Abstract
Objectives. To describe the results of a 16-year experience of a state-coverage expanded newborn screening program (NBSP) in Northeast México. Methods. Between 2002 and 2017, dried blood spots of newborns were screened for congenital hypothyroidism (CH), congenital adrenal hyperplasia (CAH), biotinidase deficiency, galactosemia, cystic fibrosis, and glucose-6-phosphate dehydrogenase (G6PD) deficiency via immunofluorescence and amino and fatty acid disorders and organic acidemias using tandem mass spectrometry. Frequency rates were determined. Results. Overall, 192 487 samples were processed; 99.4% had negative results, and 598 were diagnosed. The frequency was 3.01/1000 newborns. G6PD deficiency, CH, amino acidemia, organic acidemia, cystic fibrosis, CAH, fatty acid oxidation disorder, galactosemia, and biotinidase deficiency cases were 1:773, 1:962, 1:4277, 1:4476, 1:11,322, 1:10,693, 1:10,693, 1:38,497, and 1:64,162, respectively. Conclusion. Using different technologies in NBSP increased the number of conditions detected, facilitating infant morbidity and mortality prevention. The frequency of disorders depends on the population’s genetic background and diagnostic capacity.
Keywords
Introduction
Newborn screening (NBS) is a strategy that allows early detection and pre-symptomatic treatment for many conditions. NBS programs worldwide differ in the number of conditions detected or the extent of coverage. Therefore, starting with a pilot study to analyze the public health impact and cost-effectiveness of increasing the number of markers to be included in the NBS is recommended. 1
Inborn errors of metabolism (IEMs) are a group of genetic disorders that lead to a metabolic block in the biochemical pathway, resulting in severe complications from unnoticed clinical scenarios and early death. IEMs as an individual disease are rare; however, their overall incidence is high, affecting 6% to 8% of the world population with considerable variation among different countries. The range of incidence of IEMs is 1:667 in Saudi Arabia and 1:9330 in Japan.2-12
In México, NBS has been incorporated into the official Mexican health policy since 1988 (mandatory for congenital hypothyroidism [CH]).13-15Based on a new official Mexican health policy modified in 2013, several IEMs should be covered by an expanded NBS; remarkably, its fulfillment remains optional. 16 Currently, NBS for CH, phenylketonuria, congenital adrenal hyperplasia (CAH), galactosemia, glucose-6-phosphate dehydrogenase (G6PD) deficiency, cystic fibrosis (CF), congenital deafness, and congenital heart diseases is mandatory. However, a significant variation in the detection of diseases and methodological differences exists between leading Mexican health institutes. 17
Expanded NBS by tandem mass spectrometry (MS/MS) has been successfully applied. 18 In a previous study, we reported implementing a pilot program on IEMs, detecting amino acidemias, organic acidemias, and fatty acid oxidation disorders by analyzing amino acids (AAs) and acylcarnitines (ACs) in blood. 5 In México, awareness about this topic is insufficient, whereas epidemiological data concerning IEMs is limited.19,20
Here, we report the results of a 16-year experience of a state-coverage expanded NBS program from 2002 to 2017 using MS/MS and fluorescence techniques among 194 000 newborns in Northeast México.
Materials and Methods
Study Population
Inclusion criteria
Neonates born at the State Secretary of Health hospitals and the University Hospital “Dr. José Eleuterio González,” covering a third of births in the state, were screened between January 2002 and December 2017.
Exclusion criteria
Neonates that had received a transfusion or those whose samples could not be taken owing to their health condition were excluded from this study.
Sample collection
Blood specimens obtained between the first and fifth day of life were collected by heel puncture and spotted on filter paper S&S 903 (Schleicher & Schuell, Keene, NH) and allowed to dry for 24 hours at room temperature before being sent to the Biochemical Genetics Laboratory of the Department of Genetics School of Medicine, Universidad Autónoma de Nuevo León, México.
Laboratory methods and analysis
Dried blood spots (DBS) received from January 2002 to December 2011 were processed and extracted with a methanolic solution using 12 internal stable isotope standards of AAs and ACs for each one (NeoGram derivatized MS/MS reagent kit; PerkinElmer®, Shelton, CT, USA). Samples were derivatized and analyzed in a triple-quadrupole tandem mass spectrometer API 2000 (PerkinElmer®, Wallac, Foster City, CA, USA). Most data were acquired using “full scan,” whereas citrulline, ornithine, free carnitine, and acetylcarnitine were acquired via multiple reaction monitoring (MRM). A polypropylene glycol solution was used to calibrate the following m/z: 59, 175, 616, 906, 1254, 1545, 2010, and 2242. The software Analyst version 1.1 and database Neo Gram (PerkinElmer®, Shelton, CT, USA) were used for data acquisition, analysis, and calculation of the analyte concentrations.
