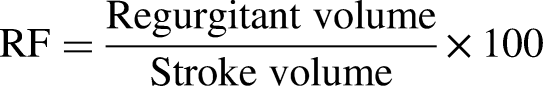Abstract
Background and objectives
Mitral valve replacement may be considered in patients with mitral valve disease when durable repair is not feasible. In recent decades, an important shift toward the use of bioprostheses for mitral valve replacement was observed and the surgical valve design has undergone several iterations to optimize the prosthesis hemodynamic performance. The objective of this study was to compare the hemodynamic performance of the stented porcine (EPIC Plus) versus the stented bovine pericardial (MITRIS) bioprostheses under standardized in vitro conditions.
Methods
Three MITRIS 27 mm and three EPIC Plus 29 mm, which are equivalent in terms of actual sizing (ie, fit into the same mitral annulus size of 38 mm), were tested in vitro in a double activation left heart duplicator system including anatomically shaped, deformable silicone moldings of left heart cavities and aorta. The valves were tested under several standardized hemodynamic conditions: heart rate: 70 and 120 bpm, mean aortic pressure: 100 and 160 mm Hg, stroke volume: 30, 70, and 100 mL. Mean mitral transprosthetic pressure gradients (mTPGs) and effective orifice areas (EOAs) were measured by continuous-wave Doppler and the geometric orifice areas (GOAs) were obtained using high speed, en-face, imaging.
Results
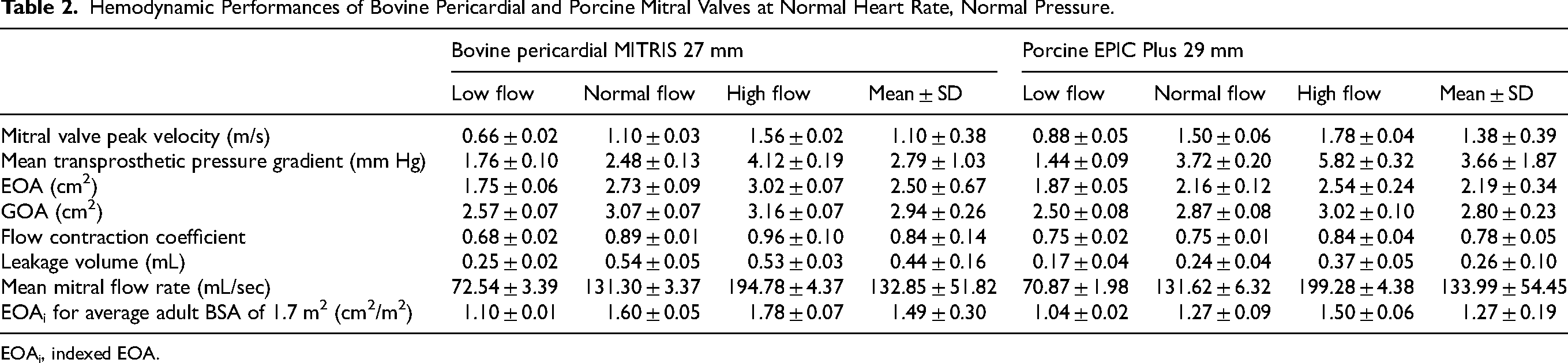
For normal heart rate (70 bpm) and normal mean aortic pressure (100 mm Hg), the mTPGs were similar (1.76 ± 0.10 vs 1.44 ± 0.09 mm Hg) in low-flow conditions, but lower (P ≤ .001) in normal (2.48 ± 0.13 vs 3.72 ± 0.20 mm Hg) and high flow (4.12 ± 0.19 vs 5.82 ± 0.32 mm Hg) in MITRIS versus EPIC Plus valves. EOAs were larger in MITRIS versus EPIC Plus valves for all, except low flow, conditions (low: 1.75 ± 0.06 vs 1.87 ± 0.05, normal: 2.73 ± 0.09 vs 2.16 ± 0.12, and high flow: 3.02 ± 0.07 vs 2.54 ± 0.21 cm2, all P ≤ .001). GOAs were also larger in MITRIS versus EPIC Plus valves (low: 2.57 ± 0.07 vs 2.50 ± 0.08, normal: 3.07 ± 0.07 vs 2.87 ± 0.08, and high flow: 3.16 ± 0.07 vs 3.02 ± 0.10 cm2, all P ≤ .001). The two bioprosthetic valves had regurgitant fraction (RF) < 5% without statistical differences between flow conditions.
Conclusions
This study demonstrates excellent valve hemodynamic performance for these two types of mitral bioprosthetic valves. However, the overall hemodynamic performances were superior with the MITRIS valve compared to the EPIC Plus valve for all flow conditions.
