Abstract
Despite significant diagnostic and therapeutic advances in valvular heart disease (VHD) care, all populations do not yet benefit equally. Established evidence reveals persistent disparities across the VHD care continuum, particularly affecting underserved populations such as women, racial and ethnic minorities, low-income individuals, and rural residents. In recent years, advancements in healthcare information technology (IT) infrastructures and the rapid emergence of artificial intelligence (AI) models have equipped us with smarter, more capable electronic health record (EHR) systems and digital tools to help tackle underdiagnosis and undertreatment challenges. However, without a specific equity lens embedded in both research initiatives and real-world implementations, these technological advancements risk leaving disparities unaddressed. In this review, we call for translating current awareness about disparities in VHD care into measurable actions. First, we review documented disparities across three main domains: diagnosis/detection, surveillance/referral, and treatment. Then, we discuss promising IT-based solutions aimed at mitigating these disparities, focusing on three key areas: AI-augmented diagnostic tools, EHR-based clinical decision support systems, and telehealth.
This is a visual representation of the abstract.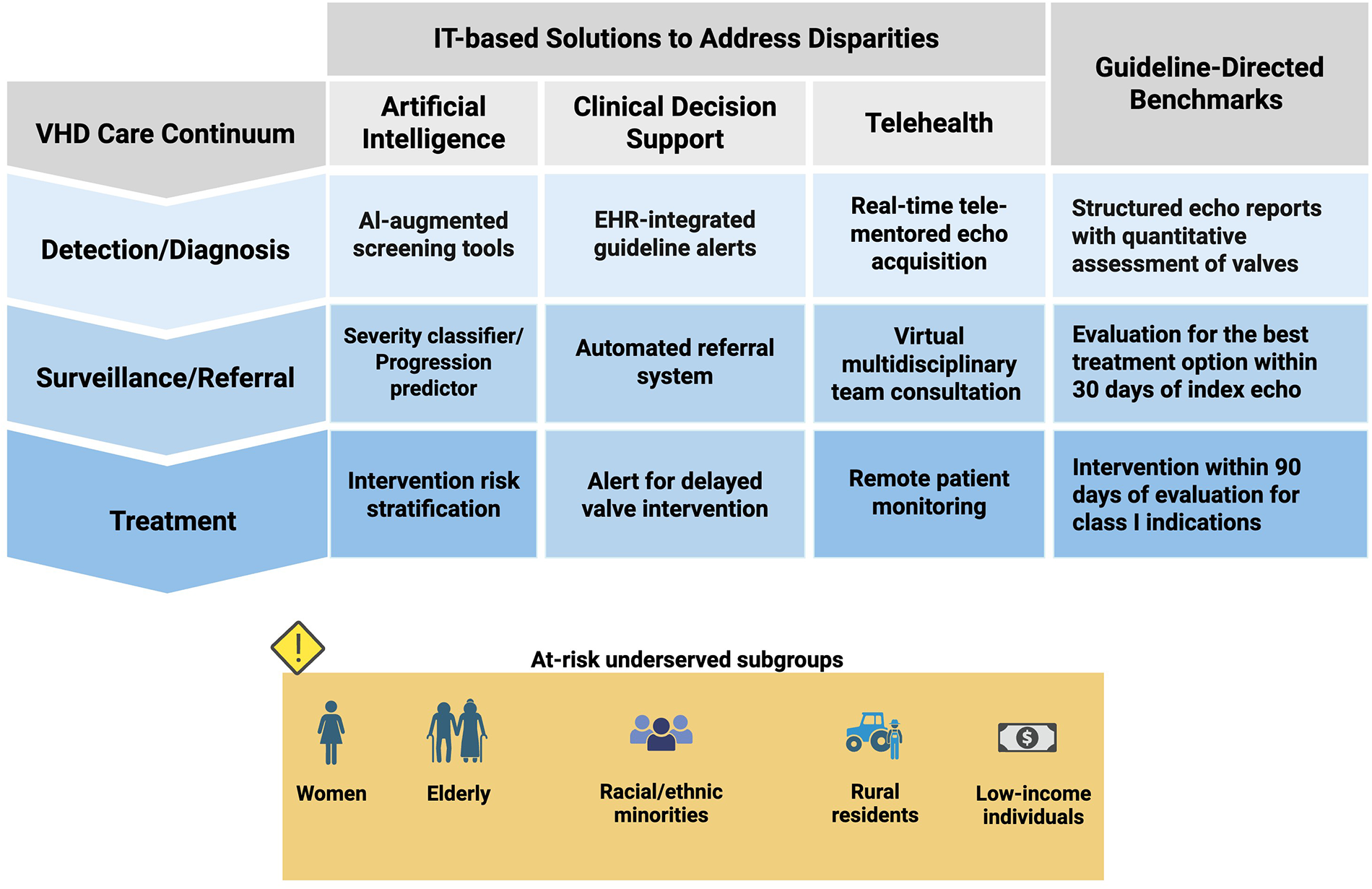
Keywords
Key Points
Despite major advances in valvular heart disease care, significant inequities persist among underserved populations. Advancements in health information technology infrastructure and the rapid emergence of artificial intelligence models present a promising opportunity to identify and address structural disparities across the care continuum. Translating this potential into measurable impact requires a deliberate equity lens embedded in both research designs and real-world implementations.
Introduction
Valvular heart disease (VHD) has grown in prevalence, primarily due to enhanced diagnostic capabilities coupled with an aging population. Yet despite an increasing prevalence, mortality rates of VHD have declined over the past two decades, likely due to improved surveillance and advances in treatment. 1 Transcatheter aortic valve replacement (TAVR) for aortic stenosis (AS) and transcatheter edge-to-edge repair for mitral regurgitation (MR) in particular have transformed the landscape of interventions in VHD. Though there has been rapid growth in intervention rates, a substantial proportion of eligible patients remain untreated, 2 and these advances have not benefited all populations equitably. 1 Gender, racial/ethnical, and socioeconomic disparities remain deeply embedded across the VHD care continuum. Numerous studies have sought to identify where and how these inequities arise, but no widely adopted, system-level approach has yet emerged to close these gaps. 3 Most efforts remain fragmented, limited to isolated pilot programs or institutional initiatives.
