Abstract
Background
Women with mitral valve prolapse (MVP) may have a higher risk of ventricular arrhythmias (VAs) and sudden cardiac death (SCD), but the impact of menopausal age remains unclear. This study examined the association between menopausal age, arrhythmic risk, and cardiovascular outcomes in women with MVP.
Methods
We included 299 female patients with MVP (mean age 58 ± 17 years) and clinical, echocardiographic, and ECG data. Menopausal status was classified as premature (<40 years), early (40-45 years), expected age (45-55 years), undetermined age, or pre/perimenopausal. Arrhythmic MVP was defined by a composite of SCD or arrest, ventricular fibrillation (VF)/tachycardia, or frequent ventricular ectopy. The study composite endpoint included cardiovascular death, heart failure hospitalization, or mitral valve intervention.
Results
Among postmenopausal women (69.2%), 3.8% had premature, 6.2% early, 49.5% expected-age, and 18.4% undetermined-age menopause. Menopausal hormone therapy (MHT) use was reported in 16.4% of postmenopausal women. There was a higher prevalence of arrhythmic MVP among women with premature-early menopause (52%) compared to other groups (P = .01). After multivariable adjustment, premature-early menopause remained significantly associated with an increased arrhythmic risk (OR, 3.97; 95% confidence interval [CI], 1.33-11.8). During a mean follow-up of 4.7 ± 3.1 years, 53 composite events occurred. Premature-early menopause without MHT was associated with an increased risk of the composite of cardiovascular events (HR, 4.10; 95% CI, 1.41-11.9).
Conclusions
Premature or early menopause in women with MVP was associated with an increased risk of arrhythmic presentation and adverse cardiovascular outcomes during follow-up. Menopausal age may serve as an additional risk stratification factor in women with MVP.
This is a visual representation of the abstract.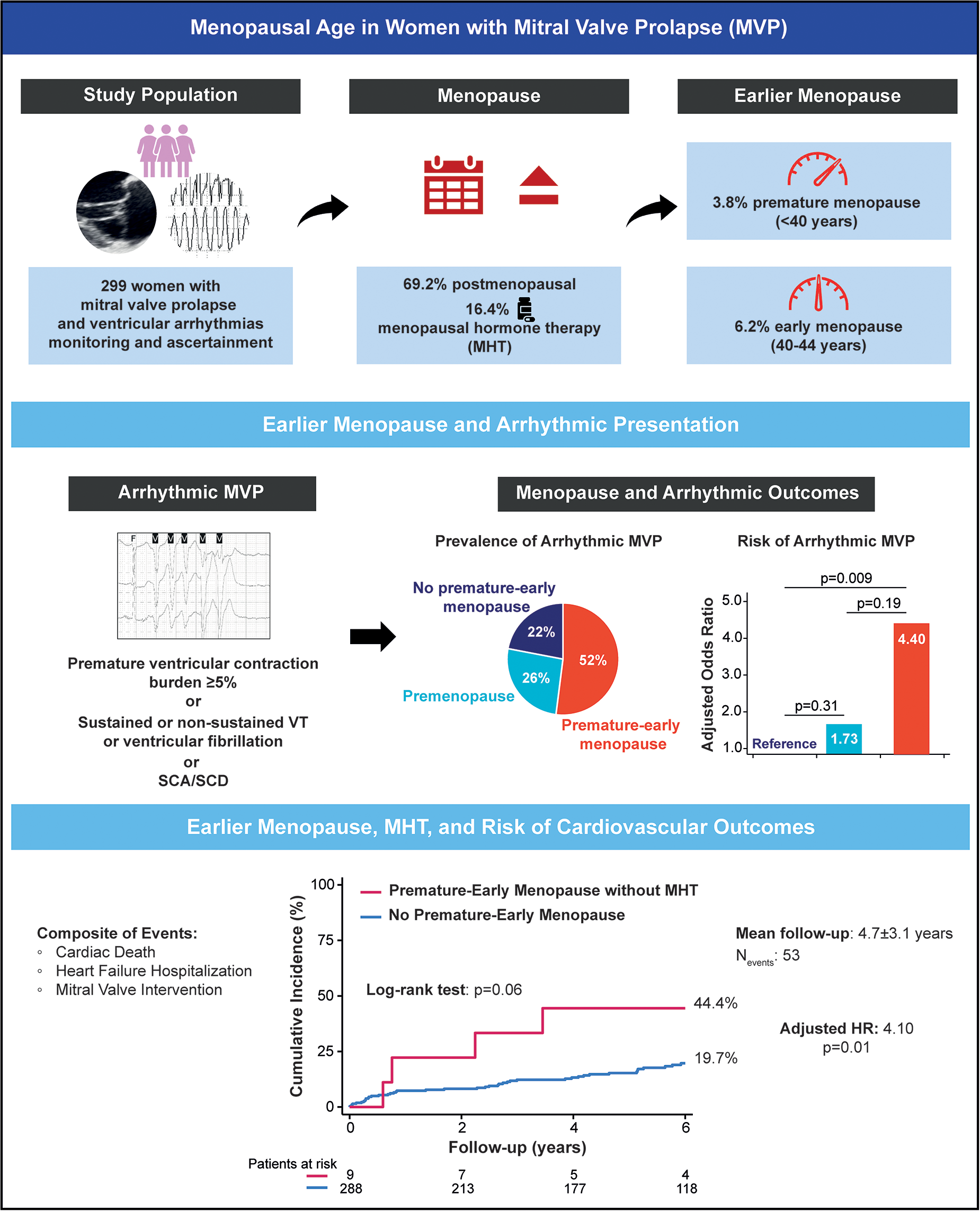
Key Points
Whether age at menopause contributes to arrhythmic susceptibility among women with mitral valve prolapse (MVP) remains unknown. Given that premature or early menopause has been associated to adverse cardiovascular outcomes in the general population, its potential role in arrhythmic MVP merits investigation. Moreover, the impact of menopausal hormone therapy on the relationship between menopausal age and long-term cardiovascular outcomes in women with MVP has not been established.
Among 299 women with MVP, premature-early menopause (<45 years). Premature-early menopause was associated to a 4-fold increased risk of arrhythmic MVP. Premature-early menopause without menopausal hormone therapy use was associated with a higher risk of cardiovascular events.
Menopausal age may serve as an additional risk stratification factor in women with MVP.