This is a visual representation of the abstract.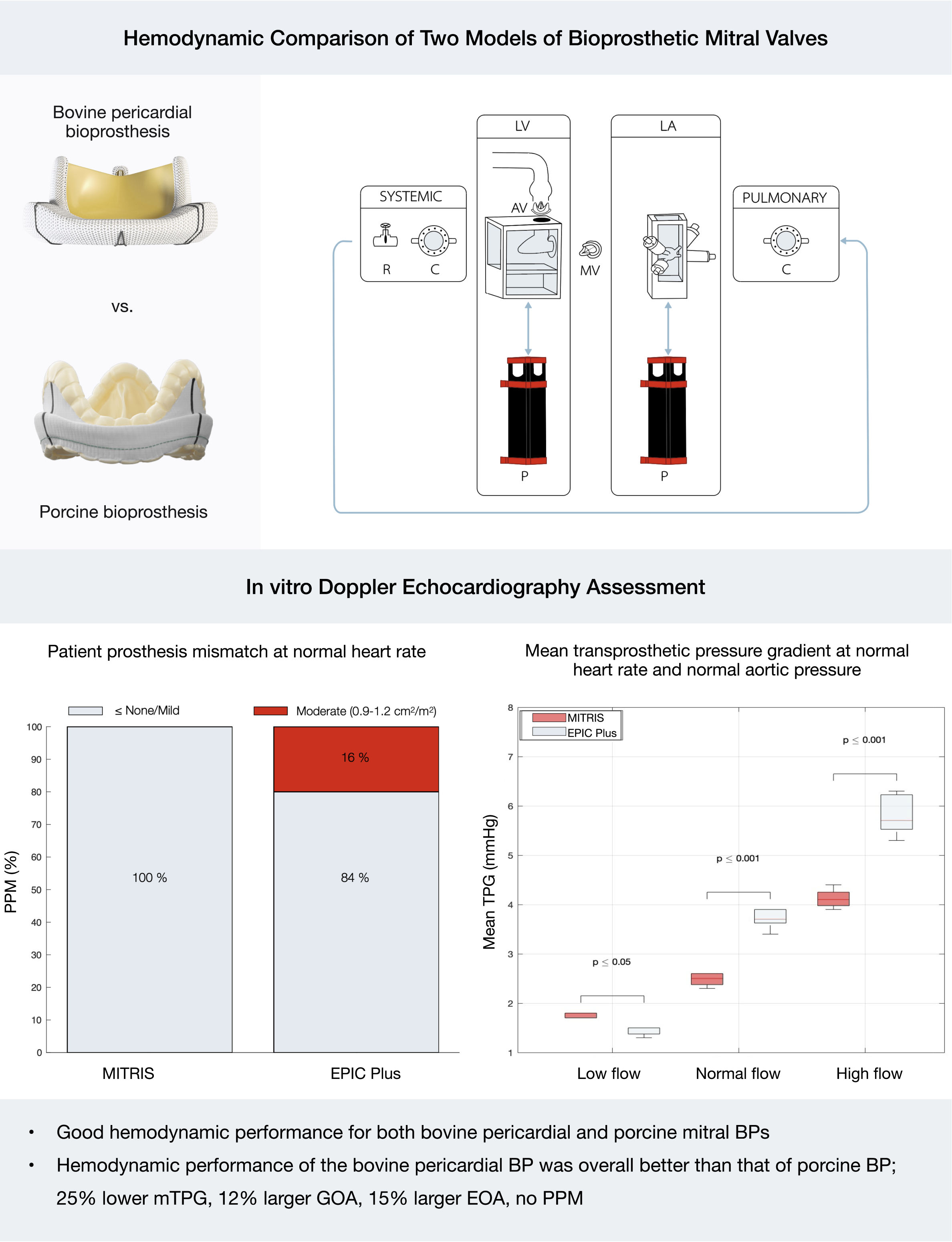
Keywords
Introduction
Mitral valve (MV) disease is the most frequent valvular heart disease in the world, with an unfavorable prognosis when left untreated.1,2 Its prevalence increases with the aging population.3–5 MV disease can manifest as mitral stenosis, mitral regurgitation, or mixed dysfunction, but over 95% of MV repair and 75% of MV replacement procedures are performed to correct mitral regurgitation. 6 Even though the MV repair is preferred over replacement in clinical guidelines, according to the analysis of Medicare claims data, the overall MV repair rate is 69% in United States cardiac surgery centers, indicating that nearly one-third of cases require alternative treatment approaches. 7 MV replacement is usually indicated when repair is not feasible, including cases of severe mitral regurgitation with unfavorable anatomy for repair, failed previous MV repair, extensive valve degeneration, and severe calcification, which prevent successful and durable repair. Furthermore, recent data failed to demonstrate superiority of repair in specific subgroups such as patients with ischemic mitral regurgitation,8–10 or with endocarditis, 11 elderly patients,12,13 and women. 14
The design of surgical mitral prosthetic valves has undergone many iterations since the first surgical valve was implanted. 15 Valve design has evolved from mechanical to bioprosthetic (BP), and ultimately to transcatheter heart valves for minimally invasive approaches.16,17 The utilization of BP valves has surpassed mechanical valve replacement over the past two decades, primarily due to the elimination of need for anticoagulation while maintaining hemodynamic parameters close to that of native valves. Despite the evolution of design helping to prevent the complications of prosthetic valves, such as prosthesis-patient mismatch (PPM), paravalvular regurgitation, thrombosis, structural valve deterioration etc, there has been a lack of direct and independent comparisons between the different platforms of BP valves.
Among the BP valves available for MV replacement, stented porcine valves Mosaic (Medtronic, Minneapolis, MN, USA) and Epic (Abbott, Chicago, IL, USA) and stented bovine pericardial valves Perimount (Edwards Lifesciences, Irvine, CA, USA), Magna Mitral Ease (Edwards Lifesciences) or newer Mitris (Edwards Lifesciences), have emerged as the most frequently used bioprostheses (BPs). The fundamental principles underlying these two types of mitral BPs are similar: that is, they are composed of a stent supporting 3 leaflets and suture ring allowing to fix the prosthesis within the mitral annulus. However, these BPs present major difference in their design with respect to the type of tissue used to fabricate the BP valve leaflets: porcine valves versus bovine pericardium. Optimization of valve hemodynamic performance of mitral BP valves is key to avoid severe PPM and high residual transprosthetic pressure gradients. At present, there is little data comparing the hemodynamic performance of bovine pericardial versus porcine mitral BPs, though bovine pericardial valves appear to offer better hemodynamic performance with improved flow characteristics and reduced pressure gradients.18,19 The objective of this in vitro study was to perform a head-to-head standardized comparison of the porcine (EPIC Plus) versus bovine pericardial (MITRIS) BPs.
Methods
A double activation simulator (Supplemental Figure 1) was used for in vitro testing of three porcine (29 mm EPIC Plus, Abbott) versus three bovine pericardial (27 mm MITRIS, Edwards) BPs. Both 29 mm EPIC Plus and 27 mm MITRIS are equivalent in terms of actual sizing (ie, internal diameter is 26.3 and 27 mm, respectively, and compatible with the same mitral annulus size of 38 mm). A 23 mm Epic-Supra aortic valve (Abbott, Chicago, IL, USA) was used in the in vitro system to ensure consistent and reproducible aortic valve function across all tests. Figure 1 presents the characteristics of the design of these BPs according to Instructions for Use of each BP. The BPs were new and used for the first time in this study. Each BP was firstly sutured on a silicone gasket (Figure 2A) to ensure the valve stability and was then inserted in custom-made and 3D-printed circular valve holder (Figure 2B).
