selection (eg, 0.9% saline vs 5% dextrose) and volume directly influence fluid balance. However, evidence suggests significant variability in practice due to gaps in guideline awareness or training. 7
Fluid overload, defined as greater than 10% cumulative fluid accumulation, is strongly linked to adverse outcomes, including prolonged mechanical ventilation, AKI, and increased mortality. 8 Neonates, with their narrow therapeutic margins, are particularly vulnerable; diluent fluids from frequent medication administrations—such as antibiotics or inotropes—can cumulatively contribute to volume overload or electrolyte imbalances. 9 Even repetitive small-volume dilutions for frequent drug dosing may further exacerbate fluid excess, particularly in fluid-restricted patients. 10
Over-administration of fluids not only contributes to fluid overload but also delays AKI diagnosis by diluting serum creatinine levels, thereby hindering early intervention.1,11,12 While fluid overload is not an independent predictor of mortality, it significantly elevates the risk of death in critically ill children.2,13 The kidneys are particularly affected, as fluid overload both exacerbates and increases susceptibility to AKI, creating a detrimental feedback loop that worsens both conditions. 14 These findings underscore the importance of vigilant, evidence-based fluid management in pediatric care.
Certain patient populations face heightened risks. For example, neonates with congenital heart disease undergoing cardiac surgery often receive large volumes of fluid pre-, intra-, and post-operatively to maintain hemodynamic stability. Despite the use of diuretics like furosemide to promote urine output, fluid overload remains common due to acute kidney injury and systemic inflammation associated with cardiopulmonary bypass.15,16 This leads to increase in-hospital mortality, prolonged ICU and hospital stays, and delayed ventilator weaning. Similarly, in preterm infants, early-life fluid management significantly influences the development of bronchopulmonary dysplasia (BPD), making precision in fluid administration crucial. 17 Yet, nurses’ choices in drug dilution can either mitigate or exacerbate these risks. 18
Beyond total volume, the composition and compatibility of IV fluids used in medication preparation also impact patient safety and therapeutic efficacy. Appropriate fluid selection is vital in complex clinical scenarios such as diabetic ketoacidosis, acute respiratory distress syndrome (ARDS), sepsis, nephrotic syndrome, and post-cardiac surgery care.19,20 For instance, furosemide requires specific diluents to remain stable, while phenytoin precipitates when mixed with dextrose solutions. 21 Despite guidelines recommending minimal diluent volumes, studies show that only 58% of pediatric nurses routinely calculate medication-associated fluid intake, and 42% lack awareness of institutional fluid restriction protocols.22,23 These knowledge gaps reflect systemic issues such as insufficient training and inconsistent implementation of clinical guidelines, which may compromise outcomes in high-risk populations.18,23
Despite the critical role nurses play in pediatric fluid management—particularly during medication preparation—research examining their knowledge, decision-making, and adherence to clinical guidelines remains limited.24,25 Consistent adherence to protocols for minimizing diluent volumes and selecting isotonic solutions is essential to prevent iatrogenic harm. However, audits reveal inconsistent practices, including the routine use of excessive diluent volumes for drug compatibility. 26
This study assesses pediatric nurses’ knowledge and practices in medication preparation and administration, focusing on adherence to evidence-based guidelines. By identifying gaps between current practices and recommendations, we aim to inform targeted interventions to improve fluid stewardship, reduce iatrogenic fluid overload, and enhance outcomes in critically ill children.
Materials and Methods
Study Design and Setting
This study is part of an ongoing research project (code: 240034) with ethical approval (IR.MUI.NUREMA.REC.1400.068). It employed a cross-sectional observational design to evaluate nursing practices and knowledge related to fluid management during medication administration in pediatric and neonatal patients in 2024. The study was conducted across 14 clinical units, categorized into 9 specialized areas for analytical purposes. These categories included: 2 pediatric internal medicine units and 1 infectious diseases unit combined under the label “Internal”; 1 pediatric emergency unit classified as “Emergency”; 2 pediatric surgical units categorized as “Surgery”; 2 pediatric intensive care units (PICUs), further classified as PICU-Cardiac and PICU-General; 1 neonatal unit labeled as “Neonatal”; 2 neonatal surgical intensive care units; and 2 neonatal intensive care units (NICUs), grouped as NICU-Cardiac, NICU-General, and NICU-Surgery. These units were selected to provide a comprehensive representation of pediatric and neonatal care settings. A convenience sampling method was used to recruit participants due to feasibility constraints, though its limitations regarding potential selection bias are acknowledged.
Participants
The study involved 200 nurses and 150 pediatric and neonatal patients (from birth to 16 years old). Nurses were included if they were employed in pediatric, neonatal, or intensive care units (PICU/NICU), provided written informed consent, and were actively working in pediatric units during the study period. Nurses who withdrew consent at any point were excluded. Patient inclusion criteria were based on their admission to the participating units and the requirement for intravenous medication administration during the study period.
Data Collection
Data were collected through a structured questionnaire and a performance checklist:
○ ○
2. ○ Type of injectable drug ○ Dosage ○ Volume of administered fluid ○ Infusion method ○ Type of solution used
Each item was scored on a three-point Likert scale: “
Nurses were observed in 3 separate medication administration scenarios, selected based on 8 specific drug administrations (vancomycin, ampicillin, meropenem, phenobarbital, amikacin, gentamicin, ceftriaxone, and paracetamol). The average score across the 3 observations served as the overall performance rating. Observations were conducted in real-time to ensure accuracy.
