Abstract
Nonocclusive mesenteric ischemia (NOMI) is a type of intestinal ischemia without organic vascular obstruction that carries high mortality rates, making it a critical condition with a poor prognosis. This report describes a case involving a 71-year-old man with esophageal cancer who developed NOMI during chemoradiation therapy. After initiation of chemoradiation therapy with combined 5-fluorouracil and cisplatin, the patient experienced acute abdominal pain and hypotension. Imaging findings suggested NOMI of the distal ileum. Early intervention rapidly decreased his symptoms and improved his vital signs, thereby avoiding surgical treatment. Thus, the patient received conservative treatment and completed his radiation therapy as scheduled without recurrence of symptoms, although chemotherapy was discontinued. To our knowledge, this is the first reported case of chemotherapy-associated NOMI successfully managed with conservative treatment alone, highlighting the potential effectiveness of early therapeutic intervention before the condition progresses to severe ischemia.
Introduction
Nonocclusive mesenteric ischemia (NOMI) refers to intestinal ischemia without an organic obstruction of the mesenteric vessels. This condition carries an extremely poor prognosis, with a reported mortality rate of ~50% due to complications such as intestinal necrosis.1-3 Although acute abdominal pain is a common symptom of this disease, it may be absent in some cases.2,3 This lack of specific clinical signs makes early diagnosis challenging, a problem compounded by the potential life-threatening outcomes of delayed diagnosis. Recently, several cases of patients developing NOMI during chemotherapy have been reported, raising awareness of its significance as a potential adverse event of cancer treatments.
We herein report a case of NOMI that developed during chemoradiation. Our patient was successfully managed through conservative treatment. To the best of our knowledge, only 4 cases of NOMI developing during chemoradiation have ever been reported in the literature.4-7
Case Report
A 71-year-old man with hypertrophic cardiomyopathy and hypertension was referred to our hospital after an upper gastrointestinal endoscopy revealed suspected superficial esophageal cancer in the middle thoracic esophagus during a routine medical checkup. A biopsy confirmed the presence of squamous cell carcinoma with a clinical depth of T1a, and a systemic workup revealed no metastasis. As such, endoscopic tumor resection was performed, with the pathological findings indicating T1a (m3), positive lymphovascular invasion, and negative resection margins. Given the patient’s refusal to undergo surgery as adjuvant therapy, the cancer board considered him a candidate for chemoradiation. Radiation therapy was planned as 40 Gy in 20 daily fractions of 2 Gy each, delivered over 4 weeks, 5 fractions/week, using 3-dimensional conformal radiation therapy applied in anteroposterior and oblique 4-port fields. Figure 1 illustrates the radiation field. Chemotherapy was planned to be composed of cisplatin (CDDP, 75 mg/m2 as a rapid intravenous infusion after rehydration on day 1) and 5-fluorouracil (5-FU, 1000 mg/m2/24 hours in a 96 hours continuous infusion on days 1-4), constituting one 28-day cycle.

The opening shape of the multileaf collimator for irradiation to the patient with esophageal cancer. 40 Gy in 20 fractions of radiation therapy was administered with concurrent chemotherapy consisting of 5-fluorouracil and cisplatin.
On the day of admission for treatment induction, the patient exhibited significant limb edema and weight gain, raising concerns about potential heart failure. As such, a diuretic was administered on the morning of the second day of treatment. However, sometime after irradiation on the same day, the patient developed sudden onset abdominal pain. His vital signs showed a blood pressure of 62/39 mmHg, pulse rate of 40/minute, oxygen saturation of 95% on room air, temperature of 35.9 °C, and respiratory rate of 20/minute.
Early diagnosis was challenging because inflammatory markers were not markedly elevated and abdominal symptoms were nonspecific at presentation. At the time of symptom onset, laboratory tests revealed an elevated serum lactate level, along with leukocytosis with neutrophil predominance, while C-reactive protein levels were not elevated at that time (Table 1). Arterial blood gas analysis demonstrated hypocapnia and reduced bicarbonate levels, indicating a mixed acid-base disturbance, which was likely attributable to hyperventilation associated with acute abdominal pain.
Temporal Changes in Laboratory Findings.

Abbreviation: “-”: not measured.
Arterial blood gas analyses were performed under room air conditions. Serum lactate levels decreased after stabilization; no further serial measurements were available.
To stabilize the patient’s hemodynamic status, a rapid intravenous infusion of 500 mL of normal saline was administered, followed by continuous infusion of noradrenaline. In addition, atropine was administered intravenously. Subsequently, hypotension and bradycardia were corrected, and the patient’s vital signs stabilized; therefore, the noradrenaline infusion was discontinued on the same day.