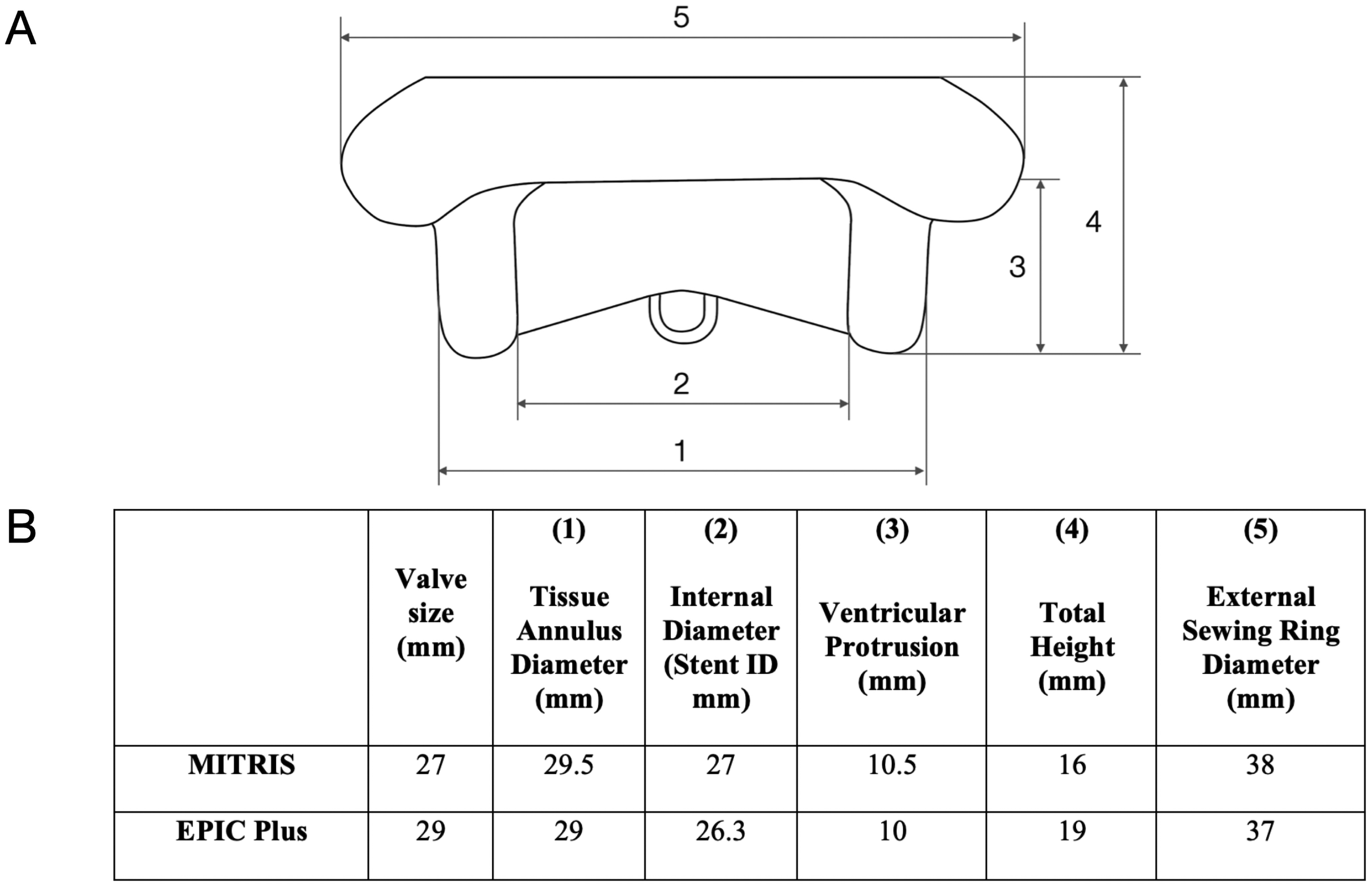
Nominal Dimensions for MITRIS and EPIC Plus Mitral Valves. Panel A: Standardized approach to present surgical heart valve physical dimensions. Panel B: Valve label size and internal diameter specifications for MITRIS 27 mm and EPIC Plus 29 mm.
