Abstract
Mitral annulus calcification (MAC) is a chronic, degenerative process characterized by calcium deposition in the mitral annulus. It is commonly observed in elderly individuals and those with chronic kidney disease, hypertension, and metabolic disorders. MAC has been increasingly recognized as a marker of cardiovascular disease and is associated with adverse clinical outcomes, including mitral valve dysfunction, arrhythmias, and increased cardiovascular mortality. The pathophysiology of MAC involves endothelial dysfunction, chronic inflammation, and osteogenic differentiation of valvular interstitial cells, driven by risk factors such as aging, chronic kidney disease, diabetes mellitus, and hypertension. Imaging modalities, including echocardiography and computed tomography (CT) play a crucial role in the diagnosis, assessment, and risk stratification of MAC. The presence of MAC complicates mitral valve interventions, making surgical and transcatheter approaches challenging. Despite advancements in imaging and therapeutic strategies, MAC remains a significant challenge in cardiovascular medicine, necessitating further research into its pathophysiology, optimal management strategies, and long-term outcomes.
This is a visual representation of the abstract.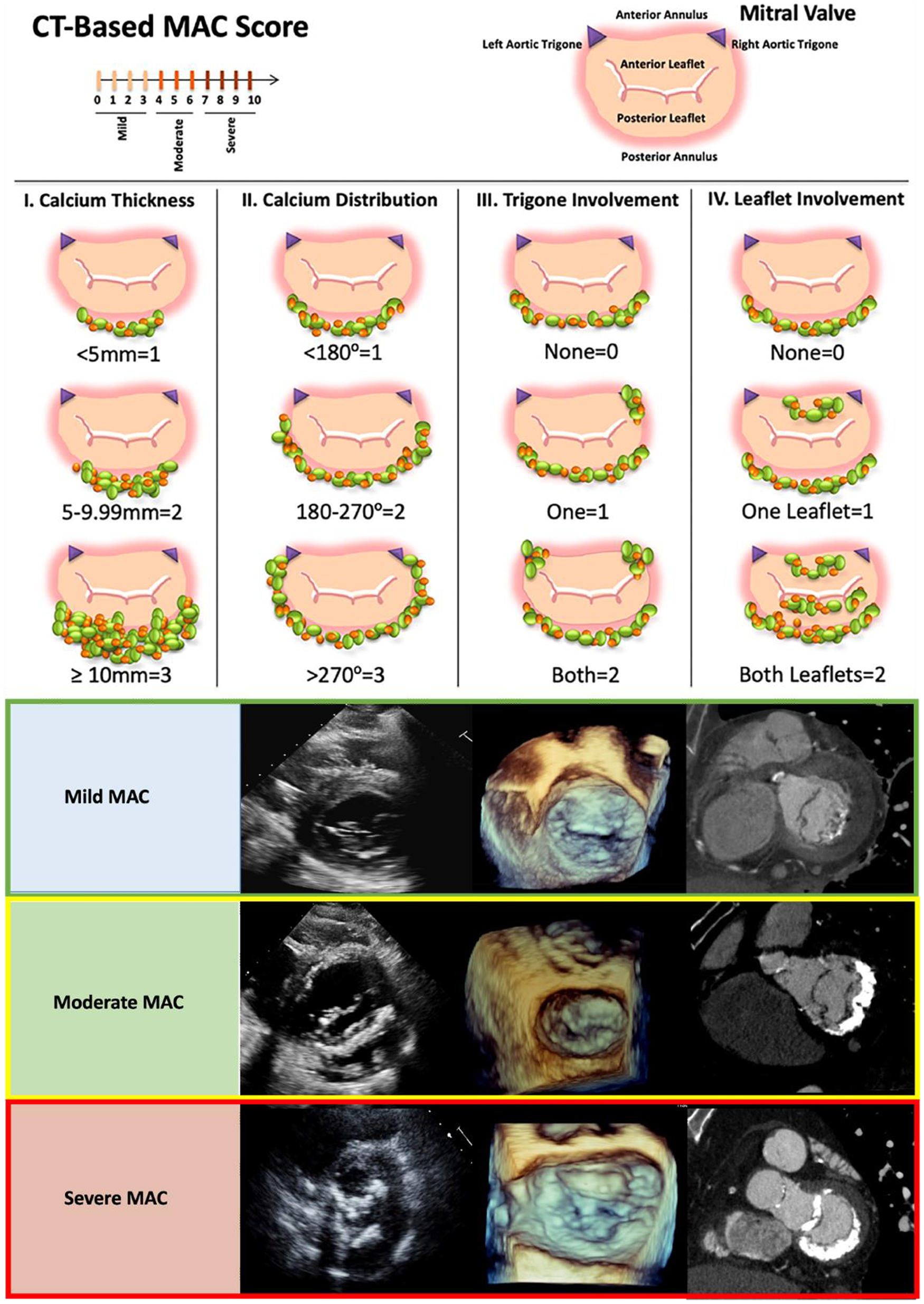
Key Points
Mitral annulus calcification is a degenerative process commonly observed in elderly individuals and increasingly recognized as a marker of cardiovascular disease. Imaging modalities, including echocardiography and computed tomography play a crucial role in the diagnosis, assessment, and grading of mitral annulus calcification (MAC) severity. The presence of MAC complicates mitral valve interventions, making surgical and transcatheter approaches challenging. While transcatheter mitral valve interventions have emerged as potential alternatives for patients with MAC, further technological advancements are needed to address the specific challenges posed by this patient population.
Definition and Epidemiology
Mitral annular calcification (MAC) refers to the deposition of calcium (along with lipid) in and around the annular fibrosa of the mitral valve. It was initially described by pathologists, 1 then using radiography and fluoroscopy before the 70s and first echocardiographic reports. Nowadays, MAC is easily diagnosed using echocardiography or computed tomography (CT) and has become more and more prevalent.2,3 Of note, MAC is independently associated with mortality, myocardial infarction, stroke and atrial fibrillation (AF). 4 Like in many cardiovascular diseases, the exact prevalence of MAC remains unknown due to several reasons (i) data often come from single-center and/or retrospective studies, (ii) prevalence varies significantly depending on the specific population explored as factor such as age, gender, comorbidities, and cardiovascular risk may influence MAC prevalence, (iii) prevalence may also differ based on the diagnostic modality used, and (iv) lastly, the definition of MAC itself may vary across studies.
