Abstract
Background. Newborn screening in Malaysia includes congenital hypothyroidism and glucose-6-phosphate dehydrogenase deficiency. Screening for inborn errors of metabolism (IEM) is typically offered only for symptomatic patients. Objective. This study aimed to review the clinical and biochemical characteristics of children who experienced early mortality because of IEM. Methods. Malaysian children who were diagnosed with IEM and died before 5 years of age, were identified through selective screening of 36 467 at-risk patients between January 2015 and December 2021. Results. Thirty-six cases were detected. The mortality rate of children under 5 years diagnosed with IEM was 1.4 per 10 000 population. Clinical symptoms overlapped across the different IEM groups, and notably, similar organic compounds were found in different types of IEM. Conclusions. The mortality rate due to IEM is significant in Malaysia and most mortalities occurring during the neonatal period.
Highlights
• What do we already know about this topic?
Under-five and neonatal mortality rates have declined globally, including in Malaysia. However, data on early mortality due to inborn errors of metabolism (IEM) are limited globally and unavailable in Malaysia.
• How does your research contribute to the field?
Our findings expand the Southeast Asia and global data on early mortality due to IEM.
• What are your research’s implications toward theory, practice, or policy?
Our study contributes to understanding the current situation of early mortality from IEM, potentially aiding decision-making and the public health policy for enhancing newborn screening.
Introduction
Inborn errors of metabolism (IEM) are a group of heterogeneous inherited diseases that are prevalent in young children. Many IEM that causes cellular energy deficiency and/or intoxication are associated with sudden infant death (SID). 1 Moreover, fatty acid oxidation disorder (FAOD), urea cycle defect (UCD) and amino acid disorders have been shown to be the main causes of deaths in young children.2 -4 Currently, the universal newborn screening program in Malaysia includes congenital hypothyroidism and glucose-6-phosphate dehydrogenase deficiency (G6PD). Services for the detection of IEM are typically offered to symptomatic patients. Currently, only 4 centers provide laboratory services for detection of IEM in Malaysia: Institute for Medical Research, Kuala Lumpur (IMR); Hospital Tunku Azizah, Kuala Lumpur; University Malaya Medical Centre, Selangor; and University Sains Malaysia, Kelantan. IMR is the national referral center for comprehensive laboratory diagnosis of IEM and has received most of the selective high-risk screening for IEM in Malaysia. In 2016, the under-five childhood mortality report in Malaysia identified conditions arising from the perinatal period (32.7%), congenital malformations, as well as deformations and chromosomal abnormalities (29.7%) as the most common causes of death among children. 5 Premature birth was the highest cause of neonatal mortality from 2000 to 2017 in Malaysia followed by congenital defects, birth asphyxia, and sepsis. 6 Despite global decline in under-five and neonatal mortality, data on early IEM-related mortality remain scarce, especially in Malaysia. Our study addresses this gap in the literature. This study aimed to review the clinical and biochemical characteristics of children diagnosed with IEM with the outcome of early mortality.
Methodology
Study Design
This cross-sectional, retrospective, descriptive study was conducted at IMR from September 2022 to August 2023. Clinical findings and relevant laboratory data were obtained from the clinician’s request forms and clinical notes in the Genetic Department, Hospital Kuala Lumpur, and Laboratory Information System at IMR. Selective screening of at-risk patients was conducted using tandem mass spectrometry (TMS) between January 2015 and December 2021. Patients eligible for selective high-risk screening were those with symptoms suggestive of IEM. The diagnosis of IEM and death status were reviewed by 2 pathologists and verified by a consultant clinical geneticist and a consultant pathologist.
Ethical Approval and Informed Consent
Ethical approval was obtained from the Medical Research Ethics Committee Ministry of Health Malaysia (MREC), REF: 22-01354-AE5 (2). This study had a waiver for patient’s informed consent from MREC.
Patients and Samples
Data were obtained from Malaysian children aged <5 years, (1 day-2 years old) diagnosed with IEM who died in Malaysian government hospitals. Table 1 shows the age details of patients. Eligible patients were selected from 36 467 samples sent to IMR. Patients with symptoms suggestive of IEM were considered eligible for the selective high-risk screening. Those with positive screening results without supporting or confirmatory results were excluded from the study.
Age Ranges in Different IEM Subgroups.

