Abstract

In this issue, Tastet et al 1 innovatively described an increased risk of ventricular arrhythmia among 21 women with mitral valve prolapse (MVP) and a history of premature or early menopause (onset before age 45) compared with 92 premenopausal women, 148 women who underwent menopause at the expected age, and 38 women with unknown age at the time of menopause. These results were independent of mitral regurgitation severity. Women with prior premature/early menopause also presented worse measures of left ventricular mechanical dispersion, and those who did not use hormonal replacement therapy (HRT) presented an increased incidence of adverse outcomes after 6 years of follow-up (mitral valve intervention, hospitalization, or cardiac death).
Beyond their novelty, the strengths of these findings lie in the comprehensive assessment of age at menopause and prior use of HRT, both of which remain frequently overlooked in clinical trials and cohort studies. 2 As well, the detailed cardiac function assessment provides hypotheses as to the underlying mechanisms potentially involved, while the cumulative events further support the clinical value of these results. On the other hand, the sample size was small, with only 21 women in the premature/early menopause group and only half of them having used HRT, thereby limiting the statistical power.
It is interesting to note that the risk of arrhythmogenic MVP was similar between the premenopausal and premature/early menopause groups, either contradicting the direct involvement of early estrogen withdrawal or implying different electrophysiological mechanisms of arrhythmia between groups. As such, perimenopausal symptoms and the use of hormonal contraception in the premenopausal group would be worth exploring. On the other hand, in the absence of systematic valvular heart disease screening programs, younger premenopausal women are more likely to be diagnosed with MVP when presenting with symptomatic ventricular arrhythmia, possibly prompting the arrhythmogenic MVP phenotype to be included in this study group. Nonetheless, these results are hypothesis-generating and may be supported by relevant theoretical explanations.
Indeed, menopause transition is a physiologically complex phenomenon leading to numerous changes in women's bodies. As highlighted by the authors, estrogens exert regulatory effects on endothelial function, inflammation, extracellular matrix turnover, and fibroblast activity.1,3,4 They also modulate neurohormonal and renin–angiotensin–aldosterone system pathways, body fat distribution, as well as glucose and lipid metabolism.3,5 Moreover, estrogens play a complex and ubiquitous role in the regulation of the cardiac rhythm. 6 Reduction in serum estrogens may directly contribute to adverse valvular, ventricular, and atrial remodelling, to myocardial ischemia, as well as arrhythmogenic susceptibility, while simultaneously impacting external factors also affecting cardiac health, such as epicardial fat accumulation, metabolism, and aortic stiffening.3,7‐9 Earlier menopause transition thus leads to earlier exposure to these adverse consequences of estrogen withdrawal, particularly in the absence of HRT use, translating into higher risk of major cardiovascular adverse outcomes such as myocardial infarction and stroke.10,11
Furthermore, upstream factors involved in the timing of menopause transition may influence women's overall cardiac health. For instance, higher prevalence of metabolic disorders, such as obesity, hypertension, diabetes, and dyslipidemia, has been linked to earlier menopause transition, thereby suggesting a bidirectional relationship between menopause and cardiovascular risk factors. 12 Likewise, poorer premenopausal metabolic health status is found in women who undergo premature surgical menopause. 13 Finally, common genetic variants linking both metabolic disorders and the timing of menopause transition could explain in part this bidirectional association. Whether shared causal mechanisms underlie the increased risk of arrhythmia and adverse outcomes associated with earlier menopause transition among women with MVP remains to be elucidated.
To conclude, the influence of sex hormones on cardiac health is a well-established fact that is no longer subject to debate. 10 Yet, the contribution of menopause and its timing to women's cardiovascular health, as well as the potential cardioprotective properties of HRT, remain widely undervalued and understudied (Figure 1). Beyond its results, the study by Tastet et al represents a major step toward closing persistent knowledge gaps in women's cardiovascular health.
