Abstract
Background
Prospective multicenter and standardized databases are essential to monitor and improve patients’ care. This article outlines the Heart Valve Society (HVS) Mitral and Tricuspid Valve Database that aims to track patients’ journeys with mitral and/or tricuspid valve disease through their lifetime.
Methods
The database focuses on the adult patients undergoing surgical or transcatheter intervention(s) for mitral and/or tricuspid valve disease. It is designed as a platform for ambispective observational cohort studies. Participating centers may contribute consecutively or based on an initiated project. The processes of ensuring high data quality are described.
Results
The database captures 78 outcome variables (14 clinical and 64 imaging), and 293 case-mix/baseline variables. Additionally, 2 patient-reported outcome measures are implemented in the database: the EQ-5D-5L and the Impact on Mental Health & Daily Activities questionnaire (IDCV). The outcomes and their respective definitions are harmonized with the International Consortium for Health Outcomes Measurement (ICHOM) Standard Set of Patient-Centered Outcome Measures for Heart Valve Disease (HVD). Participating centers can extract their own data for a local analysis, and proposals for multicenter studies are highly welcome. The database fully complies with the General Data Protection Regulation and the Health Insurance Portability and Accountability Act regulation, ensuring privacy through pseudonymized and anonymized data extracts.
Conclusions
The HVS Mitral and Tricuspid Valve Database is an interdisciplinary initiative that involves cardiologists, cardiac surgeons, and basic scientists contributing to the global standardization of data capture and governance.
This is a visual representation of the abstract.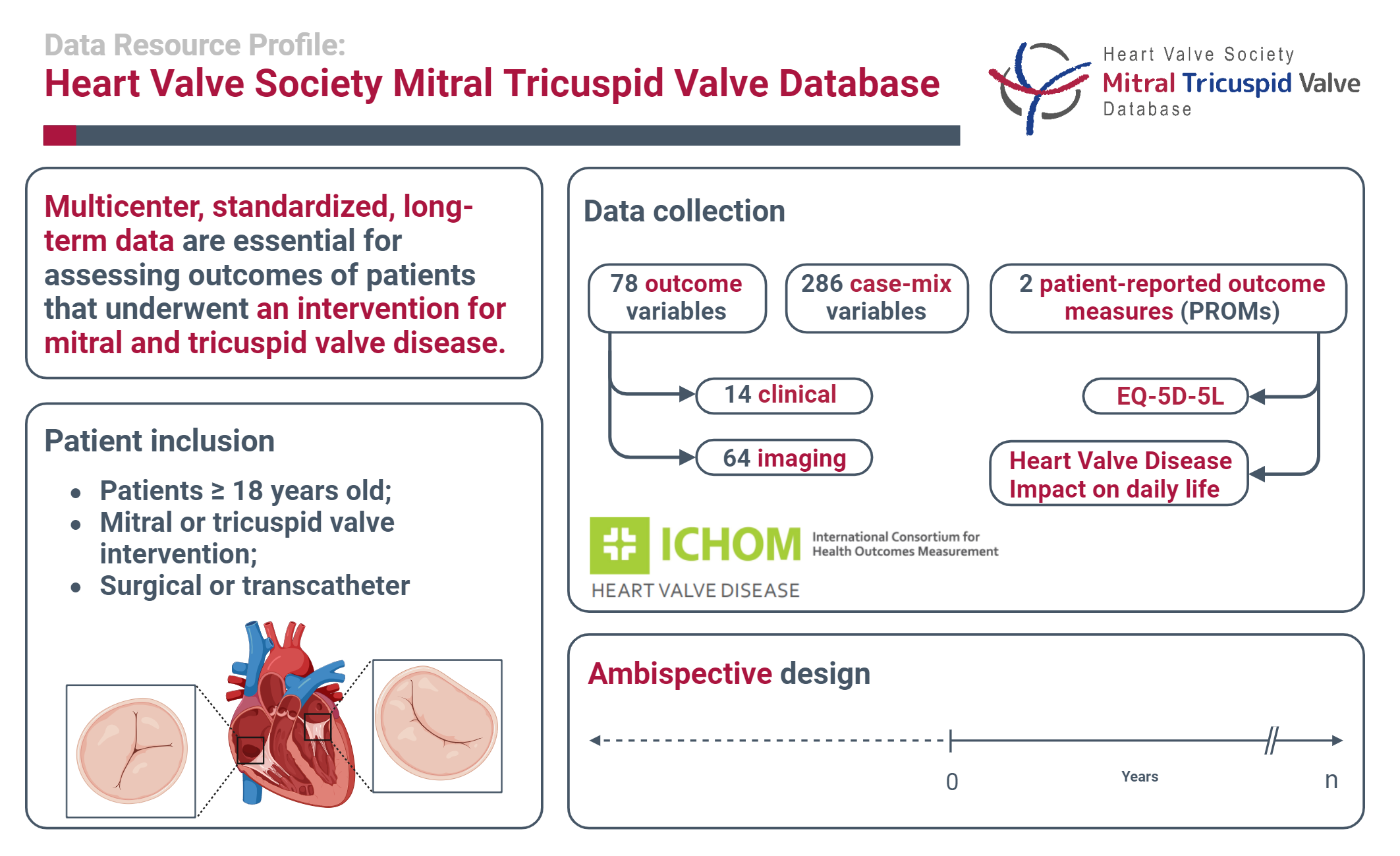
Keywords
Introduction
