Abstract
Mitral annular calcification (MAC) is an inflammatory process that shares characteristics with atherosclerosis and results in calcium deposition in the fibrous ring supporting the mitral valve. It is commonly encountered at older ages and in people with chronic kidney disease. MAC is an important prognostic marker of adverse cardiovascular events and atrial fibrillation, as well as cardiovascular and non-cardiovascular death. When severe it can cause valvular dysfunction including mitral stenosis and/or regurgitation. Aside from effects on mitral valve function, MAC is associated with abnormal compliance of both the left atrium and left ventricle. It is also associated with disruption of normal left ventricular vortex formation and increased energy losses. In combination, these factors may help explain the symptoms of dyspnea and exercise intolerance frequently seen in MAC patients.
This is a visual representation of the abstract.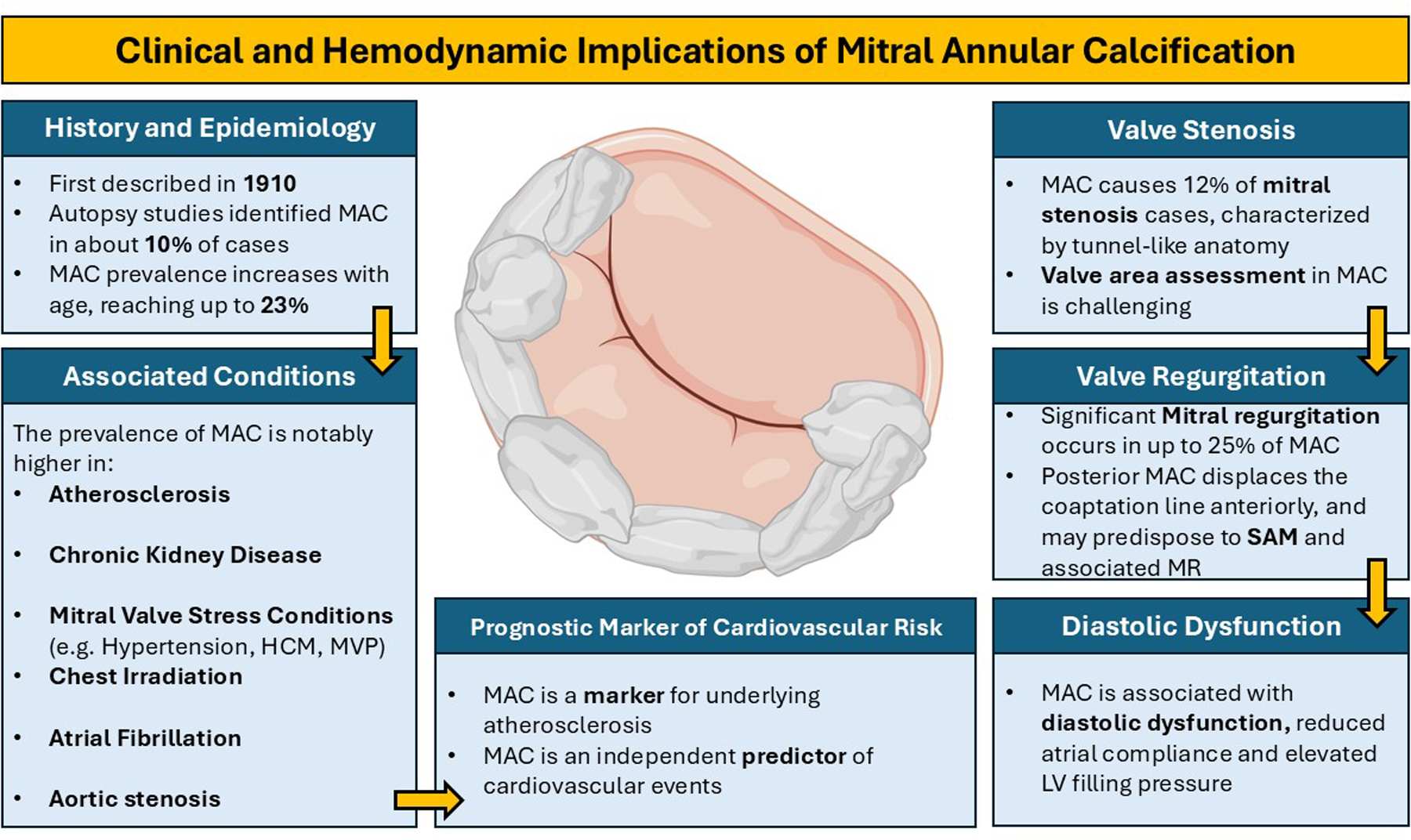
Key points
Mitral annular calcification (MAC) is a common, age-related degenerative process involving the fibrous mitral annulus, often detected incidentally.
MAC is associated with aortic stenosis, atherosclerosis, chronic kidney disease, atrial fibrillation, hypertension, and prior chest irradiation.
MAC independently predicts stroke, cardiovascular events, and all-cause mortality in population-based longitudinal studies.
By altering annular and leaflet mechanics, MAC can cause mitral stenosis, regurgitation, or mixed valvular dysfunction.
MAC is often associated with altered diastolic function due to reduced atrial and ventricular compliance.
Introduction
Calcification of the mitral annulus is commonly observed in clinical practice. It is typically diagnosed through echocardiography or computed tomography (CT) imaging of the heart but may also be detected via fluoroscopy or cardiac magnetic resonance imaging. Its presence holds prognostic significance, in addition to its effects on valvular structure and function. This review will explore the prevalence and clinical associations of mitral annular calcification (MAC), as well as its impact on cardiac physiology. Furthermore, the current limitations and challenges in quantifying these effects are discussed.
Prevalence and Associated Conditions
MAC is a chronic degenerative condition affecting the fibrous support structure of the mitral valve.1,2 MAC is often clinically silent and typically identified incidentally during echocardiography. It appears as an echo-dense, shelf-like structure giving the annulus an irregular, lumpy appearance associated with acoustic shadowing (Videos 1–3). The earliest description of this condition was tied to its clinical implications. In 1908, Bonninger reported a case of complete heart block associated with extensive calcification of the mitral annulus. 3 Subsequently, in 1910, Dewitzky was the first to formally recognize calcific disease of the mitral annulus as a distinct clinical entity. 4 He provided a detailed pathological description of 36 cases and compared this lesion to the aortic valve calcification described by Mönckeberg in 1904. 5 Dewitsky also noted a now well-established observation: the posterior portion of the annulus is more frequently and extensively involved than the anterior segment.
