Abstract
Background
Imageless robotic-assisted total knee arthroplasty (TKA) relies on intraoperative landmark acquisition and gap balancing to guide implant positioning and polyethylene insert selection. While prior studies have demonstrated high accuracy in achieving planned alignment and resection targets, limited data exist evaluating the predictability of polyethylene insert thickness using standardized pre-resection planning strategies. This study evaluated the accuracy of a standardized 10-mm polyethylene planning strategy in imageless robotic-assisted TKA and assessed whether deviation from the planned insert thickness was associated with postoperative interventions.
Methods
A retrospective review of a prospectively maintained, single-surgeon series of 400 primary TKAs performed using the imageless ROSA robotic system between 2021 and 2023 was conducted. A kinematic alignment strategy targeting a balanced 22-mm flexion and extension gap corresponding to a planned 10-mm insert was used in all cases. Exact accuracy was defined as a final 10-mm insert; acceptable accuracy was defined as ±1 mm (10–11 mm). Associations between insert thickness, intraoperative re-resection, manipulation under anesthesia (MUA), revision, and learning curve effects were analyzed.
Results
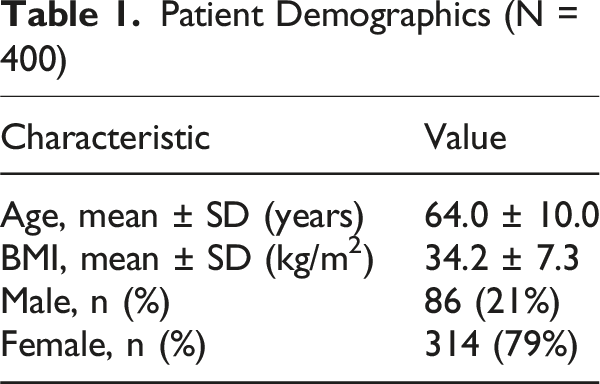
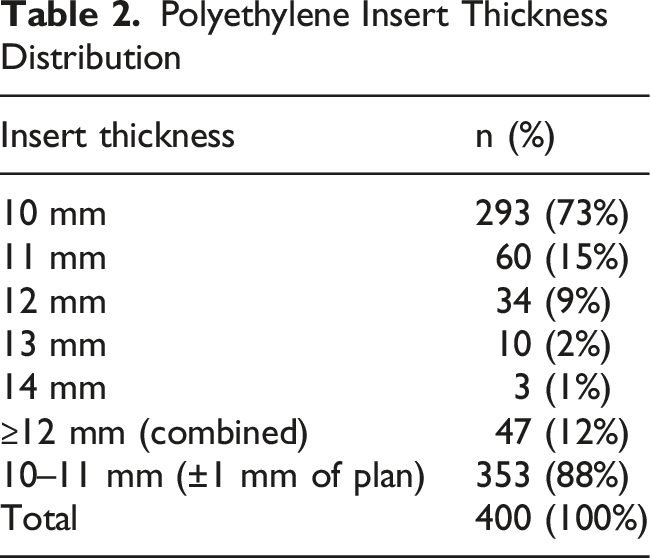
A 10-mm insert was used in 73% of cases, and 88% achieved insert thickness within ±1 mm of the planned target. Intraoperative re-resection occurred in 8% of cases, with 94% consisting of 2-mm adjustments. The overall revision rate was 5.8%, and the MUA rate was 10.3%. No statistically significant association was identified between insert thickness, re-resection, and revision or MUA (P > 0.05). Learning curve analysis of the first 30 cases demonstrated similar insert distribution and intervention rates.
Conclusion
Imageless robotic-assisted TKA using standardized 10-mm pre-resection planning demonstrated reproducible polyethylene insert selection. Deviation from planned thickness and minor intraoperative re-resections were not associated with increased postoperative intervention, supporting the reliability of imageless robotic workflows in primary TKA.
