Abstract
Rheumatic heart disease is one of the leading causes of valvular heart disease globally, with the highest disease burden occurring in young patients in low- and middle-income countries. This review explores the traditional and more recent concepts in the pathogenesis of juvenile rheumatic mitral valve disease (JRMVD). The interaction between normal mitral valve structure and function is reviewed, followed by the pathogenesis, morphological features and echocardiographic characteristics of JRMVD. Finally, the rationale for mitral valve repair techniques favored in this challenging patient population is discussed.
Keywords
Key Points
The pathogenesis of rheumatic heart disease (RHD) has been revisited, with increasing evidence that immune-mediated connective tissue damage plays a significant role in the development of valvular heart disease
Juvenile rheumatic mitral valve disease (JRMVD) should be considered a separate entity from the chronic or burnt-out valvulitis seen in older patients, with distinctive morphological and echocardiographic features
Mitral valve (MV) annular dilatation is a prominent feature of juvenile rheumatic MV regurgitation, usually associated with posterior mitral leaflet (PML) shortening and anterior mitral leaflet (AML) prolapse resulting in a characteristic posteriorly directed jet of mitral regurgitation (MR)
Juvenile rheumatic MV regurgitation is amenable to stable and durable valve repair through established techniques of annuloplasty, leaflet patch augmentation and neochordae implantation.
Juvenile rheumatic mitral stenosis (MS) is usually pliable and amenable to balloon mitral valvotomy (BMV).
Background
Acute rheumatic fever (ARF) is considered an autoimmune process that follows initial or repeated Group A Streptococcus (GAS) infection. RHD and specifically rheumatic valve disease is the most important clinical consequence, estimated to affect 30–40 million young people globally, with most afflicted in low- and middle-income countries (LMIC's). 1 Socio-economic factors, particularly malnutrition and overcrowding, have traditionally been linked to a higher incidence of RHD since the mid-20th century. 2 More recently, the lack of adherence to secondary penicillin prophylaxis is an increasingly significant contributor. Genetic and geographic factors were considered of less significance in the past, 2 however modern understanding of the disease process indicates their importance. 3
JRMVD is still commonly seen in children between the ages of 6–16 years in LMICs, particularly in Sub-Saharan Africa. These patients may present with pure MV regurgitation (MR), pure MV stenosis (MS) or a combination (mixed MV disease—MMVD) associated with cardiac failure. Occasionally, patients present with cachexia, pyrexia, and anemia in intractable cardiac failure due to acute rheumatic carditis. Cardiac surgery in the form of MV repair or replacement is a life-saving procedure.
This review explores normal MV structure and function, followed by the pathogenesis, morphology and echocardiographic features of JRMVD, and finally the rationale for advocating specific MV repair techniques in this challenging patient population.
Interactions in Normal MV Structure and Function
The MV is a complex, finely coordinated apparatus with structural integrity, functional synchrony and harmonious interplay de rigueur for normal cardiac function. The key components of the MV are the annulus, leaflets and subvalvular apparatus including the left ventricle (LV) free wall.
The MV Annulus
The MV annulus is a nonplanar, three-dimensional saddle-shaped oval orifice that does not consist of a complete fibrous ring. The anterior MV annulus is in fibrous continuity with the aortic annulus via the aorto-mitral curtain, reinforced by the dense anterolateral and posteromedial fibrous trigones. The posterior MV annulus consists of fine tenuous fibrous strands termed the fibrous fila of Henle and extend for a short distance posterior from the trigones. 4
In contrast to the protected stable anterior MV annulus, the posterior MV annulus is susceptible to massive annular dilatation when the fibrous components are disrupted by connective tissue disorders, such as rheumatic carditis and Marfan syndrome. When LV dilatation occurs in the absence of connective tissue involvement (as in dilated cardiomyopathy or severe aortic regurgitation) MV annular dilatation is not a prominent feature. 5
The posterior MV annulus merges with the basal myocardium and left atrial (LA) wall, and is considered a tissue plane adjacent to the fibroelastic fatty tissue containing the coronary sinus and circumflex coronary artery 6 (Figure 1). This area has received great interest in the study of mitral annular disjunction.6,7 The lack of a robust posterior MV annulus contributes to the complication of LV rupture that may follow MV replacement, as well as the occurrence of sub-mitral aneurysms in this region.
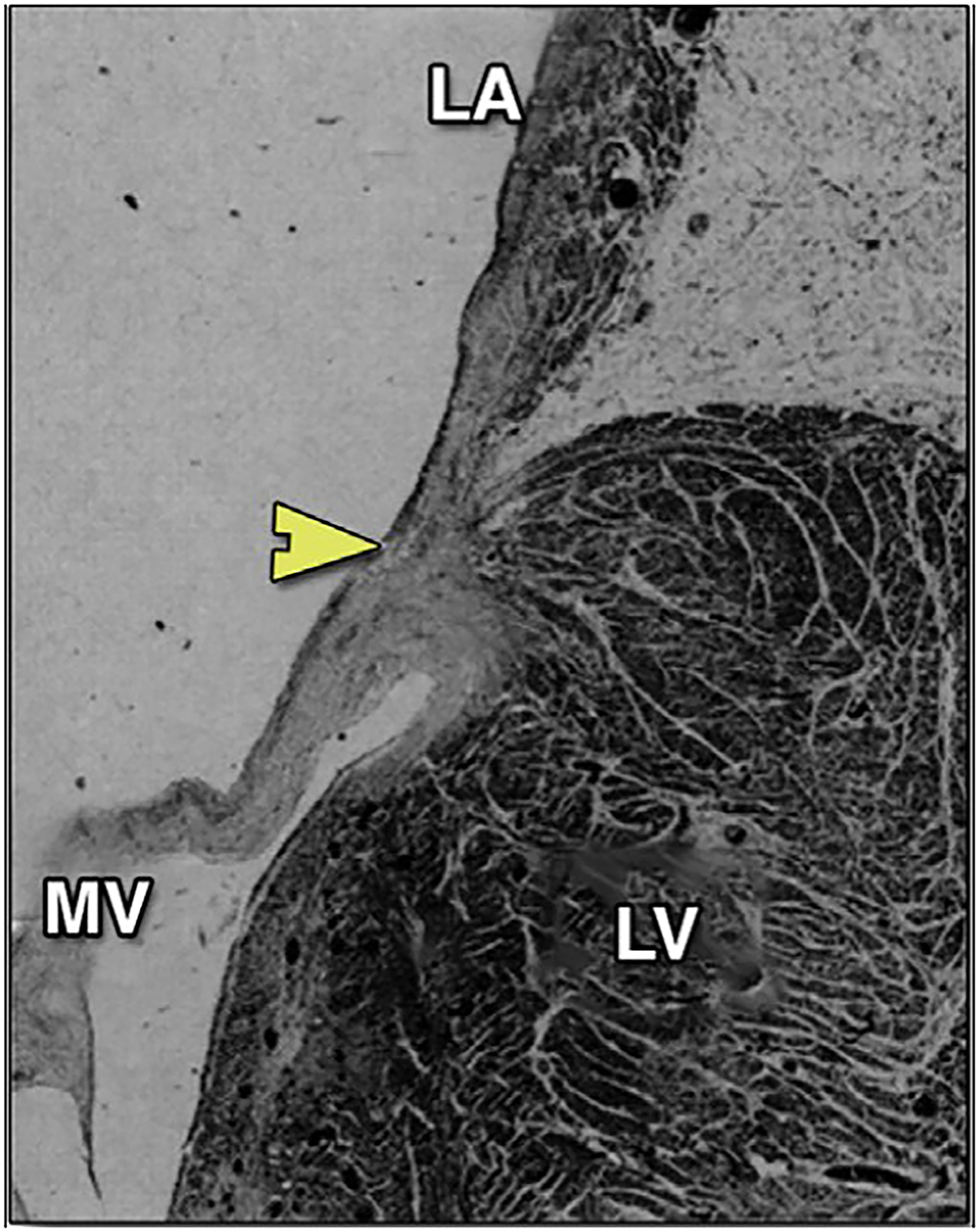
Histology section demonstrating the posterior leaflet of the MV and LA–LV interface at the posterior MV annulus (arrowhead). Reprinted with permission from Lee et al (2017). 6 MV, mitral valve; LA, left atrium; LV, left ventricle.
Functionally, the MV annulus is a dynamic structure that exhibits confirmational change in size, shape and area during the cardiac cycle.7,8 During diastole, the MV annulus dilates to facilitate LV filling, a predominantly passive process. During systole, the basal myocardium contracts and displaces the aortic-mitral curtain toward the center of the valve orifice, reducing the MV area by 30% as the anterior and posterior leaflets coapt.4,9 In systole, the transverse diameter to anterior–posterior diameter ratio is approximately 4:3. 4
MV Leaflets
The MV consist of two major leaflets. The AML is broad, trapezoid shaped and attached to the firm one-third of the MV annulus anteriorly, effectively suspended from the fibrous trigones. The PML is continuous with the LA wall and basal LV myocardium as described. The leaflets are continuous with each other at the minor commissural leaflets. Each major leaflet consists of three scallops (designated anteriorly A1, A2, A3 and posteriorly P1, P2, P3) and three zones of basal, clear and rough (free edge) with the last giving attachment to the primary chordae tendineae (CT). In the normal state, the leaflet area is 1.5–2 times the area of the MV annulus, creating an abundance of tissue available for coaptation.
Subvalvular Apparatus
CT consist of three types of fan-shaped fibrous structures that insert onto the AML and PML. Primary CT insert onto the free edge (rough zone) of the leaflet, secondary chordae are attached to the body of the leaflet, with two prominent strut chordae attached to the AML and basal CT extend from the left ventricular wall to the basal attachment of the leaflets. With normal MV structure and function, the primary CT are under virtually no tension throughout the cardiac cycle10,11 and solely function to align the delicate leaflet edges to facilitate coaptation by compression during ventricular systole.
The two main papillary muscles (PMs) are the anterolateral and posteromedial, often with multiple heads. The CT to the A1/A2 and P1/P2 scallops arise from the anterolateral PM and similarly the CT to the A2/A3 and P2/P3 scallops originate from the posteromedial PM. The LV free wall, PM and CT function as a unit to ensure MV competence at rest and with increased cardiac output states.