Sample Size Calculation
Sample size calculation was based on sample size calculations for cross sectional studies/surveys. 7 Using the formula, p denotes proportion of IEM in population which was derived from previous published findings. 8 According to Chace et al, 8 the percentage of positive IEM in the screened pediatric population under 5 years old was 1%. Whereas, d is the absolute error or precision. The researcher wants to calculate the sample size with the precision/absolute error of 5% and at type 1 error of 5%. So, if we use the above formula,
The formula for the sample size calculation is as below.
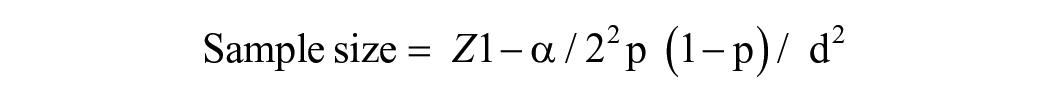
Z1-α/2 = Is standard normal variate (at 5% type 1 error(P < .05) it is 1.96 and at 1% type 1 error(P < .01) it is 2.58). As in majority of studies P values are considered significant below 0.05, hence 1.96 is used in formula.
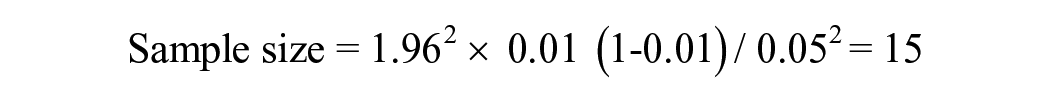
The calculated minimum sample size required for the study was 15, but we decided to include all subjects which fulfilled the inclusion criteria.
Laboratory Analysis
Tandem mass spectrometry analysis
Screening for IEM was conducted using electrospray TMS on dried blood spots (DBS). The details of the laboratory method have been published previously. 9 In brief, DBS samples received at our laboratory were checked for conformance. Next, 1 punch of 3 mm blood spot was placed in a 96 well plate. Amino acids and acylcarnitine were extracted with an extraction solvent containing an internal standard. Following evaporation at 60°C until dryness, the residues were reconstituted with a 150 µL mobile phase consisting of acetonitrile and water. Then the residues were subjected to LC MS/MS analysis using Quattro Micromass coupled with Waters Alliance 2795. Ionization was achieved by electrospray ionization using Parent 85 and Neutral Loss scan mode. The Ion spray voltage was set to 3 kV in positive ionization mode at a source temperature of 150°C and desolvation temperature at 600°C. Data acquisition and data processing were performed using MassLynx 4.0 software.
Other biochemical methods
Plasma amino acids were analyzed by ion-exchange chromatography technique using a fully automated amino acids analyzer, Biochrom 30 which uses EZChrom Elite V2B software for quantitation (Biochrom Ltd., Cambridge, UK). Physiological amino acid kits were purchased from Biochrom Ltd. (Cambridge, UK). Organic acids were analyzed using a gas chromatography system and ChemStation software (Agilent Technologies Inc., CA, USA). All standards, solvents and chemicals required for the analyses were purchased from Sigma Aldrich, (St. Louis, MO, USA) or Merck KGaA (Darmstadt, Germany). The organic solvents and other reagents used were of analytical grade.
Molecular analysis
The details of the laboratory method have been published previously. 9 Briefly, genomic DNA was extracted from dried blood spots using standard protocol in QIAmp DNA Blood Mini kit (Qiagen, GmbH), followed by DNA amplification by PCR. The PCR products were purified using the QIAQuick (Qiagen, GmbH). Cycle sequencing was performed using the BigDye Terminator cycle sequencing v3.1 chemistry (Applied Biosystems, Foster City, CA, USA) followed by purification using DyeEx 2.0 Spin Kit (Qiagen, GmbH) before loading to genetic analyser 3500 ABI (Applied Biosystems, USA) for DNA sequencing. Raw data were analyzed using SeqScape software to identify any DNA variants.
Statistical Analysis
Data from this study are presented in a descriptive form. However, simple statistics were used to estimate the prevalence of IEM among children diagnosed with the condition who died at 5 years of age or less.
Data Analysis
Descriptive analyses were performed using Microsoft Excel 2021. The mortality rate due to IEM was calculated based on the number of deaths due to IEM with the at-risk population as the denominator during the 7 years of study. The population at risk was the population of children aged <5 years in Malaysia who were suspected of having IEM. The formula used is as follows:

Results
From the 36,467 samples collected between 2015 and 2021, there were 36 cases of early mortalities with a diagnosis of underlying IEM were identified. The mortality rate in children under-five diagnosed with IEM varied between 0 to 3.5 per 10,000 population across different states with an overall rate of 1.4 for Malaysia. Approximately 69% of the patients were aged ≤1 month, whereas 31% were aged between 1 month and 3 years. Of these, 57% were male and 43% were female. Most cases were Malay (80%), with Sabah and Sarawak aborigines accounting for 10%, followed by Indians (5%) and Chinese (5%). FAOD was the most common diagnosis (51%), followed by organic aciduria (OA;21%), urea cycle defect (UCD; 16%), and congenital lactic acidosis (CLA; 12%). Figure 1 shows the distribution of the IEM groups with early mortality over a 7-year period. Figure 2 illustrates the mortality rate due to IEM across Malaysian states over the 7-year period. Figure 3 displays the distribution of clinical symptoms in patients with IEM who died early. Figure 4 shows the distribution of clinical symptoms in patients within OA, FAODs, UCDs and congenital lactic acidosis. Table 2 displays the organic compounds associated with these conditions.

Subgroups of IEM that caused early mortality between 2015 and 2021.

7-year average early mortality rate due to IEM by state (per 10 000 at-risk population) in Malaysia.

Distribution of clinical symptoms in patients with IEM who experienced early mortality.

Distribution of clinical symptoms among patients with organic aciduria, fatty acid oxidation disorder, urea cycle defect and congenital lactic acidosis.
Distribution of Organic Compounds in Organic Aciduria, Fatty Acid Oxidation Disorder, Urea Cycle Defect and Congenital Lactic Acidosis.