The prevalence of MAC typically ranges from 8% to 23% in the general population2,4–6 and up to 40% or higher in studies of elderly individuals. 7 In the Framingham Heart study, using echo (and only M-mode modality), MAC prevalence was 14%. 4 In the Multi-Ethnic Study of Atherosclerosis (MESA) cohort, the overall prevalence of MAC, using CT analysis in 6814 patients, was 9% and highest in the Caucasians (12%) followed by Hispanics (10%), African Americans (7%), and Chinese (5%). After adjustment, age, female gender, diabetes mellitus, and increased body mass index remained associated with MAC. Another study from the Mayo Clinic that included 24,414 patients with an echocardiography found that 23% of patients had MAC. 6 Overall, MAC seems to be more common in elderly patients, in women, in patients with high body mass index or several cardiovascular risk factors.4,5,7–11
In certain populations, the prevalence of MAC is even higher. Among 761 patients with severe aortic stenosis who underwent transcatheter aortic valve replacement, 49.3% were found to have MAC including mild MAC in 30.4%, moderate MAC in 9.5% and severe MAC in 9.5% of patients. 12 MAC was associated with all-cause and cardiovascular mortality as well as conduction abnormalities. Similarly, in patients with isolated severe mitral stenosis (MS), MAC was present in 41% of cases. 13 Overall, any situation that increases mitral valve stress can accelerate the development of MAC. For instance, left ventricular hypertrophy, regardless of the presence of hypertension, was associated with increased MAC severity in the MESA cohort. 14 Additionally, Patlolla et al 15 reported a MAC prevalence of 18.5% in patients with hypertrophic cardiomyopathy.
Pathophysiology and Risk Factors
Although MAC is a common finding, especially in elderly patients, the pathophysiological mechanisms that lead to induction and aggravation of MAC are incompletely understood. While MAC was once considered a passive, purely degenerative and age-related process, results of recent histological and pathological studies draw the picture of MAC as an active and regulated process showing similarities with both medial and atherosclerotic calcification.16–18
Despite novel evidence suggesting an actively regulated pathophysiology of MAC, advancing age is still strongly associated with incident MAC. Classic autopsy studies described MAC as a chronic age-related degenerative change in the fibrous support of the valve, with progressive accumulation of calcium over time. 19 Microscopic lipid deposition and calcific foci can appear in the mitral annulus by the fifth decade of life, enlarging gradually with age.19,20 Correspondingly, clinical studies show MAC prevalence rising steeply with age and nearly half of nonagenarians having macroscopic MAC at autopsy. 21 These observations support a degenerative, age-driven component in MAC pathogenesis.
Yet, the primary stimulus for the development of MAC seems to be mechanical stress leading to endothelial injury and inflammation, similar to what is known for calcific aortic stenosis and atherosclerosis.17,22 Hemodynamic factors that increase stress on the mitral valve apparatus such as long-standing arterial hypertension, hypertrophic cardiomyopathy, and mitral valve prolapse lead to excess tension on the mitral annulus and may induce calcification.17,23 Consistently, patients with these conditions have a higher incidence of MAC than those without, and severity of left ventricular hypertrophy correlates with the prevalence and extent of MAC. 23
Following mechanical injury, recent findings suggest initiation of an atherosclerosis-like process promoted by inflammatory mechanisms. Early pathological investigations noted that MAC lesions often contain atherosclerotic features, such as foam cells in early annular lesions. 24 Moreover, patients with MAC frequently have concomitant coronary and vascular disease. 25 Indeed, numerous studies have shown that MAC is strongly associated with classical cardiovascular risk factors such as hypertension, diabetes mellitus, dyslipidemia, obesity, and smoking.26,27 Endothelial injury at sites of high mechanical stress on the mitral annulus is thought to trigger an inflammatory cascade that initiates calcification. 28 Histological studies show macrophage and T-lymphocyte infiltration in early MAC lesions, similar to arterial plaques, followed by osteogenic activity in the local valve cells.28,29 Valve interstitial cells in these regions begin expressing bone-regulatory proteins such as bone morphogenetic protein-2, and the annular tissue exhibits markers of osteoblastic differentiation and organized bone formation.29,30 In addition, consistent with an active inflammatory pathogenesis, patients with MAC often have elevated circulating inflammatory biomarkers (interleukin-6, C-reactive protein, and endothelin-1) and lower levels of endogenous inhibitors of calcification (nitric oxide and fetuin-A).9,31–33
