Abstract
Heart valve disease management has entered a phase of unprecedented clinical and organizational complexity. Beyond anatomical valve lesion severity, outcomes are increasingly influenced by myocardial damage, ventricular remodeling, comorbidities, and extracardiac involvement, which often evolve independently of symptoms. This growing dissociation between valve lesions, myocardial involvement, and clinical presentation challenges traditional symptom- and procedure-centered paradigms and contributes to delayed referral, heterogeneous practice patterns, and variable outcomes, despite major advances in surgical and transcatheter therapies. Concurrently, multimodality imaging, longitudinal data acquisition, and expanding therapeutic options have transformed valve care into a data-rich, lifetime-oriented discipline. In this context, artificial intelligence has emerged as a potential facilitator of automated image analysis, phenotypic characterization, risk stratification, and longitudinal monitoring. However, the clinical value of these technologies critically depends on their integration within structured systems capable of ensuring data quality, expert validation, and accountable decision-making. This article argues that contemporary valve care requires an organizational framework that aligns biological complexity with technological innovation and proposes an integrated model combining Heart Valve Clinics, multidisciplinary Heart Teams, institutional Heart Valve Centers of Reference, and coordinated Valve Networks. Within this framework, artificial intelligence supports, rather than replaces, expert clinical judgment by enhancing consistency, reducing unwarranted variability, and enabling more timely and individualized decision-making across the disease trajectory, with the ultimate aim of improving quality, efficiency, sustainability, and equity of care.
This is a visual representation of the abstract.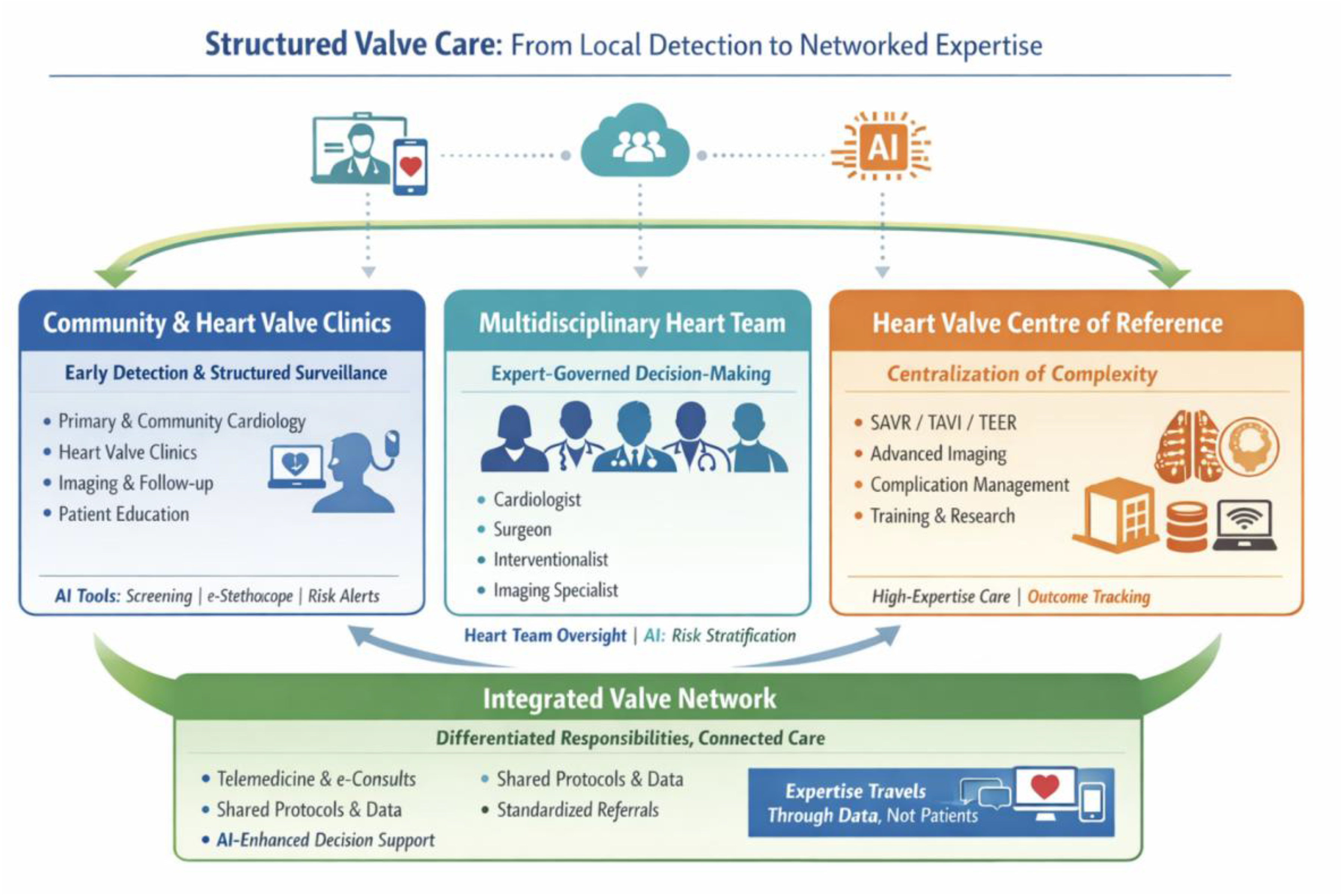
Key points
Contemporary valve care requires structured organization integrating Heart Valve Clinics, multidisciplinary Heart Teams, and institutional Centers of Reference within coordinated networks.
Artificial intelligence enhances imaging quantification, risk stratification, and longitudinal trajectory assessment but must remain embedded within expert clinical governance.
Network-based, expert-led valve care improves consistency, equity, and sustainability across the lifetime management of valvular heart disease.