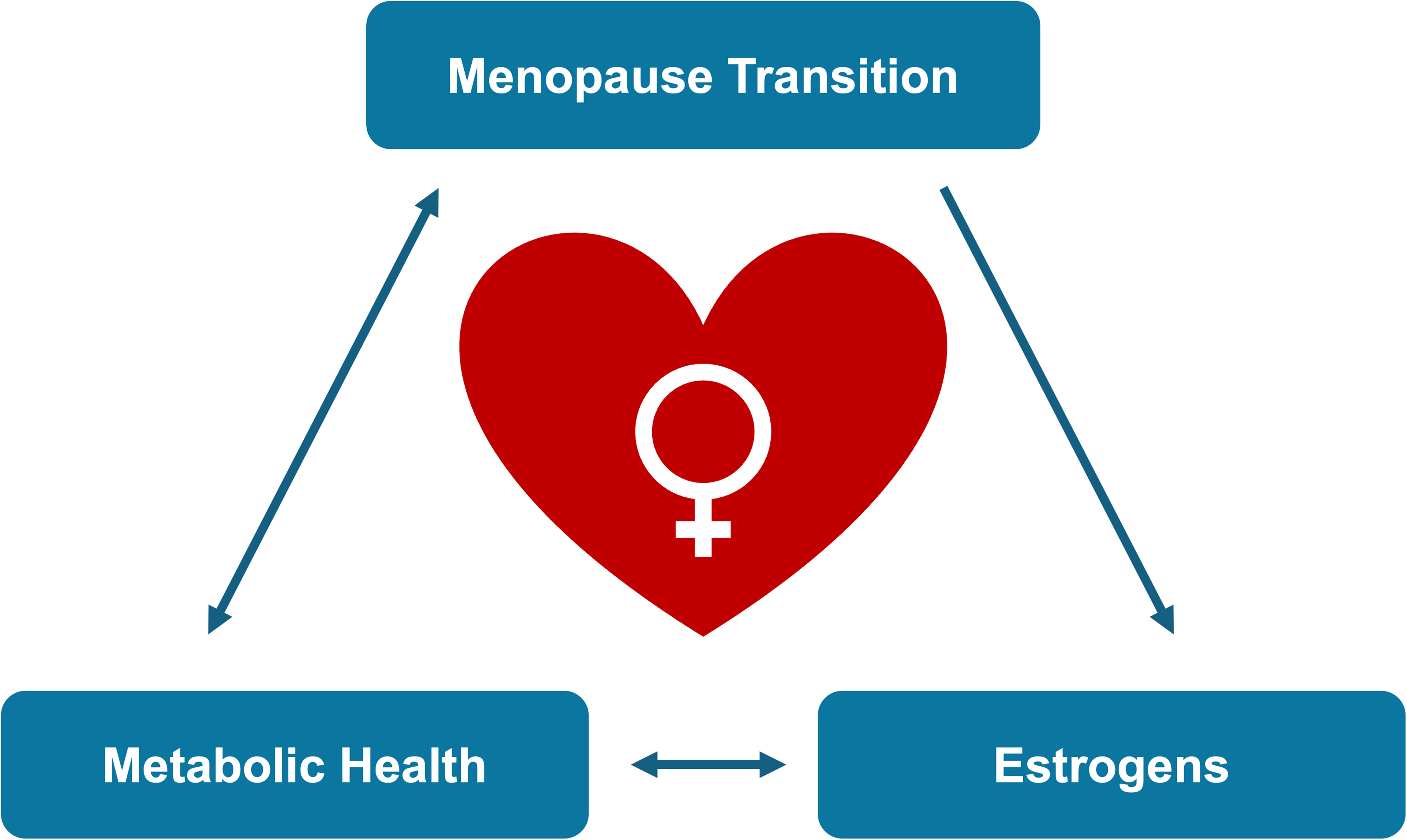
Complex bidirectional relationships linking menopause transition to women's cardiovascular health.
Footnotes
Acknowledgments
None.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contribution
AP is the sole author of this manuscript and its attached figure.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Not applicable.