The DBS received from January 2012 to December 2017 were extracted using the NeoBase™, non-derivatized MS/MS kit (PerkinElmer®) for the same AAs and ACs quantification. The samples were analyzed in a TQD MS/MS Systems (Waters®, Shelton, CT, USA), and data was acquired using an MRM. A PQ/NARBY solution was used to calibrate the following m/z: 23.06, 84.88, 172.89, and 472.67. The Software MassLynx and database Chemoview were used to acquire, analyze, and calculate the analyte concentrations. DELFIA fluorescence (Victor, Wallac, PerkinElmer®) was used to measure total galactose, biotinidase activity, G6PD, and thyroid stimulating hormone (PerkinElmer®, kits). In contrast, Auto DELFIA time-resolved fluoroimmunoassay (DELFIA, Wallac, PerkinElmer®) was employed to measure 17-hydroxyprogesterone (17-OHP) and immunoreactive trypsinogen.
Reference and cut-off values
Cut-off values were obtained for both methodologies and different kits used during the study. Mean and standard deviation (SD) were calculated. Values greater or less than 3 SD were considered as “alert” and those greater or less than 4 SD as “abnormal.” 5 Low cut-off values” of analytes, such as free carnitine, citrulline, methionine, ornithine, tyrosine, acetyl (C2), propionyl (C3), palmitoyl (C16), and stearoyl (C18), were established. Sex, gestational age, and birth weight were considered for the cut-off value of 17-OHP. G6PD was established according to the sex. According to possible false positives and negatives evaluations and a theoretical incidence detected, further adjustments were made throughout the program. Adjustments were made also for some ACs and AAs ratios (C3/C2, C8/C10, C5/C0, C5/C3, and C0/C16+C18 and Leu/Val, Phe/Tyr, and Arg/Phe, respectively), to increase the likelihood of the involved analytes in the detection of EIMs. 5
Follow-up protocol
According to the algorithm established by our NBS, 5 program, a second sample is taken 2 hours after feeding. However, this is not necessary for conditions unrelated to food ingestion, such as CH, CAH, G6PD deficiency, and CF. If the second sample results are abnormal, we proceed with confirming the disorder. If any result above 4 SD was found in the first sample, the neonate was immediately sent to a hospital for evaluation.
Confirmation tests for IEMs