The Expanding Burden of Heart Valve Disease
Heart valve disease (VHD) management is undergoing a profound transformation driven by demographic changes, expanding therapeutic options, and advances in cardiovascular imaging and data science.1,2 Although surgical and transcatheter interventions have dramatically improved outcomes across a wide spectrum of risk profiles,3–6 clinical decision-making has become increasingly complex and less linear. A central challenge in contemporary valve care is the growing decoupling between valve lesion severity, myocardial damage, and clinical presentation. Symptoms often occur late or inconsistently, while myocardial fibrosis, ventricular remodeling, pulmonary hypertension, and extracardiac organ involvement may progress silently and irreversibly despite apparently moderate valve disease.7–9 This dissociation undermines traditional symptom-driven and procedure-centered paradigms and contributes to delayed referral, heterogeneous practice patterns, and suboptimal long-term outcomes.10,11 Simultaneously, the volume and complexity of data available to clinicians have expanded exponentially. Multimodality imaging, longitudinal biomarkers, procedural registries, and lifetime management considerations now converge in each individual patient.12–14 In this setting, artificial intelligence (AI) is increasingly recognized as a potential facilitator of expert decision-making, capable of integrating high-dimensional data and supporting earlier, more consistent, and individualized therapeutic strategies.15–18 However, the clinical deployment of AI also exposes a structural paradox. While AI thrives on standardized data acquisition, procedural volume, continuous feedback, and expert validation, valve care in many healthcare systems remains fragmented and inconsistently audited.19-21 This article therefore argues that AI does not merely complement contemporary valve care, it redefines the organizational requirements necessary for safe and effective practice. Rather than reiterating the concept of Heart Valve Centers, we propose that Heart Valve Clinics, multidisciplinary Heart Teams, institutional Heart Valve Centers of Reference, and coordinated Valve Networks represent the essential infrastructure for AI-enabled valve care, aligning biological complexity, surgical and transcatheter innovation, and patient-centered lifetime management. 17