MV Competence
Physiologically, the MV allows unimpeded and mostly passive filling of the LV during diastole. This is facilitated by dilatation of the MV annulus, wide distraction of the MV leaflets, and normal mobility and relaxation of the submitral apparatus (CT and PMs) directing the inflow of blood toward the apex of the LV during diastole. In sinus rhythm, LV filling is aided by LA contraction.
During LV systole, two geo-spatial design characteristics ensure that the MV is perfectly suited to prevent the regurgitation of blood into the LA.
The first characteristic is the spatial orientation of the aortic and MV annuli, which lie at an angle of 130 degrees to each other. 5 The MV orifice is relatively anterior and on the “sidewall” of the LV, while the aortic orifice is superior. The difference in inflow and outflow angulation maintains the main vector of force—and hence blood flow—during ventricular contraction directed toward the aortic valve annulus.
The second mechanism, termed the “keystone effect,” refers to the compressive force applied between the coapting surfaces of the AML and PML. 5 At the end of diastole, the leading edges of the AML and PML drift toward each other, passively aligned by the primary CT. During systole, the force of ventricular contraction is applied along the opposite sides of the coapting leaflet surfaces, creating a tight seal by compression. A common misconception is that the MV leaflets function by rotation akin to swing doors, rather than the compression mechanism described. The leaflet tips and point of coaptation remain at a relatively constant depth within the LV cavity throughout the cardiac cycle (Figure 2).
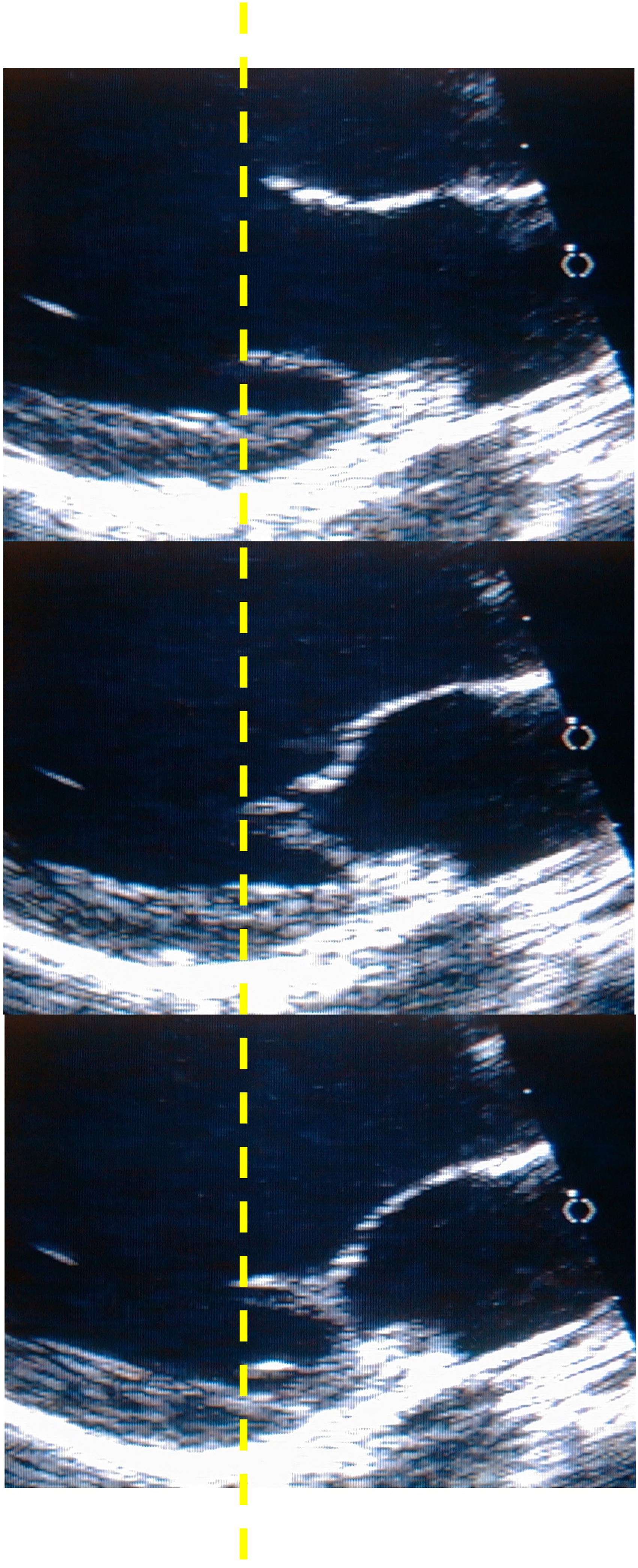
The dashed line indicates the constant position of the MV leaflet tips within the LV cavity during the cardiac cycle in diastole, early systole and late systole (top to bottom). MV, mitral valve; LA, left atrium; LV, left ventricle.
In a study of fluid-structure interactions involving the MV leaflets, it was demonstrated that under normal conditions of leaflet collagen fiber volume fraction, fiber stiffness, fiber splay and isotropic stiffness the MV leaflets are under minimal stress. Even with significant alteration of the collagen fiber volume and stiffness, the simulated valve demonstrated increased leaflet stress, yet remained competent due to the “keystone effect.”5,12
The Pathogenesis of RHD
ARF is the result of an autoimmune response to pharyngitis caused by infection with GAS. 13 The streptococcal M protein is specifically implicated in the pathogenesis of ARF, and certain M protein serotypes of streptococcus are considered rheumatogenic strains. 14
Host-streptococcal interactions resulting in ARF may be determined by the host susceptibility to the streptococcal strain, or the genetic makeup of the strain itself. Polymorphisms in genes coding for immune-related proteins have been associated with ARF susceptibility, in particular Class II human leukocyte antigen alleles. The genetic makeup of the streptococcal strain may in turn be influenced by exposure to penicillin therapy and prophylaxis over decades. 13
Two philosophies dominate theory on the pathogenesis of rheumatic carditis that follows ARF, and both are likely to play a role. 14 The original theory is based on the concept of molecular mimicry between an alpha-helical coiled coil structure of the streptococcal M protein and intramyocellular proteins myosin and tropomyosin. 15 A more recent neo-antigen theory suggests collagen-mediated heart valve damage, with primary damage of the subendothelial collagen matrix akin to Goodpasture syndrome or Alport syndrome. 16 It is likely that both doctrines are important and not mutually exclusive.17,18 The role of neoangiogenesis and lymphangiogenesis has also been emphasized in the pathogenesis of RHD. 19
The molecular mimicry and neo-antigen pathways in the pathogenesis of RHD are outlined in Figure 3. 14
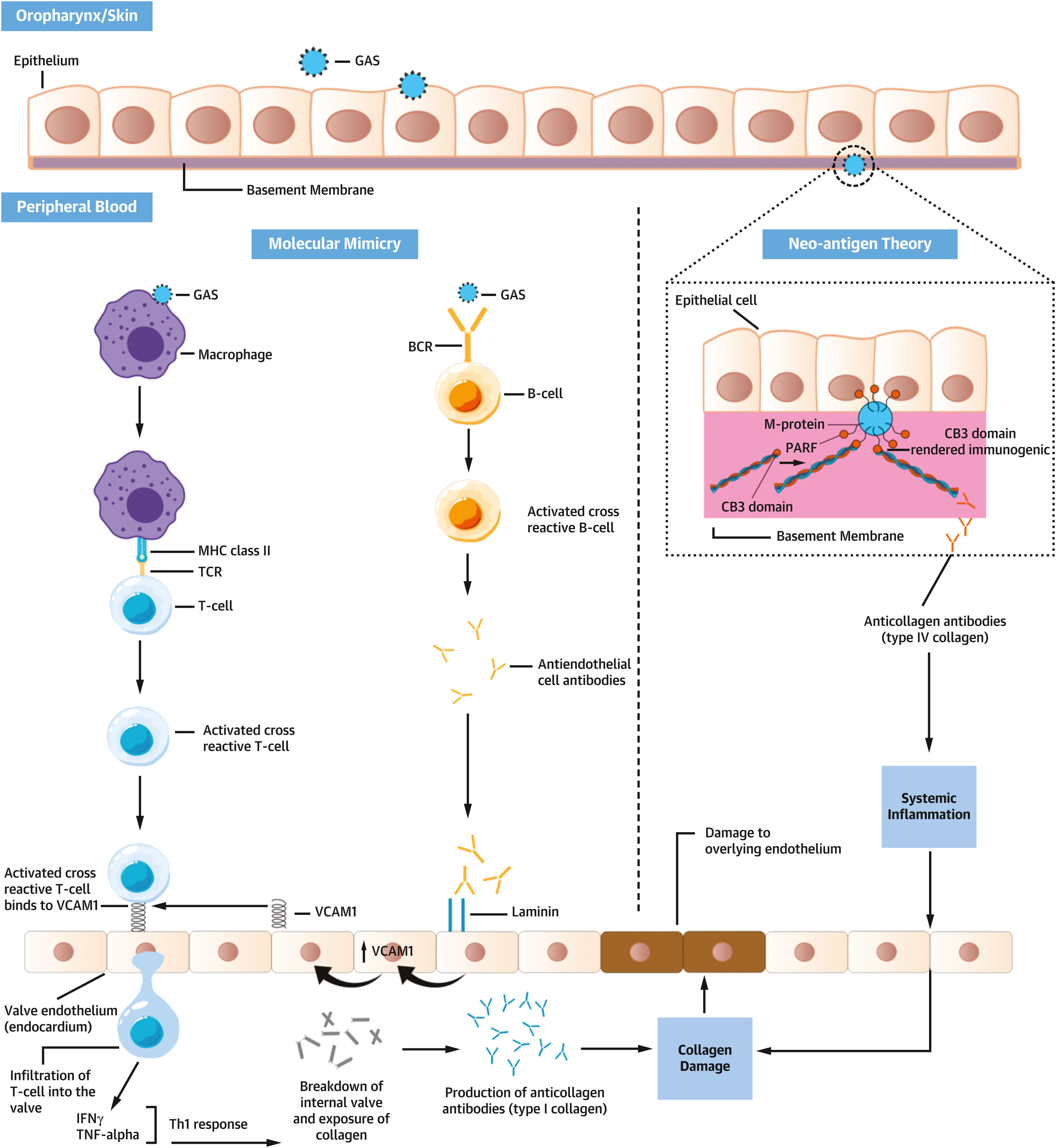
Molecular mimicry and the neo-antigen theory are both postulated to play a role in the pathophysiology ARF, either independently or as interlinked processes. ARF, acute rheumatic fever; GAS, Group A Streptococcus; BCR, B-cell receptor; IFN, interferon; MHC, major histocompatibility complex; TCR, T-cell receptor; TNF-alpha, tumour necrosis factor alpha; VCAM-1, vascular cell adhesion molecule 1.