Autopsy studies in the mid-twentieth century identified MAC in approximately 10% of cases.6,7 More recent clinical investigations have reported a prevalence ranging from 8% to 42%, depending on patient age and diagnostic method employed.8–11 The largest study to date, which examined echocardiograms in nearly 25,000 patients, found a prevalence of 23%. 12 Data from the Framingham Heart Study as early as 1983 found more than 90% of individuals with MAC were older than 59 years, highlighting a clear age-dependent risk. 13 Kanjanauthai et al, using 6814 CT scans from participants in the Multi-Ethnic Study of Atherosclerosis (MESA), confirmed that age was independently associated with MAC. 14 Given this association, it is not surprising that MAC often coexists with aortic stenosis. 15 Conversely, up to half of the patients undergoing transcatheter aortic valve replacement (TAVR) also have MAC. 16
The prevalence of MAC is notably higher in certain associated conditions:
Atherosclerosis
There is a strong association between MAC and atherosclerosis which suggests shared pathophysiological mechanisms. 17 Numerous studies have observed associations between MAC, cardiovascular risk factors, and conditions such as aortic atheroma, carotid atherosclerosis, peripheral artery disease, and coronary artery disease (CAD).18–20 This will be expanded on below. Pathological evidence further supports this link, with foam cells (a hallmark of atherosclerosis) identified in early mitral annular lesions. 21 Because of these associations it has been suggested that MAC is a marker for underlying atherosclerotic disease22,23 (Video 4). Notably, these MAC-associated conditions are often encompassed within the clinical phenotype of metabolic syndrome, which is itself associated with an increased risk of MAC. 24
Chronic Kidney Disease and Abnormal Calcium–Phosphorus Metabolism
Valvular calcifications are 4 to 10 times more common in patients with chronic kidney disease (CKD) versus those with normal renal function. 25 In a community-based sample the Framingham Heart Study found that CKD patients had 60% increased odds of MAC after multivariable adjustment when compared with non-CKD patients. 26 While this relationship is partly explained by the increased prevalence of risk factors in CKD, mounting evidence suggests the involvement of abnormal calcium–phosphorus metabolism, electrolyte imbalance, chronic inflammation, and a distinct lipid profile. 27
Clinical Conditions with Increased Mitral Valve Stress and Tension
MAC is also associated with conditions that increase stress on the mitral valve, 28 such as hypertension, aortic stenosis, and hypertrophic cardiomyopathy (HCM).29,30 These conditions lead to elevated left ventricular systolic pressure, higher mitral valve closing pressures, and increased annular stress which may promote MAC. 1 The strong association between left ventricular hypertrophy and prevalence, severity, and even incidence of MAC supports this hypothesis. 31 Additionally, disorders of mitral valve motion, such as mitral valve prolapse, also appear to promote annular calcification. Again, excess stress exerted on the annulus, this time through the valve and chordal apparatus, appears to be involved.32,33
Chest Irradiation
Radiation to the chest has been associated with MAC, particularly in cases involving the aorto-mitral curtain.34–36 Many of these patients have had “mantle” radiation in treatment of Hodgkin's lymphoma. In a study of 112 patients with MAC-related mitral stenosis (MS) undergoing invasive hemodynamic assessment, one-third had a history of thoracic radiation. 37
Atrial Fibrillation
There is a strong association between MAC and atrial fibrillation. The Framingham study, using M-mode echocardiography, observed a 1.6-fold increased incidence of atrial fibrillation in those with MAC over a 16-year follow-up period. 38 The MESA study confirmed this association in a multi-ethnic cohort where the hazard ratio for incident atrial fibrillation was 1.9 for the MAC group over 8.5 years of follow-up. 39 The same MESA investigators also observed that MAC progression was an independent predictor of incident atrial fibrillation. 40 Recently, MAC has been associated with recurrence of atrial fibrillation following catheter ablation. 41 The disruption of atrioventricular coupling and associated increased left ventricular filling pressure that occur with atrial fibrillation may, in turn, promote progression of annular calcification.39,42
Prognostic Value as a Marker of Cardiovascular Risk
Over the past decade academic interest in the detection and grading of MAC has focused on the planning and optimization of interventional or surgical mitral valve procedures, particularly as the use of these procedures has increased. Thus, younger cardiologists may not be aware that MAC was initially investigated and later validated as an important marker of cardiovascular (CV) disease and adverse outcomes. Here we discuss the association of MAC with CAD, stroke, cardiovascular mortality and all-cause mortality.
The landmark historical literature on MAC as a risk-stratification variable primarily involves the detection and grading of MAC using echocardiography. More recently the routine availability of CT imaging (ECG-triggered cardiac CT or standard chest CT) has produced significant longitudinal CT studies with sufficiently long follow-up to assess incidence of CV diseases and hard outcomes reliably.
Studies investigating MAC as a risk marker for CAD or adverse outcomes should ideally account for the fact that MAC shares many risk factors with atherosclerosis, particularly age. Therefore, a longitudinal design with rigorous multivariate analysis is essential to demonstrate an independent association between MAC and CAD, cardiovascular events, and mortality. Moreover, the most effective clinical setting to determine whether MAC is truly associated with cardiovascular disease and/or outcomes is through prospective cohort studies, rather than retrospective studies of patients with clinically driven echocardiograms, which may introduce selection bias.
Fortunately, at the beginning of the current century several large-scale longitudinal community-based studies were published, which included assessment of MAC in their imaging protocols. These studies also collected data on many other known and potential cardiovascular risk factors allowing investigation of possible independent associations of MAC with incident CV diseases, CV mortality, and all-cause mortality.
The Cardiovascular Health Study (CHS) and the Framingham Heart Study were the first such large cohort studies with long-term follow-up.43–45 They established a clear independent association of MAC with incident coronary heart disease (CHS study) as well as all-cause mortality and CV mortality (Framingham Heart Study).
Coronary events, defined as fatal coronary events, hospitalized myocardial infarction or coronary procedures, were also assessed in a substudy of the Atherosclerosis Risk in Communities (ARIC) study. 46 Here MAC was observed to be independently associated with this combined coronary endpoint. More recently, in 2010, the Study of Health in Pomerania (SHIP) confirmed that MAC was associated with all-cause and cardiovascular mortality. 47 OxVALVE (Oxford Valvular Heart Disease) is another contemporary echocardiography screening study, initially designed to detect the presence and severity of valvular disease in individuals over 65 years of age. This large study also confirmed the independent association between MAC and its severity with mortality. 48
Several multisite echocardiographic cardiovascular calcium scores have been proposed which typically include MAC and additional sites of potential calcification, such as the papillary muscles, ascending/thoracic aorta and aortic valve. These scores provide incremental value in ascertaining clinical CAD risk, and risk of CV events and mortality.49,50 They have been tested in primary prevention samples and in patients undergoing echocardiography for various clinical indications, including those undergoing stress echocardiography for suspected CAD. 51 Interestingly, there is also evidence that such global calcium scores are significantly associated with coronary calcium.52,53
Determining the presence of MAC using echocardiography is based on visual features and thus inexact. It is not always possible to distinguish calcium from sclerotic changes. In addition the severity and extent of MAC is semiquantitative and lacks precision; studies have used various definitions and classifications, and this also represents a drawback of echocardiographic assessment of MAC. These limitations don’t exist for CT determination of presence and severity of MAC. Multisite cardiac calcium scores can be utilized with SPECT-CT techniques 54 and ungated chest CT, as well as cardiac CT. In this way routine studies ordered for other indications can provide additional “free” information about cardiovascular risk.
A recent paper from the MESA study, a community-based study of individuals without CV disease at baseline who underwent CT, confirmed that MAC is a predictor of long-term stroke risk, even after accounting for the presence of atrial fibrillation/flutter. There is also a concept that MAC is a marker of biological age. In another recent paper using the MESA database MAC was independently associated with a higher incidence of all-cause, cardiovascular, and non-cardiovascular mortality. 55 MAC subjects also had a significantly higher risk of developing dementia.
It should be mentioned as well that MAC can serve as a nidus for endocarditis.56,57 In such cases it appears that S. aureus infections are usually present. On occasion, very large vegetations can form (Figure 1, Video 5). Annular complications, including perforation, can also occur and mortality rates are high.
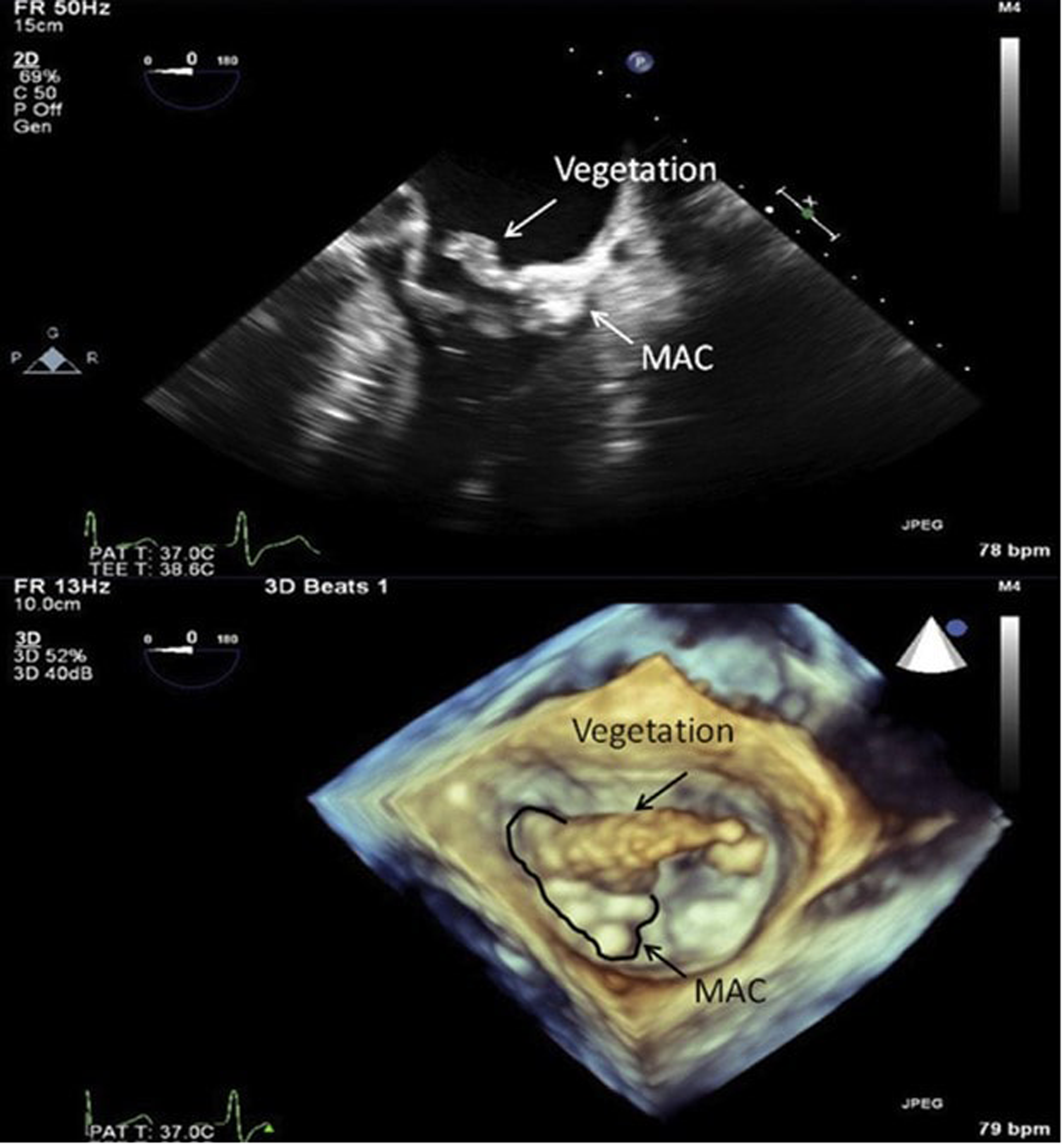
The top portion of the image is a 2D still frame from a TEE 4-chamber view. There is a large vegetation originating from the atrial surface of the calcified mitral annulus. The bottom portion of the image is a 3D still frame from the same study, looking onto the mitral valve from the left atrium. The aortic valve is at 12 o’clock and the atrial septum is at 5 o’clock. See also Video 5. Reproduced with permission from Ref. 56
MAC is a significant and robust prognostic marker regarding CAD, stroke, and mortality, both due to cardiovascular causes and non-cardiovascular causes. Information about the presence and severity of MAC, along with more global cardiac calcium scores, is readily available on clinically driven echocardiograms and CT scans. Awareness of the prognostic importance of these findings can aid the clinician in routine diagnosis and risk assessment of patients.