Mitral and tricuspid valve disease affects millions of people worldwide, significantly impacting the life expectancy and quality of life.1,2 In addition to medical management, interventions for mitral and tricuspid valve disease (ie, valve repair or replacement) may be performed surgically or by means of a catheter-based approach. Despite the rapid developments in the surgical and catheter-based field in the last decades, many knowledge gaps still exist. Large, standardized databases are needed to keep up with these rapidly evolving fields. Most published studies still only contain single-center outcome analyses and report on a limited number of devices or techniques. 3 Most recent registries aimed at quality improvement usually lack granularity and have limited follow-up.2,4–6 The underdiagnosis of the disease has resulted in late presentation and treatment and to significant gaps in management opportunities. 7 To address these gaps, the Heart Valve Society (HVS) initiated the Mitral and Tricuspid Valve Research Network (VRN), one comprehensive platform to follow patient- outcomes of patients journeys starting from first intervention, regardless of the treatment technique or approach. This paper outlines the development and structure of the HVS Mitral and Tricuspid Valve Database, and the accompanying Mitral and Tricuspid VRN. The HVS Mitral and Tricuspid Valve Database aims to compare outcomes of patients undergoing specific surgical and transcatheter interventions for mitral and/or tricuspid valve disease, to enhance uniform scientific reporting, to optimize multidisciplinary patient care, to assess quality of care, and to update and improve guidelines.
Study Design
The database is designed as a comprehensive registry platform for ambispective observational cohort studies. The patients can be included entirely retrospectively, retrospectively with both retrospective and prospective components, or entirely prospectively (Table 1). The hybrid ambispective design was chosen due to the need to assess multiple short- and long-term outcomes. Retrospective enrollment may be useful for research proposals addressing long-term outcomes, early after initiation of the database. 8 Patient inclusion in the database before a center's official entry in the Mitral and Tricuspid VRN is considered the retrospective part of the ambispective design.
HVS Mitral and Tricuspid Valve Database Summary.