As early as 1896, pathology specimens of rheumatic carditis demonstrated dilation of the left heart and the left atrioventricular orifice. Aschoff bodies, the pathognomonic histological feature of rheumatic carditis were originally described in the muscle near the left atrioventricular junction and basal left ventricular myocardium, as well as subendocardial and subpericardial layers giving rise to the concept of a pancarditis (or simply carditis). 20
Rheumatic carditis may result in pericarditis, myocarditis and valvulitis.
Pericardial involvement may range from a minor pericardial reaction with flimsy adhesions, to an acutely inflamed fibrinous pericarditis that may occasionally progress to chronic constrictive pericarditis.
Rheumatic myocarditis is evident by the presence of Aschoff bodies in the myocardium, 20 however—unlike other forms of myocarditis—cardiac myocyte necrosis is not a feature and cardiac troponin I levels are not elevated. 21 Congestive cardiac failure, LV dilatation and LV systolic dysfunction are only features of acute rheumatic myocarditis when valve dysfunction in the form of severe aortic or MR is present.5,22–25 LV dysfunction persists only in the presence of severe valve regurgitation, and appears unrelated to direct myocardial involvement. With resolution of valve dysfunction, LV dysfunction does not persist. 26
MV involvement is a consistent feature of rheumatic valvulitis, and valve damage is initiated by circulating autoantibodies that bind to the endothelial surface of the valve leading to increased expression of vascular cell adhesion protein 1 (vascular cell adhesion molecule 1—VCAM-1). Activated endothelium facilitates the infiltration of T lymphocytes into the valve subendothelium, exposing components of the extracellular matrix and thus anti-collagen antibodies are produced. 27
Brain natriuretic peptide is produced during cardiac remodeling that occurs from rheumatic MR in children and adolescents, providing a potential marker of cardiac involvement in RHD (Figure 4).13,28 Evidence of recurrent carditis confers a risk for persistence of valve regurgitation after ARF. 29 Effective antibiotic and anti-inflammatory treatment of acute rheumatic carditis is therefore critical to reduce persistence of valve disease.30,31
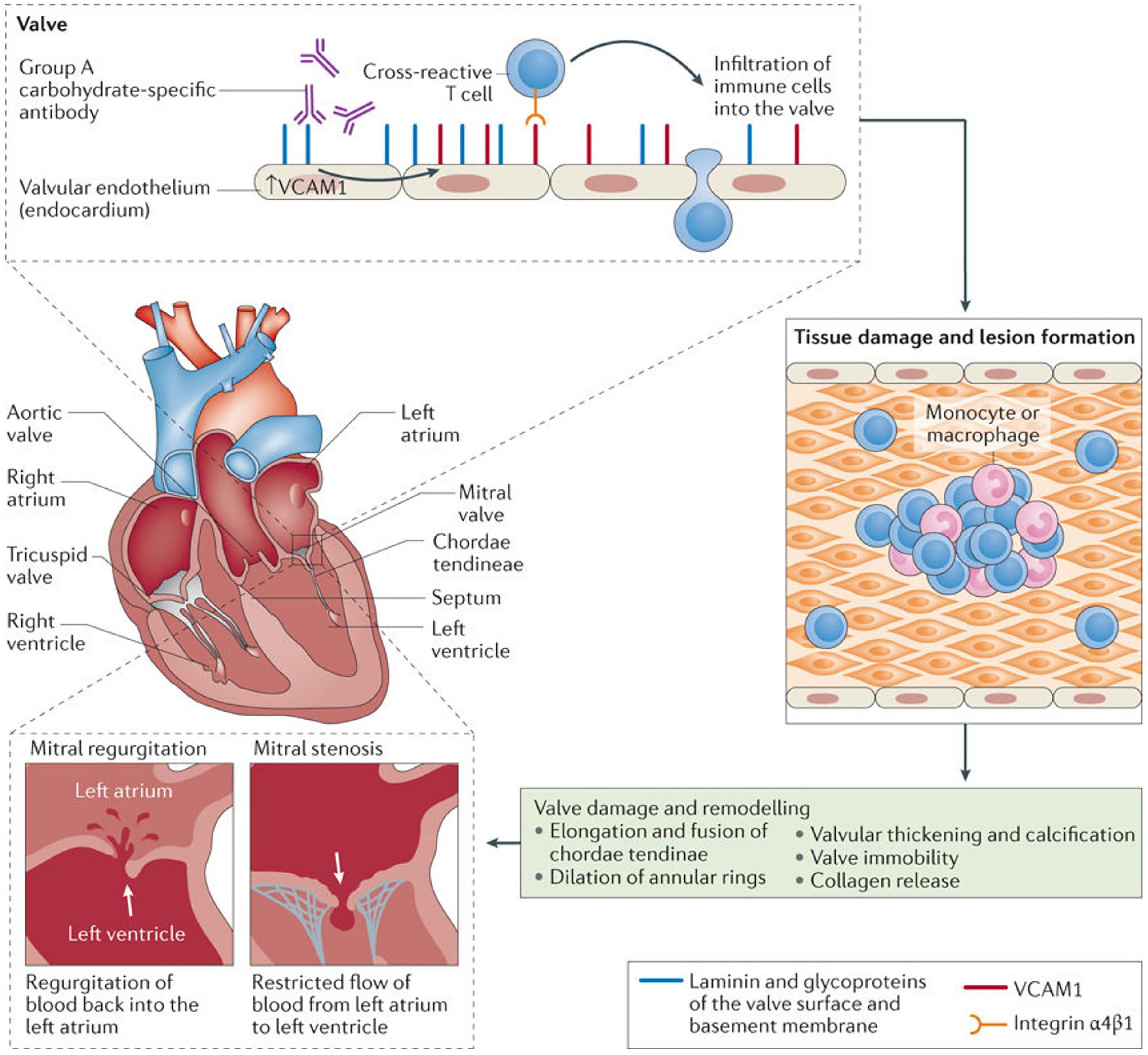
The heart is affected by antibodies (generated by B cells) against the group A carbohydrate binding to the surface of the valve and upregulating VCAM-1 on the surface of the valve endothelium. The upregulation of VCAM-1 allows T cells expressing VLA4 to adhere to the endothelium and to extravasate into the valve. The inner valve becomes infiltrated by T cells, primarily CD4+ T cells, and Aschoff bodies or granulomatous lesions form underneath the endocardium. Damage to the endothelium and infiltration of T cells into the valve remodels the valve structure, including the chordae tendineae, with malformation of the valve leading to regurgitation or stenosis of the valve. Breakdown of the valve releases collagen and results in further immune-mediated damage to the valve. VCAM-1: vascular cell adhesion molecule 1; VLA4: integrin α4β1.
Morphological Changes in JRMVD
Juvenile Rheumatic MR
The most prominent morphological feature seen in juvenile rheumatic MR is MV annular dilatation, which is present in over 90% of cases of pure MR.2,27,32–35 Severe MV annular dilatation is the usual, and the MV orifice may be up to twice the expected dimensions for the child.5,33 Valvulitis occurs to a lesser degree in young patients with pure MR, with variable leaflet thickening and small punctate vegetations evident on the leaflet edges. 36
Dilatation of the posterior mitral annulus causes tethering and retraction of the PML, with loss of the scallops resulting in progressive reduction of length of coaptation with the AML and failure of the keystone competence mechanism. Progressive dilatation of the LV creates tethering and a lateral direction of pull on the PML chordae. A variable degree of valvulitis contributes to this tissue loss, and the combination of a dilated MV annulus with paucity of leaflet tissue creates MR.
Severe annular dilatation in JRMVD results in reconfiguration of the usual spatial geometry between the mitral and aortic valve annuli. The normal angle of 130 degrees is now increased to 170 degrees. The loss of the keystone results in failure of leaflet coaptation, and the delicate leading edge primary chordae to the AML are exposed en-face to the force vector of LV ejection, placing them under non-physiological tension (Figure 5). This tension on chordae afflicted with the rheumatic process results in progressive elongation and subsequent chordal rupture, resulting in pseudo-prolapse or true prolapse of the AML.