His vital signs showed a blood pressure of 105/72 mmHg, pulse rate of 77/minute, oxygen saturation of 98% on room air, and respiratory rate of 12/minute. For abdominal pain control, intravenous acetaminophen was administered, after which the symptoms gradually resolved, and the patient was managed conservatively.
Dynamic contrast-enhanced computed tomography (CT) demonstrated gas in the intrahepatic portal vein and the superior mesenteric vein, along with the distal ileal wall thinning, suggesting ischemic bowel necrosis (Figure 2). No thrombi were visible within the vessels, consistent with the characteristics of NOMI. Differential diagnoses included acute mesenteric arterial thrombosis and acute inflammatory enteritis; however, these were considered less likely based on the CT findings, laboratory data, and clinical course, as the mesenteric arteries were patent without evidence of thrombosis and the bowel changes were suggestive of ischemia rather than primary inflammation.

Computed tomography images showing characteristic findings of ischemic bowel necrosis: (a) gas within the intrahepatic portal vein (arrows), (b) gas in the superior mesenteric vein (arrow), and (c) marked thinning of the bowel wall in the distal ileum (arrows). As no organic obstruction of the mesenteric vessels was identified, the patient was considered to have developed nonocclusive mesenteric ischemia.
A follow-up contrast-enhanced CT the next day showed that the gas in the portal and the superior mesenteric vein had resolved and that the previously thinned ileal wall was now edematous (Figure 3). This clinical course was consistent with an improvement in NOMI. During the clinical course, no marked increase in inflammatory markers was observed (Table 1). The white blood cell count, which was elevated at symptom onset, gradually decreased, while C-reactive protein levels remained low without a clear elevation. Follow-up arterial blood gas analysis demonstrated normalization of the acid-base status, and the D-dimer level was not elevated. To illustrate temporal changes in fluid balance, daily fluid intake and urine output, together with changes in body weight, are shown in Figure 4. Following initiation of 5-FU/CDDP (FP) therapy, excessive fluid intake was observed, with a transient body weight increase of 2.5 kg within the first 24 hours. The patient was kept fasting until lunch on day 6 after onset, during which time peripheral parenteral nutrition was provided.
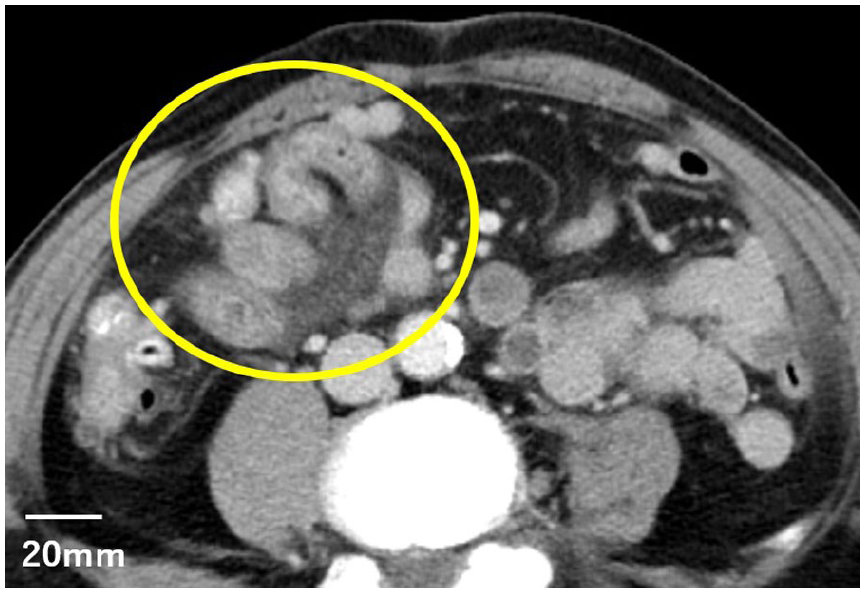
Computed tomography images on the day following symptom onset showing edematous thickening of the distal ileum wall (circle), which was previously thin at the last CT. This clinical course was consistent with the improvement of nonocclusive mesenteric ischemia.

Timeline of the clinical course showing daily fluid intake, urine output, and body weight changes.
Chemoradiation therapy was withheld on the day after onset, and radiation therapy alone was resumed on the following day (Figure 4). Radiation therapy was subsequently well tolerated and completed without further interruption.