Validity and Reliability of Instruments
The validity of the instruments was assessed by a panel of 10 experts, including 3 pediatric nursing professors, 1 clinical pharmacist, 2 pediatric medicine professors, and 4 senior nursing practitioners. The Content Validity Index (CVI) and Content Validity Ratio (CVR) were both determined to be 0.8 27 and the Face Validity was obtained above 1.5.
Instrument reliability was confirmed as follows:
Statistical Analysis
Data were analyzed using SPSS version 26. Descriptive statistics summarized demographic and clinical data. The Kolmogorov-Smirnov test (KS) was used to assess normality. Inferential statistics, including Chi-square tests, one-way ANOVA, Pearson correlation, and Spearman correlation, were applied to evaluate relationships between variables such as nurses’ knowledge, performance scores, and demographic factors (eg, work experience, education level). A P-value of <.05 was considered statistically significant.
Ethical Approval and Informed Consent
This study received ethical approval from the Regional Ethics Committee of Isfahan University of Medical Sciences (Code: IR.MUI.NUREMA.REC.1400.068) and adhered to the Declaration of Helsinki and national ethical guidelines. Written informed consent was obtained from all participating nurses prior to their involvement. Participants were informed of the study’s objectives, procedures, risks, benefits, and their right to withdraw at any time without consequences. Data were anonymized, securely stored, and measures were implemented to minimize the Hawthorne effect, ensuring observed behaviors reflected natural clinical practices. The research prioritized participant confidentiality, rights, and well-being throughout its design and execution.
Results
The study involved 200 nurses from 14 pediatric and neonatal units, with a response rate of 90.9% (200 out of 220 distributed questionnaires). Exclusions included 12 incomplete responses and 8 withdrawals of consent. Participants were predominantly female (97.5%), with an average age of 32.29 ± 4.09 years and 12.6 ± 3.12 years of work experience. Most nurses held a bachelor’s degree (91%), while 9% had a master’s degree. Work experience varied significantly across clinical units (P < .05), as determined by one-way ANOVA. Post-hoc analysis revealed that the highest proportion of experienced nurses (>10 years) was found in the neonatal surgical ICU (68.7%), whereas the lowest was observed in the pediatric surgery ward (37.1%; Table 1).
Work Experience in Pediatric Clinical Wards.

Chi-square.
Pharmacological knowledge scores also differed significantly across clinical departments (P < .05). The PICU had the highest mean score (62.18 ± 6.52), while the Pediatric Surgery Ward had the lowest (49.8 ± 8.9). Significant differences were also observed in specific domains: drug compatibility (P < .05) and storage of prepared drugs (P < .05), with both domains showing the lowest scores in surgical wards (Table 2).
Comparison of the Average Score of Nurses’ Pharmacological Knowledge in Different Scope of Pharmacological Knowledge.
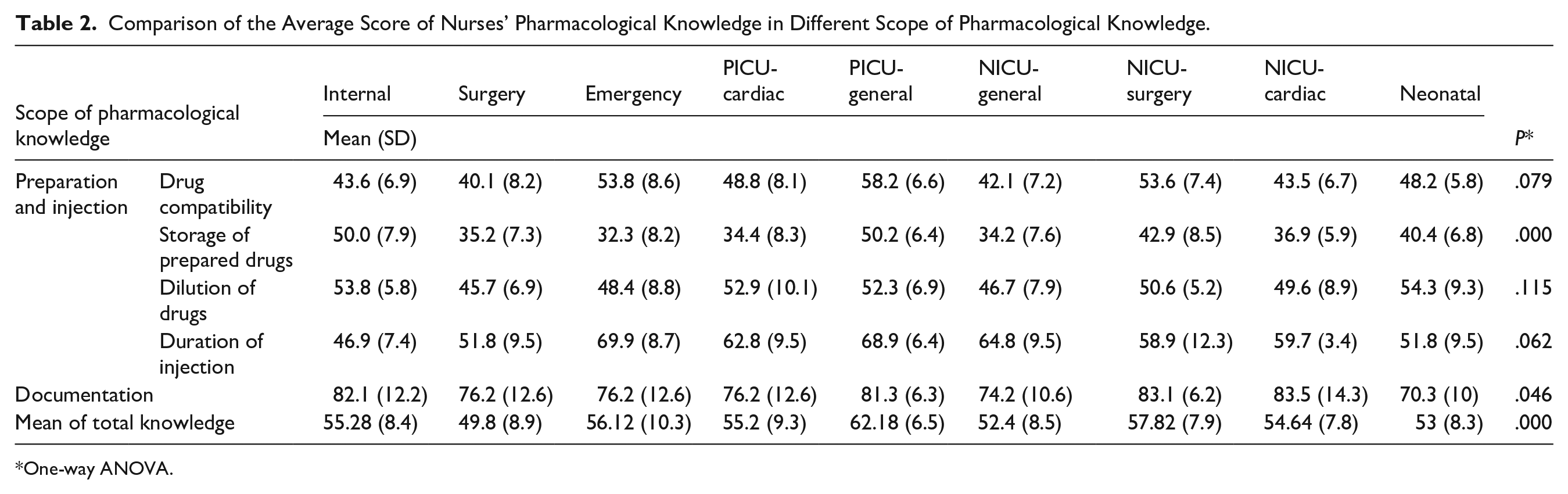
One-way ANOVA.