Discussion
The most common cause of early mortality in patients with IEM in Malaysia was FAOD, followed by OA, UCD, and congenital lactic acidosis. A systematic review revealed the association of various IEM diagnoses with early death among children with the global meta-estimate being 3.2 per 100 000 live births. 1 In another study, post-mortem metabolic screening of 793 children <3 years, gave 1% positive results, suggestive of either fatty acid oxidation or organic acid disorders. 10 Our study found a mortality rate of 1.4 per 10 000 at risk population, dominated by Northern states such as Perlis, Pulau Pinang and Kedah.
In this study, the majority (69%) of patients presenting with early mortality were aged ≤1 month, whereas 31% were aged between 1 month and 3 years. The clinical features of IEM are non-specific and the symptoms considerably overlap across the different disorders. Patients with FAOD tended to be sicker and had more variable symptoms. The most common presenting features were lethargy, respiratory difficulties, hepatomegaly, hypoglycemia, fever, septicemia-like illnesses, feeding intolerance, and seizures. Most IEM are episodic at presentation and the biomarkers may be absent when patients are not in metabolic decompensation. In our study, most of the patients presented with acute metabolic decompensation; thus, organic compounds were found in all group. Although, considerable overlap was observed between the FAOD, OA, and congenital lactic acidosis groups. Nevertheless, combinations of acylcarnitine, acylcarnitine ratios, and organic compounds in most patients facilitated specific diagnoses. In some cases with overlapping biochemical findings, molecular analysis was required for diagnosis, especially for patients with FAOD.
According to a Department of Statistic Malaysia report for medically certified conditions (2022), 37.2% of the causes of under-five mortality in Malaysia included conditions originating from the perinatal period, followed by congenital malformations, deformations and chromosomal abnormalities (29.9%); pneumonia (2.1%); Covid-19 infection (1.5%); transport accidents (1.1%); leukemia (1%); cerebrovascular diseases (0.6%); accidental drowning and submersion (0.5%); diseases of the liver (0.5%); and malignant neoplasm of the meninges, brain, and other parts of the central nervous system (0.4%). 11 A study of 1 million children born in Canada between 2006 and 2019 found that IEM were associated with preterm birth, congenital anomalies, preeclampsia, and other adverse birth outcomes. 12 In Southeast Asia, the main direct causes of under-five deaths were still neonatal (59%), including pneumonia (15%), diarrhea (9%), and birth defects (9%). 13 However, there are no specific reports on early mortalities due to IEM either in Malaysia or in Southeast Asia. This is partly because of the unavailability of expanded newborn screening or selective high-risk screening services for IEM in some regions.
The goal of newborn screening is the prevention or reduction of significant morbidity and mortality related to various disorders. 14 The introduction of TMS in the 1990s has allowed simultaneous high-throughput screening of multiple samples for several conditions. 15 Screening for IEM by TMS using DBS has the advantages of sample integrity during transportation, low blood volume requirements, and long storage stability. Advancement in newborn screening in Southeast Asia has been slow and challenging because of various factors including the economy, government stability, and health/political priorities. 16 Most of the public health expenditure in Malaysia has been spent on curative care services (68.15%), whereas only 9.76% has been allocated to public health services, including health promotion and prevention. 17 A systematic review of the clinical and cost-effectiveness of neonatal screening for IEM using TMS justified the introduction of TMS into a UK neonatal screening program for PKU and MCAD deficiency combined. 18 The Swedish newborn screening program for PKU and congenital hypothyroidism has shown substantial improvements in quality-adjusted life years. 19 As for the other IEM conditions, benefits of newborn screening may be debatable. Despite this, the value of expanded newborn screening lies in the prevention of death in young children, improvement in outcomes and long-term disability, and reduction in total medical expenditure. In an observational study of a historical cohort of patients in Mexico, mortality in the unscreened group was almost two-fold higher than that in the screened group. 20
The Malaysian healthcare system is heavily subsidized by public funding and may not be able to support expanded newborn screening. Furthermore, metabolic screening for all diseases seems unrealistic in terms of cost. Therefore, other funding options should be considered and discussed. The contribution of all sectors, including public, private, and non-governmental organizations, toward preventive healthcare for Malaysian children is essential. The expanded newborn screening in Malaysia should cover the most prevalent and treatable diseases such as such as few fatty acid oxidation disorders, urea cycle disorders and most common organic acidurias such as isovaleric aciduria, propionic aciduria and methylmalonic aciduria. However, this study has some strengths and limitations.
Strengths: This study offers critical data from Southeast Asia, contributing to the sparse global data on early mortality due to IEMs. The use of advanced screening techniques like TMS strengthens the diagnostic accuracy and insights offered by the study.
Limitations: Data from private hospitals was not included, limiting the generalizability of the findings to the entire pediatric population of Malaysia. Additionally, the unavailability of nationwide expanded screening reduces the impact of early detection and preventive strategies.
Conclusions
Most mortalities occurred during the neonatal period with considerable overlap in clinical and biochemical presentation among different IEM subgroups. The mortality rate due to IEM in Malaysia is significantly high. Initiating expanded newborn screening could be a cost-effective strategy for detecting the most common and treatable IEM.
Footnotes
Acknowledgements
We thank the Director General of Health Malaysia for permission to publish this paper. We also thank the staff of the Biochemistry and Molecular Diagnostics Unit, Institute for Medical Research, for their assistance in the laboratory work.
Author Contributions
A.H wrote the main manuscript text; N.A.A.M extract the research data and prepared figures 1-4 and ![]() ; A.H.A.Y and S.A extract and validated research data; S.A.R wrote the laboratory analysis; Y.Y wrote the molecular analysis; W.R.W.M wrote the data analysis and review the results section. N.L.H validate the clinical data. All authors reviewed and approved the final manuscript.
; A.H.A.Y and S.A extract and validated research data; S.A.R wrote the laboratory analysis; Y.Y wrote the molecular analysis; W.R.W.M wrote the data analysis and review the results section. N.L.H validate the clinical data. All authors reviewed and approved the final manuscript.
Availability of Data and Material
The datasets generated and/or analyzed during the current study are not publicly available due to the potential identification of the subjects but are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: However, authors received fund for authorship and publication from the National Institutes of Health, Setia Alam, Shah Alam, Selangor, Malaysia.
Informed Consent/Patient consent
The study had a waiver for patient’s informed consent from the Medical Research & Ethics Committee Ministry of Health Malaysia; Reference: 22-01354-AE5 (2).
Ethical Statement
This study was conducted in compliance with ethical principles outlined in the Declaration of Helsinki and Malaysia’s Good Clinical Practice Guideline.