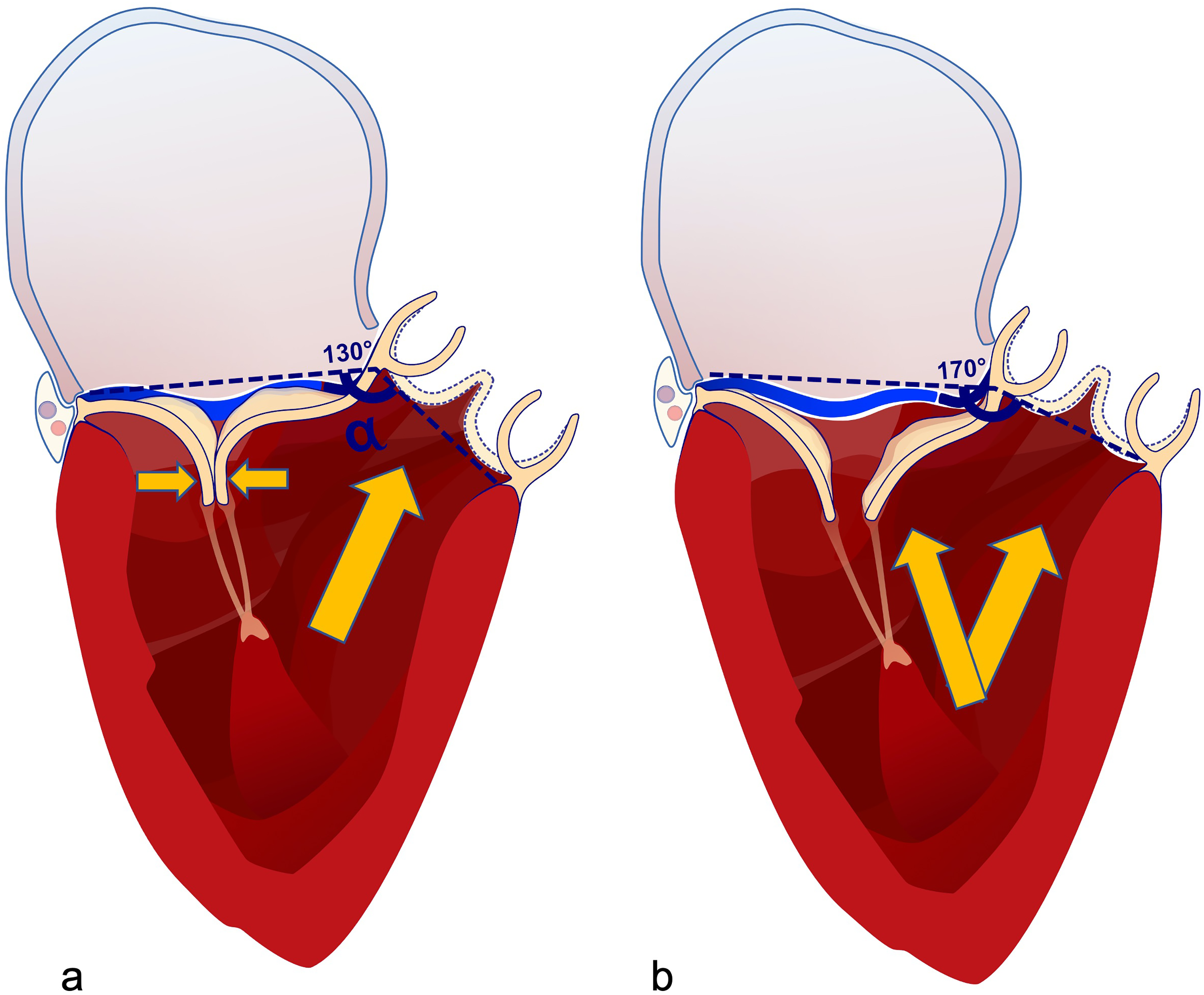
The normal spatial orientation of the aortic and MV annuli, which lie at an angle (α) of 130 degrees to each other (A). The main vector of force during ventricular contraction is directed toward the aortic valve annulus (large arrows). Compression of the MV leaflets creates a “keystone” and maintains valve competence (small arrows). In JRMVD with MR and MV annular dilatation, the angle (α) between the aortic and MV annuli is increased to 170 degrees (B). The sail-like AML is exposed en-face to the force vector of LV ejection (large arrows), placing the chordae under tension and susceptible to elongation and rupture. MV, mitral valve; JRMVD, juvenile rheumatic mitral valve disease; MR, mitral regurgitation; AML, anterior mitral leaflet.
Severe MR and progressive dilatation of the LA, MV annulus and LV results in distortion of the subvalvular apparatus, with lateral deviation of the PMs and chordae, worsening the MR. 37
Morphologically, severe MV annular dilatation causes the normal saddle shape of the MV annulus to become planar. 38 Functionally, the dilated MV annulus in JRMVD exhibits dyskinesia with paradoxical dilatation of the annulus evident in systole (Figure 6).6,39 This feature is seen in Barlow and Marfan syndromes and absent in dilated or ischemic cardiomyopathy, and is consistent with annular connective tissue disruption in JRMVD.40,41 Classical mitral annular disjunction is not a feature of JRMVD.
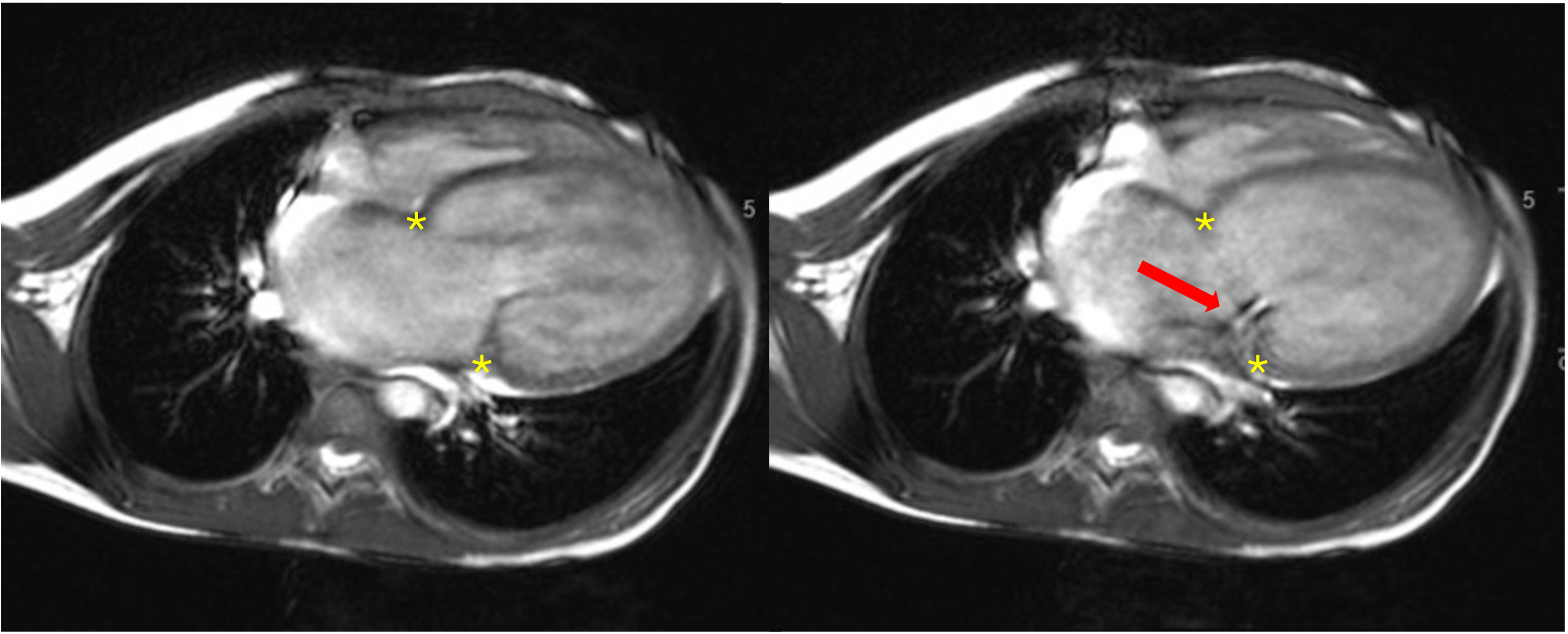
Cardiac MRI of a 10-year-old patient with JRMVD and severe MR in diastole (A) and systole (B). During systole the MV annulus (asterisks) paradoxically enlarges from 36 mm to 40 mm, with a posterolaterally directed jet of MR indicated by the arrow.
Juvenile Rheumatic MS
Rheumatic MV valvulitis may predominate over MV annular dilatation and present with the clinical features of pure or dominant MS.2,32–35 Nevertheless, MV annular dilatation is present in 30% of cases of pure MS. 27
Juvenile rheumatic MS occurs with early commissural fusion, and the free edges of the fused leaflets extend well down into the LV cavity. In some regions, rheumatic MS may present at a young age with features of atrial fibrillation, LA thrombi and airway compression due to massive LA dilatation. 42
An early study of rheumatic MV pathology specimens described the pathological involvement of the valve as either diffuse (with fibrous thickening involving the subvalvular apparatus, annulus and entire leaflet) or marginal (isolated thickening of the leaflet margins). 40
Valvulitis is usually an evolving, indolent process that worsens with repeated episodes of ARF, and may manifest in late adolescence. The early, tenuous leaflet fusion due to commissural valvulitis progresses to more extensive changes occur with thickened, matted, shortened CT, leaflet thickening and rigidity, and calcification.
MV competence is initially preserved albeit with reduced leaflet coaptation length due to early leaflet shortening and associated mitral annular dilatation. As the subvalvular disease progresses and the leaflets shorten, the keystone is lost and MS with MR results.
Echocardiographic Imaging in Juvenile RMVD
Echocardiography is a sensitive tool to detect MR in patients with ARF without clinical evidence of a cardiac murmur,43,44 or elevation of acute phase reactants. 45 In ARF, Doppler echocardiography is included in the revised Jones Criteria for the diagnosis of cardiac involvement. 46
The 2012 WHF echo criteria for the diagnosis of RHD47,48 has focused on the doppler echocardiographic features of pathological MR and valve morphology namely:
Pathological MR (All four Doppler echocardiographic criteria must be met):
Seen in two views. In at least one view, jet length ≥2 cm. Velocity ≥3 m/s for one complete envelope. Pan-systolic jet in at least one envelope.
Features in the MV:
AML thickening ≥3 mm (age-specific). Chordal thickening. Restricted leaflet motion. Excessive leaflet tip motion during systole.
Due to deficiencies in the application of the criteria, a revision of these guidelines in 2023 once again focused on mitral apparatus thickening (anterior leaflet and chordal thickening) and abnormal valve motion (restrictive leaflet motion and excessive motion of the anterior leaflet tip). 49 The authors concede that these criteria remain limited by task-sharing and hand held echocardiogram devices. It has been suggested that artificial intelligence may play a role in the detection of rheumatic MR in the future. 50
In patients with pure MR, a trans-mitral pressure gradient of ≥5 mmHg is a predictor of future MV intervention, and may be related to LA dilatation and remodeling. 51
In a South African echocardiographic study of 73 juvenile patients with acute rheumatic carditis and severe MR, the mean MV annular diameter was 37 mm compared with an age and sex matched control group MV annular diameter of 23 mm. 32 The absence of MV annular dilatation from the World Heart Federation criteria for echocardiographic diagnosis of RHD is a notable omission, as this is a consistent feature in the vast majority of JRMVD patients encountered clinically. 47 The inclusion of MV annular dilatation as a diagnostic parameter when compared with age- or body surface area (BSA) indexed thresholds may improve the relevance of these guidelines in the juvenile population.