Performance in medication administration varied significantly between clinical departments (P < .05). The PICU demonstrated the highest mean performance score (76.0 ± 9.7), while the Pediatric Surgery Ward showed the lowest (62.86 ± 12.7). Key areas such as drug compatibility and storage of prepared drugs also exhibited significant variation (P < .05), again with the lowest performance observed in surgical wards (Table 3).
Comparison of the Average Performance Score of Nurses in Different Scope of Drug Administration in Different Clinical Wards.

One-way ANOVA.
Table 4 presents the rates of drug compatibility, storage of prepared medications, dilution of drugs, and duration of injection for 8 injectable medications: Vancomycin, Ampicillin, Meropenem, Phenobarbital, Amikacin, Gentamicin, Ceftriaxone, and Paracetamol. These rates were assessed by nurses across 14 departments: Internal Medicine, Surgery, Emergency, PICU-General, PICU-Cardiac, NICU-Genera, NICU-Surgical, Cardiac NICU, and Neonatal Care. The findings indicate that the highest dilution rates were observed for Vancomycin, Ampicillin, and Ceftriaxone. However, in terms of drug compatibility, the most frequent error involved Ampicillin, which was often administered with glycosides such as Amikacin and Gentamicin, despite their known incompatibility (14.4%). Another incompatibility involving Ampicillin was noted with Dextrose 10%, which is more commonly used in neonatal units. This issue was encountered more frequently in these departments (40%). Regarding drug storage after opening the package, most medications were discarded, contrary to the manufacturer’s instructions for proper storage. Among these, Vancomycin had the highest error rate (15.6%). In terms of compliance with standards for the duration of drug administration, the highest level of negligence among nurses was associated with the medications Vancomycin, Amikacin, and Ceftriaxone.
Drug Administration Data in Different Clinical Wards.
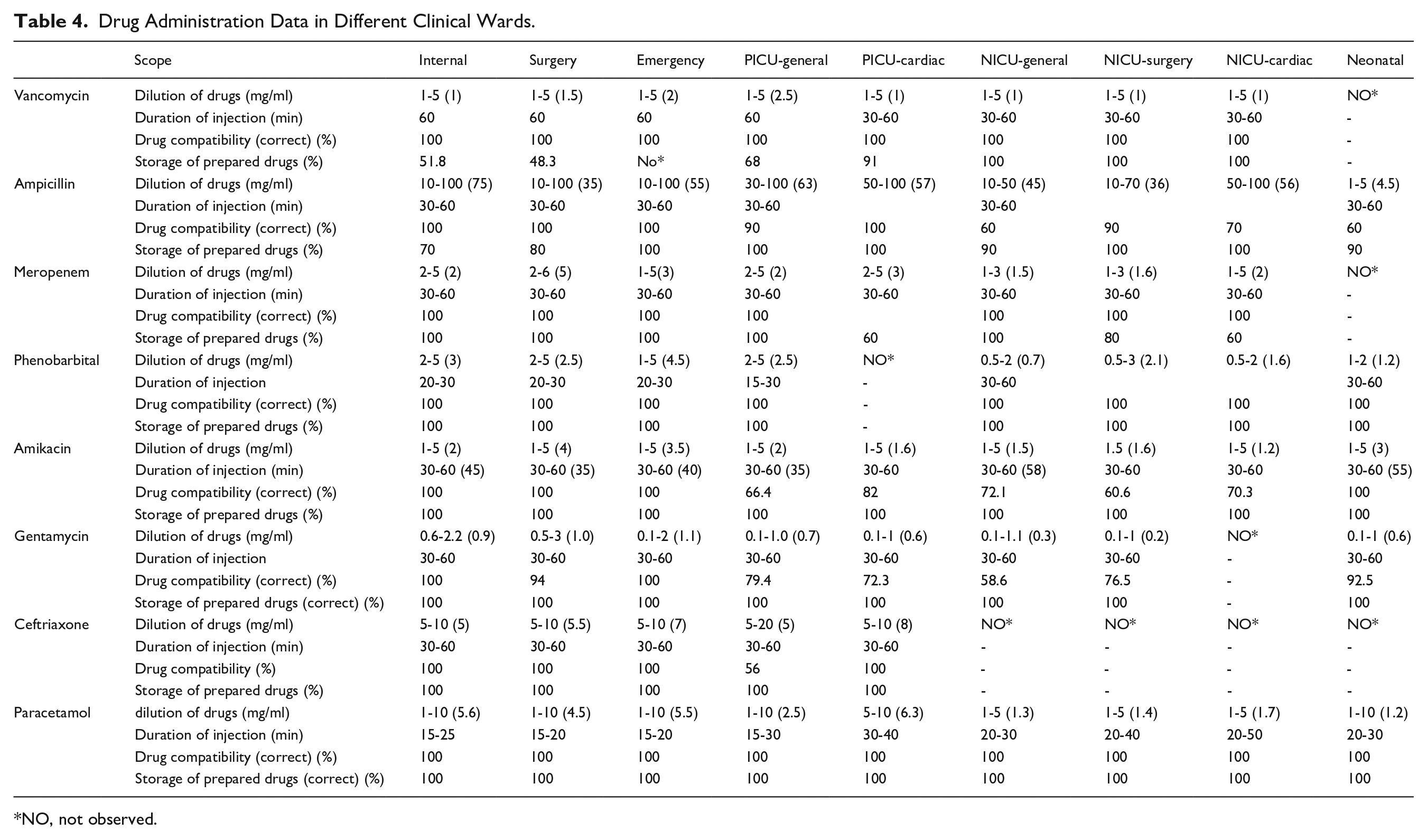
NO, not observed.