In recent years, however, a new opportunity has emerged. Advances in information technology (IT), particularly in data tracking systems and artificial intelligence (AI) applications, hold promise for transformative improvements in healthcare delivery. The growing structure and interoperability of electronic health records (EHRs), along with increasing integration of AI across clinical domains, offer powerful tools. We can now leverage these technologies to identify inequities in VHD care across the population, automating clinical workflows, facilitating timely referral to heart valve teams (HVTs), and reducing human bias. Although several IT-based initiatives have been developed in the context of VHD, few studies have systematically evaluated their impact on reducing disparities.
In this review, we aim to outline disparities across the VHD care pathway and explore the landscape of IT-based solutions, with a focus on EHR-based and AI-augmented tools and initiatives.
Disparities in VHD Healthcare Services
Inequity in healthcare is multifactorial, widespread, and frequently underrecognized. There are structural barriers that play a central role in perpetuating these inequities across the care continuum (Figure 1). Many individuals, especially underserved populations, experience barriers to timely and appropriate care, which has resulted in a detrimental impact on clinical outcomes. Growing evidence has identified older adults, women, racial/ethnic minorities, rural residents, and low-income patients as the most vulnerable subgroups. 4 In the following section, we briefly review disparities in VHD care by dividing them into three main pillars: detection and diagnosis, surveillance and referral, and treatment.
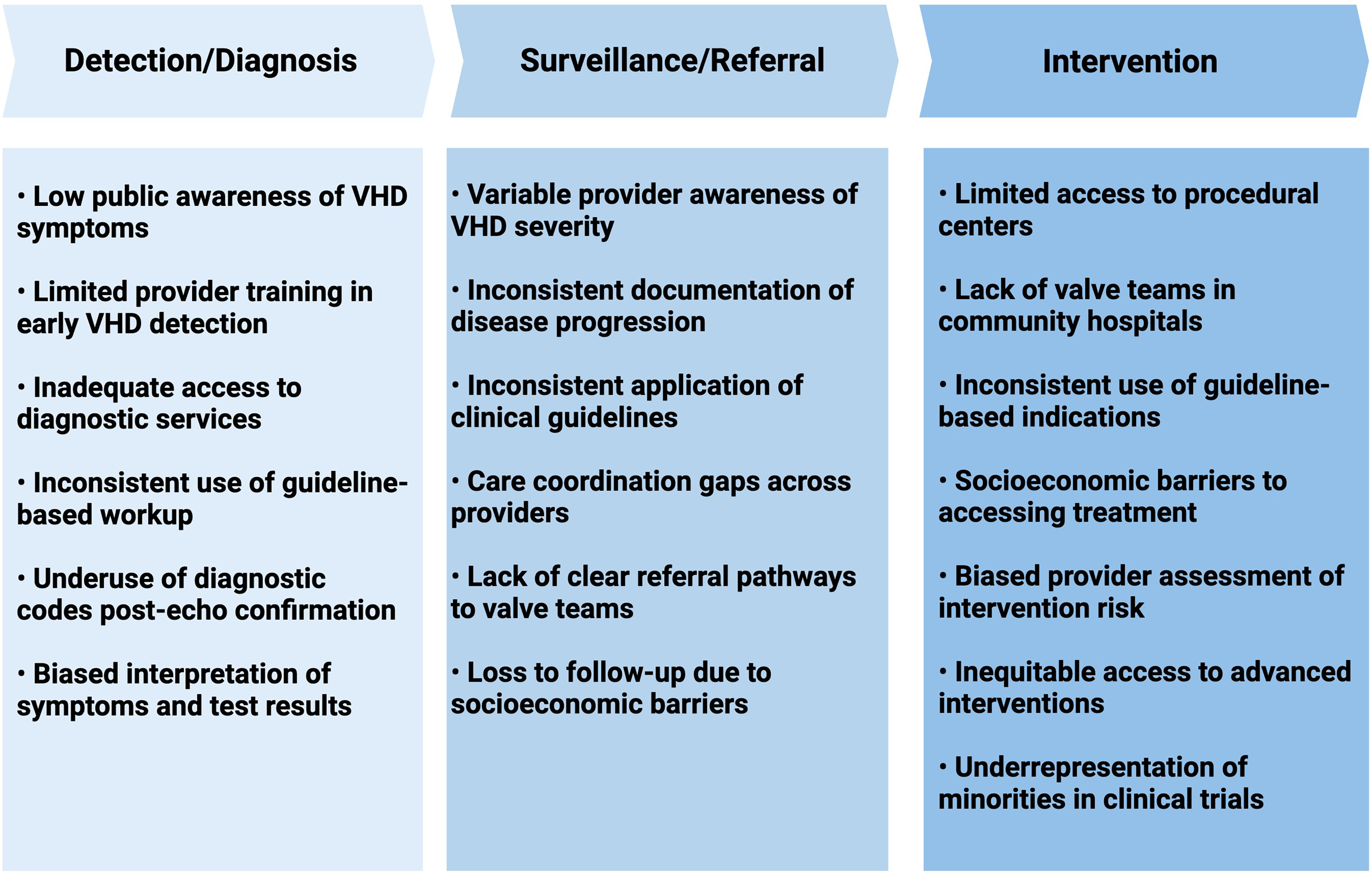
Structural Barriers to Equitable Care in Valvular Heart Disease. VHD: valvular heart disease.