Chronic kidney disease—Mineral Bone Disorder (CKD-MBD) represents another key mechanism promoting the development of MAC and defining MAC severity. CKD is a well-known risk factor for MAC, with reduced glomerular filtration and end-stage renal disease associated with markedly higher prevalence of annular calcification.34,35 This is partly explained by the high burden of atherogenic risk factors and hypertension in patients with CKD, as well as pressure/volume overload in advanced renal disease. 35 However, CKD also directly promotes calcification through mineral dysregulation. As renal function declines, phosphate excretion drops and phosphate retention ensues, leading to a compensatory rise in parathyroid hormone and mobilization of calcium from bone. The resulting elevation of the calcium-phosphate product can exceed the threshold of solubility and precipitate calcium-phosphate in tissues—essentially a form of metastatic calcification affecting the mitral annulus.17,22,34 Secondary hyperparathyroidism and hyperphosphatemia thus create a pro-calcific milieu. In fact, even in the absence of overt renal failure, elevated serum phosphate has been shown to induce vascular and valvular cells to undergo osteogenic transformation, initiating matrix mineralization in the annulus. 36 Notably, epidemiological studies indicate CKD has a stronger association with MAC than with aortic valve sclerosis, suggesting the mitral annulus may be uniquely susceptible to calcium-phosphorus disturbances.35,36
There also seems to be a particular role of sex in the context of MAC. Both risk and severity of MAC are commonly higher in female patients, despite the traditionally higher burden of atherosclerosis in males.21,26,27 For example, in the large MESA cohort, the CT-determined prevalence and incidence of MAC were 52% and 35% higher in females than in males after adjustment for other factors. 27 The impact of postmenopausal osteoporosis might be a possible explanation aligning this finding with the abovementioned concept of mineral and bone dysregulation modulating MAC. The severe bone loss in elderly women may lead to ectopic calcium deposition in the annulus, contributing to the higher prevalence and severity of MAC in women. 37 In support of this, use of bisphosphonates has been associated with a lower prevalence of MAC in elderly women. 37
Finally, certain congenital metabolic or connective tissue disorders can predispose to early-onset MAC. In Marfan syndrome, early-onset MAC occurring before age 40 has been documented and may be driven by commonly found chronic mitral valve prolapse and excess annular stress, or by an intrinsic connective tissue defect of the annulus. 38 Hurler syndrome (Mucopolysaccharidosis type I) is another example. Children with Hurler syndrome have been reported to develop MAC, presumably due to abnormal fibroblast function and accelerated degeneration of annular collagen, leading to calcific deposits even at a young age.17,38
MAC-Related MV Disease
MAC can also contribute to mitral valve dysfunction, often presenting as MS, mitral regurgitation (MR), or combination of both (mixed valve disease) (Figure 1). As a result, MAC-related mitral valve dysfunction is an important clinical consideration though its prevalence is likely underestimated. 39 Bertrand et al 40 found that among patients with MAC, 8% had MS (defined as a mean transmitral gradient [TMG] ≥ 3 mm Hg. In these patients, MR severity was none/trace in 32%, mild in 42%, moderate in 23%, and severe in 3%. Notably, higher TMG was associated with reduced long-term survival. Nevertheless, MR was associated with increased mortality in patients with low-gradient MS (TMG 3-5 mm Hg) and its prognostic impact diminished at higher gradients.
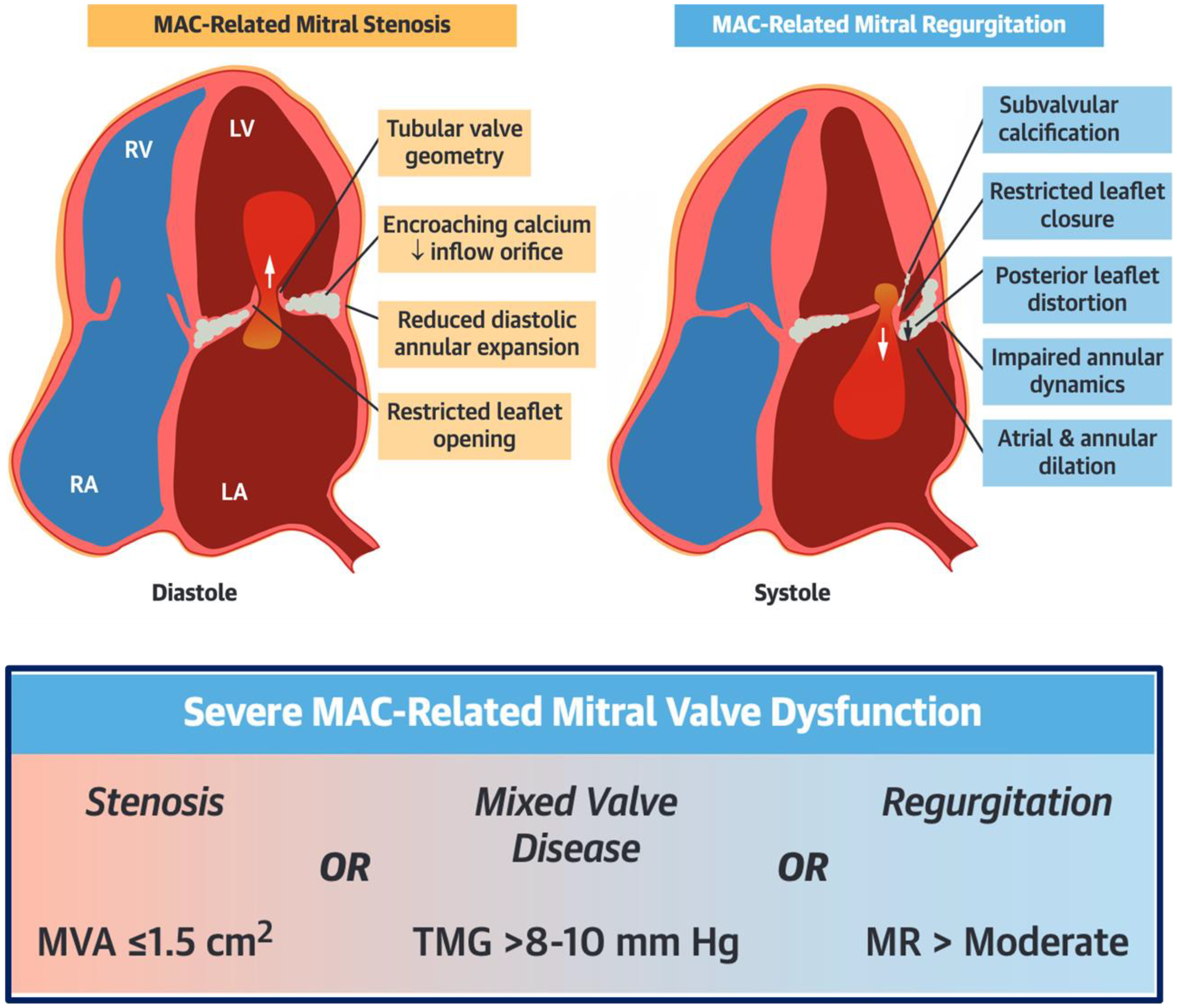
Mechanisms and definition of severe MAC-related MV dysfunction. MAC-related mitral stenosis is caused by tubular valve geometry, reduced inflow orifice and diastolic annular expansion and/or restricted leaflet opening. MAC-related mitral regurgitation is caused by subvalvular calcification, restricted leaflet closure, impaired annular dynamics and/or atrial and annular dilation. Severe MAC-related MV dysfunction is defined by a mitral valve area ≤1.5 cm2 or a transmitral gradient >8-10 mm Hg or a mitral regurgitation > moderate. Adapted from Churchill et al J Am Coll Cardiol 2022;80:739–751.