Discussion
Although the exact pathogenesis of NOMI remains unclear, one of the primary mechanisms for its development is thought to involve a reduction in cardiac output and circulating plasma volume, which triggers excessive responses from endogenous hormones and sympathetic nerves. This phenomenon promotes peripheral vasoconstriction and reduced blood flow to the intestines.1,3,8 The major risk factors for NOMI include conditions that impact circulatory dynamics, such as advanced age, cardiovascular disease, dehydration, bleeding, dialysis, and intake of cardiovascular medications and diuretics.1,8 In the current case, diuretic administration in an elderly patient with cardiovascular disease likely caused a rapid reduction in his circulating plasma volume, contributing to NOMI development.
According to the Japanese Society of Abdominal Emergency Medicine, NOMI is characterized by ischemic damage to the intestinal tract’s vascular territories despite the absence of an organic obstruction in the mesenteric vessels. 9 Typical CT findings of NOMI include various imaging features suggestive of intestinal ischemia, such as edematous changes or thinning of the bowel wall, pneumatosis intestinalis, and portal venous gas, in the absence of thrombus within the mesenteric arteries. 10 The imaging findings of the current case align with this concept.
Although specific clinical signs are often absent in the early stages of NOMI, symptoms such as abdominal pain and gastrointestinal bleeding may manifest as ischemia progresses.8,9 When intestinal ischemia becomes irreversible, surgical bowel resection becomes necessary. Thus, early diagnosis of NOMI is necessary to facilitate rapid treatment and avoid invasive surgery. Moreover, distinguishing the characteristic symptoms and image findings of NOMI, such as in the current case, is critical.
In fact, the patient was able to avoid surgery given that prompt administration of vasoactive agents and fluids rapidly restored blood flow to the ischemic bowel.
Table 2 summarizes previous reports on NOMI developing during chemotherapy. Notably, a majority of cases involved the use of platinating agents, 5-FU, and taxanes in patients with head and neck or esophageal cancers. CDDP has been implicated in most of the presented cases, with 8 out of 9 cases also involving 5-FU, similar to the current case. Additionally, 5 of these 8 cases involved docetaxel (DOC).
Previous Reports of NOMI Development During Chemotherapy.
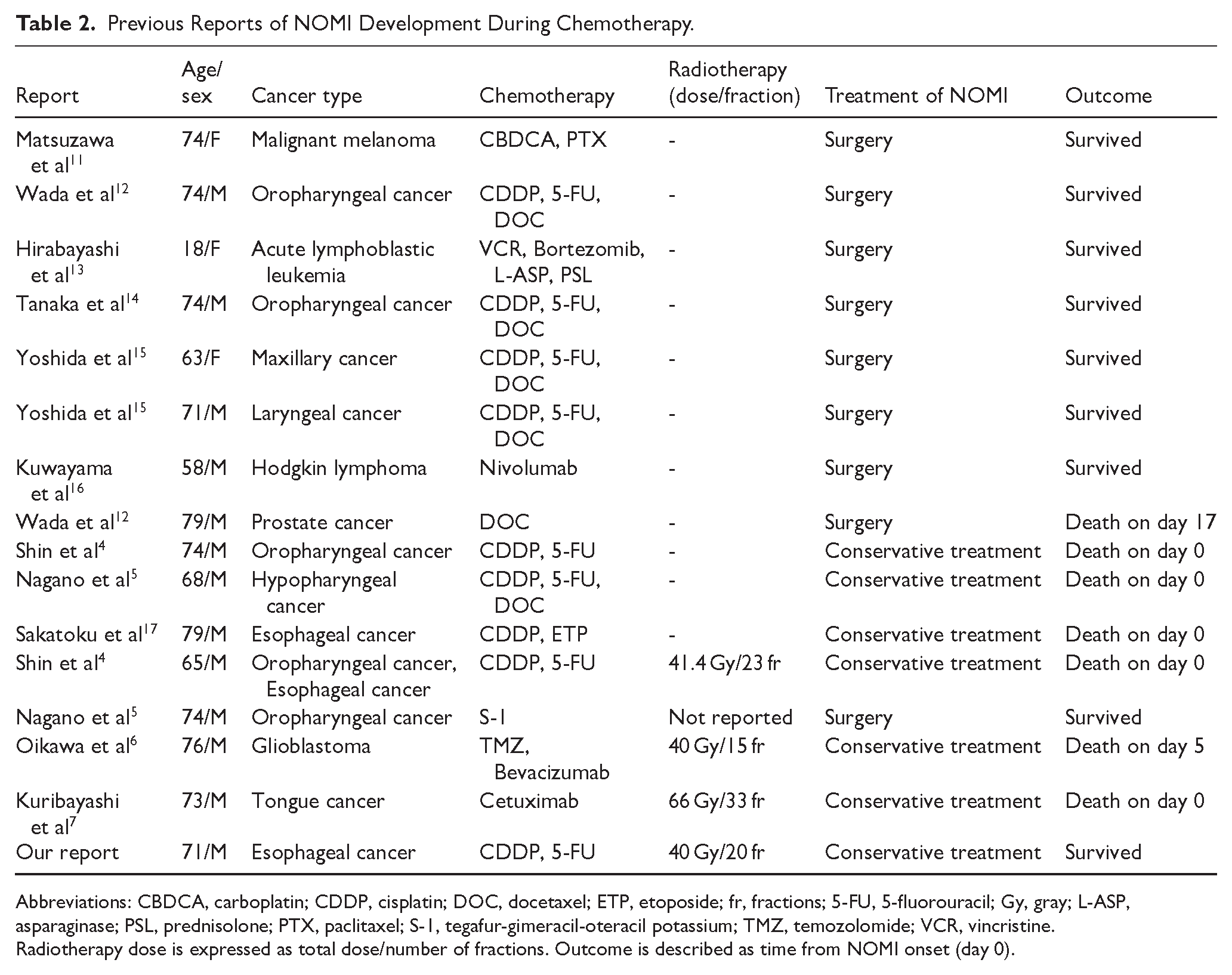
Abbreviations: CBDCA, carboplatin; CDDP, cisplatin; DOC, docetaxel; ETP, etoposide; fr, fractions; 5-FU, 5-fluorouracil; Gy, gray; L-ASP, asparaginase; PSL, prednisolone; PTX, paclitaxel; S-1, tegafur-gimeracil-oteracil potassium; TMZ, temozolomide; VCR, vincristine.
Radiotherapy dose is expressed as total dose/number of fractions. Outcome is described as time from NOMI onset (day 0).
Previous studies have discussed the mechanism behind the development of NOMI during FP therapy. Accordingly, researchers have posited that 5-FU-induced suppression of cell division, along with immunosuppression, contributes to intestinal mucosal injury and NOMI development. 4 Furthermore, the significant fluid load required during FP therapy may trigger fluid imbalance and intestinal damage due to rapid osmotic shifts. 18