The combination of mitral annular and left ventricular dilatation, together with restriction of the posterior leaflet and pseudo- or true AML prolapse result in the characteristic posterolaterally directed MR jet seen on echo doppler in JRMVD (Figure 7). 21
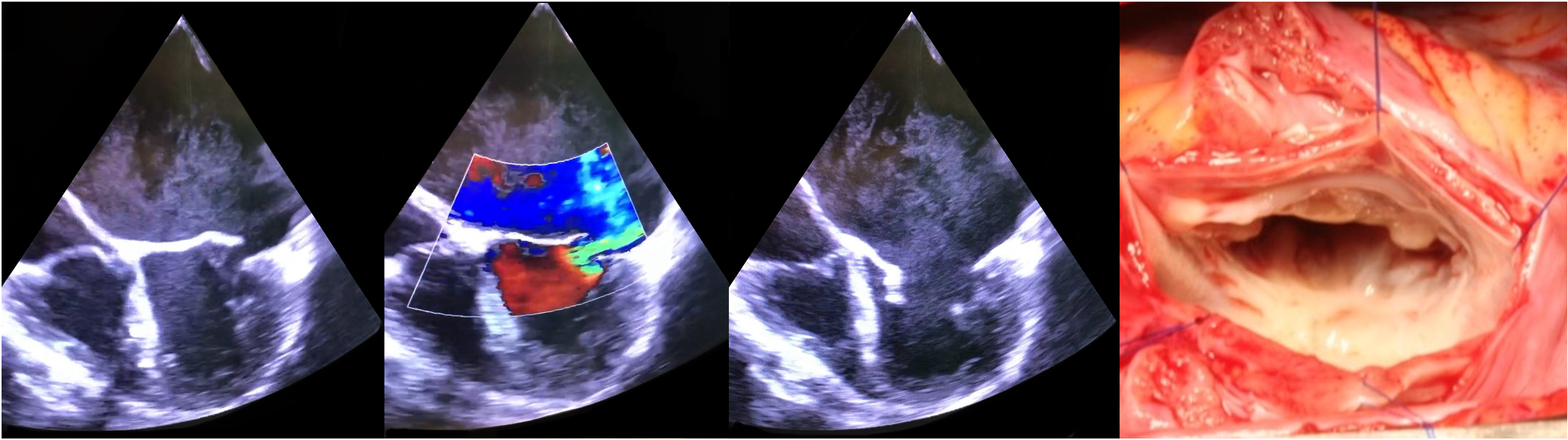
TEE images of an 8-year-old child with JRMVD and severe MR, illustrating the features of massive LA and MV annular dilatation, tethering of the PML and prolapse of the AML with a posteriorly directed jet of MR during systole (A) and (B). During diastole no features of MS are evident (C). The surgical anatomy with massive MV annular dilatation and tethering and stretching of the PML due to MV annular dilatation (D). TEE, transesophageal echocardiography; JRMVD, juvenile rheumatic mitral valve disease; MR, mitral regurgitation; LA, left atrium; MV, mitral valve; PML, posterior mitral leaflet; AML, anterior mitral leaflet; MS, mitral stenosis.
MS is evident from the limited leaflet excursion present on echocardiography (Figure 8).
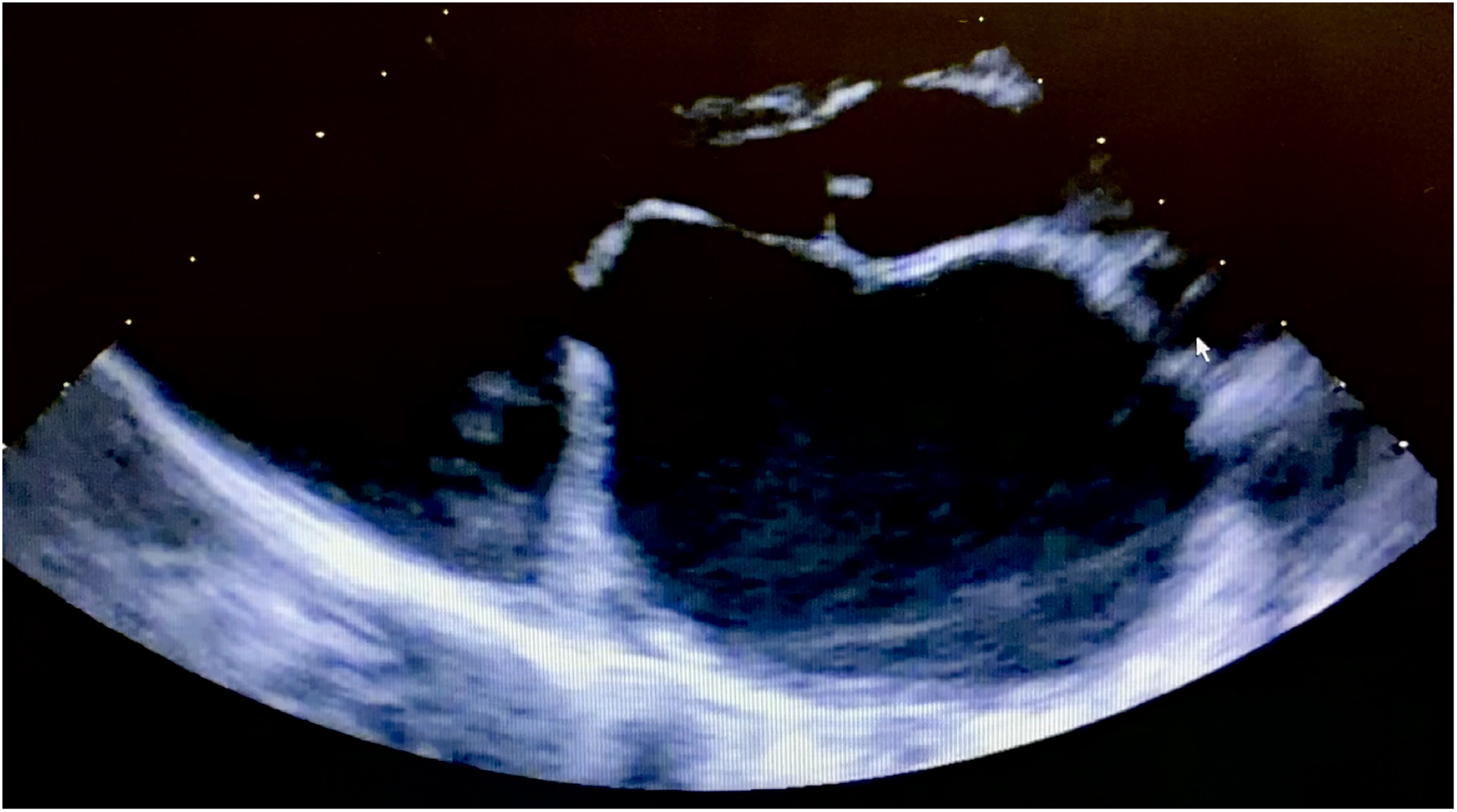
TTE image of a 14-year-old patient with severe rheumatic MS demonstrating LA dilatation with restricted opening of the AML and PML. TTE, transthoracic echocardiography; MS, mitral stenosis; LA, left atrium; AML, anterior mitral leaflet; PML, posterior mitral leaflet.
Acute Rheumatic Carditis
In patients with intractable cardiac failure due to acute rheumatic carditis, the haemodynamic lesion may perpetuate the inflammatory process, akin to a child with cardiac failure exercising continuously.2,52 The clinical course of young patients with moderate to severe carditis and significant MR treated with penicillin, corticosteroids and aspirin and strict bed rest for 30 days demonstrated regression of the MR in only 43.4% of cases. 53 The presence of severe LV dilatation conveyed an increased risk of surgical MV intervention. Medical therapy may be ineffective in severe cases and the treatment of choice is urgent MV repair or replacement.
In the early 1980s, Kinsley and Barlow described the two primary effects of acute rheumatic carditis as MV annular dilatation and an inflammatory valvulitis.5,33 These processes, in varying degrees, are responsible for the subsequent typical morphological features of juvenile rheumatic MR, MS or MMVD (Figure 9).2,32–34,54
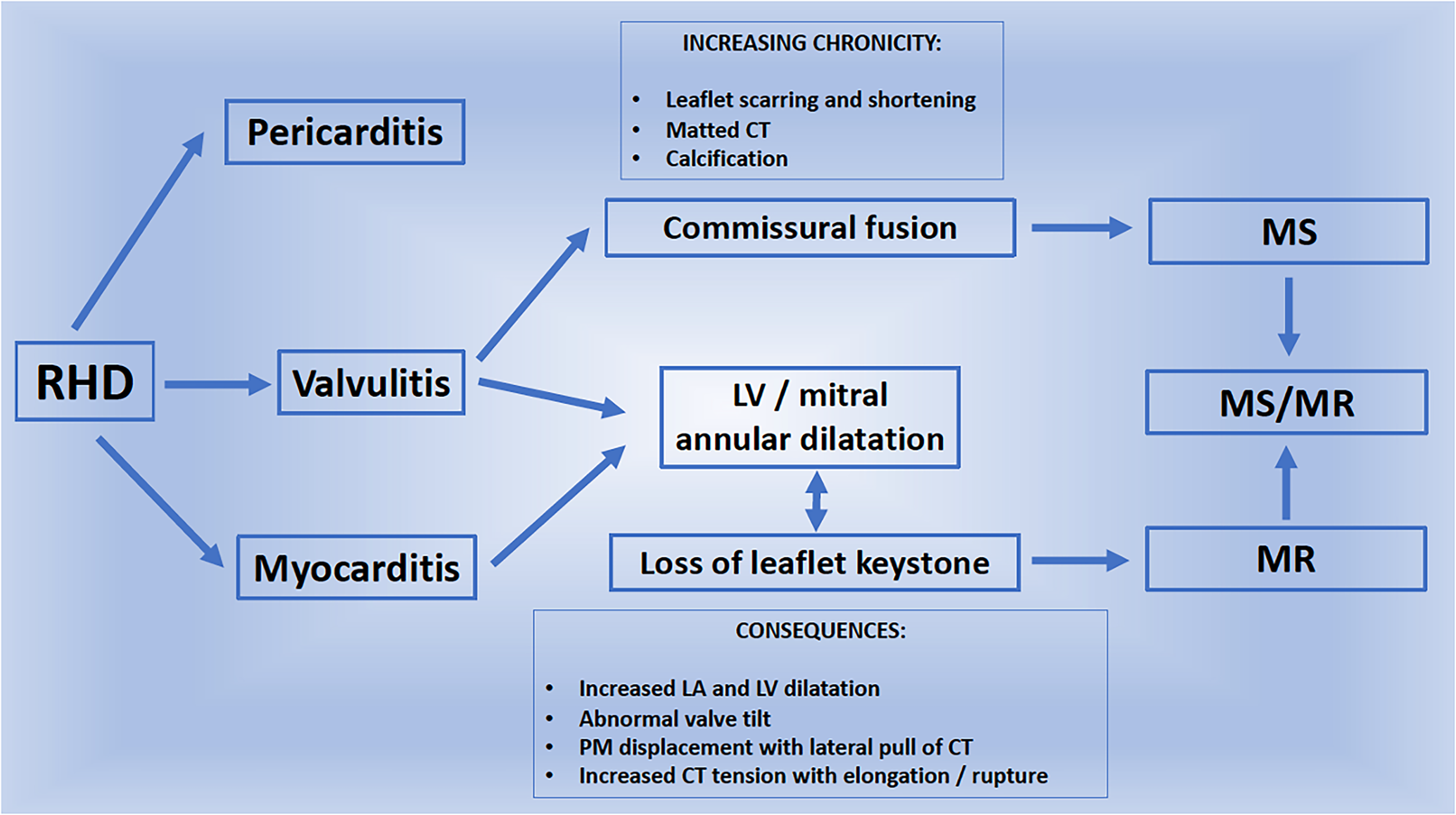
The pathogenesis of juvenile rheumatic mitral valve disease (JRMVD).