In another study, where MAC-related mitral valve dysfunction was defined as MR or MS of moderate or greater severity, the prevalence was 16%. 6 As summarized by Churchill et al, 2 MAC-related mitral valve dysfunction is higher in selected population: in patients with priori chest radiation (6%-15%) or those with aortic stenosis. A single-center study from the Cleveland Clinic reported an averaged TMG progression rate of 0.04 +/− 0.0097 mm/Hg/years with faster progression in patients who had severe MAC at baseline. 37 Yet, data remain limited regarding the natural progression of MAC-related mitral valve dysfunction.
Place of Imaging
Echocardiography
Echocardiography plays a pivotal role in assessing MAC severity and its impact on mitral valve function, offering critical insights into both structural and functional abnormalities. It enables a comprehensive evaluation of calcification, MS and MR.9,41 A systematic echocardiographic assessment of MAC begins with analyzing key visual features, including the distribution and severity of calcification, leaflet thickening, and mobility. 39 Transthoracic echocardiography (TTE) typically identifies MAC as an echodense, irregular structure with posterior acoustic shadowing, which can obscure leaflet assessment and necessitate alternative imaging angles or modalities. 41 Alternative echocardiographic views and three-dimensional (3D) echocardiography further enhance the assessment of leaflet and subvalvular structures.
The assessment of hemodynamic consequences and MAC-related mitral valve dysfunction remains challenging. For MS evaluation, color Doppler imaging ensures proper alignment of the continuous-wave Doppler beam with mitral inflow, allowing for an accurate estimation of peak and mean transvalvular gradients. However, factors such as AF, concomitant MR, heart rate, and cardiac output can influence these measurements. Pressure half-time may overestimate the mitral valve area (MVA) when left atrial or left ventricular compliance is altered—a common occurrence in this elderly population.2,42 Direct two-dimensional (2D) planimetry is often difficult, particularly in patients with acoustic shadowing due to anterior calcifications. Moreover, this measurement is debated, as, unlike in rheumatic MS, there is no commissural fusion; the orifice is nonplanar, with its narrowest part located closer to the annular plane rather than at the leaflet tips. 2 The continuity equation offers a more reliable estimation of MVA in calcific MS, yet its accuracy is compromised in cases of significant MR or AF. Three-dimensional planimetry using multiplanar reconstruction provides a more precise assessment of valve morphology but requires expertise and is more effectively performed with TEE.2,43 Assessing mitral regurgitation in MAC demands a multiparametric approach, integrating qualitative, semiquantitative, and quantitative parameters. Vena contracta measurement can be challenging and acoustic shadowing from MAC may limit the visualization of the regurgitant. In such cases, a blind continuous-wave Doppler sweep of the left atrium or a parasternal short-axis view can aid MR detection. 41 When TTE is inconclusive, TEE provides superior visualization and a better understanding of MR mechanisms, particularly with 3D color Doppler, which enhances identification of jet number and location.2,41
Beyond standard assessments, echocardiography, and particularly TEE is crucial in differentiating MAC from other pathological conditions, such as tumors or thrombi, while also identifying endocarditis, a potential complication of MAC. 39 In symptomatic patients with moderate MS, exercise stress echocardiography can provide valuable prognostic insights.39,44 Given the rising prevalence of MAC in aging populations, echocardiography remains the cornerstone and first-line imaging modality for diagnosis. However, additional imaging techniques are often required to ensure precise characterization of mitral valve anatomy and pathology and to guide therapeutic decision making. An example of an echocardiographic evaluation of a patient with MAC is provided in Figure 2.