Introduction
Mitral valve prolapse (MVP) is a prevalent valvular heart disease affecting approximately 2000 per 100,000 individuals in the general population.1,2 It is a leading cause of significant mitral regurgitation (MR) requiring surgical intervention 3 and has been traditionally considered benign in the absence of severe MR.4–6 However, a small but clinically significant subset of individuals with MVP (0.14-1.8% yearly) is at risk of arrhythmic complications, including sudden cardiac death (SCD), cardiomyopathy and excess mortality.2,7–9 Recognized risk factors for arrhythmic MVP include young age, female sex, bileaflet involvement, mitral annular disjunction (MAD), frequent or complex ventricular ectopy, and myocardial fibrosis.2,10–12
Proposed mechanisms in arrhythmic MVP involve mechanical stretch-induced myocardial remodeling, where excessive leaflet motion—often exacerbated by MAD—triggers fibrosis serving as substrate for ventricular arrhythmias (VAs). 13 However, stretch-related abnormalities, in particular focal replacement fibrosis and MAD are not consistently identified among arrhythmic MVPs and SCD victims.14–16 Furthermore, these mechanisms fail to explain a female predominance in arrhythmic MVPs,10,11,17 suggesting that additional factors contribute to arrhythmogenesis, particularly in women.
The higher arrhythmic risk observed in women with MVP is likely multifactorial and may involve sex-specific differences beyond mechanical stretch. These include differential gene expression patterns, metabolic, and systemic differences. Hormonal factors, particularly those related to menopause, may also play a role. Menopause is characterized by declining ovarian hormones, particularly estrogen and progesterone, 18 and is associated with increased cardiovascular risk, including atrial fibrillation and heart failure.19,20 In contrast, premenopausal women are thought to benefit from cardioprotective effects of estrogen, and prior studies support cardiometabolic benefits from menopausal hormone therapy (MHT).21,22 Our recent findings demonstrate diffuse interstitial fibrosis in atypical myocardial segments in women with arrhythmic MVP, suggesting a global myopathy beyond focal, mechanically induced changes.23,24 While the female predominance in arrhythmic MVP cannot be attributed to hormonal factors alone, the timing of menopause represents a modifiable or identifiable risk factor that merits investigation.
In this study, we hypothesized that premature or early menopause is associated with higher risk of VAs in MVP. We evaluated whether earlier menopause is associated with subclinical myocardial dysfunction assessed by strain analysis, and whether it increases the incidence of adverse cardiovascular outcomes, including cardiac death, heart failure hospitalization, and mitral intervention. Finally, we explored whether MHT use modifies these risks.
Methods
Study Population
From 2013 to 2023, 712 consecutive patients with MVP who presented to the University of California San Francisco (UCSF) for clinical care were enrolled in the UCSF MVP registry. Pertaining to the present study, patients were excluded if they were male, had prior mitral valve (MV) repair or replacement, greater than mild aortic valve disease, or missed comprehensive clinical and echocardiographic data. Fifty additional patients were excluded because of limited echocardiographic data, missing menopause status documentation, or incomplete clinical data. After exclusions, 299 female patients with MVP were included in this analysis (Figure 1). The UCSF Institutional Review Board approved the study and waived informed consent.
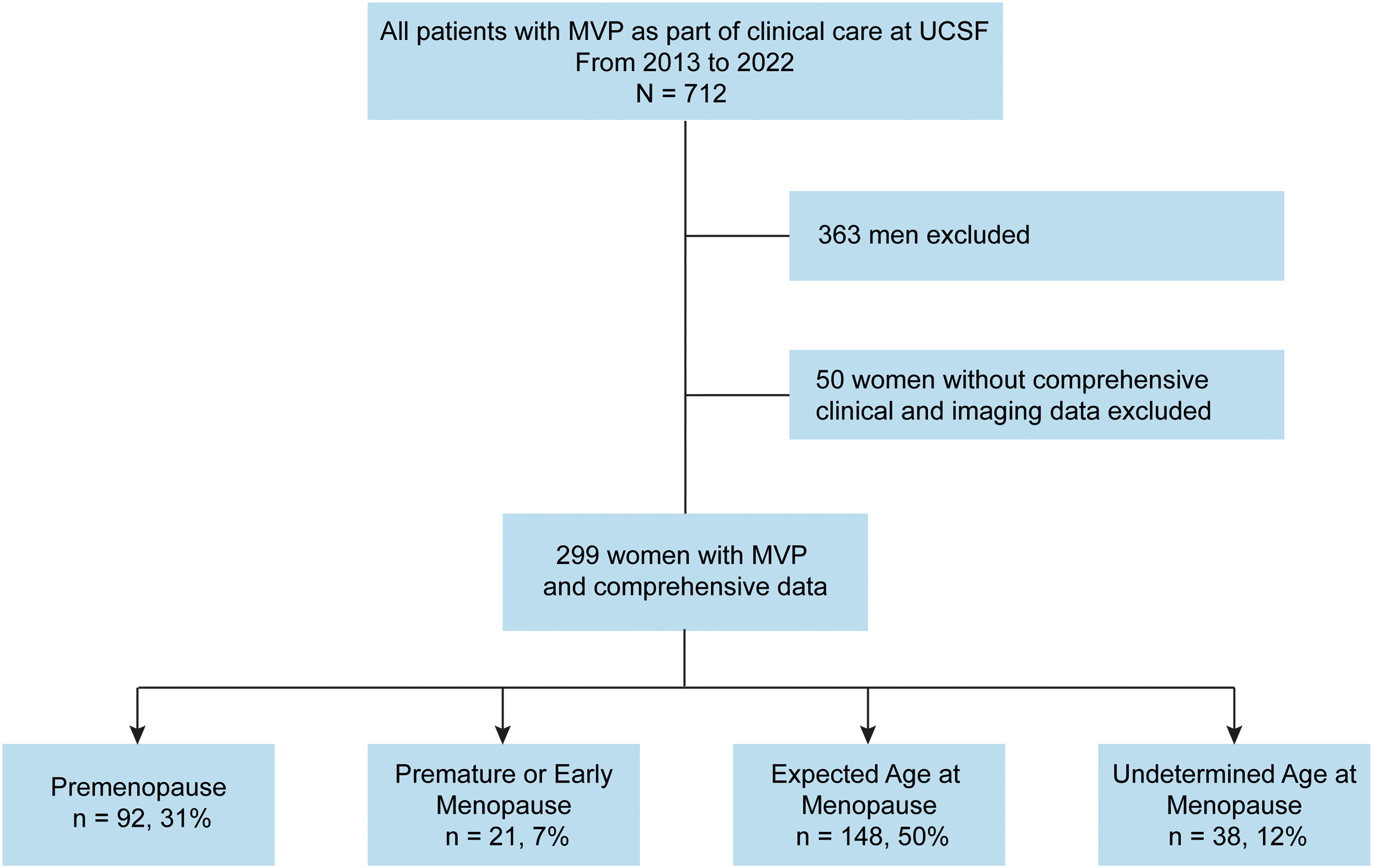
Study flow chart. Abbreviations: MVP, mitral valve prolapse; UCSF, University of California San Francisco.