Detection and Diagnosis
Best practices in VHD care begin with timely diagnosis. Early detection of valve dysfunction, ideally before symptoms develop or irreversible cardiac damage occurs, is multifactorial, depending on patient awareness, access to care, and accurate clinical evaluation. Each of these steps may be influenced by inequities. Several cohort studies have focused on the underdetection of VHD and the underlying causes of its variable prevalence among underserved populations. Still, our understanding in this area is limited and may be skewed. Black/African American (AA) patients appear to have a paradoxically lower documented prevalence of AS than White patients despite a higher prevalence of risk factors for AS. This “aortic stenosis paradox” suggests that many non-White patients with clinically significant valve disease are not being identified. 5 Patel et al identified a cohort of 272 525 patients with echocardiographic data at Vanderbilt University Medical Center, of whom 222 976 were White (81.8%) and 36 681 were Black/AA (13.5%). Severe AS was observed in 0.29% of Black/AA patients and 0.91% of White patients; after multivariate adjustment, Black/AA patients remained significantly less likely to be diagnosed with severe AS than White patients (adjusted odds ratio: 0.41; 95% confidence interval [CI]: 0.33-0.50). 6 Importantly, this pattern persists even in the assignment of diagnostic codes for AS. In another cohort study, Black/AA patients were significantly less likely to be assigned a diagnostic code for AS despite meeting echocardiographic criteria (hazard ratio [HR]: 0.65; 95% CI: 0.54-0.77; P < .0001). 7 When interpreting these findings, it is important to note that most cohorts rely on echo-based data, and access to echocardiography is not proportionate among underserved populations within the studied health systems. The rate of undetected VHD, individuals with VHD who never undergo diagnostic evaluation by echocardiography, remains unclear across populations and warrants further research.
Women with VHD are also frequently diagnosed late, presenting at older ages and with more advanced symptoms. 4 Compared with men, women are less often diagnosed with severe MR (70% vs 81%; P = .002) and severe AS (adjusted OR: 0.84; 95% CI: 0.83-0.86).8,9 Women have smaller body surface area and left ventricular dimensions, which without proper indexing of echocardiographic thresholds, lead to underestimation of VHD severity. 8 There is a higher prevalence of low-flow, low-gradient AS in women, 10 emphasizing the need to account for sex-specific differences and to use adjunctive diagnostic modalities and screening tools to improve severity assessment in VHD.
In response to these unmet needs, efforts were initiated to generate reliable, community-level prevalence estimates of VHD. PREVUE-VALVE (NCT05357404) is a prospective, population-based study designed to provide age- and gender-specific prevalence rates of VHD among older adults in the United States. To mitigate underdetection due to limited access to echocardiography in underserved populations, it conducts standardized in-home echocardiography with core laboratory adjudication for detection and grading of VHD. Concurrently, blood samples, electrocardiograms (ECGs), and structured health-status inventories are collected to support the development and validation of novel screening tools. PREVUE-VALVE has completed enrollment with 2870 participants, and results are expected in the near future.
Surveillance and Referral to HVT
Most forms of VHD are chronic and require ongoing surveillance with serial echocardiography and timely referral to HVTs when intervention is indicated. Compared to other phases of care, the surveillance and referral processes are more difficult to track, making it challenging to identify and address potential inequities.7,11 This is likely due to limitations in structured data within EHRs, data not reliably captured or entered into the EHRs, the absence of distinct billing codes for longitudinal management steps, and variation in referral workflows across healthcare systems.
Available data suggest that underserved populations are less likely to receive appropriate surveillance for VHD. In a cohort of 130 725 echocardiograms representing 42 289 unique patients at Massachusetts General Hospital, women (OR: 0.90; 95% CI: 0.86-0.95; P < .0001), Black/AA patients (OR: 0.74; 95% CI: 0.66-0.83; P < .0001), and Medicaid patients (OR: 0.85; 95% CI: 0.75-0.96; P = .009) were less likely to receive guideline-recommended surveillance. 12 Similarly, another study reported the lowest surveillance rates among Black/AA women. 13 Black/AA patients have also been shown to be significantly less likely to be referred to cardiothoracic surgery for aortic valve disease (OR: 0.48; P < .001), whereas referral rate for Hispanics was comparable to that of White patients (OR: 0.86; P = .73). 14
Intervention
Although the 2020 ACC/AHA guideline for the management of patients with VHD provides clear indications for intervention, 15 the annual rate of performed procedures remains substantially lower than the number of patients with a Class I indication.1,2 Notably, the undertreatment rates vary across demographic and socioeconomic groups, with women, racial and ethnic minorities, and low-income individuals remaining less likely to receive timely intervention. Based on National Inpatient Sample data, utilization rates of valve interventions, defined as the number of procedures per 100 000 US adults aged ≥65, were consistently higher in White patients compared to Black/AA and Hispanic patients: TAVR (43.1 vs 18.0 vs 21.1), transcatheter mitral valve repair (5.0 vs 3.2 vs 3.2), and left atrial appendage occlusion (6.6 vs 2.1 vs 3.5), respectively (P < .001). 16 Moreover, only approximately 10% of patients in the Transcatheter Valve Therapy (TVT) Registry undergoing TAVR identify as members of racial minority groups, a figure far below their estimated representation in the severe AS population. Notably, procedural outcomes are comparable across racial and ethnic groups when treatment is provided, 17 underscoring that the primary barrier is access to care, not the effectiveness or quality of the intervention itself.