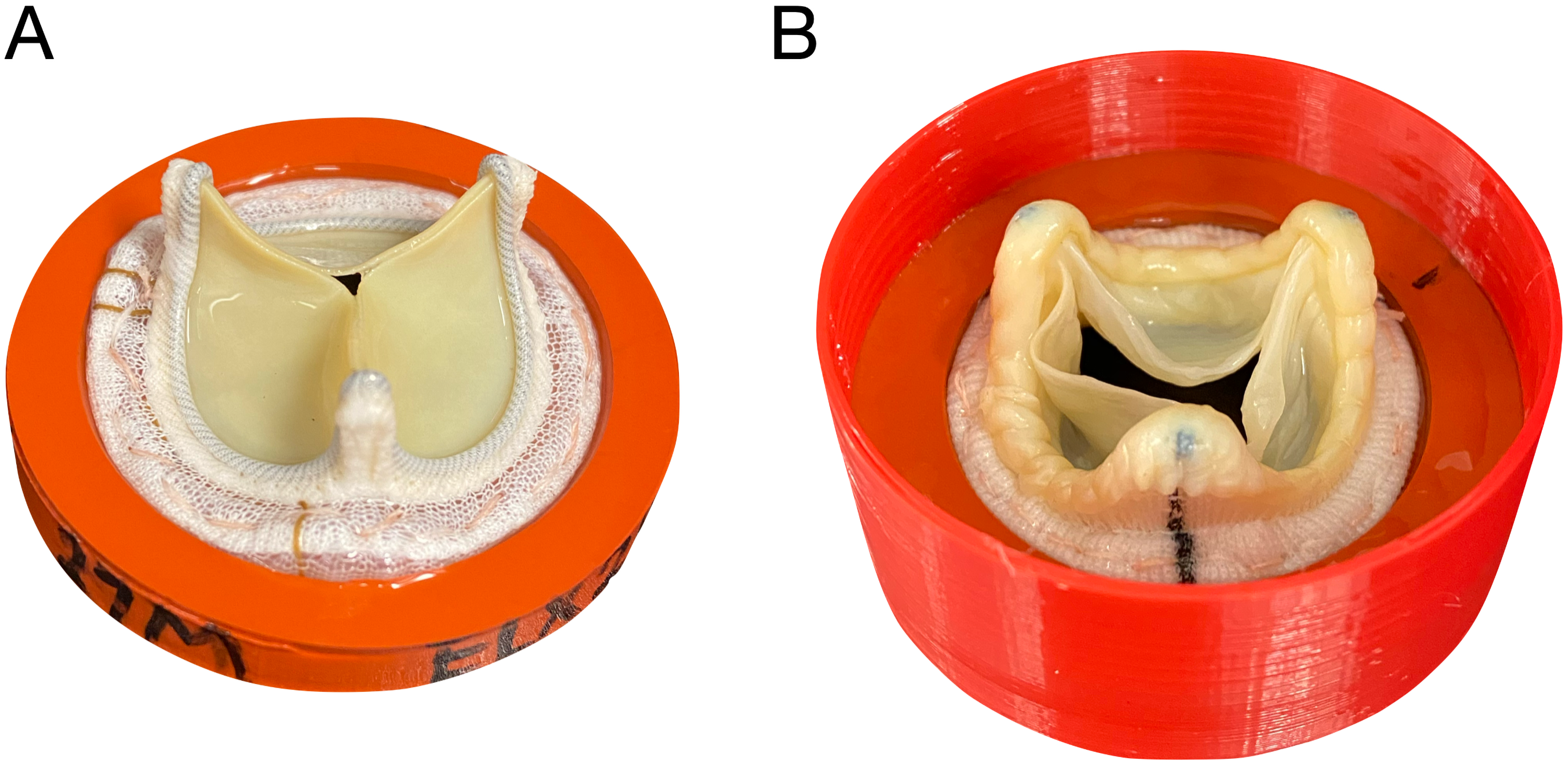
Example of the Implantation in the Circular Valve Holder. Panel A: MITRIS valve sutured on silicone gasket. Panel B: EPIC Plus valve with gasket assembly inserted in the final 3D printed valve mounting fixture.
In Vitro Cardiovascular Simulation
The double activation left heart duplicator system
20
used for this study is able to accurately reproduce the human blood circulation from the pulmonary veins to the peripheral systemic capillaries by reproducing physiological pressure and flow waveforms. The system includes: (i) anatomically shaped, deformable silicone moldings of left heart cavities; and (ii) a silicone model of the aortic root and ascending aorta; and (iii) compliance chamber and resistance valve to control the aortic pulse pressure. The deformable silicone moldings of the left atrium and ventricle are enclosed in a rectangular box filled with an activation fluid. To simulate contractions and relaxations of both left atrium and left ventricle, the moldings are independently compressed or stretched using two piston pumps (ViVitro Inc., Victoria, BC, Canada). This ensures proper atrioventricular synchronization and facilitates the movement of the circulatory fluid within the moldings. The circulating fluid was a mixed saline glycerol solution with a viscosity of 3.8 cP, similar to blood viscosity and was maintained at a constant temperature of 37 °C. The compliance chamber simulates the elastic behavior of the great arteries, whereas the resistance valve enables control of the mean aortic pressure by narrowing the lumen of the pipe, which in turn increases the aortic pulse pressure. Pressures in the left atrium, left ventricle, and the aortic root were recorded by micro-tip pressure catheters (Millar catheter and signal conditioning unit, Millar Instruments, Houston, TX, USA, accuracy ±0.5% maximum full scale). Stroke volume, closure and leakage volumes were measured with an electromagnetic flowmeter (Model 501, Carolina Medical Electronics Inc., East Bend, NC, USA, accuracy ±1% maximum full scale) positioned immediately upstream of the mitral bioprosthesis. Stroke volume and atrial, ventricular and aortic pressures were measured and averaged over 100 cycles (Figure 3). Regurgitant fraction (RF) was calculated as the ratio of regurgitant volume (including both closure and leakage volumes) to stroke volume, expressed as a percentage:
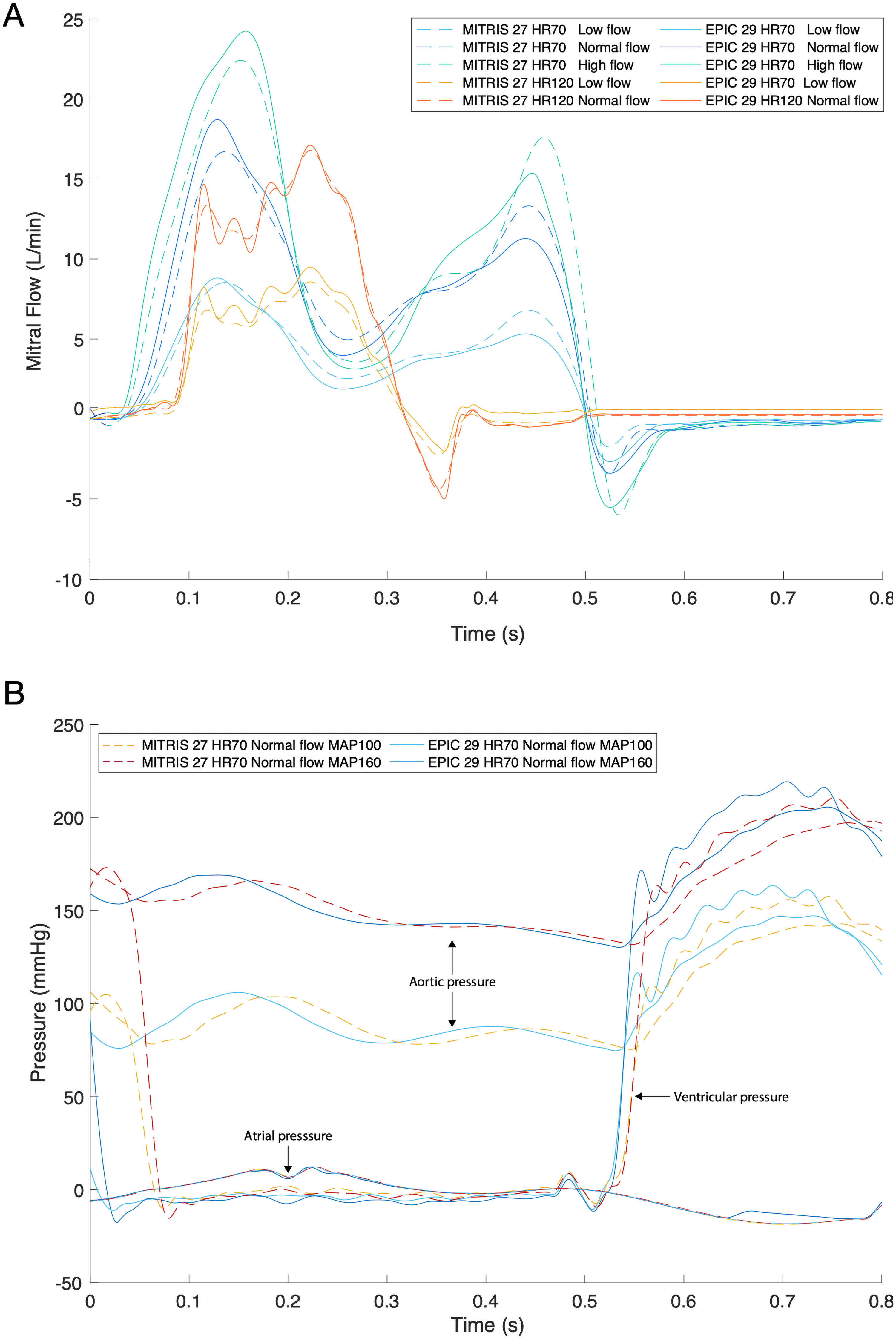
Example of All Mitral Flows for Both MITRIS and EPIC Plus Valves (A) and Different Pressure Curves (Aortic, Ventricular and Atrial) with Normal Heart Rate and Normal Flow for Both MITRIS and EPIC Plus (B). Flow and pressure curves were acquired by flow and pressure sensors and averaged over 100 cycles in custom-coded LabView program. HR = Heart Rate, MAP = Mean Aortic Pressure.