Increasing Complexity of Decision-Making in Contemporary Valve Care
Decision-making in VHD has evolved from episodic, lesion-based surgical referral toward a longitudinal, patient-centered process integrating anatomy, myocardial status, comorbidities, frailty, life expectancy, and patient preferences.13,14 This evolution is particularly relevant for surgeons and interventionists, as the durability and downstream implications of the first intervention increasingly shape lifetime outcomes. Myocardial damage has emerged as a critical determinant of prognosis. Fibrosis, impaired ventricular-arterial coupling, and extracardiac involvement may progress independently of valve hemodynamics and symptoms, reducing the benefit of late intervention even when procedural success is achieved.7–9,22 For surgeons, this challenges traditional timing thresholds and reinforces the importance of early, myocardial-informed referral. At the same time, the expansion of transcatheter therapies has multiplied procedural choices. 23 Surgical aortic valve replacement, transcatheter aortic valve implantation, and hybrid lifetime strategies must now be considered within the context of patient age, anatomical feasibility, coronary access, redo risk, and long-term durability.3–6,24,25 Similar considerations apply to mitral and tricuspid interventions, where surgical repair, transcatheter edge-to-edge repair, and emerging replacement technologies coexist. In this data-rich environment, AI-based tools may assist expert clinicians by synthesizing multimodality imaging, biomarkers, and procedural variables to refine phenotypic classification, anticipate disease progression, and support timing of intervention (Figure 1). Importantly, such tools are decision-support systems, not autonomous decision-makers, and their value depends on expert interpretation within structured clinical pathways.16,17
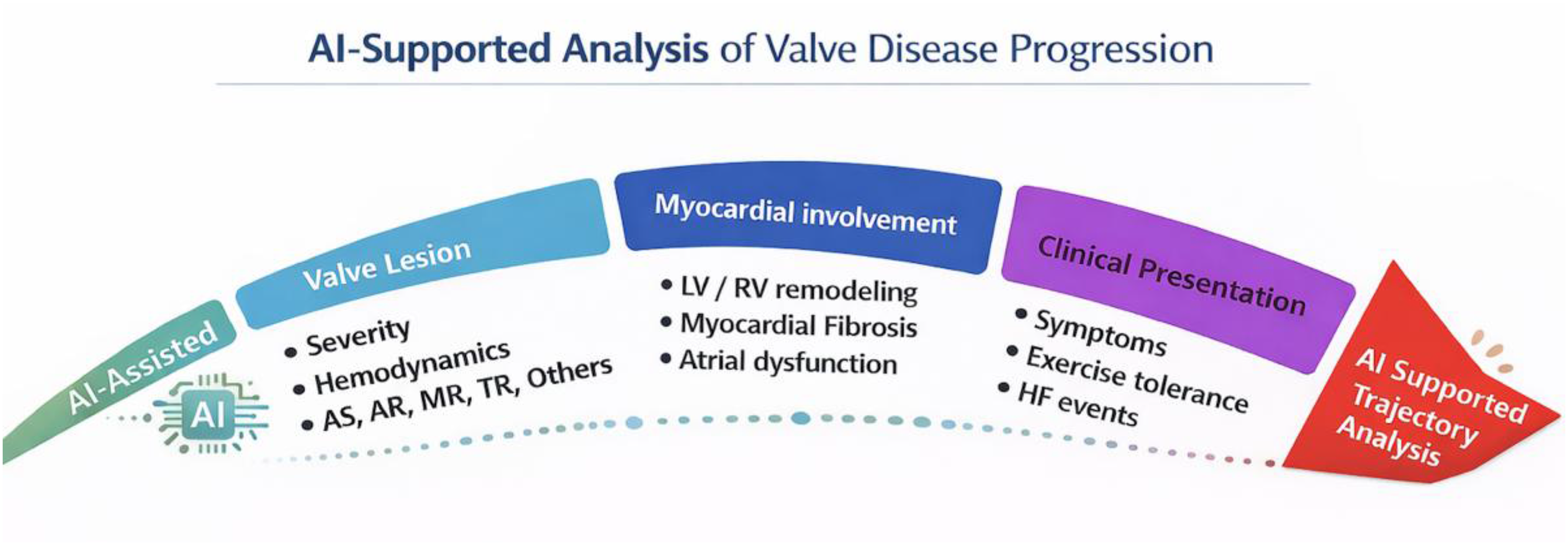
Decoupled disease trajectories in heart valve disease. Conceptual illustration of the asynchronous progression of heart valve disease. Valve lesion severity, myocardial involvement, and clinical presentation often evolve along distinct and nonlinear trajectories. Artificial intelligence supports quantitative assessment, phenotypic characterization, and trajectory analysis across these domains, helping to reveal disease progression beyond symptom-based or lesion-centered evaluation.