Anatomic Distribution and Effects on Valve Geometry
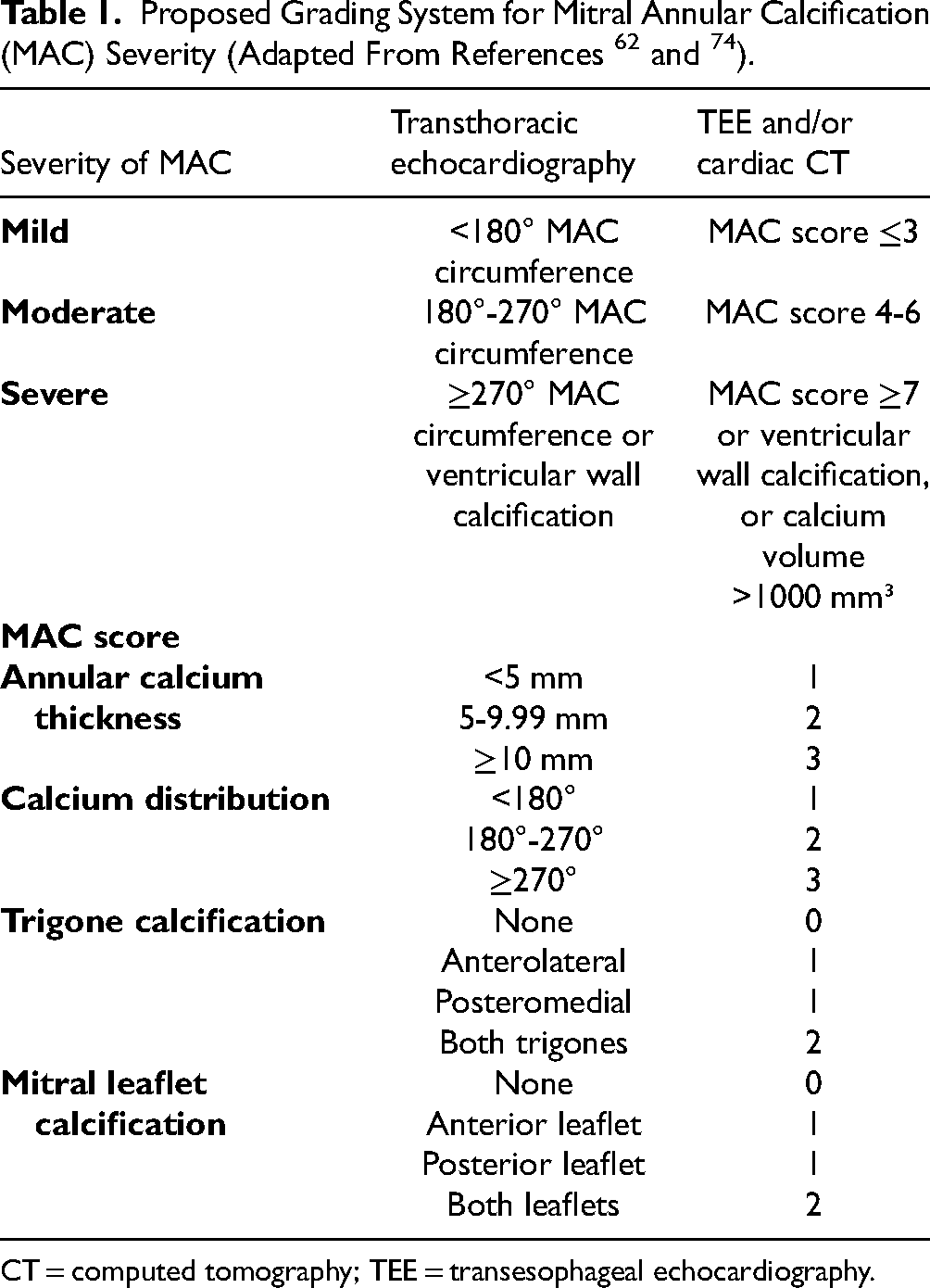
The anatomical distribution and severity of MAC can vary widely among individuals, as can its hemodynamic effects. Traditionally, semiquantitative echocardiographic scoring systems have been employed to assess severity of MAC. These mostly involve the extent of annular involvement, thickness of the calcification, and effects on valve leaflet motion.58–60 More recently, improved imaging techniques, particularly involving cardiac CT, have led to more detailed reporting of anatomical and hemodynamic characteristics of MAC. 61 With the rise of catheter based structural procedures it has become essential to have more precise assessment of MAC. The system devised by Guerrero et al 62 appears to be gaining generalized acceptance in this regard (Table 1).
CT = computed tomography; TEE = transesophageal echocardiography.