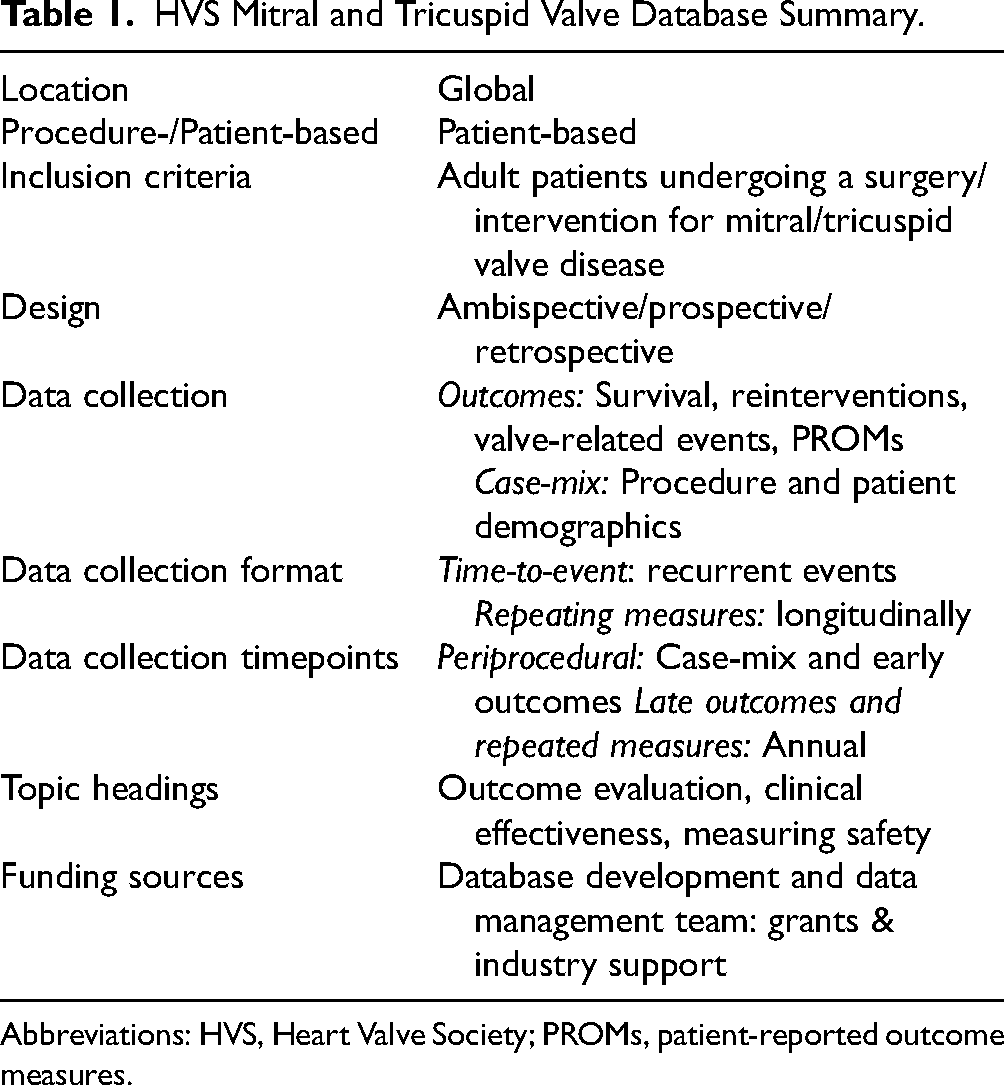
Abbreviations: HVS, Heart Valve Society; PROMs, patient-reported outcome measures.
Patient Inclusion
All adult patients undergoing surgical or transcatheter interventions for mitral and/or tricuspid valve disease, regardless of concomitant procedures, can be included in the database (Table 1).
Project-Based Versus Consecutive Inclusion
Participating centers can initiate a project with prespecified patient inclusion criteria, defined recruitment periods and limited follow-up time (Supplemental Figure 1). For example, a participating center may be interested in the 1-year outcomes of atrial functional mitral valve regurgitation patients undergoing surgical versus catheter-based intervention.
In the pathway of consecutive inclusion, centers can participate by consecutively entering all patients with mitral and/or tricuspid valve disease. Consecutive inclusion is imperative to abolish selection bias, but may pose a significant burden on the hospital's resources, as mitral and tricuspid interventions are common, and dedicated long-term follow-up entry is required. 1
This novel umbrella framework enables the Mitral and Tricuspid VRN to easily execute multicenter cohort studies using the common infrastructure, data definitions, and all data are stored on a single platform. The advantage of such a framework is that after completing a specific project, the reuse of existing data for consecutive projects is facilitated according to the FAIR principles (Findable, Accessible, Interoperable, Reusable). 9 Nevertheless, an umbrella framework has the limitation of enrolling the project-specific patients, leading to potential selection bias. The data management team includes epidemiologic expertise and oversees proper data selection for such research questions.