From Isolated Decisions to Structured Expertise in Contemporary Valve Care
The growing complexity of VHD has necessitated a shift from isolated decision-making toward structured models integrating Heart Valve Clinics, Heart Teams, institutional Centers of Reference, and Valve Networks (Table 1).11–14,19,20 The parallel expansion of multimodality imaging and AI-based tools reinforces the need for standardized, governance-driven environments to safely integrate data-driven support into clinical practice (Figure 2). 17
Artificial intelligence as a facilitator of structured valve care across the patient pathway. Structured organization of contemporary valve care integrating community-based detection, dedicated Heart Valve Clinics, multidisciplinary Heart Teams, and Institutional Heart Valve Centers of Reference, followed by rehabilitation and long-term follow-up. Artificial intelligence supports the care pathway at multiple levels by enabling imaging quantification and risk stratification, patient education and engagement, trajectory-based risk assessment with follow-up prioritization, and data integration for outcome monitoring and quality assurance. Importantly, AI functions as a transversal decision-support layer under expert clinical and Heart Team governance, complementing, but not replacing, clinical judgment throughout the disease trajectory.
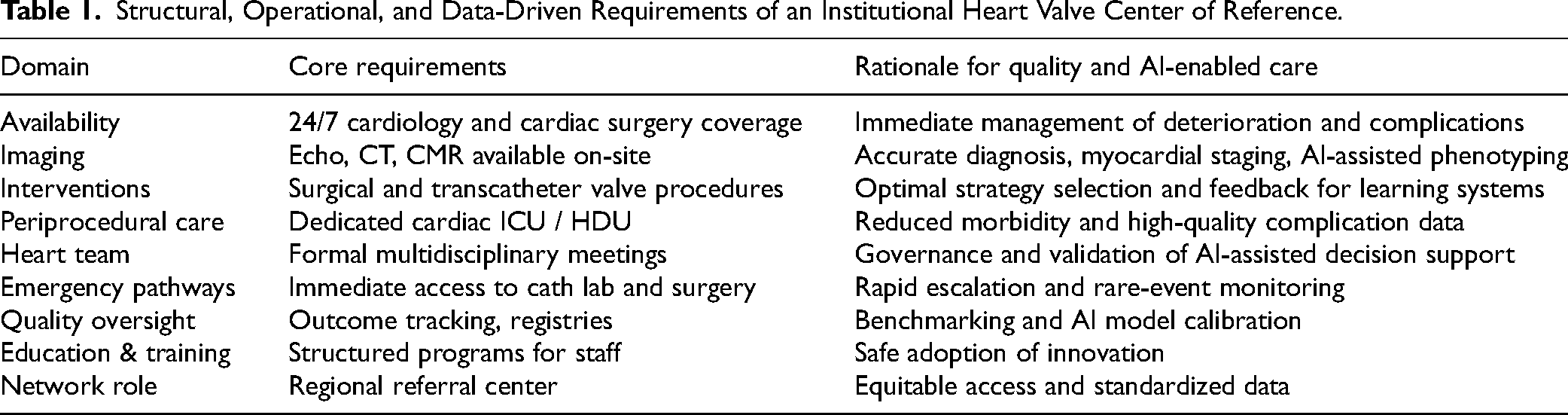
Structural, Operational, and Data-Driven Requirements of an Institutional Heart Valve Center of Reference.