Variation by Sex
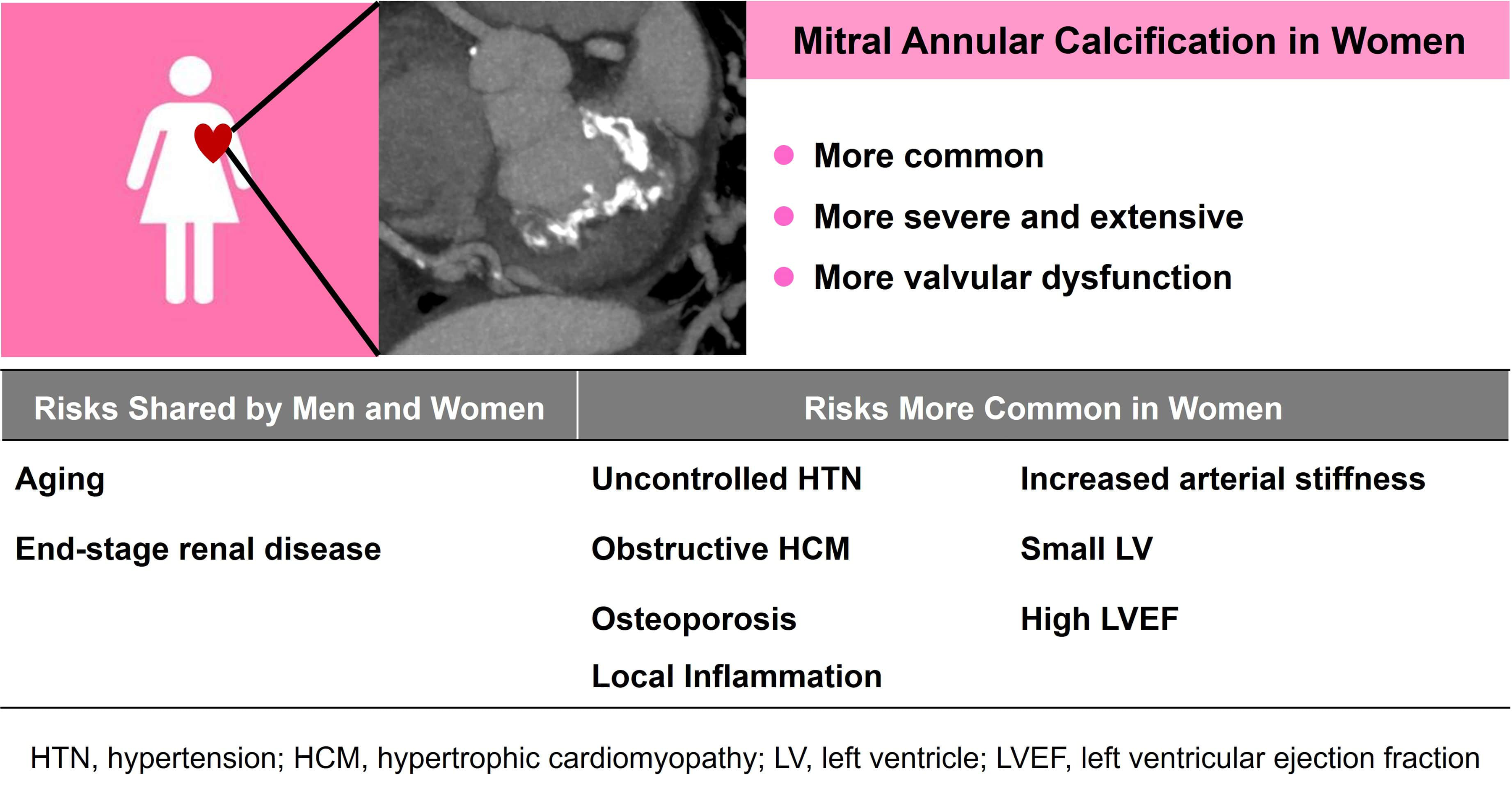
Most studies have observed a female predominance in patients with MAC.14,63,64 According to the MESA, the prevalence of MAC was higher in women than in men across all ethnic groups. 14 In addition, a previous pathologic study found that women tended to have more extensive calcium deposits as compared to men. 65 More recently, a positron emission tomography study, using 18F-sodium fluoride (as a marker of calcification activity) found that MAC activity was closely associated with female sex. 8 While MAC has been suggested to have a shared etiology with atherosclerosis, 21 these gender differences are not well explained by the traditional paradigm of atherosclerosis.14,20 Thus, other causal factors have been proposed including inflammation, mechanical stress and differences in calcium metabolism.8,66–69 (Figure 2).
Clinical and Anatomic Differences Between Men and Women with Mitral Annular Calcification.
It has been observed that MAC in elderly women is associated with severe bone loss due to post-menopausal osteoporosis. 66 Bone mineral density has also been associated with increased incidence and progression of MAC in both men and women, 70 though osteoporosis is more common in women. Additionally, the higher prevalence and more extensive nature of MAC in women could be associated with hemodynamic mechanisms. A recent study involving 537 MAC patients found that women had a higher frequency of moderate-severe MAC than men and were more likely to have mitral regurgitation. 67 In this study, risk factors for moderate-severe MAC differed significantly between women and men. While end-stage renal disease was a risk in both men and women, conditions that increase left ventricular afterload, such as uncontrolled hypertension or obstructive HCM, were independently associated with moderate-severe MAC only in women. Thus, mechanical stress associated with increased afterload and a relatively small ventricle may contribute to MAC in women.
Women with MAC had more mitral regurgitation than men. Significant MS (≥5 mm Hg) occurred in approximately 10% of both sexes but was independently associated with death only in women. Another study involving 3523 patients with extensive MAC and a mitral valve gradient greater >3 mm Hg showed similar findings. 68 In this study, using a large echocardiographic database, women significantly outnumbered men, accounting for 67% of subjects. Compared to men, women were significantly older and had fewer cardiovascular comorbidities. Women had higher transmitral gradients, more frequent concentric hypertrophy, and more frequent mitral regurgitation. In a separate study of 287 patients with severe calcific mitral stenosis associated with MAC, two-thirds of the patients were female, with this female predominance becoming particularly pronounced after age 50. 69 Furthermore, women exhibited a worse prognosis compared to men. By contrast, in a longitudinal study of MAC patients, factors such as widened pulse pressure, high ejection fraction, and the structural and functional severity of MAC at baseline were independent predictors of progression while female sex was not. 71
Posterior Annulus Versus Interannular Fibrosa
The normal mitral annulus has a saddle shape and is typically divided into anterior and posterior segments. The anterior annulus extends between the left and right fibrous trigones and is anatomically connected to the aortic annulus.1,29 The posterior annulus makes up the rest of the annular perimeter. As mentioned above MAC more commonly affects the posterior annulus than the anterior annulus.65,72,73 As the disease progresses, it can also involve the anterior annulus, or interannular fibrosa, the fibrous tissue between the anterior mitral leaflet and aortic valve. 74 Anterior annular involvement appears to be more common in cases of radiation-induced heart disease 34 but can also be present with other etiologies. When MAC is limited to the posterior annulus and non-severe, it usually does not cause functional abnormalities. More extensive MAC, particularly when it involves the anterior annulus, is often associated with functional abnormalities (see below). In the era of transcatheter mitral valve replacement, CT-scoring of MAC has prognostic value and is important for procedure planning 62 (Table 1). A key element of the CT MAC score is trigone involvement. Notably, the severity of MAC can help predict risk of valve embolization after transcatheter mitral valve replacement.
MAC, particularly when extensive, can encroach on the conduction system and produce conduction disturbances.75–77 This is because calcific deposits can extend to the atrioventricular node and the bundle of His. Interestingly, a small study that subdivided MAC by location found that AV conduction disturbances were associated with moderate to severe MAC (≥3 mm) but only when it was localized postero-medially in the mitral annulus, which is close to the AV-node and bundle of His. 78 It has also been suggested that MAC increases the risk of high-grade heart block following TAVR. In a group of 761 patients MAC was reported to be a strong independent predictor of pacemaker implantation post-TAVR. 79 However, this finding was not confirmed in another study involving 875 subjects. 80
Effects on Leaflet Function
In isolated MAC (not involving the leaflets), valve motion is maintained, allowing unhindered ventricular filling. However, calcific degeneration often progresses onto the leaflets, leading to reduced mobility and geometric distortion. These anatomic changes commonly result in the development of mitral stenosis and/or regurgitation (discussed below). In a study that performed echocardiographic evaluations on 75 consecutive hemodialysis outpatients to assess the extent of MAC, three-dimensional (3D) analysis revealed an uneven distribution of annular calcium. The middle and lateral anterior segments were less frequently calcified than the antero-medial and posterior segments. 58
Effects on Annular Function
The annulus possesses a dynamic, non-planar configuration that plays an important role in leaflet coaptation, unloading mitral valve closing forces, and promoting left atrial and left ventricular filling and emptying. 29 Recent research involving 3D echocardiography81–83 has provided insights into normal annular function and the effects of various disease processes. There has also been a study on the shape and function of the annulus using 3D echocardiography in patients with MAC. 84 This study compared 43 patients with MAC to 36 age- and sex-matched normal controls. Compared to the normal annulus, the anteroposterior diameter was larger in diastole and showed a smaller decrease during systole. Furthermore, the MAC annulus was flatter in diastole versus normal and had a smaller increase in saddle height during systole. The annular flattening and loss of systolic folding along the intercommissural axis might predispose to development of mitral regurgitation. However, these factors are opposed by the reduced motion and stiffening of the leaflets seen in MAC (see below).
Time Course and Determinants of Progression
The progression of valvular disease and its associated factors has long been of interest but remains challenging to understand. This is largely because early disease is often asymptomatic and discovered incidentally, making it difficult to study. 85 Additionally, factors that drive the initial stages of the disease may differ from those contributing to its progression.
MAC is a dynamic condition with its progression influenced by localized calcification activity and inflammation. Once present the key factors determining progression include the baseline calcium load, ethnic background, and smoking habits. 86
As is well-known, the factors associated with the incidence of MAC largely overlap with those of atherosclerosis. 87 However, these factors do not appear to be significant promoters with the exception of smoking. Conversely, ethnicity may affect disease course, with slower progression observed in Black and Hispanic patients.8,55 Notably, while CKD is a significant risk factor for developing MAC, no association has been found between eGFR levels and MAC progression8,87 though dialysis patients were excluded in these studies.