Pump activation and signal acquisition were controlled with LabVIEW8.2 (National Instruments, Austin, TX, USA) to achieve physiological flow as recommended in the ISO standards for heart valve testing in normal flow conditions (ISO 5840-1, ISO 5840-2).
The BPs were tested under several standardized hemodynamic conditions: heart rate: 70 and 120 bpm, mean aortic pressure: 100 and 160 mmHg, stroke volume: 30, 70, and 100 mL (Table 1).
Experimental Hemodynamic Conditions Used for in Vitro Testing.
Hemodynamic Performance Assessment Using Doppler-Echocardiography
Doppler-echocardiographic measurements were performed using the Philips iE33 (Philips, Eindhoven, Netherlands) with a phased array 3.5 MHz probe. The transprosthetic flow velocities, mean mitral transprosthetic pressure gradient (mTPG), and mitral Velocity-Time Integral (VTIMV) were measured ten times (on ten different cycles) per condition by continuous-wave Doppler.
The MV effective orifice area (EOA) was calculated using the continuity equation by dividing the stroke volume measured with the electromagnetic flowmeter by the echocardiographic VTIMV: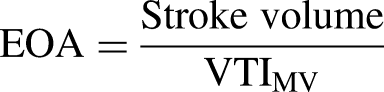
The indexed EOA (EOAi) was calculated as follows: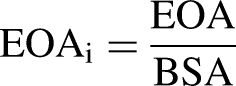
PPM is recognized as a critical factor affecting the long-term efficacy of the BPs implantation. This phenomenon occurs when the EOA of the bioprosthesis is too small relative to a patient's body size. PPM was considered moderate when the EOAi was between 0.9 and 1.2 cm2/m2 and severe if less than 0.9 cm2/m2, assuming a BSA between 1.40 and 2.30 m2 (covering the normal range for BSA in both male and female adults).
The mean mitral flow rate was calculated by dividing the stroke volume by the diastolic filling duration measured on the pulsed-wave Doppler signal of the mitral flow velocity.
Hemodynamic Performance Assessment Using High-Speed Video Camera
The geometric orifice area (GOA) was obtained by acquiring an en-face view of the valve during the cardiac cycle at a frame rate of 1000 images per second, using a high-speed camera FASTCAM SA3 (Photron, Inc., San Diego, CA, USA), equipped with 105 mm lenses (EF 24 Reflex lenses; Sigma Corporation, Kanagawa, Japan). Post-processing of the recorded images and the calculation of the GOA (Figure 4, Supplemental Figure 2) was done using MATLAB (MATLAB R2021b, The MathWorks, Inc., Natick, Massachusetts, United States). GOA was calculated for each BP by taking into account a 5-cycle average of peak GOA for each condition.