Correlation analyses revealed that pharmacological knowledge had the strongest positive correlation with overall performance across all domains. For example, knowledge of drug dilution showed a moderate positive correlation with performance (r = .432, P = .017). Work experience (r = .321, P = .021) and age (r = .341, P = .021) were also significantly correlated with better adherence to medication protocols, indicating that more experienced and older nurses performed better. Education level showed a moderate association with improved performance, particularly in drug dilution (r = .317, P = .003) and documentation (r = .315, P = .006). The ward of employment had minimal influence on overall performance (r = .121, P = .273), except for injection duration, where a weak but statistically significant correlation was found (r = .273, P = .025; Table 5).
The Relationship Between Nurses’ Mean Performance Score and Medication Knowledge Score, Education, Work Experience, Age, Ward.

Pearson correlation.
Spearman.
Discussion
This study aimed to determine the extent of fluid overload administered by nurses during medication administration to children and infants. The research focused on the injection of 8 common medications: Vancomycin, Ampicillin, Meropenem, Phenobarbital, Amikacin, Gentamicin, Ceftriaxone, and Paracetamol. It was carried out across 14 clinical departments: Internal Medicine, Surgery, Emergency, PICU-General, PICU-Cardiac, NICU-General, NICU-Surgical, NICU-Cardiac, and Neonatal Care. In addition, the study assessed nurses’ adherence to recommended infusion durations, awareness of potential drug compatibility, and compliance with proper storage conditions. Evaluating these factors is crucial for ensuring safe medication practices in this vulnerable population, given their unique physiological sensitivities. 28 According to Krzyzaniak and Bajorek, 29 preventing medication errors and promoting standardized administration practices should be a priority for all healthcare professionals. Fluid overload in infants and children, particularly during medication administration by nurses, can lead to serious complications. 30 However, data regarding fluid overload remains limited. 31 Conducting research on the current status of fluid overload in children can facilitate effective interventions to prevent complications 32 and improve necessary treatment and care. 30
The recommended concentration for vancomycin injection ranges from 1 to 10 mg/cc,33 -35 while the observed concentrations in this study ranged between 1 and 2.5 mg/cc, indicating that all units fell within the acceptable range; however, the dilution rate was relatively high. To mitigate excessive dilution, vancomycin can be administered at a concentration closer to 10 mg/cc, especially in cardiac units where fluid overload may significantly strain the heart. This increased dilution may reflect heightened sensitivity to vancomycin’s side effects among nursing staff. For ampicillin, the observed volumes of fluid used in the Internal, Surgery, Emergency, PICU-General, PICU-Cardiac, NICU-General, NICU-Surgery, NICU-Cardiac, and Neonatal departments were 75, 35, 55, 63, 57, 45, 36, 45, and 45 mg/cc, respectively. The recommended concentration range for this medication is between 30 and 100 mg/cc, with an average of 65 mg/cc.33,34,36 These findings suggest that the dilution rate for ampicillin is excessively high, leading to the administration of large fluid volumes—posing significant risks for patients with compromised heart or kidney function. Meropenem was diluted to concentrations ranging from 1.5 to 5 mg/cc across clinical departments. The recommended concentration rate for this medication ranges from 1 to 20 mg/cc, with an average of 10 mg/cc.33,34 The recommended concentration range is 1 to 20 mg/cc, with an average of 10 mg/cc. This indicates that the dilution rate is higher than necessary, resulting in unnecessary fluid burden for patients.