In a retrospective study using data from MESA, Elmariah et al observed that baseline severity of calcification was the primary determinant of MAC progression. 87 Individuals who started with more severe disease experienced more rapid progression. MAC progression was relatively independent of modifiable risk factors, further highlighting the importance of primary prevention. The one apparent exception was smoking cessation, which was associated with attenuated MAC progression and with MAC stabilization/regression. Similar observations have been made in other studies, with the fastest progression observed in patients with the largest baseline burden of MAC. 8 Cigarette smoking was again identified as a factor influencing progression of MAC.
With regards to inflammation, Massera et al utilized 18F-sodium fluoride and 18F-fluorodeoxyglucose (18F-FDG) PET to explore the relationship between calcification activity in the mitral annulus and local inflammatory signals. Increased 18F-sodium fluoride uptake (a marker of active calcification) in the mitral annulus was closely associated with 18F-FDG uptake (a marker of active inflammation) highlighting the role of inflammation not only in the development of MAC but also in sustaining the vicious cycle underlying progression of this condition. As to disease triggers the authors hypothesize that the presence of calcium within the mitral annulus increases mechanical stress and injury, thus leading to inflammation and enhanced calcification activity. 8
Lee et al investigated MAC progression by examining it from both structural and hemodynamic perspectives. 71 Structural progression was defined as an increase in MAC angle (extent of annular calcification expressed as degrees of arc) as measured via echocardiography. Hemodynamic progression was characterized by an increase in the mean Doppler transmitral pressure gradient. The authors found that MAC progression was associated with a wider pulse pressure (systolic – diastolic blood pressure), higher left ventricular ejection fraction, baseline severity of MAC, and its initial hemodynamic severity. Hemodynamic factors, including an increased pulse pressure and elevated ejection fraction, were identified as independent predictors of MAC progression (Figure 3). Pulse pressure serves as a marker of aortic stiffness, vascular aging, and associated comorbidities. An increase in pulse pressure leads to chronic elevation in left ventricular afterload, reduced longitudinal myocardial function, and compensatory enhancements in radial or circumferential contraction. The resultant increase in mechanical stress on the annulus might contribute to endothelial damage, localized oxidative stress, and inflammatory processes, which collectively drive MAC progression. 71
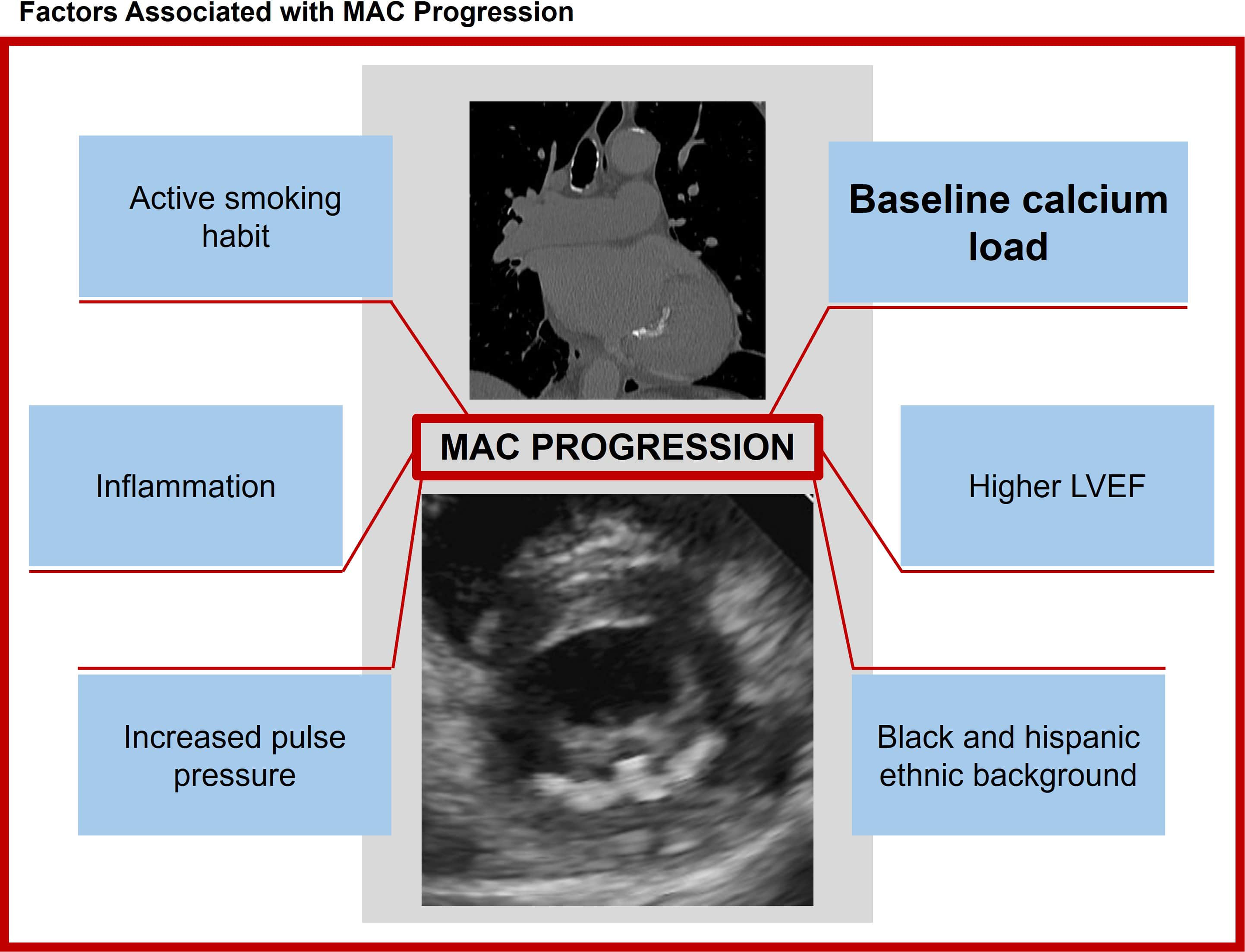
Factors Associated with Progression of Mitral Annular Calcification.
Hemodynamics—Due to MAC and Interaction with Associated Conditions
When calcific deposits extend onto the base of the mitral leaflet(s) a “bar” or ‘‘shelf’’ of calcium can form that limits leaflet motion and displaces their hinge point towards the left ventricular (LV) apex. When severe, particularly when the anterior leaflet is involved, this can produce valvular stenosis.58,88 As the population ages MAC has emerged as an important cause of mitral stenosis (MS), accounting for one in eight cases. 89
Stenosis with Comparisons to Rheumatic Disease