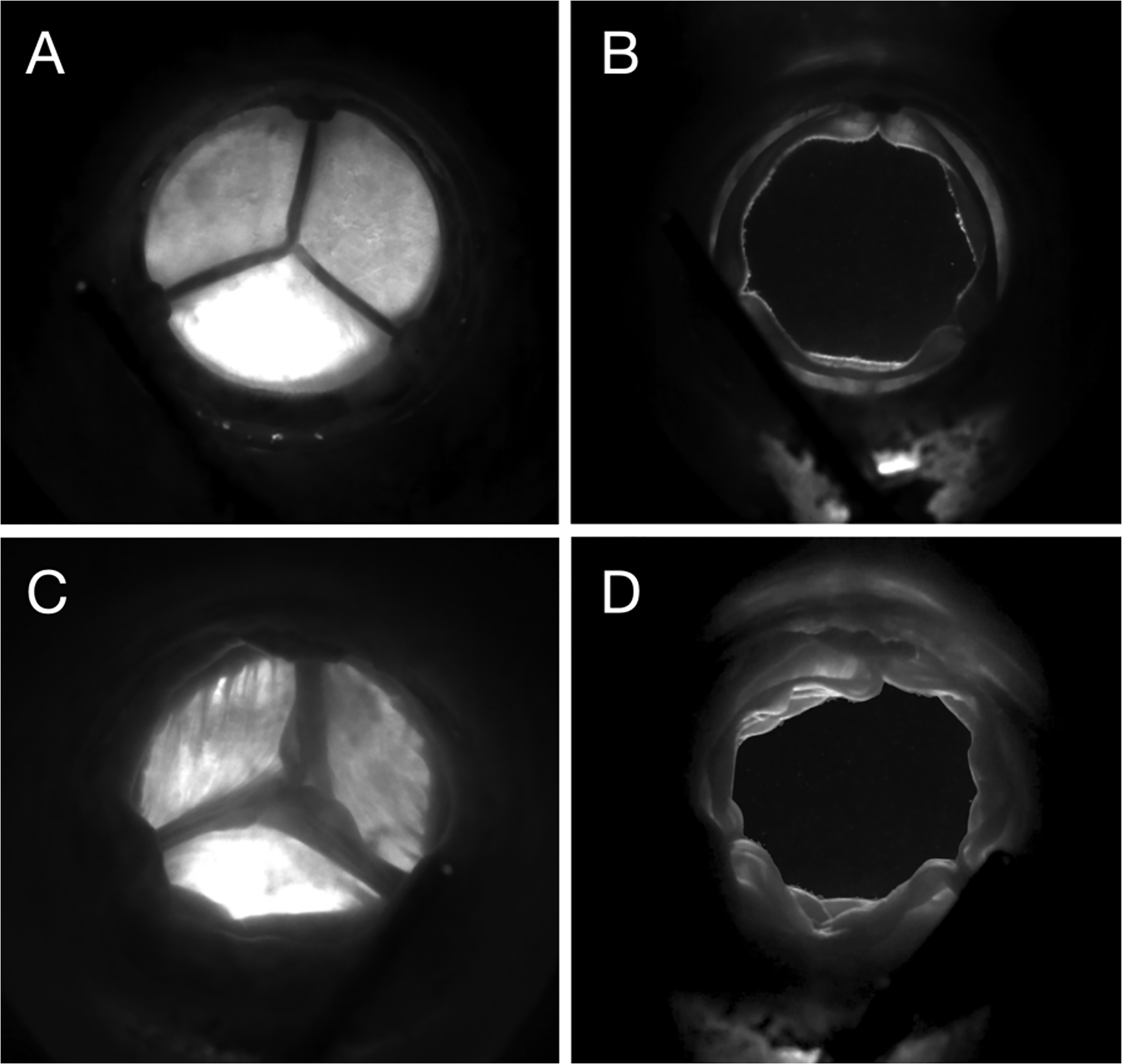
En-face High-speed Video Acquisition Used for GOA Computation. Fully Closed (A) and fully opened (B) MITRIS valve and fully closed (C) and fully opened (D) EPIC plus valve.
The contraction coefficient (Cc) of the valve was defined as the EOA at the vena contracta (the smallest cross-sectional area encountered by flow) divided by the GOA: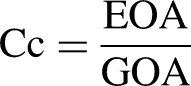
Statistical Analysis
Continuous variables were presented as mean values ± SD. To analyze the effect of valve type on EOA, GOA, Cc, and mTPG while controlling for stroke volume, heart rate, and mean aortic pressure, a multivariate analysis of covariance for repeated measures was used (SPSS; SPSS Inc, Chicago, IL, USA). When significant effects were found, separate one-way ANOVAs were performed for each outcome variable separately. Subsequently, pairwise comparisons between the two valve types within each specific hemodynamic condition were conducted using Student's t-test with Bonferroni correction. Differences were considered statistically significant at P < .05.
Results
Valve Hemodynamic Performance
Tables 2 and 3 show valve hemodynamic performance according to the type (bovine pericardial vs porcine) of BP.
Hemodynamic Performances of Bovine Pericardial and Porcine Mitral Valves at Normal Heart Rate, Normal Pressure.
EOAi, indexed EOA.
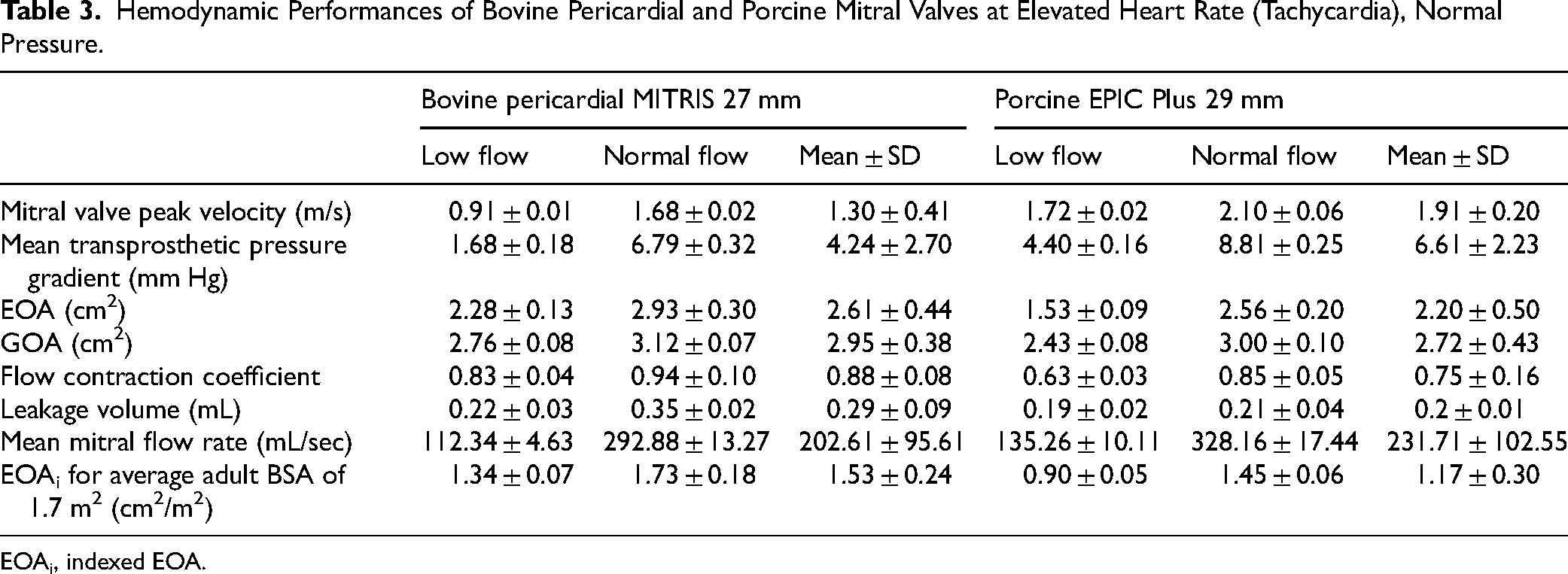
Hemodynamic Performances of Bovine Pericardial and Porcine Mitral Valves at Elevated Heart Rate (Tachycardia), Normal Pressure.