Clinical Data and Menopause Assessment
Clinical data, including age, ethnicity, and comorbidities, were retrieved from electronic health records. Menopause onset was defined as 12 consecutive months without menstruation due to permanent ovarian function loss, either naturally or surgically induced. 18 Menopause timing was categorized as: (i) premature (<40 years), (ii) early (40-45 years), and (iii) expected age (≥45 years). 18 Women who had not yet reached menopause were categorized as premenopausal, while postmenopausal women without documented age at onset were categorized as having undetermined menopause timing. MHT use was defined as any documented current or past use of estrogen-based therapy (with or without progesterone), as recorded from medical records. Formulations (estrogen-only or combined estrogen-progesterone), duration (short-term: <5 years vs long-term: ≥5 years), and route of administration (systemic [oral or transdermal] or non-systemic/local) were also recorded.
Ascertainment of Arrhythmic MVP
All patients underwent 12-lead ECG or ambulatory monitoring (48-hour Holter or 2-week event monitor) to ascertain VAs. Arrhythmic MVP was defined according to the European Heart Rhythm Association consensus statement 9 and included: (i) frequent premature ventricular contractions (PVC) ≥5% (isolated and/or in couplets/triplets), (ii) complex ventricular ectopy including non-sustained (≥3 consecutive ventricular beats at a rate >100 bpm lasting <30 s) or sustained (≥30 s or requiring intervention) ventricular tachycardia (VT), (iii) ventricular fibrillation (VF), or (iv) sudden cardiac arrest (SCA)/SCD.16,23 Patients who met multiple criteria were classified according to the most severe VA observed.
Echocardiographic Data
MVP was defined as superior displacement of one or both mitral leaflets ≥2 mm beyond the mitral annulus in a parasternal long-axis view. MVP subtypes were categorized as bileaflet, single leaflet prolapse (anterior vs posterior), or flail. MAD was identified as a distinct separation between the MV/left atrial (LA) wall junction, and the basal inferolateral segment at the posterior end of the LV myocardium.25–27 MR severity was determined using a multiparametric approach, as recommended by guidelines, 28 and graded as: none/trace (grade 1), mild (grade 2), moderate (grade 3), or severe (grade 4). LA and left ventricular (LV) end-diastolic/end-systolic volumes, and LV mass were indexed to body surface area, and LV ejection fraction (LVEF) was recorded.
Speckle-tracking echocardiography (STE) analysis was performed offline using commercially available software (TomTec V.4.6) to measure LA reservoir strain and LV global longitudinal strain (LV-GLS). Endocardial tracing was performed in apical 4-, 3-, and 2-chamber views for LA and LV strains. 25 LV mechanical dispersion, reflecting contraction heterogeneity of myocardial segments, was defined as the standard deviation of the time-to-peak strain across 16 LV segments.29,30 All STE measurements were performed during sinus rhythm. Variability in STE analyses was previously reported at our institution. 31
Study Endpoint
The study endpoint was the composite of cardiovascular events, including cardiac death (non-arrhythmic or arrhythmic), heart failure hospitalization, or MV repair/replacement. Clinical events were collected by reviewing electronic health records and the National Death Index.
Statistical Analysis
Continuous variables were tested for normality of distribution and homogeneity of variances with the Shapiro–Wilk and Levene tests, respectively, and then expressed as mean ± SD or median (interquartile range [IQR]). Continuous variables were compared between groups using ANOVA or the Kruskal–Wallis test, followed by Tukey's or Dunn's post hoc test, as appropriate. Categorical variables were presented as frequencies and percentages and were compared with χ2 test or Fischer's exact test as appropriate. Univariable and multivariable logistic regression analyses were performed to determine the association between menopausal groups and risk of arrhythmic MVP. The multivariable model was adjusted for age, MHT use, hypertension, atrial fibrillation, bileaflet prolapse involvement, MAD, MR severity, LVEF, and LV mechanical dispersion. These variables were selected based on their clinical relevance or their significant association with the risk of arrhythmic MVP in univariable analysis. Results were presented as odds ratio (OR) with 95% CIs.
The estimates of cumulative incidence of the composite of cardiovascular events were calculated using the Kaplan-Meier method and compared using the log-rank test. Univariable and multivariable Cox proportional hazards analyses were used to determine the association of menopausal groups with the composite of cardiovascular events. The multivariable analyses were adjusted for age, new menopause onset during follow-up (included as time-dependent variable), and MR severity. The variable selection for multivariable analyses was based on their clinical relevance (ie, known risk factors) or significant association with the composite of outcomes in univariable analysis. Results were presented as hazards ratio (HR) with 95% CIs. A two-tailed P value <.05 was considered significant. Statistical analyses were performed with Stata software version 18.0 (StataCorp, College Station, TX).
Results
Study Population Characteristics
The study population consisted of 299 women with MVP. The mean age was 58 ± 17 years, and 207 (69.2%) were postmenopausal. Of these, 3.8% had a history of premature menopause, 6.2% early menopause, 49.5% menopause at expected age, and 18.4% had an undetermined age at menopause. MHT use, either past or current, was reported in 16.4% of postmenopausal women, with 46.4% of these actively using MHT at baseline. Among the 34 women who used MHT, 65% received estrogen-only therapy and 35% combined estrogen-progesterone therapy, predominantly through systemic routes of administration (>90%). Additional information about therapy duration and administration route is reported in Table 1.
Study Population Characteristics According to Menopausal Status.