MS due to MAC, often referred to as degenerative mitral stenosis, has a different morphology when compared to rheumatic mitral stenosis. Calcification of the annulus and basal portions of the leaflets in degenerative MS leads to a “tunnel-like” stenosis with the greatest narrowing at the base of the valve (Figure 4, Video 6). This contrasts with rheumatic MS where commissural fusion (absent in degenerative MS) causes the valve to taper towards a narrowed orifice, yielding a characteristic “funnel type stenosis.” In addition, the MAC orifice is crescentic with open commissures while the rheumatic orifice typically has a “fish mouth” appearance bracketed by fused commissures (Figure 5). The tunnel-like stenosis of degenerative MS also produces greater flow convergence distal to the valve, resulting in a smaller contraction coefficient (effective orifice area [at the vena contracta]/anatomic orifice area). This in turn produces a higher transmitral pressure gradient for a given anatomic area and flow rate as compared to the funnel type stenosis of rheumatic MS (which has a higher contraction coefficient). 90
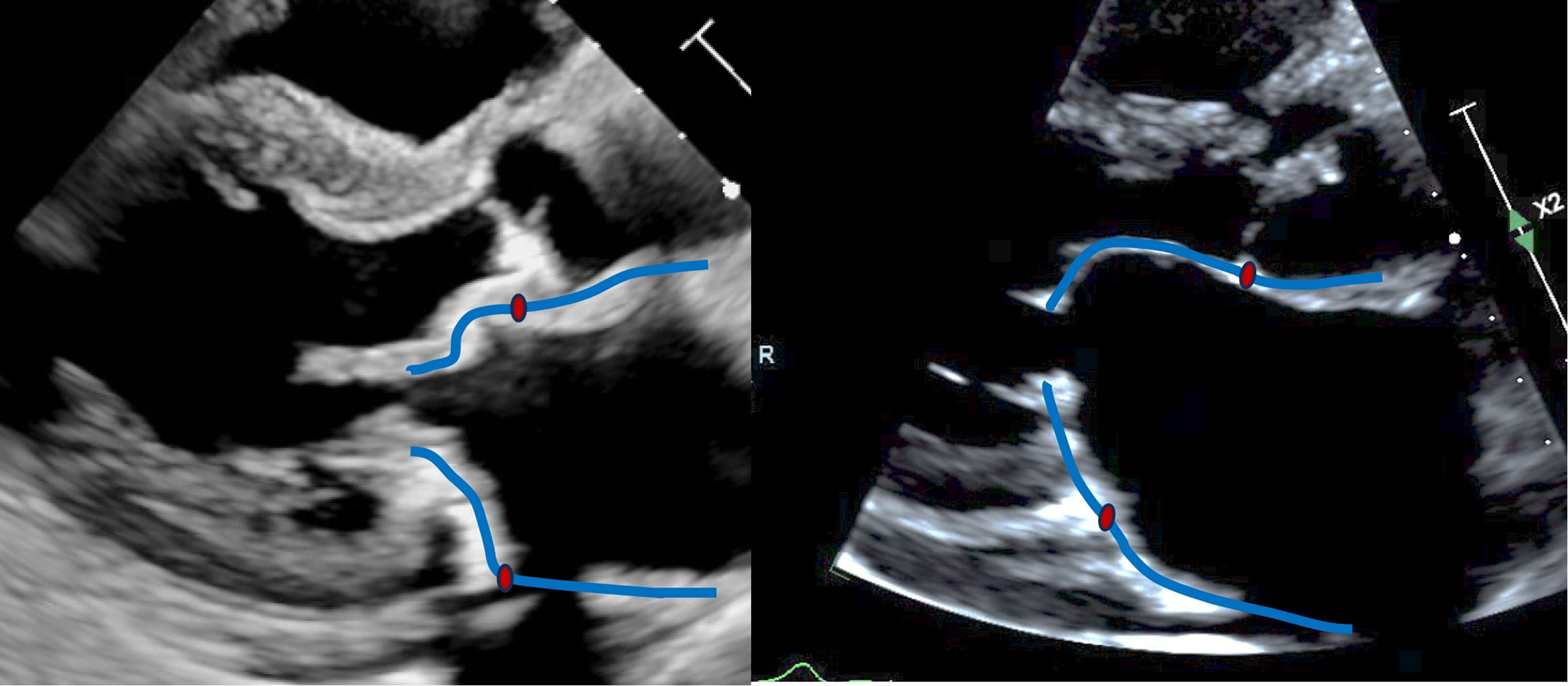
Parasternal Long-Axis Views of MAC Mitral Stenosis (left) and Rheumatic Mitral Stenosis (right). The blue lines mark the valve leaflets and left atrium, the red dots mark the mitral annulus. Note that the MAC valve has a “tunnel-like” configuration with a short distance from the annulus to the stenotic inlet of the calcium “tunnel.” By contrast the rheumatic leaflets slope more gradually to the stenotic orifice at the leaflet tips creating a “funnel” configuration.
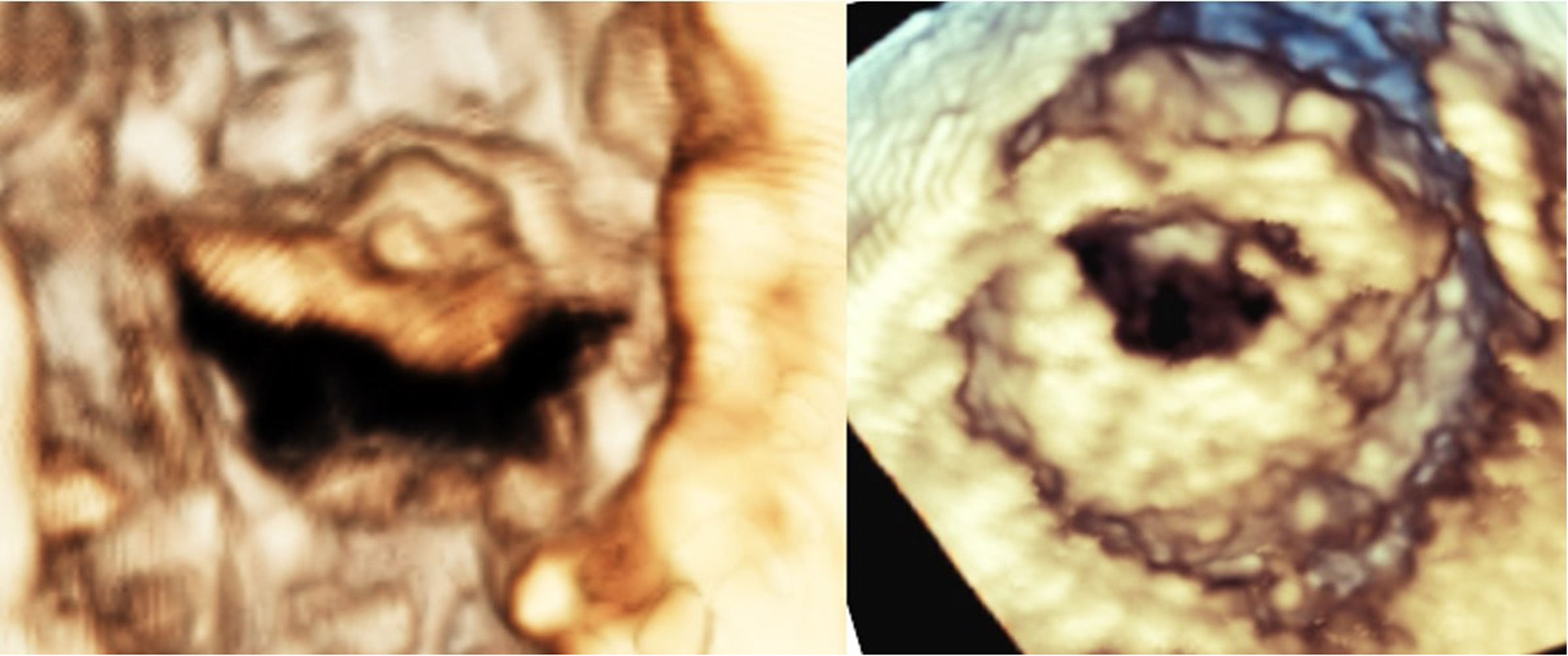
3D Diastolic Frames Obtained via Transesophageal Echocardiography. The panel on the left shows a MAC valve with prominent calcifications visualized on top of the anterior leaflet. Note the crescentic orifice and open commissures. The panel on the right shows a rheumatic valve with a “fish mouth” orifice bracketed by fused commissures.
In addition, mean gradients are dependent on the pressure difference between the left atrium and left ventricle. Degenerative MS patients are frequently elderly and have multiple comorbidities that can affect compliance of either chamber. Net atrioventricular compliance, along with valve area, determines the transvalvular gradient. 91 Poor left ventricular compliance (and high filling pressure) can result in a reduced transvalvular gradient, while poor left atrial compliance (and high atrial pressure) can increase the transvalvular gradient. Thus, although they have prognostic value, mean gradients may provide an inaccurate picture of the hemodynamic severity of degenerative MS. The cut-offs used to assess rheumatic MS severity have not been validated for degenerative MS and may not be relevant to this disease. Additionally, it should be borne in mind that transvalvular mitral gradients are greatly dependent on heart rate and flow rate across the valve.
Measurement of valve area in patients with severe MAC is challenging. However, with new transcatheter treatments becoming available it is essential to determine valve area accurately. There is no agreed upon method to measure mitral valve area in severe MAC but the pressure half-time technique should not be used.61,92 The abnormal chamber compliances noted above affect the slope of the Doppler signal making this method inaccurate. The continuity equation can be used in the absence of more than mild left-sided valvular regurgitation and is probably the most useful technique at this time. 93 A mitral valve dimensionless index 94 has also been proposed as a useful tool which eliminates the inaccuracies of left ventricular outflow tract (LVOT) measurement. 3D echocardiography, particularly via transesophageal echocardiography, can be used to directly measure orifice area via multiplanar reconstruction. However, acoustic shadowing from the annular calcium can make this difficult. In addition, the MAC orifice, as opposed to the rheumatic orifice, is non-planar. We have been working on use of virtual reality to directly measure the non-planar orifice in 3D space with encouraging preliminary data (unpublished). On occasion 3D color Doppler and multiplanar reconstruction can be used to measure the orifice area within the calcium tunnel. 95 This technique is also limited by acoustic shadowing from the annular calcium.