Other known indicators of access to care are socioeconomic status (SES) and geographic characteristics of patients. Communities with lower SES, lower household income, and rural settings are less likely to receive valvular interventions. Nathan et al evaluated the geographic distribution of TAVR program establishment in the United States between 2012 and 2018, the early years following FDA approval of TAVR in 2011, and its association with the SES of treated patients. Two key findings emerged: First, hospitals in metropolitan areas were more likely to adopt TAVR programs than those in micropolitan areas (OR: 2.70; 95% CI: 1.40-5.19; P < .001), with most growth occurring in regions where TAVR programs already existed. Second, compared with similar hospitals that did not establish a TAVR program, TAVR hospitals treated patients with higher median household incomes, lower Distressed Communities Index (DCI) scores, and fewer patients dually eligible for Medicaid. 18
A similar pattern of inequity has been reported between the patient populations of hospitals that participated in clinical trials for transcatheter valvular therapies and those of candidate hospitals that did not. Participating hospitals served patients with higher median household incomes (difference: $5261; 95% CI: $2986-$7537) and lower DCI scores (difference: −5.37; 95% CI: −8.15 to −2.59). 19 These trends underscore an important consideration for pioneers seeking to reduce care inequities with advanced, technology-based solutions: given that these innovations are often introduced by academic or high-resource centers, goal-directed site selection and proactive engagement of underserved communities will be essential to ensuring they benefit the populations most in need.
IT-Based Solutions
Role of AI
The recent surge in AI applications, particularly machine learning (ML) and deep learning (DL) models, has introduced novel opportunities for integration into clinical practice across various medical fields. In the domain of VHD, AI has demonstrated promising utility in patient screening and disease severity classification when incorporated into conventional diagnostic tools, such as digital stethoscopes, ECGs, and echocardiography. 20 Importantly, this technology also holds potential as a practical strategy for addressing disparities in care. 21 However, to date, no AI-based model has been specifically developed to mitigate disparities in VHD care. Only a limited number of studies have conducted posthoc analyses to evaluate whether AI-integrated tools reduce disparities or replicate human biases related to gender and race. In this section, we review several AI-integrated diagnostic tools with demonstrated promise in VHD detection and discuss their implications for healthcare equity.
Automated Digital Stethoscope
Since the invention of the stethoscope in the early 1800s, auscultation has remained a fundamental part of every physical examination. It is widely accessible, inexpensive, safe, easy to learn, and potentially a reasonable first screening tool for detecting murmurs caused by structural heart disease (SHD). However, interpretation of heart sounds shows variable interrater and intrarater agreement, even among cardiologists,22,23 and the ability to identify the underlying pathological features is often limited. 24 In recent years, digital stethoscopes have leveraged phonocardiograms and visual plots of heart sounds to better classify cardiac pathologies and distinguish heart murmurs from background noise.
Various research teams have explored augmenting digital stethoscopes with DL algorithms to enhance their diagnostic performance.25,26 Chorba et al evaluated the diagnostic performance of the FDA-approved Eko's automated heart murmur detection algorithm, which uses a convolutional neural network trained on over 34 h of previously annotated heart sound recordings from approximately one thousand patients, paired with echocardiogram data. All phonocardiograms were classified into one of the three categories: “heart murmur,” “no heart murmur,” or “inadequate signal.” These results were then compared to the auscultatory findings of a cardiologist who was blinded to both the algorithm output and echocardiogram results. When used at the appropriate anatomic auscultation location, the algorithm detected moderate-to-severe or greater AS with a sensitivity of 93.2% and specificity of 86.0%, and moderate-to-severe or greater MR with a sensitivity of 66.2% and specificity of 94.6%. 26 These results are notable given that conventional auscultation has shown an overall sensitivity of just 44% for detecting VHD, resulting in most patients remaining underdiagnosed. 27
Unfortunately, none of the related studies has addressed the role of their proposed tool in mitigating disparities. Where feasible, we recommend posthoc analyses of study results to assess impacts on gender and racial/ethnic disparities. AI-augmented stethoscopes hold considerable promise as first-line screening tools for earlier detection of VHD, with a potentially lower risk of disparity-related bias. They can be deployed in community health care settings as a low-cost screening intervention, particularly in primary care clinics where access to advanced diagnostic modalities is limited and clinical expertise in VHD detection may vary.
Electrocardiogram
The ECG plays an essential role in cardiovascular care. It is widely accessible, rapid, inexpensive, and safe. Compared to more advanced diagnostic modalities such as echocardiography or cardiac catheterization, ECG acquisition is less time-consuming and does not require specialized providers or equipment. VHDs often lead to cardiac remodeling during their progression, resulting in alterations in cardiac mass, chamber size, systolic and diastolic function, and secondary valvular abnormalities. Some of these changes may manifest on ECG earlier than they are detectable by echocardiography; for instance, axis deviation, increased QRS complex voltage, and right/left bundle branch block can be an early manifestation of ventricular hypertrophy/dilation. While ECG alone lacks sufficient accuracy to serve as a definitive diagnostic tool, AI-integrated ECG systems offer promise as screening tools, particularly in primary care and community-based settings where access to advanced diagnostic imaging is limited. Additionally, newer wearable devices, including smartwatches, fitness bands, and rings, are capable of capturing single-lead ECG data and may serve as valuable tools for detecting VHDs at the population level. 28
Promising efforts are in progress to enhance the potential screening application of AI-integrated ECG for VHD detection.29–32 Although none of the models have been explicitly designed to address disparities in VHD, most studies have evaluated model performance across subgroups defined by age, gender, and race/ethnicity. In general, accuracy remains consistent across these groups, highlighting the potential of AI-augmented tools to reduce existing disparities in VHD detection.
EchoNext is a robust AI-ECG screening model for SHD, including moderate or severe VHD. It was trained on more than 1.2 million paired ECG–echo studies from 230 318 patients across eight New York-Presbyterian-affiliated hospitals serving a diverse population. Across the training, validation, and test sets, racial and gender distributions closely reflected the underlying population (women, 48.7%-52.1%; White, 33.9%-35.4%; Hispanic, 19.8%-23.1%; Black/AA, 14.5%-17.8%). Incorporating EchoNext significantly improved cardiologists’ accuracy for detecting SHD from ECGs alone. Cardiologists’ accuracy differed for clinically normal versus abnormal ECGs (69% vs 62%), whereas the model performed similarly in both groups (77% each). Performance was consistent across care settings and across racial and ethnic subgroups. 33
In a pilot study of 100 patients with an ECG but no echo in the prior 3 years and no known VHD, EchoNext classified 33 as high risk (24% with left-sided VHD; 73% with SHD) and 50 as moderate risk (2% with left-sided VHD; 28% with SHD), missing no cases of VHD. 33 These findings support AI-ECG models as scalable tools for VHD screening at the population level and suggest a path to reduce existing inequities in underserved communities.