Database Variables: Outcome Measurements and Case-Mix Variables
The HVS Mitral and Tricuspid Database tracks multiple clinical/imaging outcome variables and case-mix variables to enable a wide range of research questions. Both case-mix and outcome capture, and their respective definitions, are harmonized with the International Consortium for Health Outcomes Measurement (ICHOM) Heart Valve Disease (HVD) Standard Set. The ICHOM Standard Set of Patient-Centered Outcome Measures for HVD is a multisociety initiative, initiated by HVS, to standardize data capture globally. 10
Outcome Variables
A total of 78 outcomes variables (14 clinical and 64 imaging) are tracked. The database is designed to capture repeated time-to-event outcomes, allowing analysis of repeating events (Figure 1). These events include (among others) mortality, reintervention(s), valve-related events (thromboembolism, bleeding, valve thrombosis), and heart failure hospitalization (Table 2). Furthermore, repeated measures such as valve regurgitation, valve gradients, and left ventricular ejection fraction are captured longitudinally. In the published data dictionary, all tracked outcome variables and their definitions are specified (https://heartvalvesociety.org/).
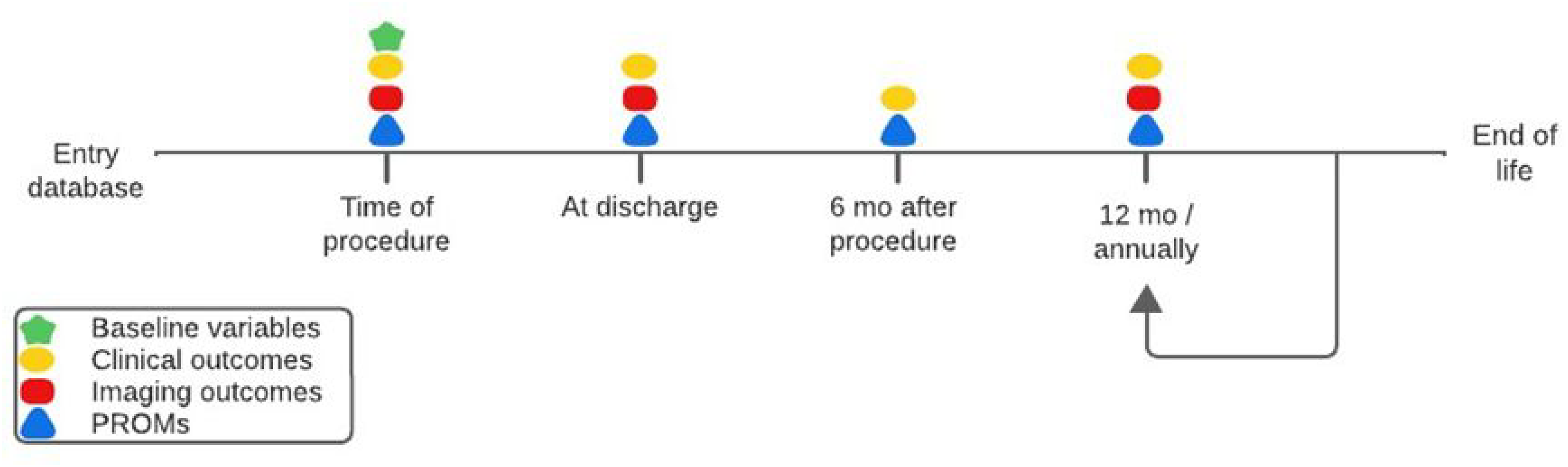
Proposed Timelines of Collecting Baseline Variables, Repeated Measurements, and Patient-Reported Outcome Measures (PROMs). Created in BioRender.
High-Level Overview of Tracked Outcomes.

Abbreviations: TIA, transient ischemic attack; PM, pacemaker; CRT, cardiac resynchronization therapy; VAD, ventricular assist device; NYHA, New York Heart Classification.
Reoperation for bleeding/tamponade, in-hospital reoperation and valve-related events, new unplanned pacemaker implantation, periprocedural myocardial infarction, acute kidney injury, low cardiac output syndrome, mechanical ventilation > 48 h, blood transfusion, and vascular complications.
Case-Mix Variables
A total of 293 case-mix/baseline variables can be tracked, detailing patient's comorbidities and treatment details. These case-mix variables have a hierarchical structure (e.g., in case of isolated mitral valve surgery the interventional variables of tricuspid valve surgery are not applicable). The HVS Mitral and Tricuspid Valve Database is a more comprehensive registry platform as compared to the ICHOM Standard Set of Patient-Centered Outcome Measures for HVD, aiming to provide more granular data for scientific research questions. All tracked case-mix variables are specified in the data dictionary (see above).