Keywords
Introduction
Robotic-assisted total knee arthroplasty (TKA) has gained increasing adoption as surgeons seek improved reproducibility in bone resection, alignment, and soft-tissue balancing. Currently, two primary robotic platforms are utilized: image-based systems, which rely on preoperative CT or MRI imaging to generate patient-specific models, and imageless systems, which depend on intraoperative acquisition of anatomic landmarks to create virtual reference planes. 1
In both platforms, final resection levels and implant positioning are guided by intraoperative assessment of ligamentous balance and implant geometry. When utilizing a functional or kinematic alignment strategy, predictable flexion and extension gap balancing is essential to optimize joint stability and postoperative kinematics.2,3 However, imageless systems rely on surgeon-dependent landmark acquisition, which may introduce variability in gap assessment and potentially affect polyethylene insert selection.1,4
Prior studies have demonstrated high accuracy of imageless robotic systems in achieving planned alignment and resection targets.4,5 However, limited data exist evaluating the predictability of polyethylene insert thickness when using standardized pre-resection planning strategies.
The primary aim of this study was to determine the accuracy of pre-resection planning using a standardized 10-mm polyethylene insert thickness in imageless robotic-assisted TKA. Secondary aims included evaluating whether deviation from the planned insert thickness was associated with manipulation under anesthesia (MUA), revision surgery, or influenced by the surgeon’s initial learning curve.
Methods
A prospectively collected, single-surgeon database of primary robotic-assisted TKA cases performed using the imageless ROSA Knee System (Zimmer Biomet, Warsaw, Indiana) was retrospectively reviewed. All procedures were performed between 2021 and 2023 at a tertiary academic center by a fellowship-trained arthroplasty surgeon. Institutional review board approval was obtained.
Inclusion Criteria
• Adults undergoing primary robotic-assisted TKA • Use of the imageless ROSA Knee System • Minimum follow-up of 1 year
Exclusion Criteria
• Revision TKA • Conversion TKA • Cases with incomplete follow-up
The surgeon’s initial 30 cases using the robotic platform were included to assess potential learning curve effects.
Surgical Technique
A standard medial parapatellar approach was used in all cases.
Femoral and tibial resections were adjusted iteratively based on real-time intraoperative laxity feedback. Final polyethylene thickness was determined following trialing and confirmation of balanced flexion and extension gaps. All cases utilized a posterior-stabilized (PS) polyethylene insert design.
Outcome Measures
The primary outcome was the accuracy of polyethylene insert thickness prediction using a standardized 10-mm pre-resection planning strategy. Polyethylene inserts were categorized as 10 mm, 11 mm, and ≥12 mm (12–14 mm). Exact prediction accuracy was defined as final implantation of a 10-mm insert. Acceptable prediction accuracy was defined as implantation within ±1 mm of the planned thickness (10–11 mm).
Secondary outcomes included intraoperative re-resection, manipulation under anesthesia (MUA), and all-cause revision surgery. Intraoperative re-resection was defined as additional femoral and/or tibial bone resection performed after initial planned cuts to optimize flexion-extension gap balancing. The magnitude and location (femoral vs tibial) of re-resections were recorded.
Manipulation under anesthesia was defined as any postoperative procedure requiring anesthesia to address arthrofibrosis. Revision surgery was defined as any return to the operating room involving exchange or removal of 1 or more prosthetic components for any indication, including stiffness, instability, loosening, or other mechanical complications. Demographic variables collected included age, sex, and body mass index (BMI).
Subgroup analyses evaluated associations between polyethylene insert thickness category and postoperative interventions. A learning curve analysis compared the first 30 cases to subsequent cases.
Statistical Analysis
Descriptive statistics were calculated for demographic and clinical variables. Continuous variables were summarized as means with standard deviations. Categorical variables were reported as frequencies and percentages. Chi-square analysis was used to evaluate associations between insert thickness and postoperative interventions. Statistical significance was defined as P < 0.05.