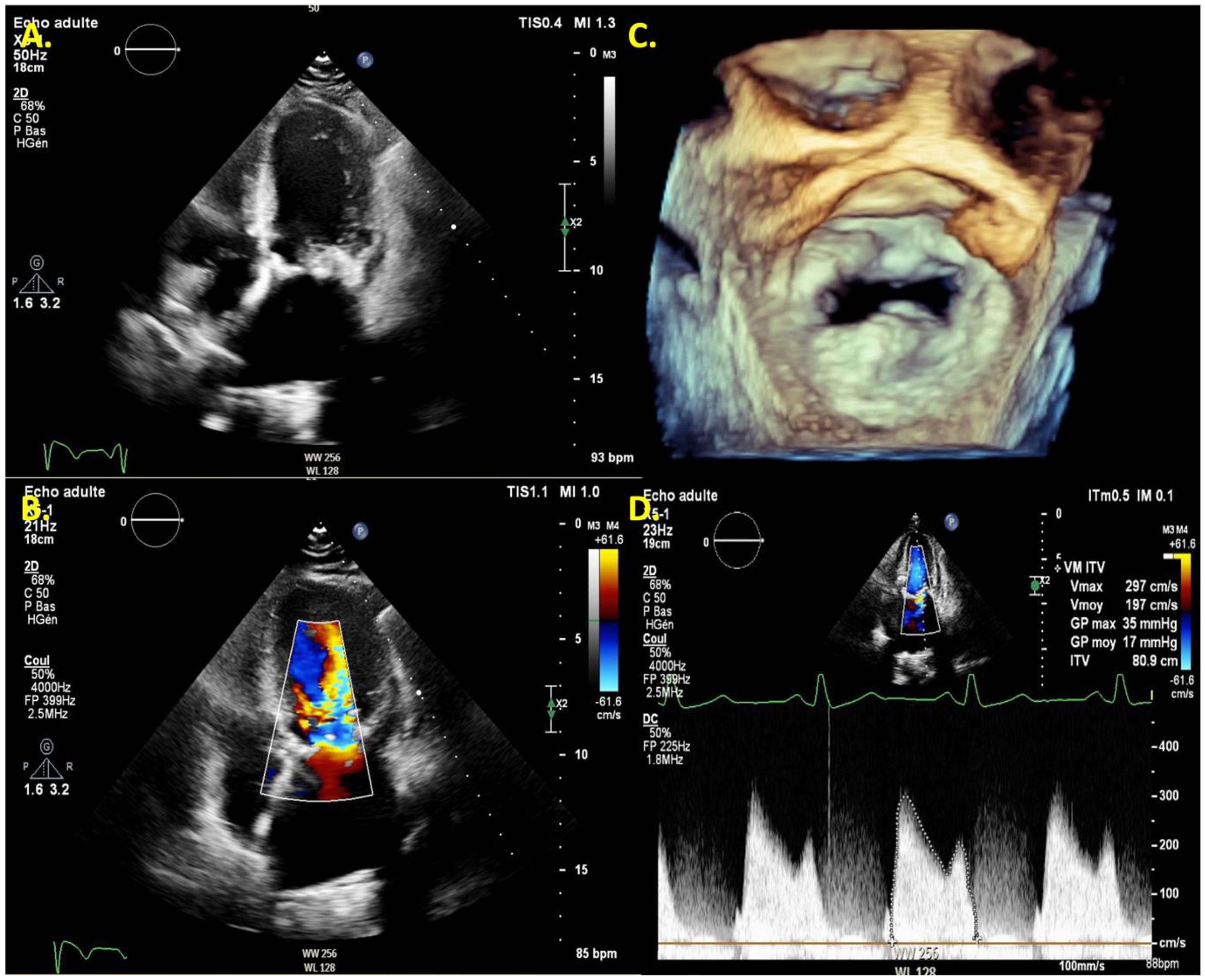
Example of echocardiographic assessment in a patient with MAC. (A) Apical 4-chamber view without color and (B) with color Doppler demonstrating extensive calcification of the mitral annulus and both leaflets, along with a reduced inflow orifice (aliasing). (C) TTE 3D en face view showing MAC-related mitral valve disease with calcification involving both leaflets and commissures. (D) Continuous Doppler recorded a mean transmitral gradient of 17mmHg which signed a severe MAC-related MV disease.
Cardiac CT
Cardiac CT has emerged as a crucial imaging modality in the comprehensive assessment of MAC, offering high spatial resolution and superior visualization of calcium burden compared to echocardiography. Beyond its role in confirming the presence and extent of annular calcification, CT provides detailed characterization of the anatomy and quantification of calcific burden, including annular circumference, thickness, and extension into adjacent structures. This allows accurate classification of MAC severity, which is critical for therapeutic planning, particularly in high-risk patients considered for surgical or transcatheter interventions. Dedicated ECG-gated CT with contrast enables evaluation of the mitral annulus geometry, degree of subvalvular and myocardial extension, and identification of caseous calcification, which may mimic mass lesions on echocardiography (Figure 3).41,45 More specifically, caseous (also called “toothpaste”) MAC is a rare form of MAC characterized by central liquefactive necrosis composed of amorphous, eosinophilic, cell-free material. This lesion is typically found in the posterior MA or periannular region with an echodense peripheral appearance and a central area with echolucency. The main differential diagnosis is endocarditis, especially in the presence of elevated and confounding inflammatory markers. This form is also associated with a high incidence of cerebrovascular events (19.2%). 46
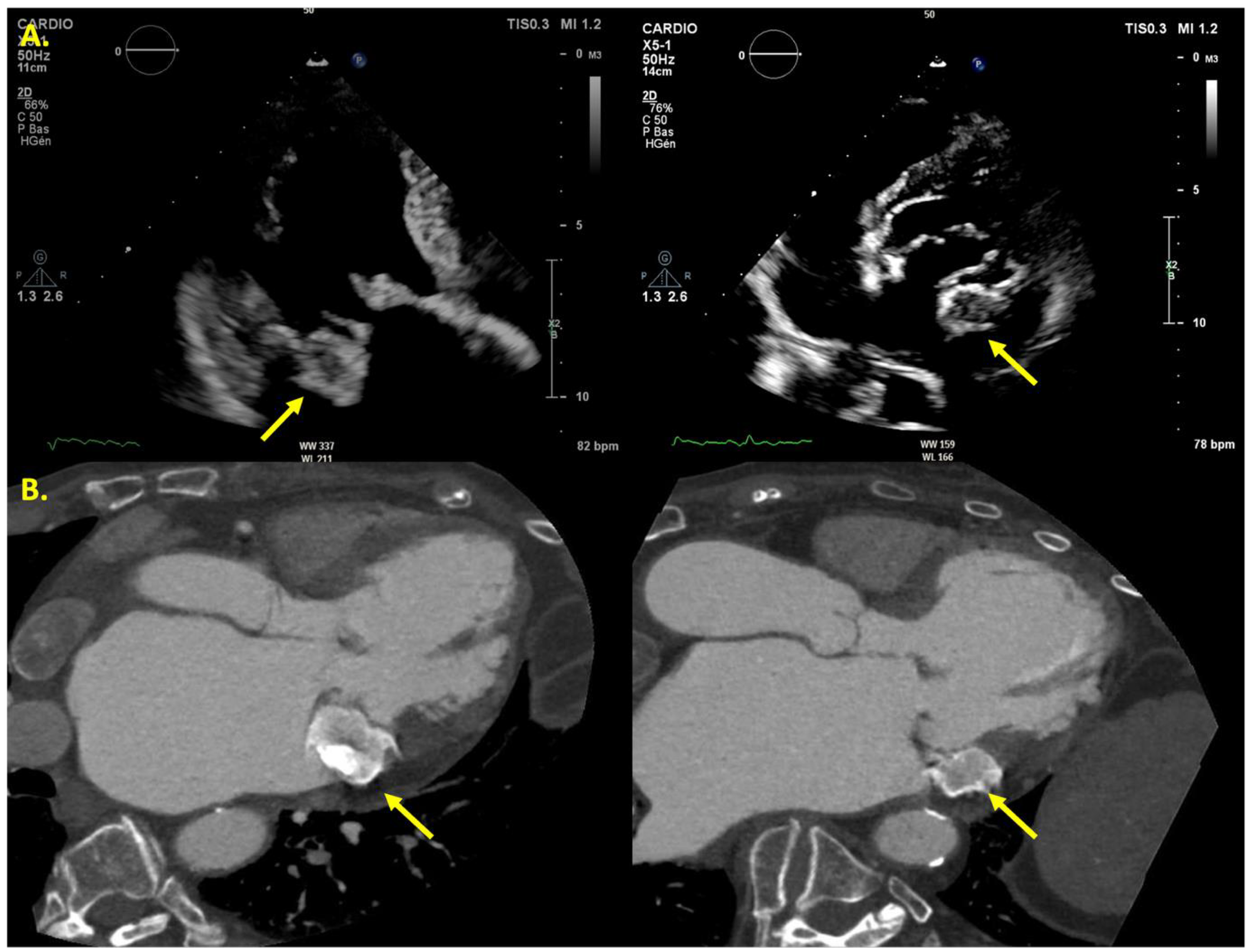
Example of a patient with caseous calcification. TTE (A) and CT (B) images of a patient with caseous calcification (yellow arrow) involving the inferolateral region of the mitral annulus. TTE demonstrates a peripheral echodense rim with a central echolucent area, while the noncontrast CT scan shows a hyperdense mass featuring peripheral calcifications and a hypodense core.
Cardiac CTA is the most important complementary imaging tool to explore caseous MAC. Without contrast enhancement, the lesion appears as a hyperdense mass with peripheral calcifications (>600 HU) and a hypodense core (<50 HU). Following contrast administration, no enhancement is seen during either the arterial (early) or venous (delayed) phases.