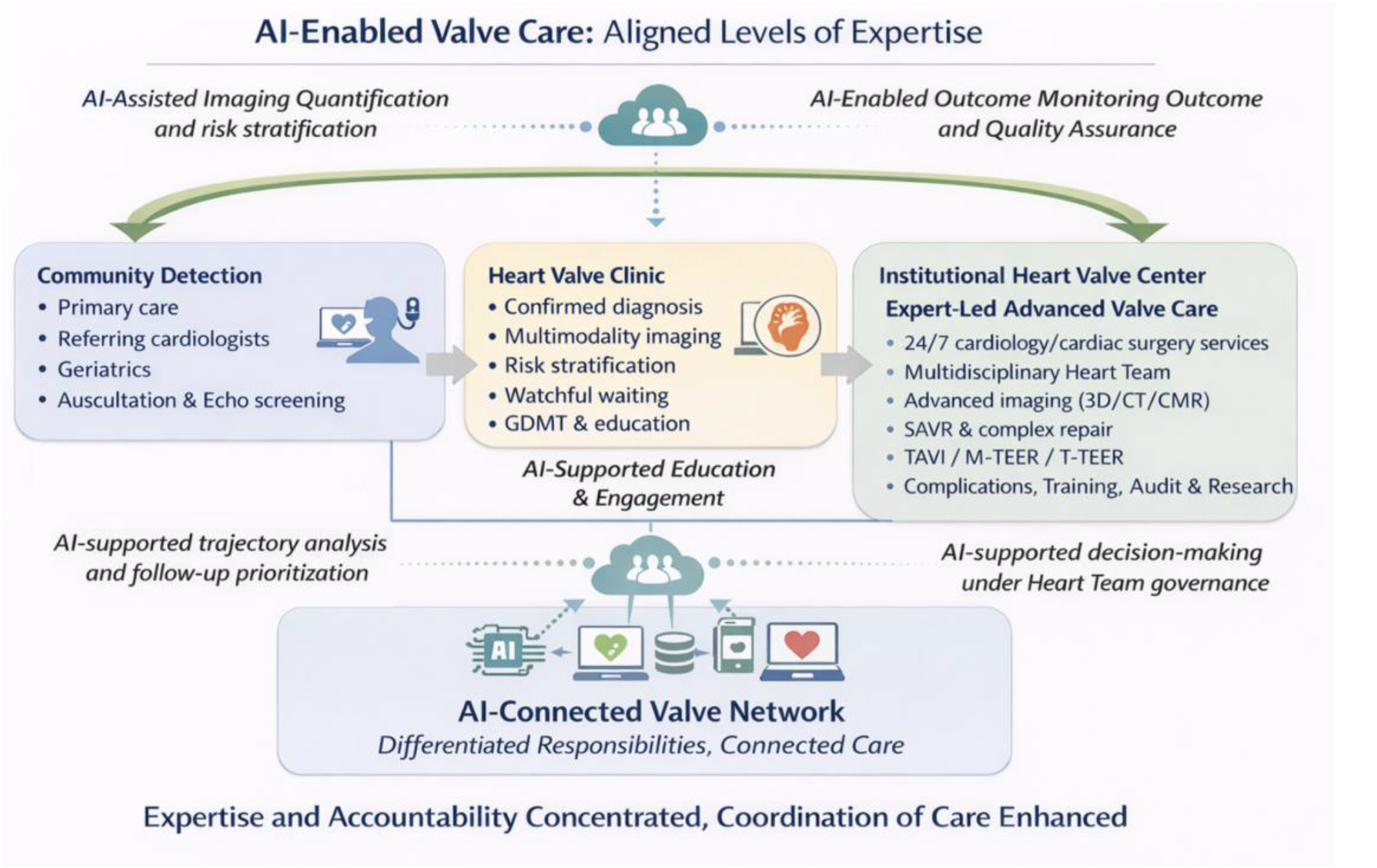
Heart Valve Clinics constitute the entry point of structured valve care and play a pivotal upstream role within this system.19,28,29 They enable early diagnosis, confirm disease severity, and ensure standardized longitudinal surveillance of patients with known or suspected valve disease, including those who are asymptomatic or present with borderline findings. Beyond lesion assessment, dedicated clinics support longitudinal evaluation of ventricular involvement and systemic consequences, consistent application of contemporary recommendations, optimization of guideline-directed medical therapy, coordination with referring physicians, and recruitment into clinical trials, thereby reducing the risk of delayed referral or loss to follow-up.13,14 From a surgical and interventional perspective, this upstream organization facilitates earlier referral and more appropriate timing of intervention, allowing treatment before irreversible myocardial damage has occurred. In this context, digital tools may further reinforce the role of Heart Valve Clinics by improving understanding of disease status, recognition of clinically relevant symptoms, and engagement with structured follow-up pathways. When embedded within standardized workflows, emerging AI-enabled tools, such as ECG-based detection, digital auscultation, automated echocardiographic quantification, and risk stratification or alert systems, may further enhance early identification of clinically relevant disease and timely referral, provided they operate under expert clinical supervision and governance.30,31
Institutional Heart Valve Centers of Reference represent organizational entities rather than individual teams or operators. 