Another DL-ECG model developed based on diverse populations from five Mount Sinai hospitals within New York City achieved area under the receiver operating characteristic curve (AUROC) ranges of 0.81 to 0.88 for MR and 0.86 to 0.89 for AS in both internal and external validations. Performance detection was reported to be constant across age, racial/ethnical, and gender subgroups. 31
Ulloa-Cerna et al employed a robust dataset comprising ECG traces, EHR, and echocardiogram-confirmed VHD condition from more than 434 000 patients to develop their ML-based ECG model. 32 After testing various input combinations, they found that a model incorporating only age, sex, and ECG traces provided the best balance of performance, objectivity, and portability. The model achieved an AUROC of 0.91, 42% positive predictive value, and 90% sensitivity. Inclusion of age and sex, both widely available in any standard ECG system, improved detection accuracy and allowed the model to account for physiological differences, which may help reduce bias across these demographic groups.
Echocardiography
Echocardiography remains the gold standard for the diagnosis and assessment of severity for VHD. Hemodynamic measurements obtained via Doppler imaging, combined with anatomical assessments from two-dimensional (2D) and four-dimensional modalities, provide a comprehensive evaluation of valve function and its hemodynamic consequences. However, image acquisition, quantitative measurement, and interpretation require a highly skilled echocardiographer and experienced valve specialist, and the process can be time-consuming. Moreover, assessing VHD severity can be challenging when quantitative parameters are discordant; for example, in low-gradient severe AS, with preserved ejection fraction, such discrepancies may lead to the diagnosis being missed or delayed. These may be more pronounced in low-resource settings, high-volume centers, and among underserved populations.
AI has already been integrated into echocardiography imaging to support automated image quality assessment, view classification, region segmentation, volume and ejection fraction calculation, and disease classification. While most current applications emphasize improvements in image acquisition and measurement precision, there is a growing need for AI models that assist in higher-order interpretation, such as flagging clinically relevant findings or identifying missed diagnoses.
In recent years, several research teams and AI companies have focused on developing models to address the underdetection of VHDs, aiming to improve the diagnosis and classification of VHDs based on echocardiographic data. Yang et al developed an automatic screening tool for echocardiographic videos that classifies echocardiographic views, detects the presence of VHDs, and, when present, quantifies key metrics related to valve disease severity. The model was then tested in a prospective real-world setting, and results were compared to interpretations by experienced physicians. Disease classification accuracy was high, with AUROC of 0.99 (95% CI: 0.97-0.99) for mitral stenosis, 0.88 (95% CI: 0.86-0.90) for MR, 0.97 (95% CI: 0.95-0.99) for AS, and 0.90 (95% CI: 0.88-0.92) for aortic regurgitation in the prospective test dataset. The model performance also demonstrated acceptable agreement with expert physicians’ estimates of valve lesion severity metrics. 34 Unfortunately, no subgroup analyses were performed to assess potential disparities in model performance across demographic groups.
In addition to a screening role, AI-based models have shown improved classification of VHD severity. Sengupta et al developed an ML-based classifier for echocardiographic grading of AS severity, validated by cardiac magnetic resonance and computed tomography data. Compared with conventional strategies for AS severity classification, ML-based reclassification has shown improved discrimination for AS severity, especially in cases with discordant echocardiographic findings. 35
There are also other AI-based efforts aimed at facilitating VHD diagnosis in the setting of limited echocardiographic data, such as when Doppler images or gradient measurements are unavailable.36–38 In one example, Wessler et al developed an automated AS detection model using limited 2D transthoracic echocardiographic datasets. External validation in a cohort of 8502 outpatient transthoracic echocardiograms showed that screening for AS can be achieved using parasternal long-axis imaging alone, with an AUROC of 0.91. 36
Such models could also be a scalable adjunct to portable point-of-care devices with more limited measurement capabilities than comprehensive echocardiography evaluation, or to studies performed for unrelated clinical indications in emergency or critical care settings, enabling earlier identification of patients at risk for VHD and prompting timely referral for further evaluation. However, they still require real-world validation and regulatory approval before being adopted into routine clinical practice.
Furthermore, while all these modalities show promise in improving population health screening for VHD, any proposed solution must be integrated into clinical workflows in a way that does not overwhelm the health system with the need for confirmatory testing. Given that none of these screening tools offers definitive diagnostic accuracy for VHD, health systems will also need to address downstream challenges, such as elevated false-positive rates and increased demand for confirmatory echocardiography. In systems already struggling to meet existing demands and undertreating patients with known severe VHD, this may unintentionally widen disparities in care.
EHR-Based Clinical Decision Support System
Clinical decision support (CDS) tools have demonstrated a promising role in cardiovascular care. In heart failure management, CDS has already been integrated into routine workflows in several settings, improving patient identification, risk stratification, and adherence to guideline-based management, ultimately reducing hospitalizations and mortality.39–41 Similar approaches are now being extended to VHD; digital health platforms such as CardioCare (Egnite), Next (Tempus), and Echo Surveillance Manager (Biome) leverage natural language processing (NLP)-enabled algorithms to analyze multimodal patient data from EHRs to identify high-risk, missed, or clinically prioritized patients with significant VHD. NLP has demonstrated strong performance in extracting large-scale data from unstructured clinical documents, such as echocardiographic reports and clinical notes, where structured tabular data may be limited or unavailable. For example, in a study by Généreux et al aimed at quantifying the burden of untreated severe AS, an NLP-based algorithm developed by the Egnite platform was applied to more than 1.6 million echocardiographic reports across 24 centers to classify AS severity. Model performance was validated against manual review of 8000 reports, achieving excellent accuracy (99.88% [99.78%-99.94%]). 42 While these platforms hold promise for reducing diagnostic and treatment delays, their real-world impact on addressing disparities in VHD care remains insufficiently understood.