Patient-Reported Outcome Measures
Two patient-reported outcome measures (PROMs) are currently implemented in the database. These include the EQ-5D-5L and the Impact on Mental Health & Daily Activities questionnaire (IDCV), as proposed by the ICHOM Standard Set of Patient-Centered Outcome Measures for HVD. Other PROM questionnaires that may be required for specific projects can be easily implemented as specific project modules. Nevertheless, this practice is discouraged, as it contradicts the aspiration to achieve global data standardization.
Governance
The database is governed by a group of core leaders with a rotating chair, a data management team, and a scientific committee, all overseen by the HVS board (Figure 2). The core leaders serve as the executive body of the Mitral and Tricuspid VRN, leading its general direction. The data management team is responsible for developing and maintaining the database, including guiding application processes, data selection, data quality monitoring and data quality improvement initiatives. The scientific committee reviews project applications, research proposals and proposed abstracts and manuscripts originating from the registry.
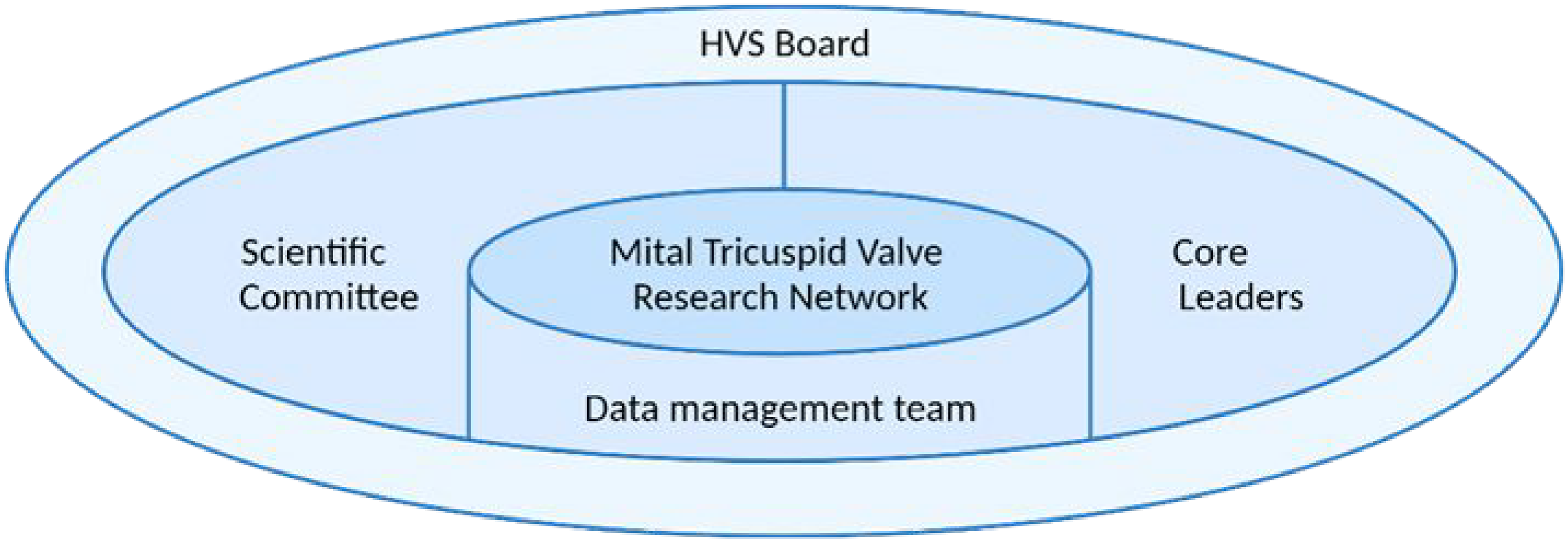
Governance. Created in BioRender. HVS: Heart Valve Society.