The Rationale for Valve Repair
The presence of moderate to severe MR, MS or a combination thereof (MMVD) requires intervention to relieve symptoms and to prevent progressive LV dysfunction and pulmonary hypertension. While pure MS may be treated by catheter intervention, MR and MMVD require surgery in the form of MV repair or replacement.
MV replacement in young patients in LMICs is fraught with increased morbidity and reduced life expectancy due to complications related to anticoagulation. Poor compliance with warfarin (coumadin) therapy and lack of access to monitoring of anticoagulation levels lead to increased risk of valve thrombosis and bleeding complications (Figure 10).55–59 Although bioprosthetic valves may be used in exceptional circumstances, their use in young patients is generally discouraged due to early structural valve degeneration and limited access to repeat valve replacement in LMICs.58,60,61
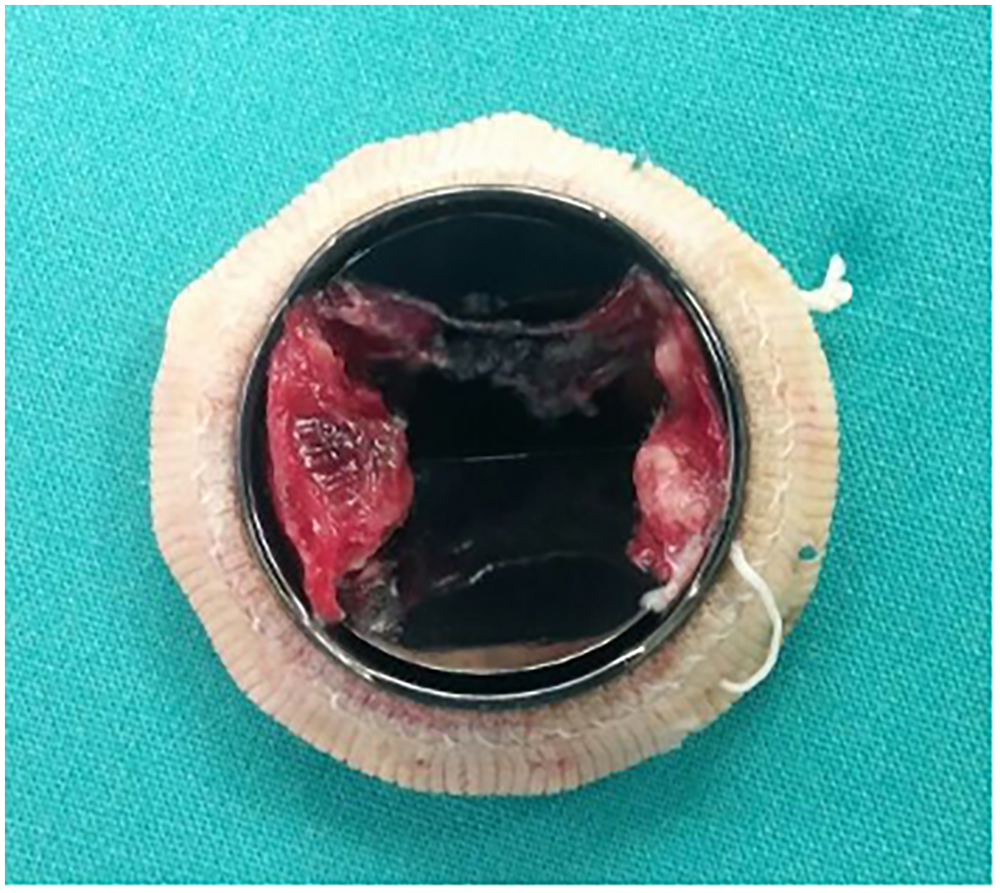
A mechanical mitral valve (MV) prosthesis explanted from a 12-year-old patient, demonstrating thrombus at the hinge points.
Attempt at valve repair should be the surgical strategy in every young patient requiring MV surgery in LMIC's. Successful repair is achievable by a thorough understanding of the mechanisms of MV dysfunction and the use of established surgical approaches to restore MV competence. If a durable MV repair is not possible, a mechanical valve prosthesis is preferred in children or teenagers.
Principles of Valve Repair for Based on Valve Morphology and Dysfunction
The initial step in planning MV valve repair is thorough analysis of transthoracic echocardiography (TTE) and intraoperative transesophageal echocardiography (TEE). In addition to MV analysis, echocardiography is used to exclude concomitant aortic valve involvement, LA thrombus, or pericardial effusion. JRMVD is extremely rare below the age of 5 years, accounting for less than 5% of cases in some series. 62 In endemic areas in LMIC's, differentiating the diagnosis of late-presenting congenital MV disease and JRMVD may be difficult, and they may co-exist owing to the high prevalence of RHD.
The principles of MV repair in JRMVD have been developed from the experience of surgeons working in LMICs, including our own group, and follow the Carpentier methodology. 56 The Carpentier classification is useful to describe functional MV abnormalities from a surgical perspective—mitral annular dilatation (Type 1), prolapse or pseudo-prolapse of the leaflet (Type 2), and restricted leaflet motion of the leaflet (Type 3). 63
While a simplified approach to “correct what is wrong” is applicable in degenerative MV regurgitation, in JRMVD extensive tissue distortion involving the LA, MV apparatus and the LV precludes achieving anatomical normality. No surgical guidelines exist for a standardized MV repair technique in this patient population, and a combination of repair techniques is often necessary to create a functional and durable repair (Figure 11).
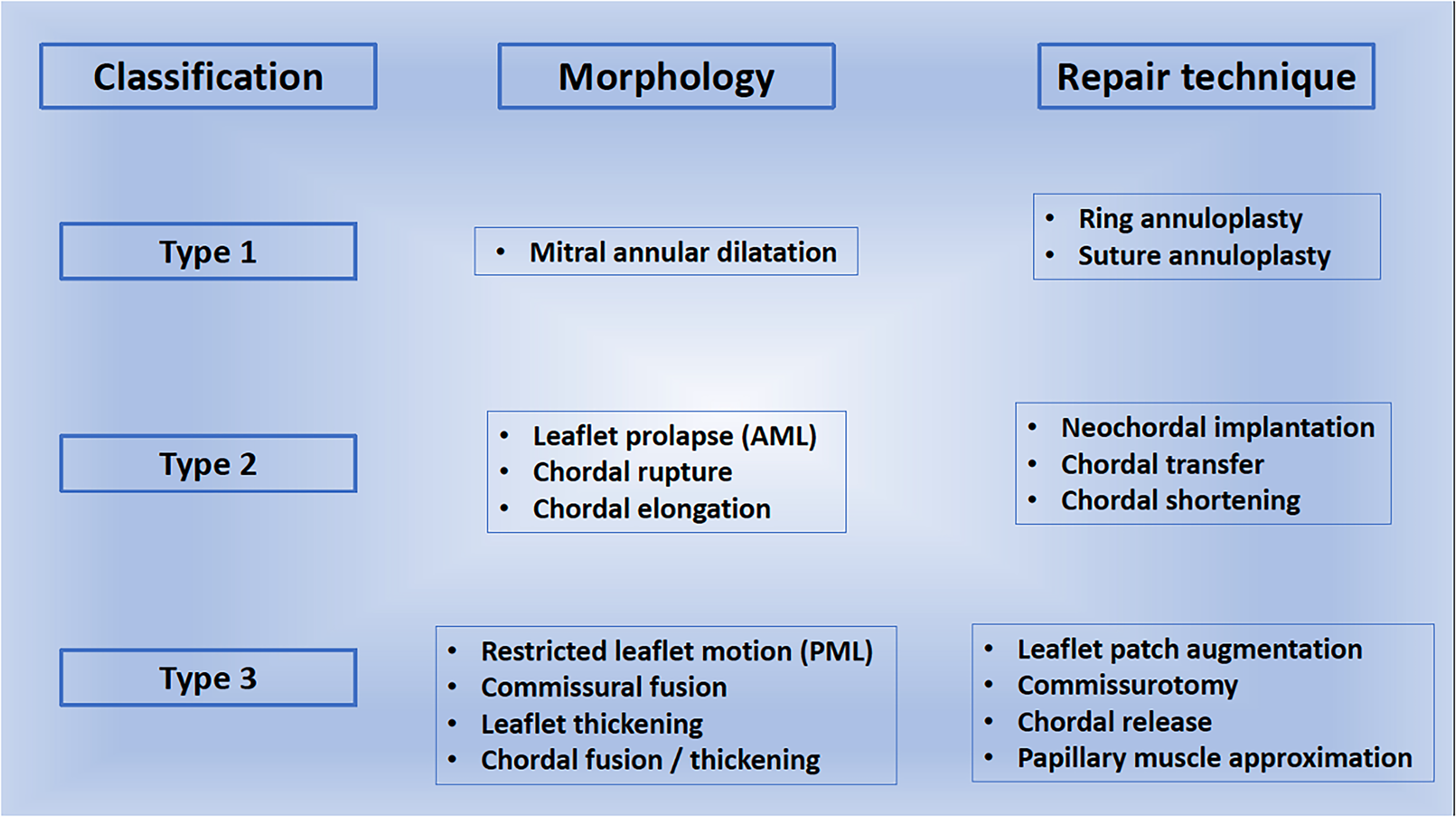
The classification and morphology of lesions in juvenile rheumatic mitral valve disease (JRMVD) and choice of repair techniques.