The Electronic Physician Notification to Facilitate the Recognition and Management of Severe Aortic Stenosis (DETECT AS) trial is the first randomized trial evaluating an electronic provider notification system for patients with VHD. 43 In this trial, clinical providers were randomized to either usual care or receive an electronic physician notification (EPN) when a patient's echocardiogram met criteria for severe AS (aortic valve area ≤1.0 cm2). A total of 144 providers were assigned to the intervention arm (n = 496 patients) and 141 to the control arm (n = 443 patients). The primary endpoint, rate of aortic valve replacement (AVR) within 1 year following the index echocardiogram, was significantly higher in the EPN group (48.2%) compared with usual care (37.2%). Within 1 year, patients in the EPN group had longer life in both the overall cohort (by 12 days; P = .04) and by nearly a month among symptomatic patients (by 23 days; P = .01).
Notably, the benefit of EPN was more pronounced in underserved populations (Figure 2). Among patients aged >80 years, the odds of undergoing AVR were twice as high with EPN (OR: 2.00; 95% CI: 1.17-3.41; P = .01), and the effect was even greater among women (OR: 2.78; 95% CI: 1.69-4.57; P < .001). 43 These findings suggest that CDS tools, such as DETECT AS, may play an impactful role in mitigating disparities in care for patients with severe AS. The next phase of the project, currently in its pilot phase, will further evaluate the impact of an active intervention that integrates CDS with a facilitated referral system to HVTs for patients identified with severe AS. Notably, after a window of time, the intervention will automatically refer patients for whom the provider does not either accept or reject HVT referral when recommended by the CDS tool.
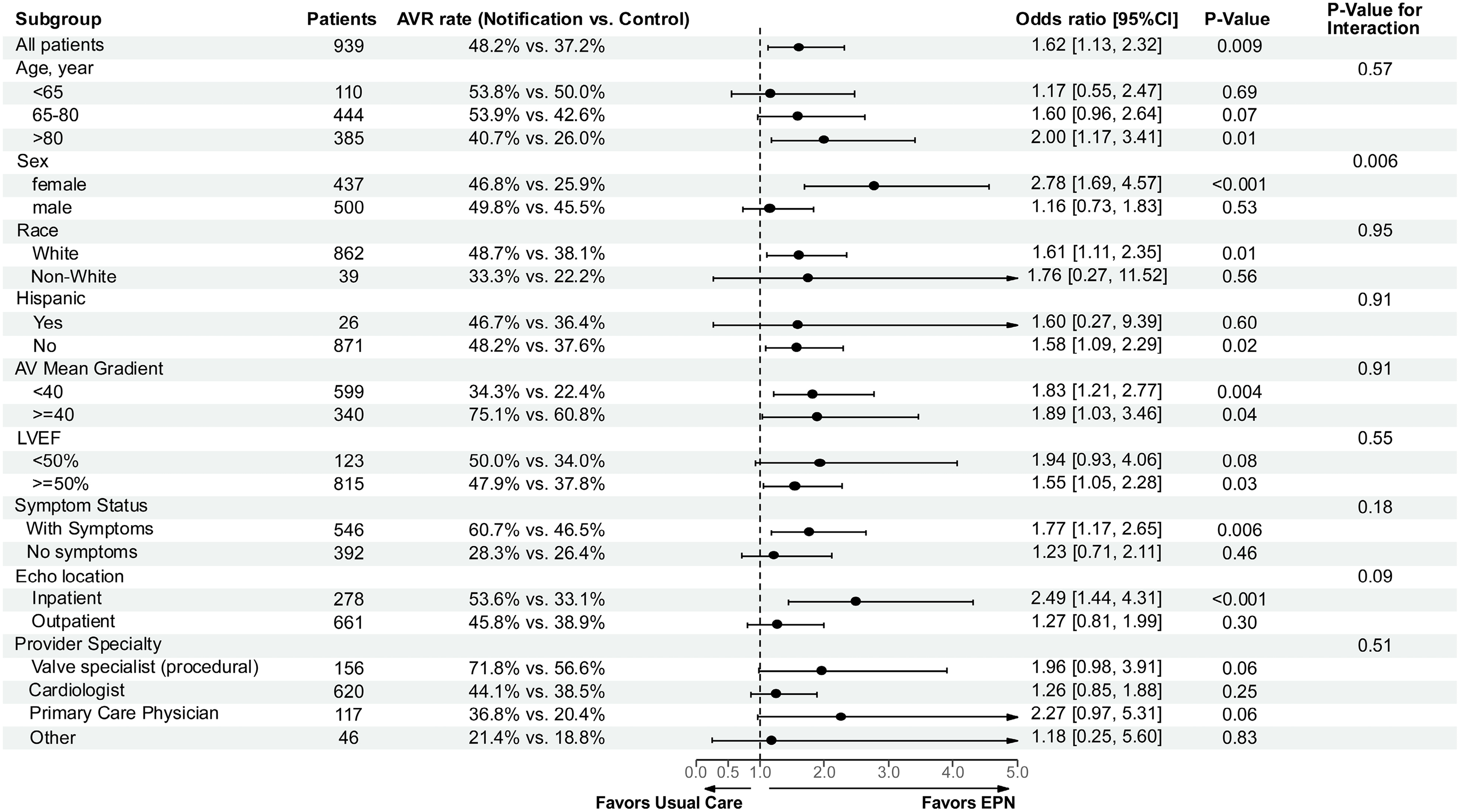
Impact of Electronic Physician Notification on AVR Rates Across Prespecified Subgroups. Reprinted with permission from Tanguturi et al. Circulation. 2025;151(21):1498-1507. ©2025 American Heart Association. https://doi.org/10.1161/CIRCULATIONAHA.125.074470. Abbreviations: AV: aortic valve; AVR: aortic valve replacement; EPN: electronic provider notification; LVEF: left ventricular ejection fraction.