Regurgitation
Mitral regurgitation (MR) occurs through multiple mechanisms: (a) MAC can interfere with the sphincter-like function of the annulus, (b) it can directly restrict leaflet motion thus interfering with coaptation, or (c) it can push the posterior leaflet towards the atrium reducing the area of coaptation.73,96 Calcification of the chordae tendineae and disruption of normal annular dynamics can also contribute to MR. 84 The frequency with which significant regurgitation occurs in MAC has not been well studied. In one series of 138 MAC subjects, 22 (16%) were reported to have moderate or severe MR. 71 Another study looked at a group of 56 younger patients with MAC (<age 50) and found moderate or severe MR in 14 (25%). 97 By contrast in a study of 75 dialysis patients, 64% of whom had MAC, none had severe MR and severity of MAC was not predictive of presence or absence of MR. 58 Finally, in a large observational study of 24,414 subjects who underwent clinically indicated echocardiography, there was no significant association between presence of MAC and presence of MR. 12
Recently, a staging system has been proposed which assesses extra-mitral cardiac damage in MAC patients with mitral valve dysfunction. 98 This is similar in concept to the staging system used for patients with calcific aortic stenosis. In stage 0 there is no extra-mitral cardiac damage. Stage 1 is defined by the presence of left ventricular remodeling with sex-specific thresholds for LV end-systolic and end-diastolic volumes. Stage 2 occurs when there is left atrial remodeling defined as indexed left atrial volume >34 mL/m2 and/or the presence of atrial fibrillation. Development of pulmonary hypertension and/or ≥ moderate-severe tricuspid regurgitation denotes the presence of stage 3 disease with the addition of right ventricular dysfunction defining stage 4. Using this system graded relationships were found between higher stages and mortality as well as heart failure hospitalization.
Systolic Anterior Motion (SAM) of the Mitral Valve
SAM of the mitral valve is a characteristic finding in patients with dynamic outflow tract obstruction. While typically found in patients with HCM it is not universally present in this entity, and it can occur in other clinical scenarios. 99 SAM involves movement of one or both mitral leaflets into the LVOT during systole. If septal contact occurs, particularly if it is prolonged, a pressure gradient develops in the LVOT. In severe cases this gradient can be quite high and can be associated with syncope. Mechanistically, the valve leaflets are “pushed” into the LVOT, although there may be some contribution to SAM by the Venturi effect as the pressure gradient increases. 100
Various anatomic features of the left ventricle, mitral valve, and LVOT predispose patients to the development of SAM. These include protrusion of the septum into the LVOT, elongated mitral leaflets, abnormally positioned papillary muscles, and anterior displacement of the mitral coaptation line towards the interventricular septum. While these features predispose to SAM the resultant outflow tract gradients are dynamic and vary according to hemodynamic conditions such as left ventricular preload, afterload, and inotropic state. A recent paper described a series of patients with severe posterior MAC that appeared to contribute to development of SAM. In these patients the bulky calcium deposits formed in such a way that the mitral coaptation line was displaced anteriorly, effectively narrowing the LVOT and allowing SAM to emerge 101 (Video 7). Many of these patients had other anatomic features often associated with SAM including an elongated anterior mitral leaflet and a hyperdynamic LV.
Left Ventricular Diastolic Function
Patients with MAC are often elderly and have many comorbidities. The left ventricle is typically small and hypertrophied while the left atrium is often dilated and may be fibrotic as reflected by reduced atrial strain. 102 Patients with moderate or severe MAC often have reduced tissue Doppler velocities 103 and it is difficult to accurately assess diastolic function in these patients. 104 Whether the reduced values are due to the annular calcification or reflect true diastolic dysfunction is uncertain. A framework for assessing left ventricular diastolic dysfunction in patients with substantial MAC has been proposed. 105 The authors note that MAC can affect mitral E velocity and annular e′ thus making the E/e′ ratio unreliable in this setting. Their algorithm focuses instead on the E:A ratio and isovolumic relaxation time (IVRT). In an initial cohort of 60 subjects plus a validation cohort of 21 subjects they observed a sensitivity of 85% and specificity of 95% for detecting high LV filling pressure (>12 mm Hg). However, a subsequent study from a different center 106 was unable to demonstrate comparable sensitivity, specificity, and accuracy. Further work needs to be done in this area.
Interpretation of Doppler Gradients in Severe MAC
Patients with severe MAC often have a resting gradient across the mitral valve, and this gradient has prognostic value.107,108 It should be recognized, however, that the Doppler velocity envelope in these patients may have a configuration that differs from that in rheumatic mitral stenosis (Figure 6). Left atrial compliance in patients with severe MAC is often reduced (ie, the atrium is “stiff”) and this can alter the Doppler signal across the valve. A high peak E-velocity occurs in early diastole due to high left atrial pressure at valve opening. Left atrial pressure then drops quickly as blood leaves the atrium, resulting in a short deceleration time. These effects can potentially increase the transvalvular gradient as determined by the simplified Bernoulli equation 109 but are unrelated to valve obstruction. In a study comparing MAC MS with rheumatic MS we observed that, for similar valve areas, patients with MAC had higher transvalvular gradients than did patients with rheumatic disease. 110 While that difference may have been related to higher stroke volume in the MAC patients, effects of differing chamber compliances may also have played a role. In addition, Doppler echocardiography, particularly using the simplified Bernoulli equation, has not been validated in MAC.
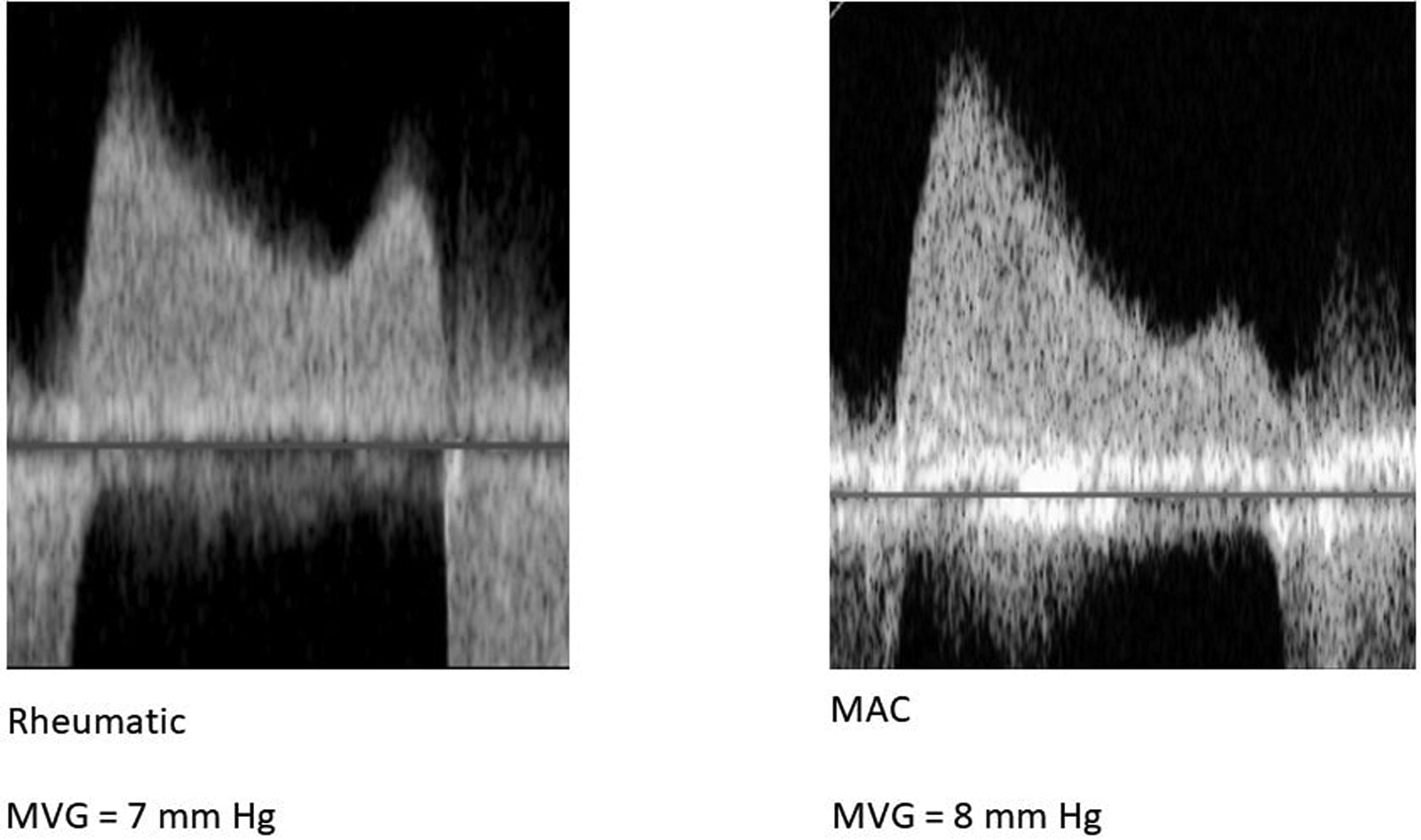
Representative Continuous-Wave Doppler Signals from a Subject with Rheumatic Stenosis (left) and Stenosis Due to Mitral Annular Calcification (MAC, right). Note that the MAC patient has an E-wave dominant profile and a shorter deceleration time as compared to the rheumatic patient.