Results
Patient Demographics (N = 400)
Polyethylene Insert Thickness Distribution
Implant Fixation and Construct Characteristics

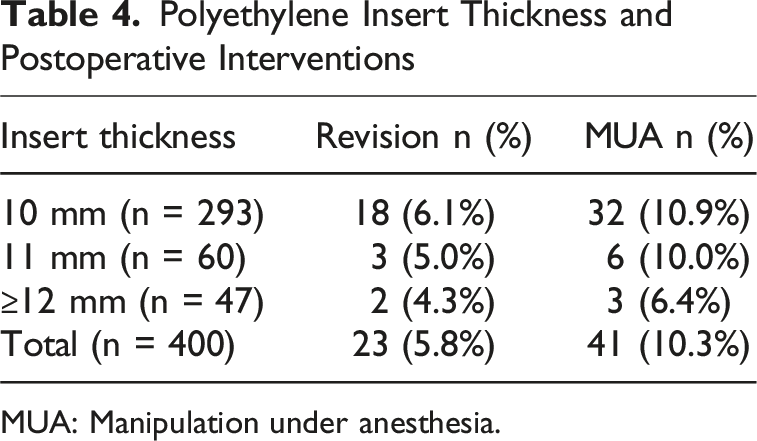
Polyethylene Insert Thickness and Postoperative Interventions
MUA: Manipulation under anesthesia.
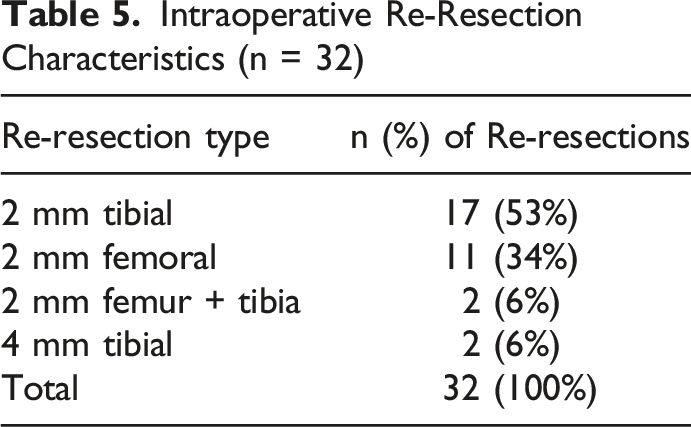
Intraoperative Re-Resection Characteristics (n = 32)
A total of 41 patients (10.3%) underwent manipulation under anesthesia (MUA). MUA occurred in 32 of 293 cases (10.9%) with 10-mm inserts, 6 of 60 cases (10.0%) with 11-mm inserts, and 3 of 47 cases (6.4%) with inserts measuring 12 mm or greater. There was no statistically significant association between polyethylene insert thickness and MUA (P > 0.05) (Table 4).
Revision Indications

Learning Curve Analysis (First 30 Cases)

Discussion
In this single-surgeon series of 400 primary total knee arthroplasties performed using an imageless robotic system, pre-resection planning with a standardized 10-mm polyethylene insert resulted in exact prediction in 73% of cases and acceptable prediction within ±1 mm in 88% of cases. These findings demonstrate reproducible polyethylene insert selection and consistent soft-tissue balancing when utilizing an imageless robotic-assisted workflow.