20 Embedded within hospitals or academic medical centers, they provide continuous access to cardiology and cardiac surgery services, advanced multimodality imaging, comprehensive periprocedural care, and structured pathways for complication management. Their mission extends beyond procedural delivery and includes responsibility for training and education of surgeons, interventional and imaging cardiologists, specialized nurses, and allied professionals, as well as systematic monitoring of outcomes and quality indicators in complex, multidimensional valve disease.13,14 Such centers also provide the critical infrastructure required for the responsible deployment of AI, including standardized data acquisition, multidisciplinary validation of algorithmic outputs, and continuous outcome-based feedback. For surgeons, this environment facilitates complex procedures, redo surgery, hybrid lifetime strategies, and training in evolving techniques, while allowing AI systems to function as learning tools rather than static predictors. 32
At the core of both the clinic and the center lies the multidisciplinary Heart Team. By integrating surgical, interventional, imaging, anesthetic, and geriatric expertise, Heart Teams mitigate individual bias and align therapeutic strategies with person-centered goals, particularly when evidence is limited or when clinical profiles differ from those represented in randomized trials.13,14 Artificial intelligence–based risk models and imaging analytics may support this process by providing objective estimates of procedural risk, durability, and lifetime management strategies; however, algorithmic outputs must be interpreted in the context of surgical judgment, anatomical nuance, and individual preference. 33 In this sense, the Heart Team functions as both a cognitive and governance structure, ensuring that AI-assisted decision support enhances, rather than replaces, expert clinical reasoning. Real-world practice remains heterogeneous, and some centers still operate without formal multidisciplinary Heart Teams, often favoring a single therapeutic modality. In this context, AI may support more consistent triage and decision-making. However, it cannot replace multidisciplinary expertise, which remains essential for intervention planning.