Preprocedural CT assessment is also essential in procedural planning for transcatheter mitral valve replacement (TMVR) in MAC, providing measurements of the mitral annular area, left ventricular outflow tract (LVOT) dimensions, and simulated valve placement to predict the risk of LVOT obstruction. Quantification of calcific burden has demonstrated prognostic significance, as higher calcium volumes are associated with procedural complexity and increased risk of paravalvular leak, device malposition, and mortality.39,41 Moreover, observational studies have highlighted the association between extensive MAC detected on CT and adverse cardiovascular outcomes, including AF, stroke, heart failure, and all-cause mortality, suggesting that MAC burden could be also an interesting marker of atherosclerotic burden. 39 Thus, cardiac CT not only refines anatomical diagnosis but also offers risk stratification in MAC, guiding therapeutic strategies and potentially anticipating procedural risks.
Finally, the emergence of photon-counting CT offers improved visualization of calcifications by reducing artifacts and potentially enabling assessment of calcification quality. Examples of its application are shown in Figure 4.
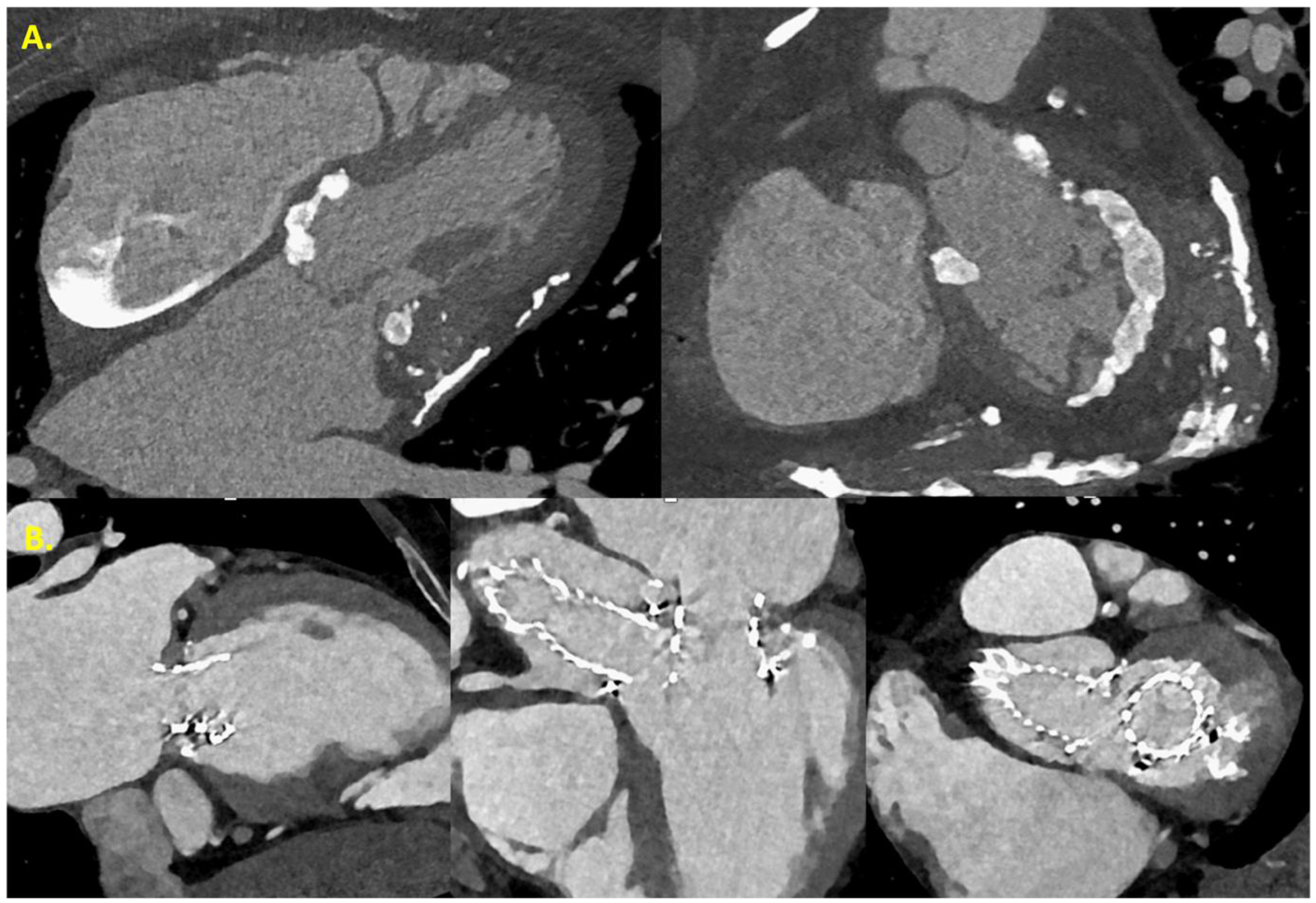
Examples of photo-counting CT applications. (A) Photon-counting detector CT. Ultra high resolution mode (0.11 × 0.11 × 0.2 mm3). Patient with severe MAC and pericardial calcifications. The UHR mode enables a more precise delineation of mitral annulus calcification. Constriction is suspected due to the tubular configuration of the left ventricle. (B) Photon-counting detector CT. Standard resolution mode (0.24 × 0.24 × 0.4 mm3). TAVR in aortic prosthetic valve and TAVR in mitral prosthetic valve. High tension acquisition to limit metal artifacts. MAC remains nicely depicted.
Cardiac MRI
The role of cardiovascular magnetic resonance imaging (CMR) in the assessment of MAC is limited compared to echocardiography and CT due to intrinsic limitations in calcium detection. Conventional CMR sequences do not allow for reliable visualization or quantification of calcific deposits, given their low signal intensity and susceptibility artifacts.39,41 Therefore, CMR is not recommended for structural assessment or procedural planning in MAC. Nevertheless, CMR can provide precise evaluation of mitral valve function in cases of multiple and/or mixed valvular disease, especially when acoustic shadowing from calcifications limits Doppler assessment. CMR offers accurate MR quantification using phase-contrast velocity sequence and volumetric techniques, independent of jet eccentricity or image quality limitations inherent to echocardiography. 39 This is crucial in patients with MAC-associated MR, where multijet or eccentric regurgitation is frequently under-quantified by Doppler echocardiography. Additionally, CMR can detect functional consequences of MAC, such as LA and LV remodeling, 47 which have been associated with adverse prognosis. Beyond structural evaluation, late gadolinium enhancement (LGE) imaging may identify concomitant focal myocardial fibrosis, 47 providing incremental prognostic information in patients with MAC, as ventricular fibrosis correlate with higher arrhythmic risk and mortality. 