Phenobarbital was diluted to concentrations ranging from 0.7 to 4.5 mg/cc across clinical departments, compared to a recommended maximum concentration of 10 mg/cc.33,34 This excessive dilution raises concerns, especially for patients under fluid restrictions. Amikacin concentrations ranged from 1.2 to 4 mg/cc, while the recommended concentration range is 2.5 to 10 mg/cc, with an average concentration of 6.25 mg/cc.33,34 Similarly, gentamicin concentrations varied between 0.2 and 1.1 mg/cc, compared to a recommended range of 2 to 10 mg/cc. Both drugs showed suboptimal dilution rates, contributing to unnecessary fluid intake in young patients.33,34
Ceftriaxone was diluted to 5 to 8 mg/cc, whereas the recommended concentration range is 10–40 mg/cc.33,34 Paracetamol concentrations also fell below the recommended concentration, with values ranging from 1.2 to 6.3 mg/cc depending on the department. The recommended concentration for this medication is 10 mg/cc.33,34 These findings highlight that many medications are being diluted beyond recommended levels, leading to potentially harmful fluid overload in children and infants. Fluid overload poses significant risks in this population, including AKI. 37 A review by Matsushita et al 38 found that fluid overload is associated with AKI in critically ill infants, emphasizing the importance of careful fluid balance management. Multiple studies have shown correlations between fluid overload and poor respiratory outcomes.39,40 Research by Black et al 41 revealed that fluid overload occurring after the fourth day of ARDS is linked to worse clinical outcomes. Additionally, Matsushita et al 42 found that initial fluid overload in very low birth weight infants was associated with higher mean airway pressure and prolonged mechanical ventilation during the first week of life. Hayes et al 43 reported that patients experiencing more than a 20% increase in fluid volume had longer stays in the PICU, delayed renal recovery, and extended hospitalization. Additionally, Ingelse et al 44 suggested that avoiding early fluid overload could reduce the duration of mechanical ventilation in children.
Fluid overload is also poses a significant challenge in children and infants with cardiac conditions, making fluid management particularly crucial for infants with congenital heart disease undergoing cardiac surgery. 45 Postoperative fluid overload following congenital heart surgery is associated with increased mortality and morbidity, 46 prolonged mechanical ventilation, extended PICU stays, and delayed sternal closure. 47 Numerous studies have established a link between fluid overload and mortality in both children and adults. 48 Abulebda et al 49 found that fluid balance significantly affects the likelihood of 28-day mortality. Additionally, the study by Flori et al 50 noted that a positive fluid balance of 10 ml/kg over 24 hours correlates with increased mortality and longer mechanical ventilation duration. A meta-analysis by Fernández-Sarmiento et al 51 found that children with fluid overload had higher mortality rates than those without. Similarly, a systematic review by Alobaidi et al 52 reported that each 1% increase in fluid overload corresponded to a 6% rise in mortality risk.
Managing fluid overload in critically ill pediatric patients remains complex due to the lack of a definitive method for calculating it. Given existing knowledge gaps, further research is essential to develop targeted interventions for early detection and prevention of fluid overload-related complications.2,30
Vancomycin should be administered over a minimum of 60 minutes.33,34 Observations indicated that this recommended time was met in Internal Medicine, Surgery, Emergency, and PICU-General departments. However, administration times in NICU and PICU-Cardiac units ranged between 30 and 60 minutes. Adhering to the recommended infusion time is critical due to the drug’s potential complications. Red man syndrome (RMS) is the most common adverse effect of vancomycin, characterized by itching of the face, neck, and upper body. Severe cases may result in angioedema, cardiovascular collapse, 53 fever, anaphylactic shock, 54 or even cardiac arrest. 55 According to a study, administering 1 g of vancomycin over 10 minutes can cause a 25% to 59% drop in systolic blood pressure. Therefore, the infusion should never last less than 1 hour. 56 Additionally, Children and infants are particularly susceptible to RMS, with incidence rates as high as 37.4% in infants, according to Fernandes et al. 57 Thus, vancomycin should be administered over at least 60 minutes, with some studies recommending up to 90 minutes in neonatal and pediatric wards. 58
Based on the observations, no drug incompatibilities were identified during vancomycin administration in this study. However, concurrent use with aminoglycosides such as tobramycin or amikacin increases the risk of nephrotoxicity and ototoxicity. Monitoring renal function and hearing is essential, especially in patients with pre-existing renal impairment. 59 Interprofessional collaboration and pharmacist involvement can help mitigate these risks. 60
Vancomycin should be stored at 2°C to 8°C for up to 4 days.33,34 This guideline was strictly adhered to in the NICU-General, NICU-Surgery, and NICU-Cardiac departments. Compliance rates in the Internal, Surgery, PICU-General, and PICU-Cardiac departments were 51.8%, 48.3%, 68%, and 91%, respectively. Proper storage is essential to maintain drug efficacy, as improper storage can reduce potency by up to 9.4%. 61 According to a study by Nguyen et al, 62 the potency of vancomycin stored at 5°C diminishes after 28 days. Additionally, Mehta et al 63 have noted a decline in the potency of vancomycin stored at 4°C over time.
Ampicillin, when administered at a dosage of 500 mg, can be injected over a period of 3 to 5 minutes, while a 1 g dose should be administered over 10 to 15 minutes. 33 However, observed infusion times in this study ranged from 30 to 60 minutes across departments. Prolonged infusion can delay therapeutic effect and may contribute to unnecessary fluid administration. Proper injection techniques are essential to prevent complications such as gastrointestinal upset, rash, urticaria, elevated liver enzymes, and maculopapular rashes. Proper administration techniques are vital to minimizing these risks. 64 Drug incompatibility rates varied markedly across clinical departments. No incompatibilities were reported in Internal Medicine, Surgery, Emergency, or PICU-Cardiac units, whereas rates reached 40% in NICUs and PICU-General units. Ampicillin is incompatible with multiple medications, including amikacin, adrenaline, amphotericin B, calcium gluconate, cisatracurium, dopamine, erythromycin lactobionate, fluconazole, gentamicin, hydralazine, hydrocortisone sodium succinate, metoclopramide, midazolam, ondansetron, tobramycin, verapamil, and dextrose. 65 According to Medina-Barajas et al, 59 most amikacin incompatibilities arise during co-administration with ampicillin, a concerning finding given their frequent combined use as a first-line empiric regimen. Notably, the absence of visible incompatibility signs may obscure reduced drug efficacy, underscoring the need to avoid concurrent administration. Additionally, dextrose use is common in NICUs and PICUs, potentially increasing incompatibility risks in these units. Nurses must exercise heightened caution when administering ampicillin in pediatric and neonatal settings, particularly with aminoglycosides or dextrose-containing solutions, to ensure safe and effective treatment delivery.