Confirmation of IEMs was made using recommended methodologies (Table 1).
Confirmatory Tests for Suspected IEM Used in This Study.
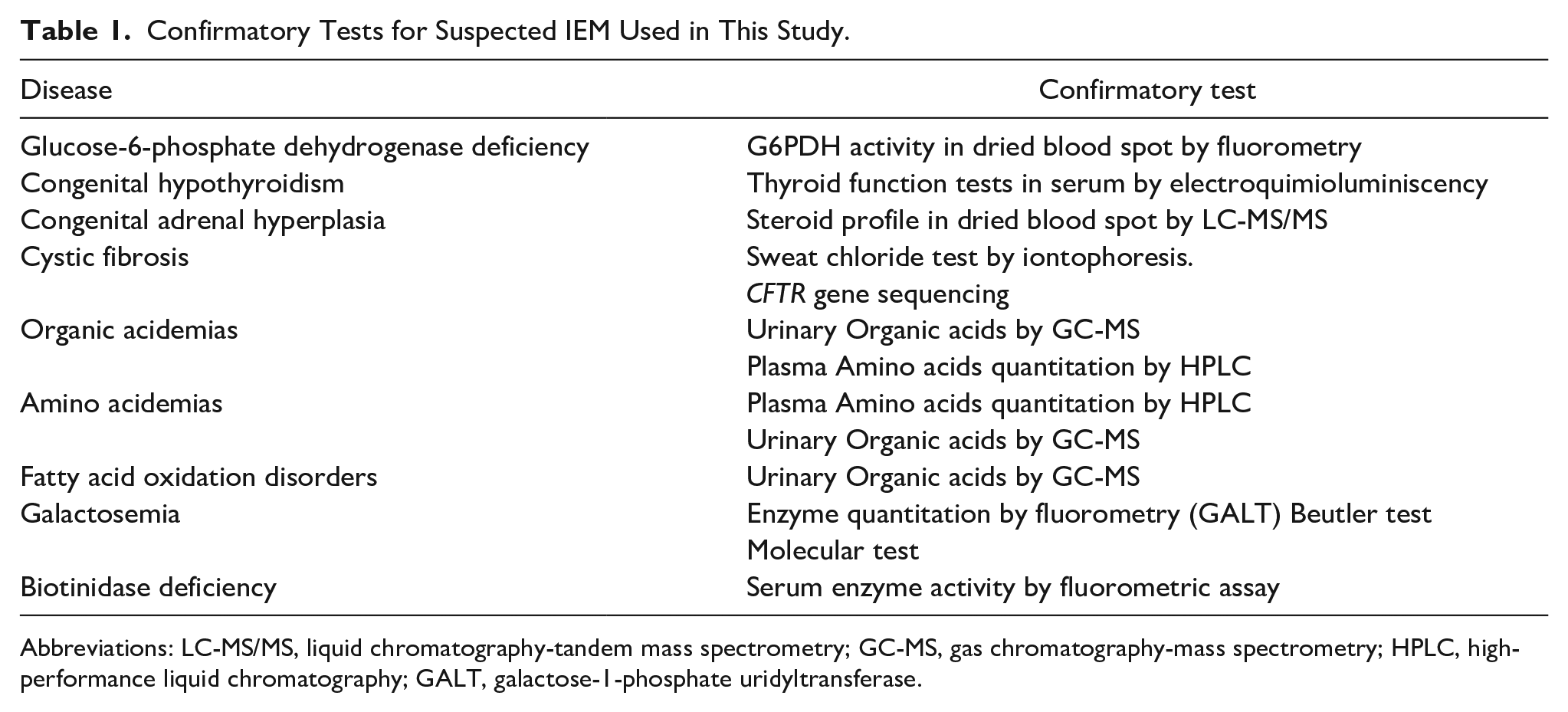
Abbreviations: LC-MS/MS, liquid chromatography-tandem mass spectrometry; GC-MS, gas chromatography-mass spectrometry; HPLC, high-performance liquid chromatography; GALT, galactose-1-phosphate uridyltransferase.
Quality control
Our NBS program participates in the Newborn Screening Quality Assurance Program of the Centers for Disease Control and Prevention.
Statistical analysis
Basic statistics were performed. Mean and SD for the cut-off values of analytes were obtained, logarithmic transformation of raw values was used to normalize data, and percentages and frequencies of abnormal results and diseases were obtained. Sensitivity and specificity values, as well as positive predictive value (PPV) and negative predictive value (NPV) were obtained. SPSS program was used [IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY, USA)].
Ethical approval and informed consent
The NBS program in Nuevo León is a mandatory governmental program, and no personal information is shared; therefore, ethics approval is not needed, and informed consent is not necessary.
Results
Table 2 shows the specific cut-off values obtained using the different methodologies from over 30 000 samples in our population according to the mean and ±3 SD. Sensitivity and specificity values, as well as PPV and NPV, are shown in Table 3.
Cut-off Values Established for Some Analytes Obtained From Over 30 000 Samples in the Present Study. a

Units: Amino acids, acylcarnitines, and 17OHP (μmol/L), total galactose (mg/dL), TSH (μU/mL), IRT (ng/mL), biotinidase (nmol/min/dL), and G6PD (U/gHb).
Abbreviations: Ala, Alanine; Leu, Leucine; Phe, Phenylalanine; Tyr, Tyrosine; Cit, Citrulline; Orn, Ornithine; C0, Free carnitine; C2, acetyl; C3, propionyl; C5, butyryl; C8, octanoyl; C10, decanoyl; TSH, thyroid stimulating hormone; IRT, Immunoreactive trypsinogen; 17-OHP, 17-hydroxyprogesterone; G6PDH, Glucose-6-phosphate dehydrogenase; M, Male; F, Female.
Reference values were established from the mean ± 3 SD.
Low cut-off values.
Performance of the Tests Employed in the Study. a

Missing values were not considered in the analysis, thus, parameters may change.