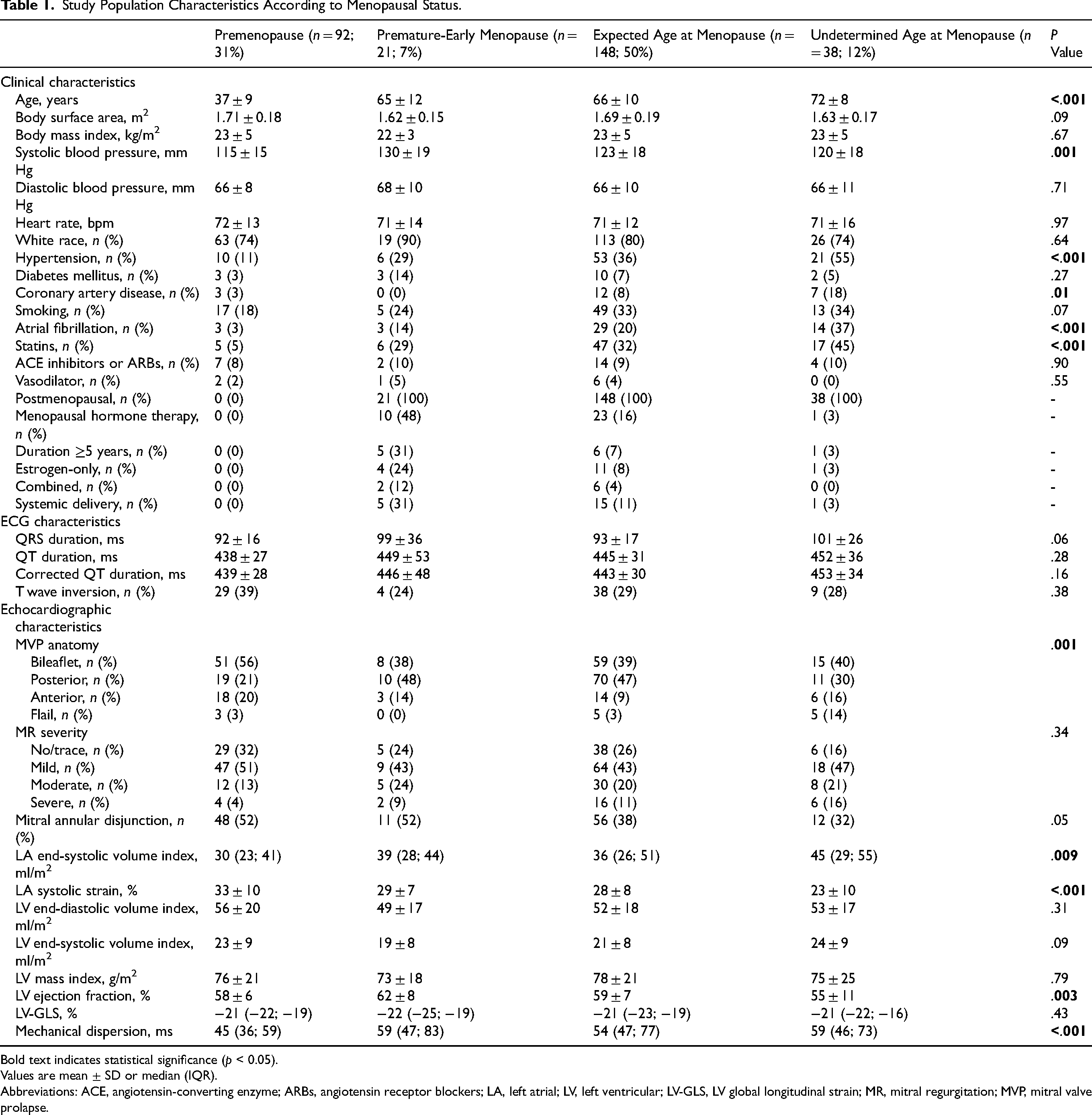
Bold text indicates statistical significance (p < 0.05).
Values are mean ± SD or median (IQR).
Abbreviations: ACE, angiotensin-converting enzyme; ARBs, angiotensin receptor blockers; LA, left atrial; LV, left ventricular; LV-GLS, LV global longitudinal strain; MR, mitral regurgitation; MVP, mitral valve prolapse.
Baseline clinical, ECG, and echocardiographic characteristics according to menopausal status are presented in Table 1. Women with undetermined age at menopause were significantly older, and more likely to have hypertension, coronary artery disease, and prior atrial fibrillation (all, P < .05). However, women with premature-early menopause had significantly higher systolic blood pressure compared to other groups (P = .001) (Table 1). There were no significant differences in ECG intervals (Table 1). Medication use, including ACE inhibitors or ARBs and vasodilators, was similar between groups, with higher statin use in older postmenopausal women, as expected (Table 1).
By echocardiography, bileaflet prolapse was more common in premenopausal women, whereas flail leaflet was more frequent in the undetermined age group (Table 1). MR severity was similar across menopausal groups (P = .34). Similarly, women with undetermined-age menopause had larger LA size and lower LA reservoir strain (all, P < .01). Compared to premenopausal women, those with premature-early menopause demonstrated increased LV mechanical dispersion and lower LA strain, despite similar LVEF (Table 1).
Menopausal Status and Arrhythmic MVP
Of the 299 women, cardiac monitoring modalities were as follows: 12-lead ECG only in 124 (41%) patients, 48-hour Holter monitoring only in 35 (12%) patients, 2-week event monitoring only in 5 (2%) patients, ECG with Holter in 26 (9%) patients, ECG with event monitor in 68 (23%) patients, Holter with event monitor in 4 (1%) patients, and all three modalities (ECG, Holter, and event monitor) in 37 (12%) patients. A total of 76 (25%) patients were diagnosed with arrhythmic MVP. The specific components of arrhythmic MVP diagnosis across menopausal groups are detailed in Supplementary Table S1. Figure 2 shows the distribution of arrhythmic MVP and sustained VAs by menopausal status and MR severity. Women with premature-early menopause had a significantly higher prevalence of arrhythmic MVP (52%) compared to those with premenopausal (26%), expected-age (23%), and undetermined-age menopause (16%) (P = .01). Similarly, sustained VAs were more prevalent in premature-early (4.8%) and premenopausal (6.5%) women, where there were no prevalent cases in the expected-age or undetermined-age menopause groups (P = .004).