Ampicillin can be stored at room temperature for up to 1 hour. 33 Compliance with this guideline was high in the Emergency, PICU-General, PICU-Cardiac, NICU-Surgery, and NICU-Cardiac units. However, compliance was lower in some NICUs, with rates of 70%, 80%, 90%, and 90% observed in the Internal, Surgery, NICU-General, and Neonatal wards, respectively. According to a study by Huskey et al, 66 maintaining ampicillin at room temperature for up to 72 hours prior to continuous infusion for 24 hours results in a potency loss of less than 10%. Similarly, research by Mitchell et al 67 indicated that beta-lactam antibiotics undergo hydrolysis under ambient pH and temperature conditions. Therefore, nurses should pay closer attention to the recommended 1-hour storage limit for ampicillin to ensure its stability and therapeutic effectiveness.
The recommended infusion time for meropenem is more than 30 minutes.33,36 All departments adhered to this guideline, with infusion times ranging from 30 to 60 minutes. Zhao et al 68 found that extended infusion improves therapeutic outcomes, especially in patients with reduced creatinine clearance and elevated uric acid levels. Therefore, nurses should be particularly vigilant regarding the infusion duration of this medication in children and infants with kidney diseases.
No drug incompatibilities were observed during the study. However, Meropenem is incompatible with Amphotericin, calcium gluconate, diazepam, ondansetron, and pantoprazole. 65 Healthcare providers, including nurses and doctors, should avoid simultaneous administration.
The storage conditions for Meropenem at room temperature are limited to less than 30 minutes, 33 a guideline that was fully adhered to in the Internal, Surgery, Emergency, PICU-General, and NICU-General units. However, in the NICU-Surgery department, compliance was observed at a rate of 80%, while the PICU-Cardiac and NICU-Cardiac departments demonstrated compliance rates of 60%. Research supports the importance of proper storage conditions. A study by Foy et al 69 found that approximately 10% of the meropenem concentration was lost after 144 hours at 6.7°C, and after 72 hours following 24-hour storage at 22.5°C. Similarly, Fawaz et al 70 reported that meropenem concentration decreased to 90% of its initial level within 7.4 hours at 22°C and within 5.7 hours at 33°C. These findings confirm that meropenem degrades rapidly at higher temperatures, highlighting the critical need for strict adherence to recommended storage guidelines to ensure drug stability and therapeutic efficacy.
The recommended infusion duration for amikacin ranges from 60 to 120 minutes33,71; However, in this study, observed infusion times were significantly shorter, ranging from only 30 to 60 minutes. Utami et al 72 found that infusion duration has a significant impact on plasma levels, thereby influencing both the drug’s therapeutic efficacy and potential toxicity. Intravenous amikacin should not be administered as a bolus; instead, it is recommended to use intermittent infusion over at least 60 minutes, especially considering that the drug is primarily excreted through the kidneys. Rapid infusion increases the risk of AKI 72 and ototoxicity. 73 Therefore, nurses must ensure that amikacin is administered within the recommended time frame to minimize these risks. Notably, no drug incompatibilities were observed in the Internal Medicine, Surgery, Emergency, or Neonatal units. However, higher rates of drug incompatibilities were reported in the PICU-General (33.6%), NICU-General (27.9%), NICU-Surgery (39.4%), and NICU-Cardiac (39.7%) departments. Amikacin is known to be incompatible with several medications, including: potassium chloride, aminophylline, amphotericin B, ampicillin, benzylpenicillin, cefotaxime, ceftazidime, ceftriaxone, cefuroxime, gentamicin, heparin sodium, Pabrinex, pantoprazole, phenytoin sodium, propofol, and tobramycin. 65 Heparin is routinely used in neonatal and pediatric intensive care units (NICUs/PICUs) to maintain central venous catheter (CVC) patency, typically through heparinized saline flushes (1-10 units/ml). However, its co-administration with amikacin poses significant incompatibility risks, particularly in these settings. The high frequency of heparin uses for CVC maintenance, combined with the administration of multiple medications in critically ill children, amplifies these risks. To mitigate adverse events, protocols such as flushing lines thoroughly with normal saline before and after incompatible medications, using separate catheter lumens for heparin and antibiotics, and staff education on compatibility guidelines are essential.
Regarding storage conditions, full compliance was observed across all clinical units. According to Kothari and Kowalski, 61 both temperature and storage duration can significantly affect the stability of amikacin. It was reported that aqueous solutions of amikacin sulfate at concentrations ranging from 37.5 to 250 mg/ml retained over 90% stability for up to 36 months at 25°C, 12 months at 37°C, and 3 months at 56°C. 74 These findings underscore the importance of maintaining appropriate storage conditions to preserve drug potency and ensure safe administration.