EOAi, indexed EOA.
Overall EOAs were larger, and mTPGs were lower with the bovine pericardial MITRIS versus porcine EPIC Plus BP. No difference in RF was found between the two BP types and all tested BP valves had RF < 5%. Each valve induced a physiological response at normal heart rate with E/A ratio of 1.40. Fusion of E and A waves on mitral inflow Doppler was observed for each valve at high heart rates, as expected.
Mean Mitral Transprosthetic Pressure Gradient
The mTPGs were significantly lower (P < .001) for bovine pericardial versus porcine BP under all hemodynamic conditions except at normal heart rate, low flow condition where they were similar (1.76 ± 0.10 and 1.76 ± 0.09 vs 1.44 ± 0.09 and 1.43 ± 0.11 mm Hg, P > .05 for normal aortic pressure vs hypertension) (Tables 2 and 3, Supplemental Tables 1, 2 and Figure 5). As expected, the mTPGs increased with increasing stroke volume for both bovine pericardial and porcine BPs. The pressure level (hypertension vs normal aortic pressure) did not have a significant effect on mTPGs (Figure 5). A significant increase (P < .001) was observed in the mTPG at higher heart rate compared to normal heart rate (2.1 ± 0.4 and 2.6 ± 1.2 vs 4.2 ± 2.7 and 6.6 ± 2.2 mm Hg for bovine pericardial and porcine BPs).
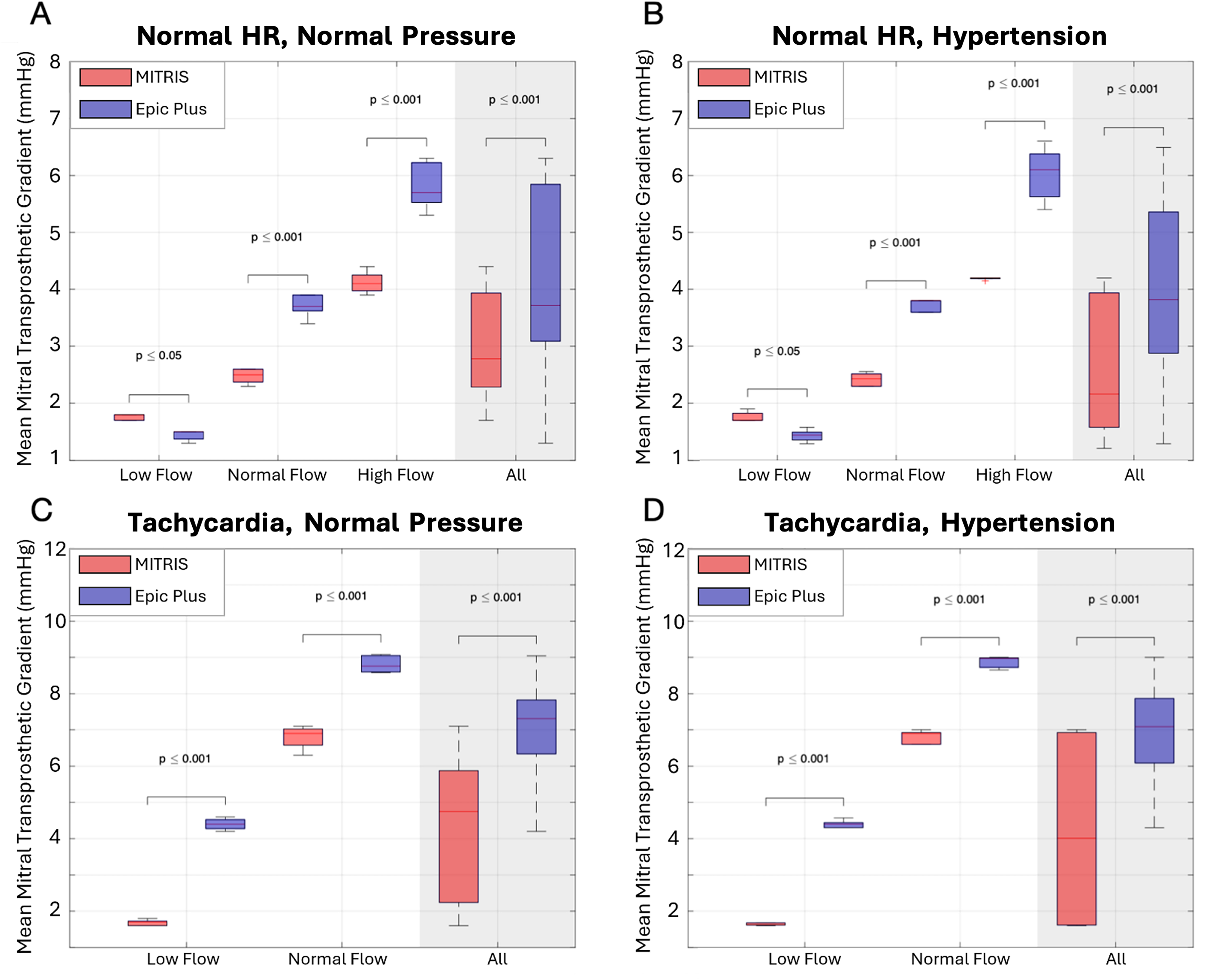
Mean Pressure Gradient in MITRIS (red) Versus EPIC Plus (blue) Valves for all Tested Hemodynamic Conditions. Panel A: Normal heart rate (70 bpm), Normal mean aortic pressure (100 mm Hg), Panel B: Normal heart rate (70 bpm), Hypertension (160 mm Hg), Panel C: Tachycardia (120 bpm), Normal mean aortic pressure (100 mm Hg), Panel D: Tachycardia (120 bpm), Hypertension (160 mm Hg).