Mitral Regurgitation
The goal of valve reconstruction in MR is the creation of a predominantly monocuspid competence-producing mechanism that will accommodate patient growth and avoid progressive MS. A tailored approach is based on analysis of the mechanism of MR, and the geometric and functional anatomical changes of the MV.
The commonest morphological pattern encountered in JRMVD is a dilated MV annulus (Type 1), retraction and tethering of the PML (Type 3), and prolapse or pseudo-prolapse of the AML, which may be associated with chordal elongation or rupture (Type 2). 64
As MV annular dilatation is invariably present, MV annuloplasty is required in over 90% of cases to ensure leaflet coaptation. 35 In older patients, a semi-rigid complete ring is preferred to correct the annular deformity and ensure complete reduction of leaflet stress.4,65 A C-shaped ring fashioned from Teflon felt is an inexpensive alternative to a semi-rigid annuloplasty ring, but may be less effective in patients with massive MV annular dilatation and anatomical distortion.4,66–68
JRMVD patients often have a MV annulus diameter far in excess of the predicted Z-score based on BSA, it is often possible to implant an adult sized (30–32 mm) MV annuloplasty ring which accommodates patient growth. 32 In younger patients requiring annuloplasty in the range of 28– 0 mm, an alternative to ring implantation is a suture commissuroplasty which will allow for annulus growth. 34 The use of a biodegradable annuloplasty ring has also been proposed to allow for growth in the pediatric population, however this has not been widely adopted in clinical practice.57,69
Reduction of the MV annulus diameter as a sole intervention seldom relieves MR, and may exacerbate PML tethering and AML prolapse, changing the nature of the MR jet. 41 In a situation analogous to ischemic MR, isolated aggressive MV annuloplasty contributes to the poor durability of repair in that patient population. 70 Annular diameter reduction is therefore usually accompanied by leaflet augmentation and chordal procedures to address these secondary effects. 71
The PML is typically retracted and stretched thin due to the LA and MV annular dilatation, and may require augmentation to achieve a suitable surface of coaptation. Leaflet delamination or fibrous peeling in older patients, as popularized by Kumar and Chotivatanapong, delivers less yield on leaflet surface area in the juvenile population, and patch augmentation is usually preferred.67,72 Leaflet patch augmentation is used to avoid the implantation of a small MV annuloplasty ring to achieve competence.34,64,73,74
PML patch augmentation is widely used, with the natural concavity of the leaflet allowing for ready advancement and improvement in the coaptation length without the need for a bulky patch. 76 Patch augmentation of the PML simulates a kite on a string, with the native free edge of the leaflet rotated into the ventricular cavity and forming the zone of coaptation with the AML, creating a monocuspid mechanism of valve competence.
As a guide, the PML patch may contribute to 2/3 of the diameter of the MV orifice and should not be not oversized, else the pericardial patch creates a cup-like “aortic cusp effect” in diastole, resulting in an increased trans-mitral pressure gradient. 77 AML patch augmentation is also used effectively, provided that the lateral extent of the patch extends beyond the fibrous trigones to the commissures to allow the convex leaflet to “drop” into the LV cavity to improve coaptation length. 78
Glutaraldehyde-treated autologous pericardial patch augmentation of the MV leaflets have demonstrated good durability as initially described by Carpentier and Dubost.76,78–80 Brief immersion in glutaraldehyde is considered sufficient to obliterate interstitial cells and reduce subsequent fibrosis, although the duration of exposure and concentration of glutaraldehyde varies widely without consensus.71,76–78,81–85 Bovine pericardium may be used when autologous pericardium is unsuitable or deficient.
The use of fresh autologous pericardium for leaflet patch augmentation in adult MV repair has been described, however in the pediatric population concerns exist regarding early fibrosis, retraction and calcification.82,86 In the future, the use of decellularized scaffolds for leaflet augmentation or even leaflet replacement may greatly enhance the durability of valve repair by diminishing the risk of calcification and fibrosis. 87
In a situation analogous to extreme undersizing of the annuloplasty ring creating competence of the MV valve at the expense of MS, extreme oversizing of the leaflet augmentation patch produces MV competence but yields poor long-term outcomes. 75 Judgement is required to find the appropriate balance between the maximal diameter of annuloplasty ring that can be used with the least amount of patch material, allowing for durability of the repair.
Alternatives techniques to restore the keystone and MV competence include posterior MV leaflet resuspension to the contralateral annulus, although this non-anatomical solution may result in increased leaflet stress and abnormal fluid dynamics limiting durability. 88
After annular reduction and leaflet augmentation have been achieved, an accurate assessment of the degree of AML prolapse is possible. Leading edge CT at A1/A2 and A2/A3 are often found to be elongated or completely ruptured. Various techniques of chordal repair such as chordal shortening or chordal transfer have been described, and the implantation of polytetrafluoroethylene (PTFE) artificial neochordae may be preferred in cases of acute rheumatic carditis or endocarditis with inflamed and friable native CT.65,73,74
Under normal conditions, leading edge CT are not under tension, and this is borne in mind when calibrating neochordae length. Overcorrection of prolapse that results in excessive CT tension is avoided and associated with repair failure.11,89,90 PM approximation for functional MR has been of interest recently as a means of counteracting CT tethering from the lateral wall of the dilated LV, avoiding the use of an undersized annuloplasty ring.. 91 This technique may be applicable to the JRMVD population.
Following MV repair, an adequate length of leaflet coaptation length of more than 6 mm correlates with lower postoperative MR and improved durability of repair. 40 A review of preoperative echocardiographic characteristics demonstrated that the preoperative length of AML of greater than 18 mm/m2 predicts a satisfactory post-repair coaptation length and durability of repair. 92 The presence of a restrictive AML and the requirement for PML patch augmentation in juvenile rheumatic MV repair proved to be risk factors for recurrence.66,92–94
Mitral Stenosis
In pure MS, the focus of intervention is to relieve obstruction to LV inflow without creating new or further MR. BMV has been shown to be safe and effective for the relief of juvenile rheumatic MS, as well as adults with late presenting pliable MS.95–98 This procedure is especially useful as a temporizing measure in symptomatic young women presenting ab initio with MS during pregnancy.
In patients who are unsuitable for BMV, MV repair is necessary. The pathology in young patients is commonly commissural fusion at leaflet level, amenable to commissurotomy and limited PM splitting.67,73 In patients with more severe subvalvular involvement, fenestration of matted CT and papillotomy may be required to improve diastolic flow. 72 Calcification, leaflet thickening and fibrosis amenable to delamination is seldom seen in juvenile rheumatic MS. Extensive fibrosis and calcification occurs with chronicity and confers a lower rate of valve repair in adults. 99
Surgical Outcomes
MV repair in JRMVD often requires a combination of surgical techniques that differ from those used in degenerative MV repair, largely due to the paucity of valve tissue in relation to the surface area of the MV annulus. As a result, the substrate for systolic anterior motion of the AML-induced left ventricular outflow tract obstruction is not present following JRMVD repair. Significant residual MR or MS following MV repair cannot be accepted, and residual regurgitant MR jets are interrogated carefully and may cause post-operative haemolysis warranting reoperation. The early and late echocardiographic imaging of repair techniques used in JRMVD are illustrated in Figures 12 and 13.