A total of 194 000 newborns were screened; 192 487 (99.2%) were processed, and 1513 (0.77%) were rejected. Among the newborns processed, 8044 (4.1%) and 184 443 (95.8%) were positive and negative, respectively. A second sample was obtained in 6306 (78.3%) of the 8044 (4.1%) positive cases. Of these, 5297 (84%) cases were classified as false positive, and 1009 (16%) were suspected of having one of the disorders screened and were recalled for further confirmation assessment. From these, 598 cases were ultimately confirmed for an overall rate of 3/1000 neonates (Figure 1). We did not receive 21.7% of the samples for confirmation owing to false information, change of address, or parental refusal. Additionally, we did not have a report regarding death as the cause of non-found cases.

Results obtained according the follow up protocol of the study.
Among the 598 infants, cases detected by fluorescence technique were 200 (1:962) CH, 249 (1:773) G6PD deficiency, 18 (1:10,693) CAH, 17 (1:11,322) CF, 5 (1:38,497) galactosemia, and 3 (1:64,162) biotinidase deficiency, whereas those identified by MS/MS were 45 (1:4,277) amino acid disorders, 43 (1:4,476) organic acidemias, and 18 (1:10,693) fatty acid oxidation disorders. Phenylketonuria and hyperprolinemia were the commonest amino acidemias (1:24,060), whereas 3-methylcrotonyl-CoA carboxylase deficiency was the most frequent organic acidemia (1:13,749), and medium-chain acyl-CoA dehydrogenase deficiency was the most common fatty acid oxidation disorder (1:19,248) (Table 4). The frequency of disorders identified via MS/MS is shown in Table 5.
Frequency and Rates of IEM in 192 487 Screened Newborns in Nuevo León, Mexico.

Abbreviations: G6PD, glucose-6-phosphate dehydrogenase deficiency; 3-MCC, 3-methylcrotonyl-CoA carboxylase deficiency; CAH, congenital adrenal hyperplasia; FAO, fatty acid oxidation disorder; MCADD, medium-chain acyl-CoA dehydrogenase deficiency; CF, cystic fibrosis.
Overall rate (males and females).
Frequency and Rates of Disorders Detected by Tandem Mass Spectrometry (Amino Acid Disorders, Organic Acid and Fatty Acid Disorders, and Mitochondrial Disorders).

Transient Tyrosinemias. Patients were followed only with HPLC test.
Discussion
For over 16 years after its launch, the expanded NBS in our state has shown great success, registering approximately 600 newborns with 30 different disorders. The overall frequency was 1 per 321 newborns (3.01/1000 newborns), even though G6PDH, 17-OHP, IRT, total galactose, and biotinidase were not assessed between 2002 and 2011. Worldwide frequency of IEMs and other diseases identified through NBS depends on the capability of detection and diagnosis in different countries, as well as the genetic background of the population, since most of the diseases screened in neonates are of genetic origin. Our incidence rate of disorders corresponds with that reported by Cantú-Reyna et al 21 of 3.4 per 1000 neonates in the same population.
The frequency of IEMs obtained was 1 in 1798 neonates, higher than the global prevalence of 1 in 1964 newborns reported by Waters et al 22 but lower than the prevalence of 1 in 1315 neonates reported in the Eastern Mediterranean population, which can be explained by the higher rate of parental consanguinity in this region.
CH was the second most common disorder found in this study (1:962), which is higher than the frequency of 1 in 1373 newborns recently reported by Hinojosa Trejo et al 23 among the population across the country. Nevertheless, 2 studies from the same population in Nuevo León reported lower frequencies of CH cases, 1 in 5000 neonates, 24 and none in 10 000 samples. 21 In these studies, samples were taken from venous puncture and referred to a laboratory outside México. Capillary and venous blood differ in hematocrit concentration, 25 and cut-off values may vary according to population; therefore, these may affect the results.
The frequency of CH varies among different ethnicities. Frequencies of 1:1,600; 1:1,757; 1:2,380; 1:3,533; and 1:11,000 have been reported in Hispanics, Asians (Indian), Asians (Chinese and Vietnamese), non-Hispanic whites, and non-Hispanic blacks. 26
G6PD deficiency is the most common metabolic disorder of erythrocytes that affects over 400 million people worldwide. In Eastern Saudi Arabia, the overall prevalence of G6PD deficiency is 25%, whereas it is 3.2% in Iran. 27 G6PD deficiency is reportedly more common in the Mediterranean population, Africans, or Asians, with rates ranging from 1:4 to 1:24 cases, owing to genetic changes that appeared in these populations, for protection against malaria, 28 compared to Latin American countries where significantly lower rates have been reported in Argentina, Bolivia, México, Peru and Uruguay, but not in Curaçao, Ecuador, Jamaica, Saint Lucia, Suriname and Trinidad, Brazil (Amazonas), Colombia, and Cuba, where the prevalence is >10%. 29 Both studies mentioned above reported a higher rate than ours, with 1 per 773 neonates.