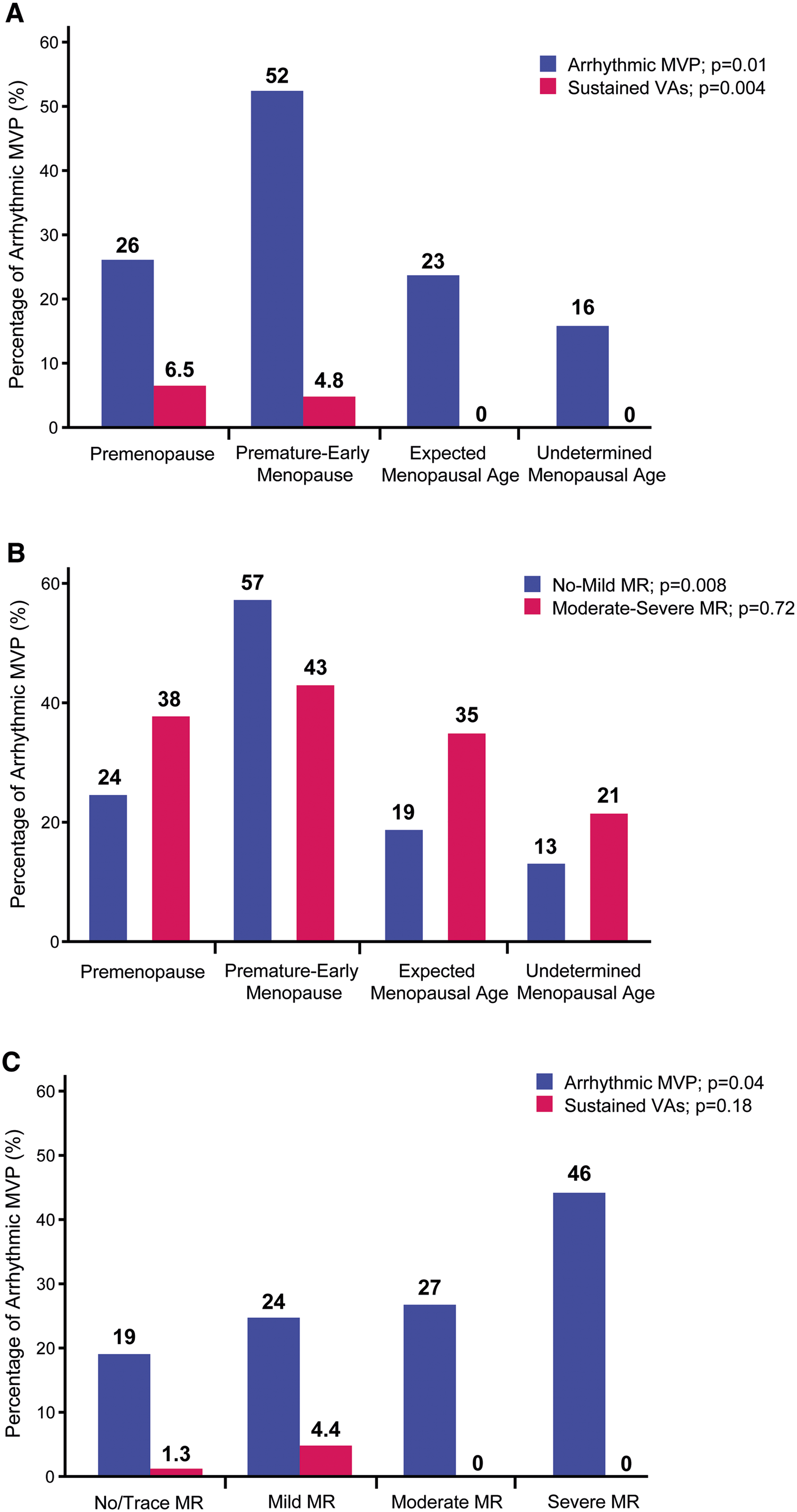
Distribution of arrhythmic MVP and sustained ventricular arrhythmias by menopausal status and mitral regurgitation severity. Proportion of arrhythmic MVP and sustained ventricular arrhythmias (sustained ventricular tachycardia or ventricular fibrillation) across menopausal groups (A). Proportion of arrhythmic MVP by MR severity across menopausal groups (B). Proportion of arrhythmic MVP and sustained ventricular arrhythmias according to MR severity (C). Abbreviations: MR, mitral regurgitation; VAs, ventricular arrhythmias; other abbreviations as in Figure 1.
When compared by MR severity, the prevalence of arrhythmic MVP remained significantly higher in premature-early menopause (57%) among those with no or mild MR (P = .008), whereas this difference was less evident among those with moderate-to-severe MR (P = .72) (Figure 2). Furthermore, the prevalence of arrhythmic MVP significantly increased with worsening MR severity (Figure 2). However, sustained VAs were exclusively observed in patients with mild MR (4.4%), followed by those with no/trace MR (1.3%), with no cases observed among those with moderate or severe MR (Figure 2).
Table 2 shows the univariable and multivariable logistic regression models to predict the risk of arrhythmic MVP. After multivariable adjustment for known risk factors and potential confounders, including MHT use, premature-early menopause remained significantly associated with an increased risk of arrhythmic MVP compared to all other menopausal groups (OR, 3.97; 95% confidence interval [CI], 1.33-11.8). This association remained significant when LV mechanical dispersion was included instead of LVEF (OR, 13.1; 95% CI, 2.61-65.8). Similarly, sensitivity analysis including ACE inhibitor or ARB uses instead of hypertension provided similar results for the association between premature-early menopause and arrhythmic MVP (OR, 3.99; 95% CI, 1.34-11.8). In addition, using expected-age and undetermined-age menopause as a reference group, premature-early menopause remained significantly associated with an increased risk of arrhythmic MVP (OR, 4.40; 95% CI, 1.45-13.3), while premenopausal status was not (OR, 1.73; 95% CI, 0.59-5.08) (Table 2).
Menopause Transition and Risk of Arrhythmic Mitral Valve Prolapse.

Results are odds ratio (OR) with 95% confidence interval (CI). Multivariable analyses adjusted for age, hypertension, menopausal hormone therapy use, atrial fibrillation, bileaflet MVP, mitral annular disjunction, MR severity, and LV ejection fraction.
Abbreviations as in Table 1.
Menopausal Status and Risk of Cardiovascular Outcomes
During a median follow-up of 5.0 years (IQR: 1.7 to 7.5 years) (mean 4.7 ± 3.1 years) after the index echocardiogram, the composite endpoint of cardiovascular events occurred in 53 (18%) patients. Two patients lacked information about clinical events or last known date alive and were thus excluded from the survival analysis. Of the composite events, there were 11 (21%) cardiac deaths, 36 (68%) MV interventions (32 MV repairs and 4 replacements), and 6 (11%) heart failure hospitalizations.
Figure 3 shows the cumulative incidence of the composite events according to menopausal status and MHT use. Among women with known menopausal age (ie, excluding women with undetermined-age menopause, those with premature-early menopause without MHT had a significantly higher 6-year incidence of the composite endpoint compared to premenopausal and expected-age menopause groups, regardless of MHT use (44.4% vs 18.8%; P = .04) (Figure 3A). Similar findings were observed when including women with undetermined-age menopause (44.4% vs 19.7%; P = .06) (Figure 3B). In addition, when stratifying premature-early menopause by MHT use, women without MHT use had a higher incidence of the composite events, although this did not reach statistical significance (Figure 3C and D). The main clinical characteristics comparing patients with premature-early menopause with versus without MHT use are reported in Supplementary Table S2. There were no significant differences between groups.