Data Use
Each center can extract its own data for local analysis. To conduct multicenter studies in the project-based pathway, a project proposal must be submitted to the scientific committee. The project proposal will be reviewed by the scientific committee for scientific validity and by the core leaders for project feasibility. After acceptance of the proposal, the data management team will launch the project through the Mitral and Tricuspid VRN to contact additional centers willing to participate in the proposed project (Supplemental Figure 3). The research team submitting the project proposal is responsible for the data analysis and leads the writing process of the project-based publication. In all research endeavors, centers that contributed data to the specific project are eligible for scientific publication co-authorship. To conduct multicenter studies using existing data in the database, a research proposal is submitted to the scientific committee (Supplemental Figure 2). Each center with suitable data for the accepted research proposal participates in the study and will be notified about the proposed study, and their data will be used automatically (nonopposition procedure). In the case a center opposes the data usage for this study the data will not be included.
Data Quality
High data quality and adequate (follow-up) completeness in databases is of paramount importance for obtaining valid conclusions. 11 We implemented several data checks, procedures, and feedback processes in the HVS Mitral and Tricuspid Valve Database to ensure high-quality data acquisition. These steps include the use of data dictionary, monitoring of the timely data entry, data validation, feedback reports, and data cleaning/correction, all summarized in Figure 3. By having clinicians in the lead regarding the data dictionary and harmonizing with ICHOM Standard Set of Patient-Centered Outcome Measures for HVD, care is taken to use unambiguous outcome definitions, thereby minimizing center heterogeneity. In the web-based electronic clinical report form, several validation steps are built in the system to prevent incompatible responses. In batch uploads, data cleaning steps will be carried out by the Mitral and Tricuspid VRN data management team. Furthermore, biannual internal audits will be performed to discuss any discrepant results with the Mitral and Tricuspid VRN members.
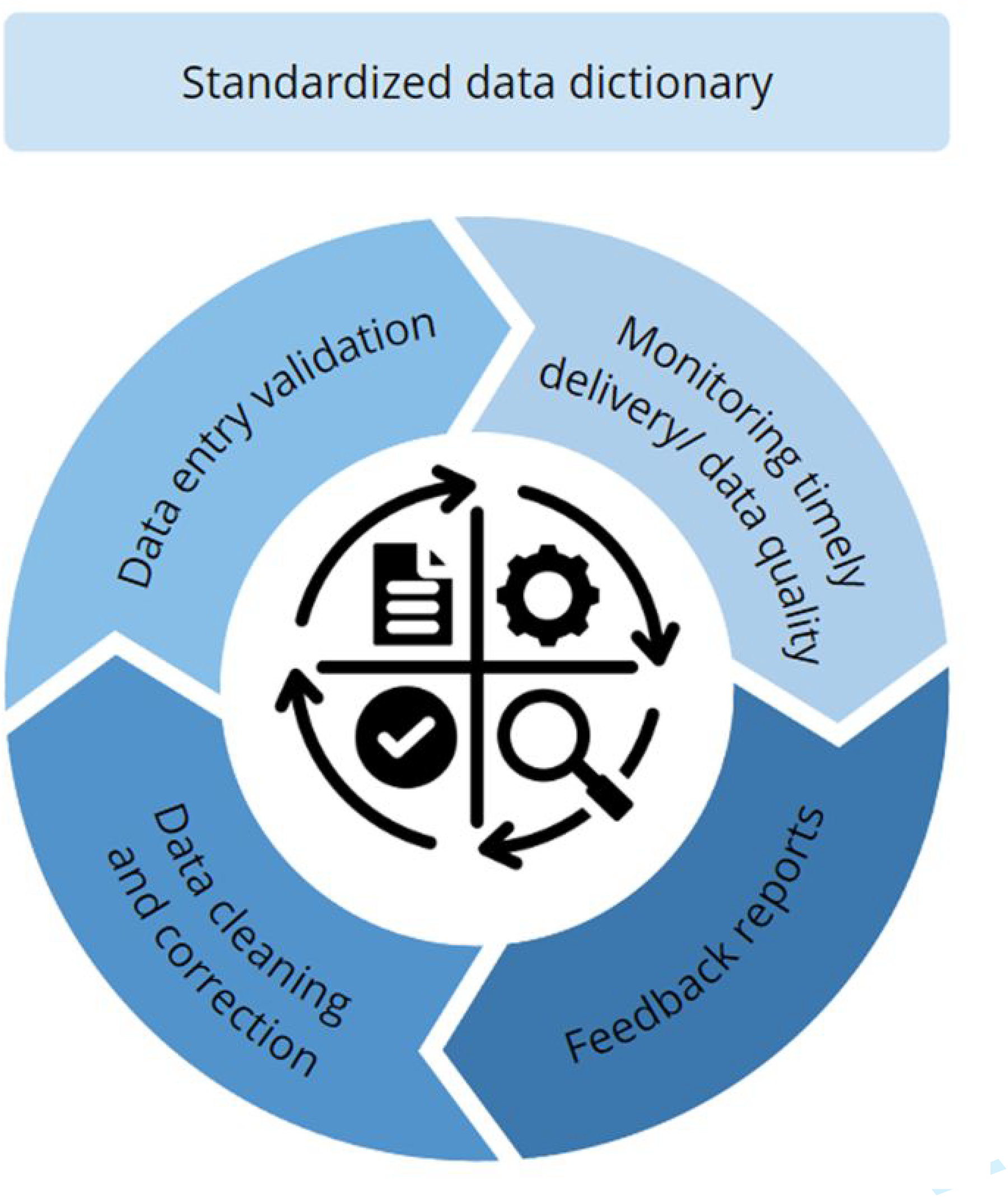
The Standardized Steps of Data Quality Control. Created in BioRender.