Prior investigations of imageless robotic systems have demonstrated high accuracy in achieving planned alignment and resection targets. Vaidya et al. reported improved component positioning accuracy using an imageless hand-held robotic system compared to conventional techniques. 4 Rossi et al. demonstrated high reproducibility of alignment parameters with robotic assistance. 6 Schrednitzki et al. confirmed accurate in vivo reproduction of planned bone resections using an imageless platform. 7 More specifically, Gamie et al., in a prospective single-surgeon series evaluating the imageless mode of the ROSA system, reported mean differences between planned and actual resection depths ranging from 0.67 to 1.10 mm across femoral and tibial landmarks, indicating a high degree of intraoperative precision. 5 While these studies primarily focused on resection thickness and alignment accuracy, the present study extends this body of literature by evaluating the predictability of polyethylene insert thickness. The 88% rate of achieving final insert thickness within ±1 mm of the planned target in this cohort is consistent with the millimeter-level reproducibility reported in prior imageless robotic investigations.4,5
Intraoperative re-resection was performed in 8% of cases to further optimize flexion-extension gap balancing. Notably, 94% of these recuts consisted of 2-mm adjustments, suggesting that re-resections primarily reflected iterative fine-tuning rather than substantial deviation from planned resections. Prior studies evaluating imageless robotic systems have demonstrated millimeter-level accuracy in reproducing planned resections, with mean deviations typically less than 1 mm.5,7 Additionally, functional and kinematic alignment philosophies emphasize incremental, millimeter-based adjustment of bone resections to achieve balanced extension and flexion gaps.2,3 The present findings are consistent with these principles, suggesting that controlled re-resection represents part of an iterative balancing workflow rather than correction of major planning error. Importantly, re-resection was not associated with increased revision rates and was not concentrated during early platform adoption. 8
Deviation from the planned insert thickness was not associated with increased rates of revision or manipulation under anesthesia. Revision rates were similar across insert thickness groups, and MUA rates did not differ significantly between exact-match and non-exact groups. These findings suggest that intraoperative adjustments guided by robotic feedback to achieve balanced gaps do not appear to compromise short-term clinical outcomes. Long-term survivorship data comparing robotic-assisted and conventional TKA have demonstrated comparable or improved outcomes with robotic assistance, further supporting the reliability of robotic workflows in achieving durable implant constructs. 9
Postoperative stiffness represented the most common indication for revision in this series. Given concerns that fixed-thickness cementless single-peg patellar components may contribute to patellofemoral overstuffing and stiffness, we evaluated revision rates according to patellar fixation type. Although stiffness-related revisions were numerically higher in the cementless single-peg subgroup compared to cemented patellae, the difference was not statistically significant. These findings suggest that postoperative stiffness in this cohort is likely multifactorial and not attributable solely to patellar fixation strategy or insert thickness selection. Stiffness following TKA has been well described as a complex and multifactorial complication influenced by patient factors, surgical technique, and implant design. 10
Subgroup analysis of the surgeon’s initial 30 cases demonstrated consistent insert distribution, MUA rates, and re-resection rates compared to later cases. Although revision rates were numerically higher early in the series, re-resections were not clustered during the learning curve. These observations suggest that polyethylene insert predictability and intraoperative refinement remain stable even during early experience with the imageless robotic system.
Collectively, these findings contribute to the evolving understanding of imageless robotic-assisted TKA by demonstrating that standardized pre-resection planning strategies can produce predictable polyethylene insert selection while allowing controlled intraoperative refinement when necessary. While prior literature has emphasized alignment and resection accuracy, the present study highlights the reproducibility of gap balancing and implant construct parameters, reinforcing the reliability of imageless robotic systems in contemporary TKA practice.
Future studies should further evaluate the relationship between polyethylene insert selection, re-resection frequency, and long-term patient-reported outcomes and implant survivorship. Comparative analyses between imageless and image-based robotic platforms may also clarify whether differences in preoperative planning methodology influence insert predictability and intraoperative adjustment patterns.
This study has several limitations. First, it represents a single-surgeon experience, which may limit generalizability to other surgeons or institutions. Second, although the database was prospectively maintained, the analysis was retrospective in nature and subject to inherent selection and reporting biases. Third, detailed implant construct variables such as baseplate configuration and peg design were not systematically captured, limiting granular implant-specific subgroup analysis. Additionally, detailed radiographic assessment of patellofemoral offset and patient-reported outcome measures were not available for this analysis. Finally, no direct comparison with conventional or image-based robotic systems was performed. Future multicenter and comparative studies are warranted to validate these findings.
Conclusion
Imageless robotic-assisted total knee arthroplasty using a standardized 10-mm pre-resection planning strategy demonstrated reproducible polyethylene insert selection, with 88% of cases achieving final insert thickness within ±1 mm of the planned target. Intraoperative re-resections were infrequent and primarily consisted of small, 2-mm adjustments to optimize gap balancing. Neither deviation from planned insert thickness nor re-resection was associated with increased revision or manipulation under anesthesia. These findings support the reliability of imageless robotic systems in achieving predictable soft tissue balancing during primary TKA.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