The recommended infusion duration for gentamicin ranges from 30 to 60 minutes.33,75 This duration was consistently observed across all clinical wards. As gentamicin is primarily excreted through the kidneys, appropriate administration timing is essential to minimize the risk of nephrotoxicity.75,76 Nurses should be particularly mindful of this concern when administering the medication. During the study, no drug incompatibilities were identified in the Internal Medicine and Emergency departments. However, high rates of incompatibility were observed in NICUs and PICUs. Gentamicin is incompatible with several medications, including: amoxicillin, amphotericin B, ampicillin, cefotaxime, cefradine, ceftazidime, ceftriaxone, cefuroxime, clindamycin phosphate, drotrecogin alfa (activated), flucloxacillin, furosemide, heparin sodium, propofol, sodium bicarbonate, and sodium fusidate. 65 These elevated incompatibility rates in NICUs and PICUs may stem from the frequent use of multiple medications in these units and should be carefully monitored to avoid adverse interactions.
Gentamicin should be stored at room temperature,33,34 which must be consistently maintained across all units. A study by Naveed et al 77 found that exposure to heat and ultraviolet (UV) light at a maximum wavelength of 243 nm for 30 minutes significantly reduced the drug’s effectiveness. Similarly, Curti et al 78 reported a decline in gentamicin stability after 30 days of storage. Therefore, maintaining appropriate storage conditions is crucial to preserving the drug’s potency and ensuring safe administration.
The recommended duration for ceftriaxone administration is at least 60 minutes.33,36 However, in the present study, infusion times ranged from 30 to 60 minutes across all departments. Adherence to the recommended infusion duration is critical due to the potential for adverse effects. According to Fesharakinia et al 79 ceftriaxone should be administered with caution, under supervision, and at an appropriate rate—particularly in children with kidney stones, nephrolithiasis, or gallbladder pseudolithiasis. Similarly, a systematic review by Donnelly et al 80 reported complications associated with ceftriaxone use, including elevated serum bilirubin levels, perinatal asphyxia, pulmonary hypertension, and thrombocytosis. These findings underscore the necessity for careful administration under supervision and at the correct speed.
No drug incompatibilities were observed during the administration of ceftriaxone in this study. However, it is essential to note that ceftriaxone is incompatible with several medications and solutions, including calcium-containing solutions, Ringer’s solution, aminophylline, amphotericin, clindamycin phosphate, fluconazole, gentamicin, labetalol, tobramycin, and vancomycin. 65 Healthcare providers should remain vigilant about these incompatibilities to prevent harmful incompatibilities.
Ceftriaxone can be stored at room temperature for up to 2 days. 33 This guideline was strictly followed in all clinical units during the study. Herrera-Hidalgo et al 81 indicates that ceftriaxone’s stability decreases after 30 hours when stored at 30°C. Therefore, adherence to recommended storage conditions—including both temperature and duration—is essential to preserve the drug’s potency and ensure safe administration.
The duration of paracetamol injection should exceed 15 minutes, as observed consistently across all clinical departments. Rapid administration of paracetamol has been associated with several adverse effects, including gastrointestinal bleeding, a mild increase in systolic blood pressure (approximately 4 mmHg), hepatotoxicity, 82 mild asthma, 83 rash, thrombocytopenia, leukopenia, and neutropenia. 65 Therefore, adherence to the recommended infusion duration is essential to ensure safe administration and minimize the risk of complications.
No drug incompatibilities were observed in any of the departments during the study, likely because paracetamol was consistently administered separately using normal saline in all clinical settings. Information regarding drug incompatibilities involving paracetamol remains limited. 65 However, some sources suggest that diazepam and chloramphenicol are incompatible with paracetamol. 74
Regarding storage conditions, guidelines indicate that if the ampoule has been opened, paracetamol can be stored at room temperature for up to 6 hours. In contrast, if the medication is drawn into a sealed syringe, it may be stored for up to 48 hours.65,84 These storage recommendations were consistently followed across all wards during the study.
In the present study, the highest scores for knowledge and safe medication performance were observed among nurses working in the Pediatric Intensive Care Unit (PICU) and General wards, while the lowest scores were recorded in the Surgery ward. According to Kane-Gill et al, 85 safe medication practices are especially critical in PICUs due to the heightened sensitivity of patients, the frequent use of multiple medications, and the need for meticulous administration to prevent complex complications in children.
This highlights the vital role of nurses in ensuring medication safety, as they are responsible for the final step in the medication administration process and remain continuously present at the patient’s bedside. 86 Despite this, the high incidence of reported medication errors across all pediatric wards underscores the necessity of implementing standardized safe medication practices in every clinical setting—particularly considering the unique physiological sensitivities of pediatric patients. 87 This is especially pertinent in surgical wards, where the risk of infection is elevated and the safe administration of antibiotics becomes a priority.