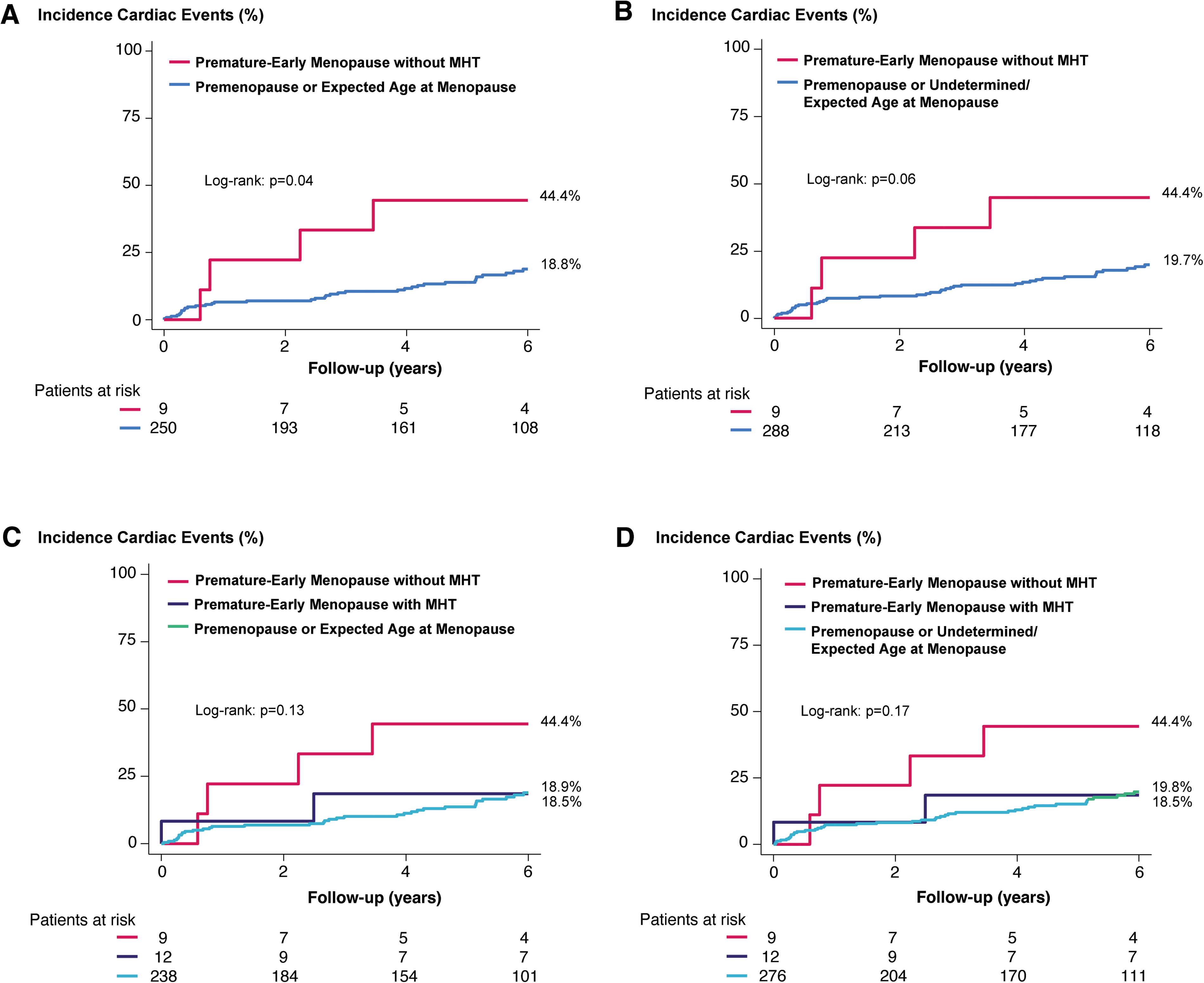
Risk of cardiovascular outcomes according to menopausal groups. Kaplan–Meier curves with the incidence of cardiovascular events (cardiac death, HF hospitalization, or MV intervention) stratified by menopausal groups. Incidence of events in women with premature-early menopause without MHT versus other menopausal groups (A) and after excluding the undetermined-age menopause group (B). Incidence of events in women with premature-early menopause, stratified by MHT use, compared to other menopausal groups (C) and after excluding the undetermined-age menopause group (D). Next to each curve are the cumulative incidence of events at 6-year. Bottom numbers refer to patients at risk at each interval time. Abbreviation: MHT, menopausal hormone therapy.
Table 3 shows the univariable and multivariable Cox regression models to predict the risk of composite events according to menopausal status. When stratifying by MHT use and after multivariable adjustment, premature-early menopause without MHT remained significantly associated with an increased risk of composite events compared to other menopausal groups (HR, 4.10; 95% CI, 1.41-11.9; P = .01) (Table 3). Similarly, when using undetermined-age or expected-age menopause as the reference group, premature-early menopause without MHT use remained significantly associated with an increased risk of events (HR, 4.01; 95% CI, 1.37-11.6; P = .01), whereas premature-early menopause with MHT was not (HR, 0.33; 95% CI, 0.04-2.39; P = .27) (Table 3).
Menopause Transition, Hormonal Therapy and Risk of Cardiovascular Outcomes During Follow-Up.