NOMI has also been reported to occur in association with DOC. One previous study suggested that myelosuppression, a common adverse event of DOC, may also play a role in NOMI development, similar to 5-FU. 12
In prior cases, conservative management was reserved for patients deemed unfit for surgery due to poor general condition or for those who declined surgery, and no survivors were reported. A favorable outcome with conservative therapy, as in the present case, has not been documented in those reports. In most prior cases, patients had fever and laboratory evidence of systemic inflammation at onset or had preceding symptoms such as diarrhea. By contrast, our patient exhibited almost none of these findings, suggesting that NOMI was recognized at an earlier stage before progression to severe disease. It is likely that early diagnosis enabled prompt hemodynamic optimization, allowing recovery with conservative treatment.
In the present case, FP therapy was administered to a patient with a history of hypertrophic cardiomyopathy. The FP therapy is one of the standard treatments in Japan as an additional therapy following endoscopic resection for T1a esophageal cancer. Although 5-FU is generally known to have cardiotoxicity, the patient was in good general condition, and its use was determined at the discretion of the attending medical oncologist. The FP regimen required the infusion of over 3000 mL of intravenous fluids on the first day of treatment, which worsened the patient’s edema and necessitated diuretic administration. According to prior reports, splanchnic venous capacitance is reduced in patients with heart failure, and, as a result, even small volume shifts may be redistributed centrally, compromising mesenteric perfusion. 19 Therefore, the rapid changes in circulating plasma volume caused by this fluid shift and subsequent diuresis likely contributed to the development of NOMI in our patient. In particular, among patients with hypertrophic cardiomyopathy, excessive reduction in preload may lead to hemodynamic instability due to impaired ventricular filling and reduced cavity size; thus, diuretic administration in this case may have contributed to hemodynamic deterioration. 20 As such, administering chemotherapy to patients with underlying cardiac conditions requires careful monitoring to anticipate and manage potential NOMI development.
Footnotes
Ethical Considerations
This case report is a retrospective medical record review.
Consent to Participate
Written informed consent was obtained from the patient before initiating the treatment.
Consent for Publication
Written informed consent was obtained from the patient to publish this case report and any accompanying images. A copy of the written consent is available for review by the editor-in-chief of this journal.
Author Contributions
S.I. designed the study and prepared the manuscript. S.W., S.N., K.K., T.K., K.M., N.A., A.H., and O.D. contributed to the data acquisition. G.S., H.Y., and K.Y. supervised the study. All authors have read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI grant no JP24K10789 (grant-in-aid for scientific research).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