The findings of this study highlight several key factors that influence nurses’ performance in medication administration within pediatric and neonatal care settings. Pharmacological knowledge emerged as the strongest predictor of performance across all domains, demonstrating significant correlations with competence in drug compatibility, dilution techniques, and documentation practices. This underscores the critical importance of a strong theoretical foundation in pharmacology to ensure safe and effective medication administration. Nurses with greater pharmacological understanding were better equipped to make informed clinical decisions—such as identifying drug incompatibilities and adhering to recommended storage conditions. These results are consistent with previous research underscoring the role of education in improving clinical outcomes. 88
Work experience was also found to significantly influence performance, particularly in areas such as drug compatibility assessment, storage of prepared medications, and infusion duration adherence. This suggests that hands-on clinical experience contributes to the development of practical skills and confidence, reinforcing the value of structured mentorship programs and comprehensive onboarding for newly hired or less experienced staff. Similarly, age showed a positive correlation with performance in drug compatibility and infusion timing, indicating that older nurses may benefit from accumulated clinical experience and professional maturity.
Education level was moderately associated with improved performance, especially in drug dilution and documentation, emphasizing the need for ongoing professional development and access to advanced training opportunities. Interestingly, the ward of employment had minimal impact on overall performance, except for infusion duration, where a weak but statistically significant association was observed. This implies that individual competencies—rather than the specific clinical setting—are the primary drivers of safe medication practices, although differences in local protocols, resources, and institutional support may still exert some influence. Collectively, these findings emphasize the importance of implementing targeted interventions, such as continuing education programs, mentorship initiatives, and standardized clinical protocols, to enhance nursing performance and promote safe, effective medication administration in high-risk pediatric and neonatal populations. Addressing these factors can ultimately lead to improved patient safety, reduced complications related to fluid overload, and fewer medication errors in vulnerable patient groups.
Limitations
The topic of intravenous fluids in pediatric patients is vast, yet in many cases, there are no specific clinical guidelines for drug dilution. This lack of standardization makes it challenging to accurately determine the type and volume of fluids to be administered. Additionally, in numerous instances, the type of fluid prescribed for fluid resuscitation is incompatible with the injectable drugs, forcing nurses to administer excess fluids, which complicates fluid management in these patients.
In this study, we faced similar challenges in identifying clinical guidelines for fluid administration, as no unified clinical guideline was available. Consequently, we had to rely on textbooks rather than standardized clinical guidelines to develop the questions and checklists used in our research. This reliance on these resources may have introduced some degree of subjectivity into the assessment process.
Conclusion
Our findings reveal that nurses frequently dilute medications using empirically determined fluid volumes, without adequate consideration of the child’s clinical condition or specific fluid restrictions. This practice commonly results in the administration of 10% to 30% excess fluid, primarily due to gaps in pharmacological knowledge regarding appropriate drug dilution volumes. Such fluid overuse contributes to fluid overload, a preventable yet often overlooked complication in pediatric care that significantly impacts patient outcomes.
Fluid overload is associated with prolonged ICU and hospital stays, extended duration of mechanical ventilation, and increased vulnerability to complications such as acute kidney injury and respiratory distress. Although not an independent predictor of mortality, fluid overload exacerbates illness severity and may indirectly increase mortality risk in critically ill pediatric patients. Moreover, current practices often exclude medication-related fluid intake from routine fluid balance calculations, further compounding the problem.
Given these findings and the existing knowledge gap in pediatric fluid management, we propose several actionable recommendations to improve nursing practices and patient safety. First, standardized clinical guidelines should be developed through collaboration with pediatric pharmacists, intensivists, and nursing experts, ensuring that protocols for medication dilution and infusion volumes are evidence-based and tailored to each child’s age, weight, and clinical condition—such as cardiac status, renal function, and fluid restrictions. Second, pediatric-specific fluid management training should be integrated into both pre-service nursing education and continuing professional development programs, with a focus on safe dilution techniques and accurate fluid tracking. Third, point-of-care tools such as standardized dilution charts, mobile apps, or EHR-based alerts should be implemented to assist nurses in calculating appropriate dilution volumes based on individual patient factors. Additionally, interdisciplinary collaboration, particularly between pharmacists and nurses, should be strengthened, especially in high-risk units like PICUs and NICUs, to review dilution practices and monitor for signs of fluid overload. Lastly, further research should be supported to develop and validate tools for comprehensive fluid balance monitoring—including medication-derived fluid input—and to assess the impact of targeted interventions on clinical outcomes. By addressing fluid overload through structured guidelines, enhanced education, and interdisciplinary teamwork, healthcare institutions can significantly reduce preventable harm and enhance the safety and quality of care provided to vulnerable pediatric and neonatal patients.
Footnotes
Acknowledgements
The authors would like to thank the nurses who participated in this study for their valuable time and contributions. We also gratefully acknowledge Isfahan University of Medical Sciences for providing the institutional support and platform necessary for conducting this research.
Consent for Publication
Not applicable.
Author Contributions
ASH, FK, and MN designed the study. ASH, FK, AS, FJ and SKH collected the study data. AS, FK, and ASH performed data analysis and interpretation. ASH and FK, prepared the manuscript, and all authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed by the Vice Chancellor for Research of Isfahan University of Medical Sciences (Project number 240034).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Informed Consent
Both written and verbal consent was obtained from the nurses.