Finally, Valve Networks extend the benefits of specialized centers to broader populations by linking community-based clinics with Heart Valve Centers of Reference through structured referral pathways. In line with ESC/EACTS recommendations, these networks should not be conceptualized primarily as instruments of care centralization but as organized systems of differentiated responsibilities across levels of care. This model preserves local access while concentrating clinical complexity, advanced interventions, and governance of clinical algorithms in high-expertise centers, while surveillance, longitudinal follow-up, and patient engagement remain locally accessible. This coordinated and connected structure allows expertise to circulate through shared protocols, data exchange, and specialist oversight rather than through patient transfer alone, thereby improving outcomes, consistency, and equity of care across healthcare systems. From a health-system perspective, such networks support sustainability by reducing delayed referrals, avoidable hospitalizations, and late-stage interventions, while enabling lifetime valve management strategies aligned with long-term outcomes and the responsible integration of digital health, telemedicine, and AI-enabled decision support within governed referral frameworks. 17
Procedural Volume, Outcomes, and AI-Enabled Quality Assurance
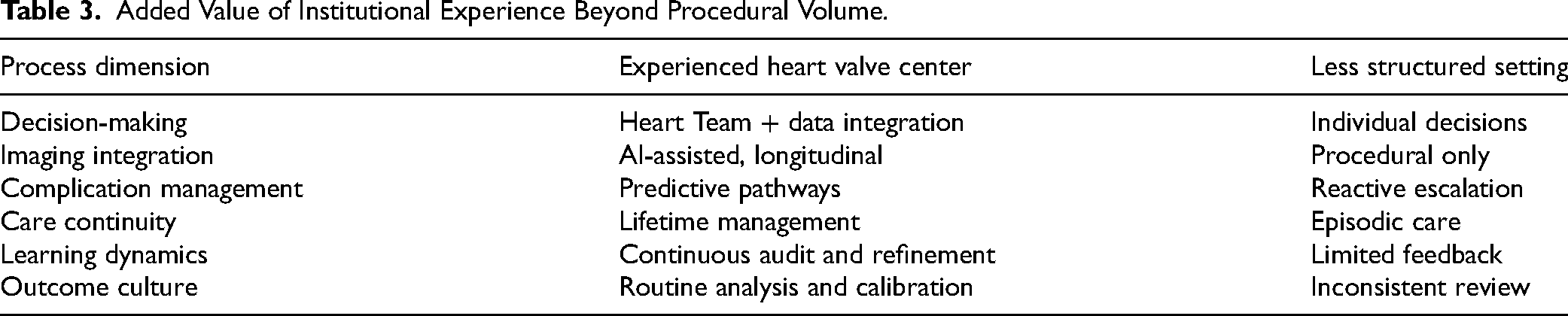
A consistent association between procedural volume and clinical outcomes has been demonstrated across a wide range of valve interventions, with higher institutional volumes linked to improved technical success, fewer complications, and more effective management of adverse events, particularly for complex surgical and transcatheter procedures requiring advanced imaging and multidisciplinary coordination (Table 2).34–36 However, procedural volume should be regarded less as a numerical threshold than as a surrogate marker of cumulative institutional expertise and organizational maturity. Team stability, imaging proficiency, standardized periprocedural pathways, and structured complication management are central determinants of outcome quality and are reinforced through continuous feedback and learning (Table 3). In this setting, AI-based analytical tools may support quality assurance by enabling automated outcome tracking, detection of practice variability, and benchmarking at institutional or network level, without replacing clinical accountability. Concentration of complex procedures within experienced centers embedded in coordinated networks—combined with systematic outcome monitoring, internal audit, and participation in registries—offers a more robust framework for quality assurance than volume metrics alone. 28 Beyond clinical outcomes, such structured models also support sustainability by reducing avoidable hospitalizations, late-stage interventions, and complication-related costs, while facilitating lifetime management strategies aligned with long-term resource efficiency.15,17
Procedural Volume Benchmarks Supporting Outcomes and Data-Driven Quality Improvement.

SAVR, surgical aortic valve replacement; TAVI, transcatheter aortic valve implantation.
Added Value of Institutional Experience Beyond Procedural Volume.