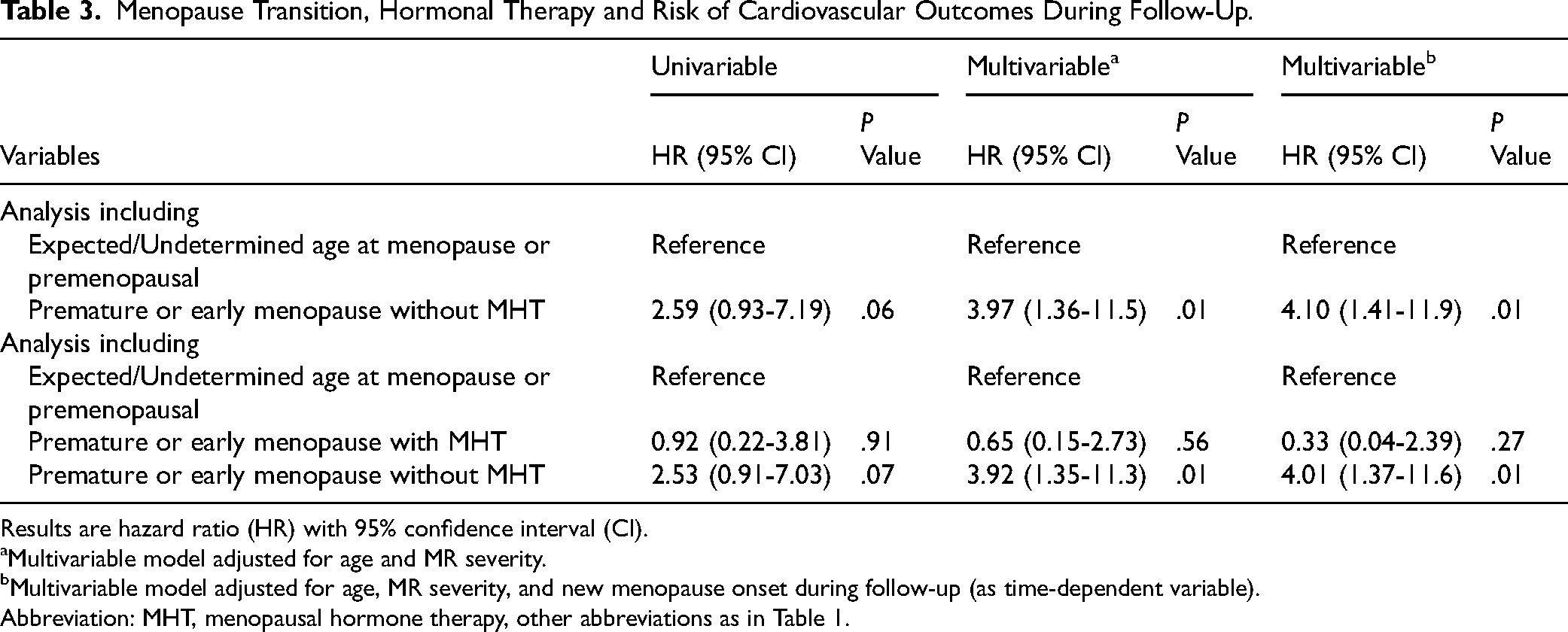
Results are hazard ratio (HR) with 95% confidence interval (CI).
Multivariable model adjusted for age and MR severity.
Multivariable model adjusted for age, MR severity, and new menopause onset during follow-up (as time-dependent variable).
Abbreviation: MHT, menopausal hormone therapy, other abbreviations as in Table 1.
Regardless of MHT use, univariable analysis showed that, when using premenopausal women as the reference group, both premature-early menopause (HR, 3.67; 95% CI, 1.56-8.67; P = .003) and undetermined-age or expected-age menopause (HR, 3.63; 95% CI, 1.11-11.9; P = .03) were significantly associated with an increased risk of composite events (Supplementary Table S3 and Supplementary Figure S1). However, after multivariable adjustment for risk factors and onset of menopause during follow-up (as a time-dependent variable), these associations were no longer significant (Supplementary Table S3).
Discussion
In this cohort study of women with MVP, we evaluated the association of menopausal age with arrhythmic presentation and long-term clinical outcomes. Among postmenopausal women, approximately 10% had a history of premature (<40 years) or early (<45 years) menopause, and notably, this subgroup demonstrated an increased risk of MVP-related VAs. Of importance, women with premature-early menopause who did not use MHT were at increased risk of cardiovascular events, including cardiac death, HF hospitalization, and MV intervention. To our knowledge, this study is the first to report significant association between menopausal age and both arrhythmic complications and major adverse events in MVP. These findings have significant clinical implications, emphasizing the need for improved risk stratification in women with MVP, particularly those at higher risk of arrhythmic events.
Menopausal Transition and Prevalence
In our study, 3.8% of women had a history of premature menopause (either naturally or surgically induced), and 6.2% had early menopause. These findings align with previous large studies reporting a similar prevalence of earlier menopausal transition.19,22 Among postmenopausal women, 16% used MHT, with approximately half of those with premature or early receiving MHT. The prevalence of MHT use in this subgroup varied when compared to previous studies, with some reporting higher rates, 22 while others observed similar or lower rates in more recent analyses.19,32 These differences may reflect variation in prescription patterns, patient characteristics, or treatment preferences.
Premature-Early Menopause and Ventricular Arrhythmias
Our findings identify two distinct high-risk phenotypes for VAs in women with MVP, each with different underlying mechanisms. First, premenopausal women demonstrated a high prevalence of sustained VAs, comparable to premature-early menopausal women, but with important mechanistic differences. Premenopausal women had a significantly higher prevalence of bileaflet MVP (56% vs 38% in premature-early group), suggesting that mechanical factors (excessive leaflet motion and valvular-ventricular stretch) could be driving severe arrhythmic events in this group. This aligns with prior studies identifying young age, bileaflet involvement, and mechanical stretch as key risk factors for malignant MVP.7,10,11
Second, premature-early menopausal women presented a different phenotype characterized by the highest overall prevalence of arrhythmic MVP (52%), despite lower rates of bileaflet MVP. This dissociation between arrhythmic burden and classic mechanical risk factors suggests that hormonal mechanisms, specifically estrogen deficiency, contribute to arrhythmic susceptibility in this group, representing a previously unrecognized high-risk phenotype. Further prospective studies are needed to determine whether this phenotype may reflect a predominance of frequent PVCs and non-sustained VT rather than sustained arrhythmias, potentially indicating diffuse myocardial substrate rather than focal mechanical triggers.
The precise mechanisms underlying the association between premature-early menopause and VAs in MVP are unclear. Earlier menopausal transition has been associated with an increased risk of atrial fibrillation in postmenopausal women, 19 likely driven by hormonal dysregulation. Early loss of endogenous estrogens may contribute to endothelial dysfunction, increased expression of inflammatory cytokines, vascular dysfunction, and maladaptive cardiac remodeling.18,33 The hyperadrenergic state in arrhythmic MVP may be exacerbated by hormonal withdrawal, 34 particularly in younger menopausal women.
Interestingly, we observed that the association between premature-early menopause and arrhythmic MVP was stronger in women with no or mild MR, whereas this difference was less evident in those with moderate or severe MR. In addition, sustained VAs were only observed in women with mild or no/trace MR. In patients with moderate-to-severe MR with arrhythmic MVP, the diagnosis was based on frequent PVCs rather than sustained arrhythmias. This suggests that mechanisms beyond MR severity, may contribute to arrhythmogenic susceptibility in this subgroup. Another potential mechanism involves myocardial remodeling and altered myocardial mechanics. Compared to premenopause or expected menopausal age, women with premature or early menopause had greater LV mechanical dispersion, a measure of heterogeneous contraction and electrical dispersion, and known predictor for VAs in MVP. 29 We previously demonstrated that increased LV mechanical dispersion correlates with the extent of myocardial fibrosis in sudden arrhythmic death victims regardless of MVP status. 30 Furthermore, women with arrhythmic MVP have a greater expansion of interstitial diffuse myocardial fibrosis, as quantified by extracellular volume fraction on cardiac magnetic resonance. 23 Whether premature or early menopause may accelerate myocardial fibrosis in MVP is unclear. Mechanistic studies are needed to understand how hormonal changes during menopause contribute to LV myocardial remodeling and arrhythmic vulnerability. Additionally, these findings provide a rationale for early screening of arrhythmias and targeted anti-arrhythmic therapies in this population.