Data Controllership and Privacy
The participating centers operate under a joint controllership agreement, wherein each center has the responsibility for safeguarding the personal data recorded in the database. The database was developed in accordance with Good Clinical Practice (GCP) guidelines and adheres to the General Data Protection Regulation (GDPR) and the Health Insurance Portability and Accountability Act (HIPAA). Upon eCRF data entry, a unique patient code is generated, with no identifiable information. Identifying key files remain solely with participating centers, inaccessible to the central database or governing bodies (Supplemental Figure 4).
Strengths and Weaknesses
The harmonization of the HVS Mitral and Tricuspid Valve Database with the ICHOM Standard Set of Patient-Centered Outcome Measures for HVD represents a crucial step toward global data standardization. Aligning data capture practices facilitates uniform scientific reporting across diverse healthcare settings. 7 The databases' rich data collection allows for comprehensive information capture, minimizing confounding factors. This depth of data collection supports multiple subgroup analyses and enables the assessment of longitudinal trends, providing deeper insights into the outcomes of heart valve diseases. 12 However, some inherent challenges are encountered. Similar to other multicenter international registries, we will presumably encounter data gaps and incomplete follow-up entries. These gaps, particularly in follow-up data acquisition, may significantly impact the estimates of long-term outcomes. 13 Vigilant monitoring and incentivizing participation are essential to maintain data accuracy. Furthermore, our approach empowers participating centers to independently initiate projects with prespecified patient inclusion criteria and predefined follow-up periods. By adhering to the standardized protocols and rigorous data collection practices in the project-based pathway, all participating centers ensure robust and reliable information, enhancing the overall integrity of the registry.
Database Access
The database hosting system, scientific protocol, and application documents can be found on the HVS website (https://heartvalvesociety.org/). Standardized templates for project- and research proposals are provided. All centers performing mitral and/or tricuspid interventions are highly welcome join the database and Mitral and Tricuspid VRN- free of charge. Currently, the only prerequisite is that at least one researcher in the participating center is an active member of HVS. Collaboration with other registries is highly encouraged and interested researchers can contact the data management team for further questions or requests.
Supplemental Material
sj-docx-1-hvs-10.1177_30494826241313425 - Supplemental material for Data Resource Profile: The Heart Valve Society (HVS) Mitral and Tricuspid Valve Database
Supplemental material, sj-docx-1-hvs-10.1177_30494826241313425 for Data Resource Profile: The Heart Valve Society (HVS) Mitral and Tricuspid Valve Database by Kevin M. Veen, Donna de Geest, Rebecca T. Hahn, Hoda Hatoum, Victoria Delgado, Vinod H. Thourani, Meindert Palmen, Jasimuddin Ahamed, Marianna Adamo, Marta Sitges, Moritz C. Wyler von Ballmoos, Ralph Stephan von Bardeleben, Michele De Bonis and Evaldas Girdauskas in Journal of the Heart Valve Society
Footnotes
Abbreviations
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Evaldas Girdauskas received a research grant from Edwards to support the development of the Mitral/Tricuspid VRN database (Grant ID: SURG-I23-111).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
No new data were generated or analyzed in support of this research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