41 Moreover, in rare cases of caseous mitral annular calcification, CMR can help differentiate this benign entity from infective masses or tumors by demonstrating characteristic imaging features, such as central liquefaction surrounded by a calcified rim. While CMR does not play a frontline role in MAC detection, its unique strengths in functional and tissue characterization offer complementary diagnostic and prognostic information.
MAC Grading
In recent years, several MAC grading classifications have been proposed. Among these, Guerrero's classification—based on four parameters (calcium thickness, calcium distribution, trigone involvement, and leaflet involvement) assessed by CT imaging—was originally developed to predict the risk of transcatheter heart valve embolization and remains the most widely used (Central Illustration). 48
In addition, using TTE short-axis views to improve visualization of annular calcification, several authors have suggested alternative scoring systems to grade MAC severity.2,39,41
Outcomes
MAC is a chronic, degenerative process commonly seen in older adults and associated with a range of cardiovascular risk factors. The outcomes of patients with MAC can vary significantly depending on whether they have concomitant MV disease, such as mitral stenosis, regurgitation, or combined MV disease.
Outcomes of Individuals With Mitral Annular Calcification
The presence of MAC is not benign. Several studies have shown that MAC is associated with worse clinical outcomes compared to a noncalcified MV annulus. In the Framingham Heart Study, a total of 1197 subjects (445 males and 752 females) were assessed by M-mode echocardiography and classified according to the presence of MAC. 36 The prevalence of MAC was 14% and its presence was independently associated with increased risk of mortality (all-cause and cardiovascular), myocardial infarction, heart failure, stroke, and AF. This increased risk was found to occur in a graded fashion by MAC severity.
In the MESA, a cohort study of 6814 men and women aged 45 to 84 years without a known history of CVD at baseline, MAC was assessed with the use of CT. 49 Of the 6814 participants (mean age 62.2 ± 10.2 years), 644 (9.5%) had MAC. Patients with MAC were older, more likely to be women (10.7% vs 8.0%, women vs men, respectively), white, and former smokers. In this multiethnic cohort, the presence of MAC was associated with increased risk of incident renal failure, dementia, all-cause (HR 1.47, 95% confidence interval [CI] 1.29 to 1.69), cardiovascular (HR 1.39, 95% CI 1.04 to 1.87), and noncardiovascular mortality (HR 1.35, 95% CI 1.14 to 1.60) compared to those without MAC.