Women with premature-early menopause had lower LA reservoir strain compared to premenopausal women, but values were similar to those in expected-age postmenopausal. Although indirect contribution may not be excluded, worst LA function was observed in women with undetermined-age menopause, likely reflecting age-related changes and higher comorbidity burden. We recently demonstrated that LA function can be significantly altered in MVP even without significant MR, suggesting the existence of a primary atriopathy. 25 However, our data do not support a specific additional contribution of premature-early menopause to LA dysfunction. The underlying mechanisms for LA impairment in MVP appear multifactorial, potentially involving abnormal valvular-ventricular mechanics, and other systemic changes associated with MVP. Whether earlier menopause may accelerate LA remodeling and dysfunction via different mechanisms remains unclear and merits further investigation.
Several other important confounding factors merit consideration in interpreting these results. First, LV and atrial mechanical abnormalities observed in postmenopausal women may be influenced by factors beyond MVP itself, including hypertension, arterial stiffening, metabolic syndrome, and diastolic dysfunction. In our cohort, women with premature-early menopause had significantly higher systolic blood pressure compared to other groups, which could have affected ventricular loading, afterload, and arrhythmic substrate. Second, the interplay between menopausal status, hypertension, and metabolic dysfunction is complex, as estrogen deficiency itself may promote hypertension and metabolic dysregulation. However, the association between premature-early menopause and arrhythmic risk remained significant after adjusting for hypertension, suggesting this association is not solely explained by blood pressure/hypertension status.
Menopausal Status, Hormonal Therapy, and Cardiovascular Outcomes
Our findings add to the existing literature on the adverse cardiometabolic consequences of premature and early menopause.35–37 Notably, we observed a 5-fold increased risk of the composite outcomes of cardiovascular death (including arrhythmic-related death), HF hospitalization, and the need for mitral intervention in women with premature or early menopause who did not receive MHT. This association remained significant after adjusting for known risk factors, including diabetes, coronary artery disease, atrial fibrillation, and MR severity. On the other side, women with premature or early menopause who used MHT had similar outcomes risk as compared to those in other menopausal age groups, suggesting a potential protective effect of MHT in this high-risk subgroup.
The cardiovascular benefits of MHT remain debated, with prior studies reporting neutral or even non-protective effects.20,38,39 In particular, the type of MHT (estrogen-alone vs progestin-containing therapy) may influence the risk of cardiovascular events. 39 Nonetheless, contemporary evidence supports the potential benefit in selected patients, especially women with premature or early menopause, when initiated within an optimal therapeutic window.21,22,40 While our study was not designed to evaluate the effect of different MHT formulations, future studies should examine whether specific therapies influence the arrhythmic and cardiovascular risk in MVP.
Beyond its impact of the LV myocardium, earlier menopause transition appears to have consequences at the valvular level. The strong association between premature or early menopause and cardiovascular outcomes in our study aligns with recent findings demonstrating a faster progression rate of aortic valve stenosis in women with early menopause who did not receive MHT. 41 This raises important questions about whether MHT could mitigate or modulate disease progression and need for surgery in women with pre-existing valvular heart disease, including MVP. While these findings need to be corroborated in additional studies of MVP patients, they also emphasize the importance of menopausal age as a determinant factor for adverse clinical outcomes in women MVP. Larger, prospective studies are required to validate these findings and explore the potential protective effects of MHT, as well as determine optimal management strategies in MVP.
Study Strengths and Limitations
Our study has several strengths and limitations. We excluded 50 women (14%) due to incomplete data, which could potentially introduce selection bias. However, the observed proportion of menopausal status in our analysis was comparable to prior populations. This emphasizes the need for prospective studies with standardized, complete data collection. In addition, the relatively small number of women with earlier menopause using MHT restricted further analyses on formulation, duration, and timing of MHT use. The sample size in the premature-early menopause group precluded comprehensive multivariable adjustment for all potential confounders, including hypertension, and other cardiovascular risk factors. Diastolic function parameters were not systematically assessed in this cohort. Potential confounding effect of diastolic dysfunction on the association between menopausal status and outcomes cannot be fully excluded. Information on beta-blocker and antiarrhythmic medication use, which could potentially modify arrhythmic risk, was not systematically collected and therefore could not be included in multivariable models. Future studies are needed to comprehensively evaluate medication effects on arrhythmic risk in women with MVP. Despite these limitations, this study is the first to identify a high-risk phenotype for VAs and adverse cardiovascular outcomes in women with MVP. Prospective longitudinal studies are needed to investigate the impact of menopausal hormonal changes on MVP progression and arrhythmic risk. Additionally, mechanistic studies are required to elucidate the interplay between hormonal status, MV apparatus and myocardial remodeling, and arrhythmogenic susceptibility. Given the complex relationship between menopausal status, optimal MHT use, and risk stratification, these results highlight the importance of a multidisciplinary approach between care practitioners and heart valve specialists to optimize risk assessment and management of MVP.
Conclusion
In this study, premature or early menopause was associated with an increased risk of arrhythmic presentations, including sustained VAs, among women with MVP. A history of premature or early menopause without menopausal hormonal therapy was associated with an increased risk of adverse cardiovascular events during follow-up. These hypothesis-generating findings suggest that menopausal age may represent an additional clinical consideration in the risk stratification of women with MVP. Further studies are needed to unveil the mechanisms linking VA and menopause in MVP.
Supplemental Material
sj-docx-1-hvs-10.1177_30494826261419380 - Supplemental material for Menopausal Age, Ventricular Arrhythmias, and Cardiovascular Outcomes in Mitral Valve Prolapse
Supplemental material, sj-docx-1-hvs-10.1177_30494826261419380 for Menopausal Age, Ventricular Arrhythmias, and Cardiovascular Outcomes in Mitral Valve Prolapse by Lionel Tastet, Luca Cristin, Rohit Jhawar, Amy H Rich, Dwight Bibby, Qizhi Fang, Anoop Muniyappa, Viktória Stanová, Marie-Annick Clavel and Francesca N Delling in Journal of the Heart Valve Society
Footnotes
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Health NHLBI R01HL153447. Dr Clavel holds the Canadian Research Chair on Women’s Cardiac Valvular Health from Canadian Institutes of Health Research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