Another ongoing multicenter, randomized controlled trial, Addressing Undertreatment and Health Equity in AS and MR Using an Integrated EHR Platform (ALERT trial), is evaluating the impact of a CDS platform that delivers automated, EHR-integrated notifications on the management of patients potentially indicated for valve intervention. In the intervention group, clinical providers receive automated alerts for patients with possible or definitive severe AS or MR identified on echocardiography. The trial aims to assess whether these alerts increase the rates of (1) transcatheter or surgical valve intervention and (2) timely clinic visits with members of the multidisciplinary heart team. Outcomes will be analyzed across intervention and control arms (no alerts sent), as well as within and between groups stratified by race, ethnicity, sex, and geography. The findings of this trial are expected to provide further insight into the potential of CDS tools to reduce undertreatment and address disparities in VHD care (NCT06099665).
Telehealth
With the rapid growth of digital infrastructure, internet connectivity, and portable technologies, telehealth has become a scalable solution across a range of clinical settings. Integrated with point-of-care technologies, telemedicine has demonstrated promising potential in resource-limited settings by expanding access to care and reducing geographic barriers that often delay diagnosis and treatment.44–46
Point-of-care ultrasound (POCUS), a portable echocardiographic device initially introduced in emergency medicine, is now emerging as a screening tool for VHD. When performed by experienced sonographers, POCUS has demonstrated diagnostic performance comparable to standard echocardiography and significantly greater accuracy than auscultation in detecting VHD. 47 However, its generalizability, accuracy when used by nonexperienced providers, and cost-effectiveness as a screening modality remain to be determined.
In a randomized clinical trial conducted in a low-resource setting among patients with known SHD, adding POCUS along with smartphone-connected mobile health devices to clinical evaluations by local nonspecialist providers led to earlier referrals for valve intervention (83 ± 79 vs 180 ± 101 days; P < .001) and reduced risk of hospitalization and/or mortality (15% vs 28%; adjusted HR: 0.41; 95% CI: 0.21-0.83; P = .013). 44
Real-time telementored echocardiography has also been explored as one of the solutions, enabling remote experts to guide local providers in image acquisition and interpretation. This approach has the potential to improve diagnostic accuracy and expand access to specialist input. However, its effectiveness varies depending on the imaging modality, clinical context, and specific cardiac parameters being assessed. 48
Together, a range of IT-based strategies may support more equitable VHD care, each designed to address specific care gaps across the continuum of care (Table 1). The most effective solution for a given health system will depend on its specific needs and constraints. Identifying data-driven bottlenecks within the care continuum is essential to selecting the tool best aligned with local capacity, patient population, and clinical workflow. These trade-offs must be carefully considered when implementing interventions in real-world settings.
IT-Based Solutions Across the Valvular Heart Disease Care Continuum.
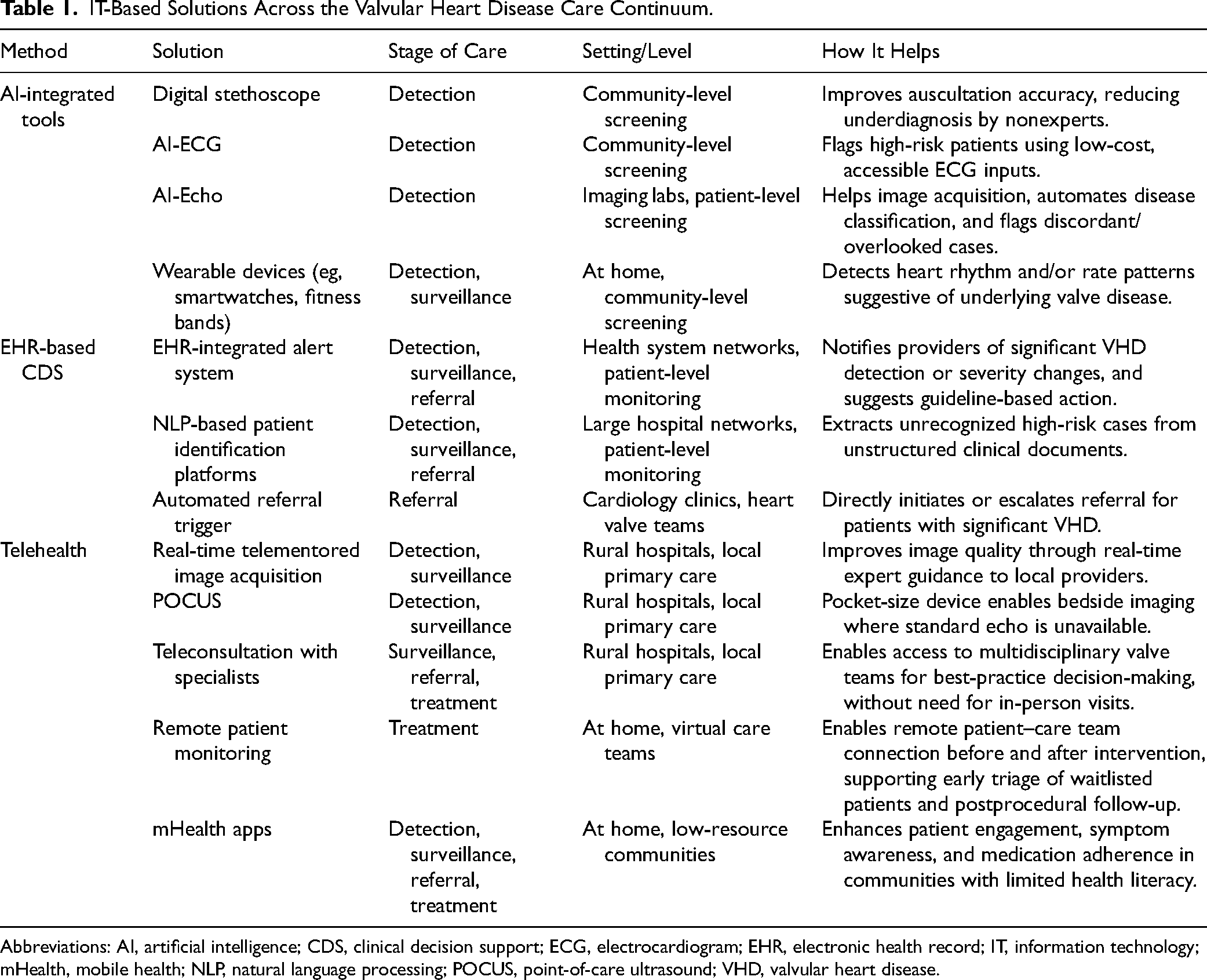
Abbreviations: AI, artificial intelligence; CDS, clinical decision support; ECG, electrocardiogram; EHR, electronic health record; IT, information technology; mHealth, mobile health; NLP, natural language processing; POCUS, point-of-care ultrasound; VHD, valvular heart disease.