MAC is a chronic degenerative process that may progress. 50 MAC progression can be structural (greater extent of MAC in the MV circumference) or hemodynamic (increased MV mean diastolic pressure gradient). The progression of MAC was observed in one-third of patients (31.2%) with baseline MAC, with structural being more common than hemodynamic progression. Patients who had progressive MAC demonstrated increased mortality rates compared to patients with stable MAC
Therefore, MAC is recognized as a marker of increased cardiovascular risk. Patients with MAC have a notably high prevalence of AF, which is likely due to structural changes and fibrosis caused by calcification that disrupts normal electrical conduction pathways in the heart. The increased risk of mortality is particularly pronounced in patients with MAC, who are elderly and often have additional adverse prognostic factors. The association of MAC with other comorbidities further exacerbates the cardiovascular risk profile of these patients.
In terms of clinical management, there is no intervention specific to the calcification itself. Instead, the primary focus is on managing associated cardiovascular risk factors, such as optimizing treatments for hypertension, hyperlipidemia, and diabetes. Monitoring for the development of AF and other complications is also crucial. Regular echocardiographic assessments are recommended to detect any progression to mitral valve disease.
Outcomes of Individuals With Mitral Annular Calcification and Mitral Valve Disease
MAC is not always associated with MV disease. However, as MAC progresses, it can involve the components of the MV complex, including the leaflets, trigones, and subvalvular apparatus. This progression can lead to the development of MR, MS, or a combination of both conditions. The combination of MAC with significant MV disease is associated with higher morbidity and mortality rates compared to MAC alone.
In a retrospective study of 41,136 patients who underwent transthoracic echocardiography, MAC was identified in 2855 (6.9%) patients. 51 Significant MR and/or MS were found in 434 (1.1% of total; 15.2% of MAC) patients: median age [interquartile range, IQR], 80 [73 to 87] years; 63% women. Patients with mixed MR and MS had the most extensive involvement of MAC (location, trigone involvement, circumferential distribution, and leaflet disease) and the highest risk of mortality compared to those with either MR or MS (MR + MS 18.5 [11.3, 29.6], MR 15.1 [12.5, 19.1], MS 14.9 [10.4, 21.7], per 100 person-year follow up). A total of 59 (14%) of patients underwent MV intervention. The incidence of all-cause death was 7.3 [IQR: 3.4 to 13.4] per 100 person-year in patients with MV intervention versus 16.8 [14.3 to 20.4] in those without MV intervention. Although infrequently performed and limited to selected patients, MV intervention is associated with improved survival.
The outcomes of patients with MAC vary significantly based on the presence and severity of associated MV disease. While patients with MAC alone face increased cardiovascular risks, those with concomitant MV disease experience higher morbidity and mortality. Management strategies need to be tailored to the individual patient, taking into account the severity of calcification, the presence of MV dysfunction, and the overall cardiovascular risk profile. Advances in surgical and transcatheter therapies offer promising options for high-risk patients, but careful evaluation and selection are essential to optimize outcomes.
Future Perspectives for MAC Assessment and Management
Looking ahead, imaging tools should enable the identification of patients who are rapidly progressing to severe MAC, allow for accurate and reproducible evaluation of MAC-related MV dysfunction, and provide precise quantification and characterization of calcifications to support optimal management. At the same time, research should focus on developing strategies to slow disease progression and on identifying patient phenotypes most likely to benefit from invasive interventions.
While surgical mitral valve interventions remain the standard of care for many patients, severe MAC is associated with increased perioperative morbidity and mortality, often rendering patients inoperable or at prohibitive surgical risk, or CV surgeons reluctant to operate on patient with severe MAC. Indeed, MAC predominantly affects elderly subjects (> 70 years old), with multiple comorbidities, 52 and is often observed in those with history of mediastinal radiation therapy—a particularly vulnerable subgroup due to the frequent coexistence of coronary artery disease, aortic stenosis, and porcelain aorta.45,53 Moreover, while mild MAC is generally not affecting repair or replacement feasibility, 54 mitral valve surgery in subjects with significant MAC is associated with a higher risk of atrioventricular groove rupture, circumflex artery injury, embolic events, and significant paravalvular leaks (PVL).55,56
In this context, transcatheter mitral valve interventions have emerged as potential alternative strategies. However, the rigid and often asymmetric anatomy imposed by MAC introduces unique procedural complexities, including difficulty in device anchoring, increased risk of PVL, and the potential for LVOT obstruction.
Mitral transcatheter edge-to-edge repair (M-TEER) presents substantial technical and clinical hurdles that can limit procedural success and long-term efficacy. While TEER is technically feasible in selected patients with MAC with predominant MR, 57 thorough preprocedural imaging is essential to evaluate leaflet mobility, the extent of calcification, and the remaining valve area, ensuring an optimal balance between the risks of residual regurgitation and procedure-induced stenosis. Balloon mitral valvuloplasty widely adopted for the treatment of rheumatic MS, has historically demonstrated limited efficacy in the setting of MAC-related mitral obstruction due to fundamental differences in pathophysiology.
Finally, TMVR seems a promising solution for patients with severe MAC and complex mixed mitral valve disease, especially when other interventions are contraindicated. However, success requires rigorous preprocedural planning, advanced imaging, highly experienced multidisciplinary teams, and continued device innovation. Ongoing trials and real-world registries will be instrumental in defining optimal patient selection and refining procedural techniques to enhance outcomes in this high-risk cohort.
Overall, while transcatheter mitral valve interventions have emerged as potential alternatives for patients with MAC, they require careful patient selection, meticulous preprocedural planning, and procedural refinement. Further technological advancements are needed to address the specific challenges posed by this patient population.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflict of Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Augustin Coisne has served as consultant for Abbott Vascular and GE Healthcare and received speaker fees from Abbot Vascular, BMS, Edwards Lifesciences, GE Healthcare, MSD, Oseus and Pfizer. Andrea Scotti has served as a consultant and received consulting fees from NeoChord Inc. Sebastian Ludwig received research grants from the German Heart Foundation and 4C Medical, speaker honoraria from Abbott, advisory fees from New Valve Technology and Bayer, and travel compensation from Edwards LifeSciences and Shockwave Medical. Charles Fauvel has received speaker fees from GE Healthcare. The remaining authors have nothing to disclose.
Adapted from Guerrero et al. 8