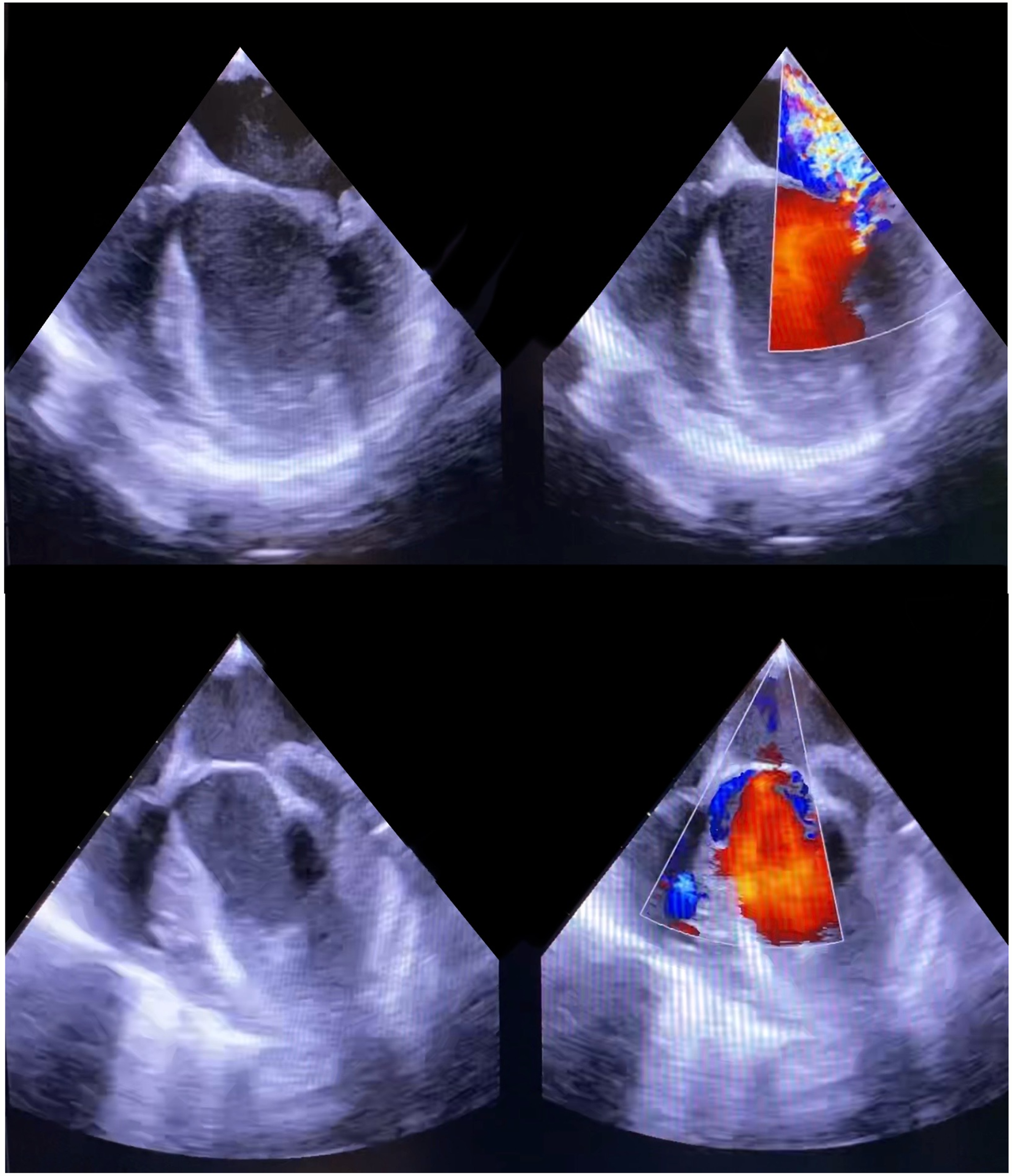
TEE images of an 11-year-old year old patient with JRMVD and severe MR illustrating a dilated LA and MV annulus, with thickening and tethering of the PML and failure of coaptation with the AML resulting in the typical posteriorly directed jet of MR (A). Following MV repair, the TEE demonstrates the “keystone” length of coaptation creating a competent valve (B). TEE, transesophageal echocardiography; JRMVD, juvenile rheumatic mitral valve disease; MR, mitral regurgitation; LA, left atrium; MV, mitral valve; PML, posterior mitral leaflet; AML, anterior mitral leaflet.
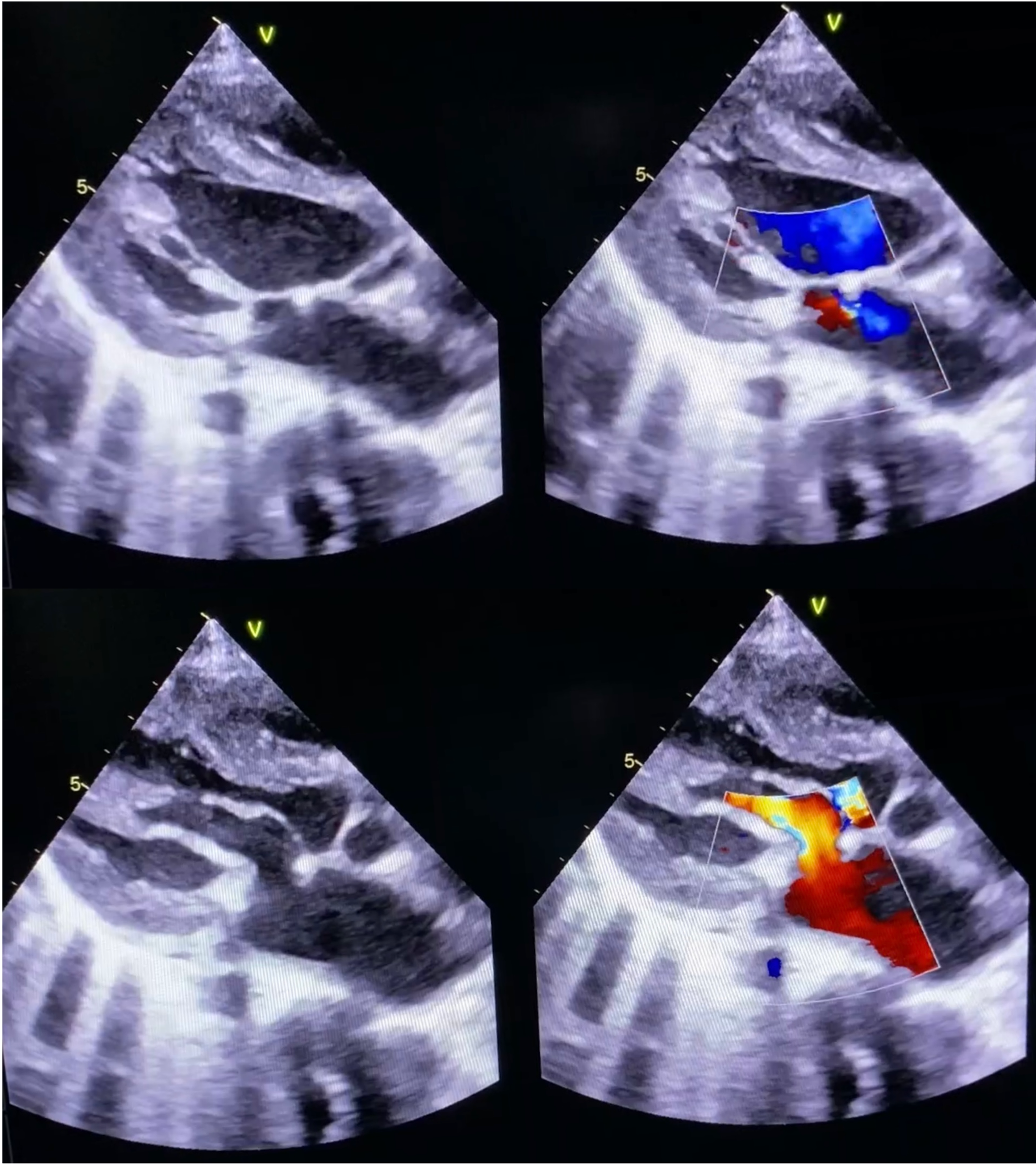
TTE images of a 14-year-old child following MV repair for JRMVD at two-year follow-up, illustrating trivial to mild residual central MR (A) with no significant MS despite thickening and echogenicity of the patch augmented PML leaflet (B). TTE, transthoracic echocardiography; MV, mitral valve; JRMVD, juvenile rheumatic mitral valve disease; MR, mitral regurgitation; MS, mitral stenosis; PML, posterior mitral leaflet.
MV repair for the chronic sequelae of rheumatic valvulitis in adults has been well described, with excellent results achievable in expert hands. 72 The focus of MV repair for JRMVD predominantly addresses MV annular dilatation and can be undertaken safely with good early and long-term outcomes.58–60,65,74
The Royal Children's Hospital in Melbourne reported a series of 79 patients (median age 11.4 years) that underwent MV repair for JRMVD. Early surgical outcomes were excellent, however freedom from valve deterioration at 5, 10 and 15 years was 75%, 36% and 28%, respectively. 93 Factors associated with repair failure were the requirement for PML patch augmentation, associated with AML restriction. 94
In a retrospective review of 116 patients (mean age 12.6 years) with JRMVD at Hospital de Santa Marta, Lisboa, MV repair was possible in 86 (74%) patients. Freedom from reoperation at six months, 5 years and 10 years was 96%, 72% and 44%, respectively. MS was the primary risk factors for reoperation. 73
A retrospective-prospective series of 106 patients (mean age 13.1 years) in Ahmedabad underwent MV repair for JRMVD. The indication for surgery was MR in 80 (78.4%) patients, MS in 8 (7.8%) patients, and MMVD in 14 (13.7%) patients. Of note, 30 (29.6%) patients had recent ARF (< 8 weeks), and 11 patients underwent emergency surgery for intractable heart failure. Operative mortality was 1% and freedom from reoperation at 2.5 years was 97.1%, highlighting the excellent early outcomes. 71
Although rheumatic MV repair in children and teenagers may be considered less durable than MV repair for congenital MV disease, the benefit over mechanical MV replacement is significant. 73 An additional factor associated with failure of MV repair for JRMVD is ARF recrudescence, and the importance of secondary penicillin prophylaxis cannot be overstated.100,101
Future Outlook
Globally, ARF and RHD are regarded as a predominantly LMIC problem, and the declining incidence has resulted in the neglect of resources for research. Initiatives from Africa—including the Cape Town Declaration and more recently the Accra Declaration—have highlighted the disease burden of RHD in Sub-Saharan Africa and beyond.102–106 Specific areas identified for research opportunities include demographic studies, screening echocardiography, rapid streptococcal antigen tests, GAS vaccine development, the study of tissue and plasma proteomics to further understand the physiology and to improve therapeutics, and the development of longer-acting penicillin formulations to improve secondary prophylaxis. 13
Recent interest in the identification of an accurate biomarker for ARF included the analysis of serum plasma samples from patients in Uganda and Pakistan, suggesting that immunoglobulin G3 (IgG3) may be a more useful marker of ARF than prothymosin alpha (PTA). 107
The recognition of epitope spreading as a contributor to the heterogeneity of immune response of individuals are leading to a deeper understanding of the pathogenesis of ARF, and this research may yield biomarkers for diagnosis in the future. 108
Echocardiographic predictors of the resolution of RHD after the first episode of ARF remain limited, with an emphasis on valvulitis and leaflet motion.109,110 MV annular dilatation seen in JRMVD should be recognized as a distinct entity from valvulitis, and echo diagnosis guidelines developed to facilitate early detection of MV annular dilation as a substrate for MR.
Durable MV repair is achievable in most patients with JRMVD in experienced centers, and surgeons in LMICs (either resident or members of visiting teams) should have the requisite skills for MV repair in the juvenile population. This is achievable using a hub-and-spoke model of focused training and skills transfer at centers of excellence. 111
Footnotes
Acknowledgments
The authors wish to acknowledge Gabriella Ricciardi MD and Raoul Biondi MD for the development of the artwork used for this article.
Informed Consent
Owner, institutional and patient permission was received for the publication of all images.
Author Contributions
The authors have contributed equally to the conception, development and synthesis of this manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