Equity Considerations and Future Research Directions
In the field of VHD, comprehensive national registries—such as the Society of Thoracic Surgeons (STS)/American College of Cardiology (ACC) TVT Registry and the STS Adult Cardiac Surgery Database—offer valuable insights into procedural and postprocedural outcomes. However, these registries are largely limited to surgical and transcatheter interventions and do not capture the broader continuum of VHD care. As such, they potentially overlook key sources of disparity across earlier stages of the care pathway. 49
In recent years, multiple cardiovascular societies have called for the need for quantitative metrics to track the quality of care and identify opportunities for improvement in the continuum of VHD management. AHA/ACC has introduced a list of 11 clinical performance and quality measures based on clinical practice guideline recommendations for VHD, intended for use by practitioners and institutions in national quality improvement efforts, public reporting, and pay-for-performance programs. Of note, 8 out of 11 metrics are related to medical treatment and interventions for VHD, and three pertain to patient education and monitoring with echocardiography. 50 Another international consortium initiated by the Heart Valve Society introduced a set of measures to monitor the patient's journey from diagnosis to treatment, with the aim to shift focus from procedure to patient-centered outcomes. Built through a collaborative review of the literature and relevant trials, this dataset is intended to introduce standardized outcome measures globally, enable cross-study comparisons, and guideline evaluation, which ultimately improve VHD care. 51
The American Heart Association Target Aortic Stenosis initiative is the first national quality initiative focused on patients with VHD. 52 This program serves as a compelling example of how registry-based quality platforms can be utilized to trace the patient journey, from initial echocardiographic diagnosis to intervention. Beyond evaluating intervention rates, the program enables a more comprehensive assessment of upstream phases of care, with a particular focus on the timeliness of surveillance, referral, and intervention. This framework aims to identify and address potential care gaps, barriers, and disparities along the care pathway. In the pilot phase of the program, 15 hospitals representing diverse geography, size, and teaching status enrolled a total of 2510 unique patients with AS. Care process in three primary domains: (1) echocardiographic surveillance of AS, (2) completeness of echocardiography reports, and (3) multidisciplinary team (MDT) evaluation and treatment of AS were collected from each participating center. Overall, patients with moderate and severe AS received follow-up echocardiography (95.6% within 24 months, 90.8% within 12 months, respectively). Echocardiography reports were largely complete (91.8%), and 68.4% underwent MDT evaluation for asymptomatic severe AS within 60 days. However, only about half of the patients received AVR within 90 days of diagnosis (51.8%), highlighting ongoing gaps in timely referral and intervention. 52
For the next phase of the program, the study team plans to expand participation to 80 hospitals, with revised metrics building on lessons learned from the pilot phase. Target AS seeks to identify system-level changes that enhance care quality and disseminate best practices to underperforming sites. Importantly, to address existing gender and racial/ethnic disparities in the care pathway, the program will assess whether improvements in quality are equitably distributed across diverse patient subgroups. 52
Efforts to improve the care pathway will remain incomplete without explicitly addressing disparities related to age, sex, race and ethnicity, and SES. 53 Equity considerations require a multifaceted approach throughout the research process, including trials’ study design, predefined endpoint selection, stratified analyses, and transparent reporting. 54 Future research in IT-based solutions and tool implementation efforts should prioritize the deliberate use of these innovations to reduce disparities and promote equitable care delivery.
Conclusions
Despite growing awareness of disparities, inequities in VHD care persist across the patient journey. This review underscores the promise of IT-based initiatives to translate that awareness into measurable improvements in access, consistency, equity, and timeliness of care. However, realizing this potential will require a deliberate equity lens in both research and implementation. Research efforts need to define equity as a core objective with prespecified equity-focused endpoints, inclusive study designs that represent underserved populations, and integrating stratified analyses. Likewise, real-world implementations should aim from the outset to address structural inequities, with embedded mechanisms to monitor, evaluate, and transparently report outcomes across demographic subgroups. A clear and sustained commitment to these principles is essential to ensure that technological innovations do not simply scale care but actively drive more equitable outcomes.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Elmariah has received consulting fees from Edwards Lifesciences and holds equity in Prospect Health. Zier holds equity in Prospect Health. Other authors have reported no conflicts of interest.