Heart Valve Networks as Enablers of Scalable and Governed AI Integration
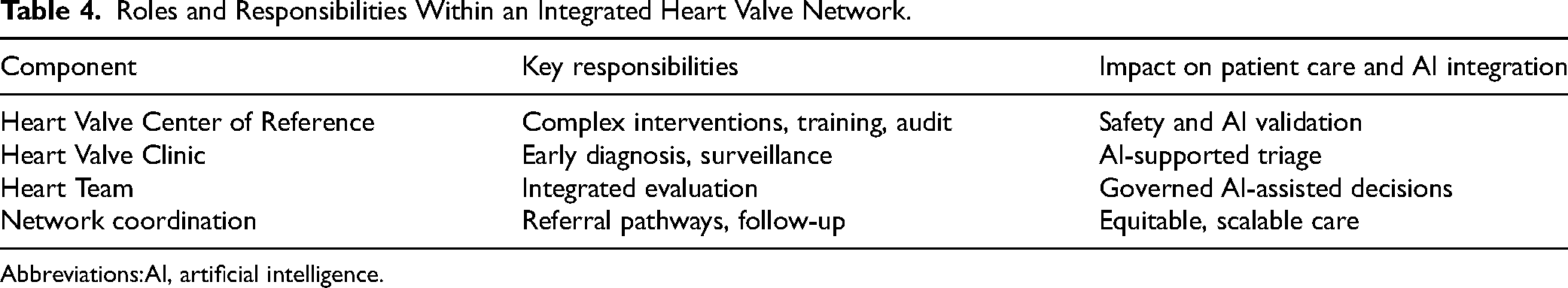
Integrated Heart Valve Networks linking community-based clinics to institutional Heart Valve Centers of Reference structure the critical interface between early disease recognition and access to advanced valve expertise (Table 4). By organizing referral pathways and longitudinal follow-up across healthcare levels, these networks reduce delays in diagnosis and intervention while preserving proximity of care.13,14 Beyond logistical coordination, they provide a scalable framework for consistent patient stratification, standardized data capture, and coordinated management of increasingly complex valve disease. In this setting, digital platforms and AI-enabled data integration may support risk stratification and referral prioritization at scale, particularly for patients who are clinically silent but biologically progressing.31–33 The clinical adoption of AI in valve care, however, is constrained less by algorithmic performance than by challenges related to data quality, interoperability, and integration into clinical workflows. Heterogeneous acquisition protocols, incomplete longitudinal datasets, limited external validation, and the evolving nature of valve disease complicate model calibration and generalizability. In a clinical domain where decisions often lead to irreversible interventions, AI outputs therefore require careful interpretation and robust governance. Artificial intelligence deployment also raises important data security and privacy issues. The use of large-scale multimodal data requires secure infrastructures, clear governance, and regulatory compliance to ensure data protection and maintain trust. Structured valve care systems, characterized by standardized workflows, multidisciplinary oversight, and continuous outcome monitoring, provide the environment in which AI can be validated, recalibrated, and meaningfully integrated into expert Heart Team decision-making, reinforcing rather than replacing clinical judgment. At scale, the effectiveness and sustainability of Heart Valve Networks and AI-supported care pathways also depend on alignment at health-system level. Coordinated national or regional frameworks can facilitate data interoperability, accreditation of specialized centers, harmonized reimbursement models, and equitable access to advanced valve care, while preserving clinical autonomy and expertise-based governance. Such alignment positions Heart Valve Networks not only as organizational solutions but as enabling infrastructures for responsible, data-driven innovation in contemporary valve care.
Roles and Responsibilities Within an Integrated Heart Valve Network.
AI, artificial intelligence.
Conclusions
Heart valve disease has become a multidimensional condition in which valve lesions, myocardial damage, and clinical presentation frequently diverge. In this context, isolated and procedure-focused decision-making is no longer sufficient. Heart Valve Clinics, multidisciplinary Heart Teams, Heart Valve Centers of Reference, and coordinated Valve Networks provide the organizational framework required to manage this growing complexity. When embedded within such structured systems, AI acts as a facilitator of expertise-based, longitudinal decision-making, enhancing consistency, timeliness, and equity of care, while remaining subordinate to clinical judgment and supported by robust governance and data protection frameworks.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