Chamber Compliance, Vortex Formation, and Energy Losses
Calcification causes the annulus to become more rigid and less compliant which reduces the ability of the LA to expand and contract during the cardiac cycle. 96 Reduced annular compliance also impedes valve opening in diastole, contributing to elevated left atrial pressure.58,111 This elevated pressure increases the risk of pulmonary congestion and development of pulmonary hypertension over time. 112 This might also contribute to the observed increased risk of atrial fibrillation.39,42
Severe MAC can cause mitral stenosis and/or regurgitation, both of which affect left ventricular filling.61,113 Mitral stenosis impedes diastolic filling of the LV while regurgitation causes backward blood flow, potentially leading to increased LV filling. Both conditions can disrupt normal flow patterns, resulting in absence of a transmitral vortex and reduced forward stroke volume.114,115 If the LV becomes hypertrophied in response to increased workload, its compliance may be further reduced, exacerbating the hemodynamic impact.116,117
Reduced compliance of both the atrium and the ventricle leads to a decrease in overall cardiac efficiency. 118 The heart struggles to maintain an adequate stroke volume and may exhibit signs of heart failure, particularly in the presence of hypertension or CAD.
In diastole, when blood flows into the LV, vortex formation facilitates optimal filling, ensuring that the ventricle can expand and accept a proper volume of blood.119,120 Moreover, the diastolic vortex ring plays a crucial role in facilitating the exchange of flow at the apex, promoting efficient ventricular fluid washout during systole. In the absence of this diastolic vortex ring, apical fluid washout is significantly reduced, which increases the risk of thrombus formation.121,122
The diastolic vortex also plays a key role in transformation of kinetic energy and momentum transfer. It redirects flow forces while simultaneously facilitating the rotation and movement of blood as the cardiac cycle transitions from diastole to systole.123,124 During systole, as blood is being ejected from the LV, a well-coordinated vortex forms in the ventricle, aiding in efficient blood propulsion. This vortex helps create smooth laminar flow that minimizes energy loss.125–127
When MAC stiffens the mitral annulus, it can lead to incomplete or irregular mitral valve opening 96 which disrupts normal vortex formation.128,129 The resultant turbulent flow leads to inefficient heart pumping via increased friction and energy dissipation130,131 (Figure 7). Disruption of vortex flow due to MAC results in higher levels of energy loss, particularly during diastole when the ventricle should be passively filling. 132 In the absence of a well-formed vortex, there is a greater resistance to blood flow, which forces the heart to expend more energy to achieve the same level of filling and pumping.133–135 As a result of inefficient filling and increased energy demands, the LV may undergo hypertrophy to compensate for the reduced efficiency. 136 Over time, this can further reduce LV compliance, creating a cycle of increasing hemodynamic strain.
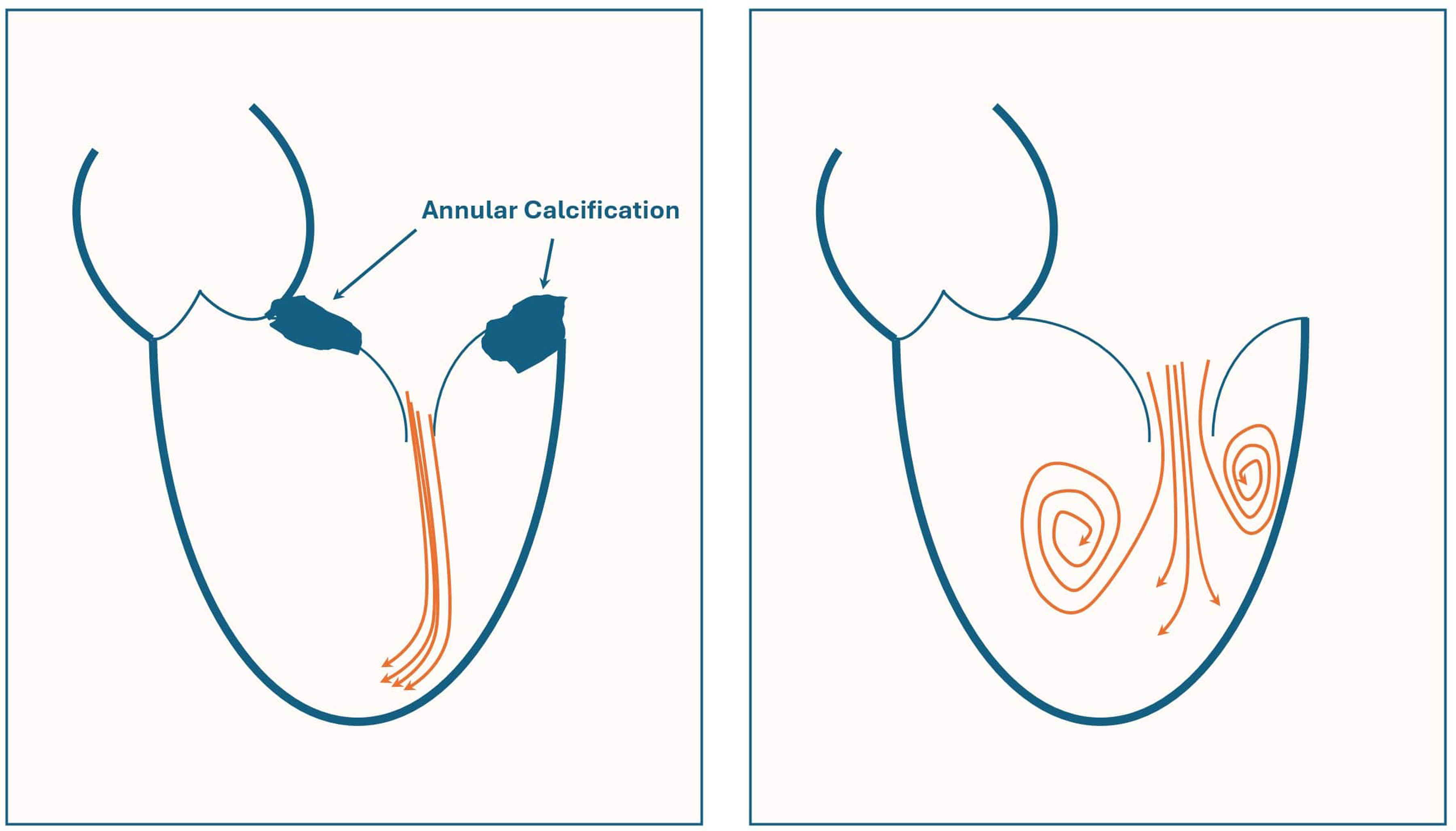
Sketch Illustrating Normal Vortex Formation in Diastole as Blood Crosses the Mitral Valve and Enters the Left Ventricle (right). In the setting of severe mitral annular calcification this vortex formation is absent (left).
The combination of disrupted vortex flow, turbulent filling, and increased energy loss likely contribute to limitations in cardiac output and symptoms of dyspnea and exercise intolerance. These changes in myocardial energetics may also help explain associations of MAC with increased risk of arrhythmias, heart failure, and other cardiovascular complications.137,138
Conclusion
In summary, MAC is a common finding in clinical practice, particularly in the elderly and those with CKD. It serves as an important marker of increased risk for adverse cardiovascular outcomes and mortality. Its presence should thus prompt consideration of aggressive risk factor modification. When annular calcification is extensive, particularly when it migrates onto the valve leaflets, it produces valvular dysfunction. While mitral regurgitation is a common consequence it is typically mild. By contrast, MAC can produce significant, even severe stenosis. Associated abnormalities in compliance of the left atrium and left ventricle, combined with valvular dysfunction, can significantly impact hemodynamics and increase myocardial energy losses.
Supplemental Material
sj-docx-1-hvs-10.1177_30494826251347539 - Supplemental material for Clinical and hemodynamic implications of mitral annular calcification
Supplemental material, sj-docx-1-hvs-10.1177_30494826251347539 for Clinical and hemodynamic implications of mitral annular calcification by Andrea Faggiano, Bianca Rocca, Chi Young Shim, Samer Farhud, Geu-ru Hong, Nicola Gaibazzi, Pompilio Faggiano, Giovanni Benfari, Salima Qamruddin, Arash Kheradvar and Gregg S. Pressman in Journal of the Heart Valve Society
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.