Amino acid disorders accounted for 7.5% of all cases, and phenylketonuria was the most common type, with a frequency of 1 in 24 060, in agreement with previous studies.1,4,6,8,10,11 Worldwide phenylketonuria prevalence is 1 in 23 930 live births, with the highest prevalence reported in Turkey (1 in 2622 neonates) and the lowest in Thailand (1 in 333 333 neonates). 30 The incidence in Latin American countries is estimated at 1:23,000 live births, similar to that found in our study and the incidence previously reported in México (1 in 27 546 neonates). 31
Hyperprolinemia was also common. Two types of hyperprolinemia exist as follows: Types I (hyperprolinemia I) and II ( hyperprolinemia II) caused by pathogenic variants in PRODH (22q11.21) and ALDH4A1 (1p36.13) genes, respectively. A relationship has been identified between 22q11 microdeletion syndrome and hyperprolinemia. Therefore, evaluating neonates with hyperprolinemia to rule out 22q11 syndrome would be interesting. 32 However, in our study, we could not perform chromosomal or molecular studies in these cases.
Organic acidemias accounted for 5.9% of the cases, and 3-methylcrotonyl-CoA carboxylase deficiency was the most common type. In contrast, fatty acid oxidation disorders accounted for 2.4% of the cases, and medium-chain acyl-CoA dehydrogenase deficiency was the most common type. These results are broadly different from those of Saudi Arabian, American, British, German, Italian, Chinese, Japanese, and Egyptian populations, including 204 cases from the México City cohort.1,4,6,8,10,11,20,33,34 3-Methylcrotonyl-CoA carboxylase deficiency is apparently detected more frequently among the Hispanic population.35,36
CAH accounted for 2.4% of the cases. However, its frequency of 1:6578 compares to those detected in Chinese, Indian, and other lower-incidence populations ranging from 1:6084 to 1:26,727. 37 CF accounted for 2.3% of the cases (1:6965), except for Caucasians (1:3000), our finding revealed even higher frequency than those reported in newborns from southeastern México (1:13,724), 38 Hispanics (1:9200), Native Americans (1:10,900), African Americans (1:15,000), and Asian Americans (1:30,000). 39 Galactosemia and biotinidase deficiency accounted for 0.6% (1:23,681) and 0.4% (1:39,469) of the cases, respectively. Notably, the reported incidence of galactosemia ranges from 1:10,000 to 1:77,500,1,6,11 whereas that of biotinidase deficiency ranges from 1:55,000 to 1:120,000.1,6
The strength of this study lies in the size and breadth of the sample, and our analysis of IEMs using MS/MS is the largest in México. However, the interpretation of our findings should consider some limitations, such as the geolocation of the study, since most of the disorders identified through the NBS follow an autosomal recessive inheritance pattern, in which the effect of consanguinity is an issue to consider. México is genetically heterogeneous; consequently, the frequency of the diseases identified in our study does not reflect the one in the country. Regionalized studies should be conducted to determine whether there are differences. Finally, another limitation is that the sample size was not calculated, which could also bias the estimated frequency of the disease. The expansion of screening to all newborns in México will be very useful not only for the prevention of disability or death and genetic counseling, but also for knowing the frequency of the disorders.
Conclusions
Using MS/MS and immunofluorescence increased the detection rate of disorders in our population. Our findings contribute to the knowledge concerning the incidence of these diseases and justify the implementation of an expanded NBS program for preventive and treatable disorders, which, combined with genetic counseling, early detection, and pre-symptomatic treatment, will enhance the quality of care of patients.
Footnotes
Acknowledgements
We would like to acknowledge all health workers from the State Secretary of Health and University Hospital who participate in the Newborn Screening Program.
Author Contributions
MDR T-S: Contributed to analysis, drafted the manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
LEMDV: Contributed to conception and design, critically revised the manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
JZV-P: Critically revised the manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MDCRH: Contributed to analysis, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
PDCAV: Contributed to conception and design, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
AKT-M: Drafted the manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Data Availability Statement
The data is stored in the laboratory of the Genetics Department at the University Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
The newborn screening program in Nuevo León is a governmental program, and no personal information is shared; therefore, ethics approval is not needed, and Informed consent is not necessary.
Consent to Participate
Not applicable.
Consent for Publication
All authors are informed and had given their consent for publication.