Mitral Valve Area
Compared to porcine BPs, the bovine pericardial BPs had significantly higher EOAs (P < .001) (Tables 2 and 3, Supplemental Tables 1, 2 and Figure 6) at different heart rate, flow, and pressure conditions except for the low flow condition at normal heart rate, normal aortic pressure where the EOA was higher for the porcine BP (1.87 ± 0.05 vs 1.75 ± 0.06 for porcine vs bovine pericardial BPs respectively). The EOAs increased with increasing stroke volume and heart rate for both bovine pericardial and porcine BPs. The pressure level did not have a significant effect on EOAs.
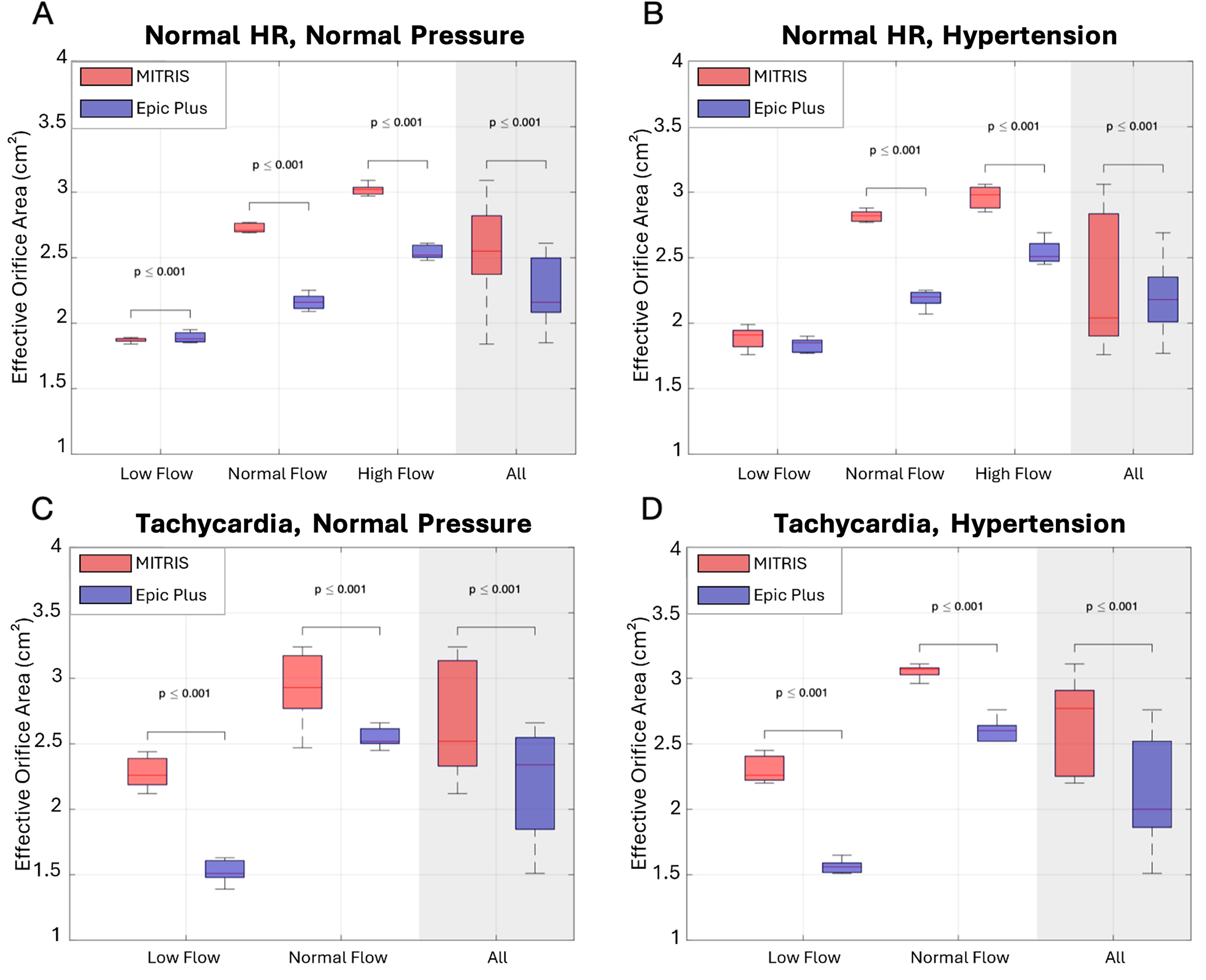
Effective Orifice Area in MITRIS (red) Versus EPIC Plus (blue) Valves for all Tested Hemodynamic Conditions. Panel A: Normal heart rate (70 bpm), Normal mean aortic pressure (100 mm Hg), Panel B: Normal heart rate (70 bpm), Hypertension (160 mm Hg), Panel C: Tachycardia (120 bpm), Normal mean aortic pressure (100 mm Hg), Panel D: Tachycardia (120 bpm), Hypertension (160 mm Hg).
Similar findings were observed for the measured valve GOA, with bovine pericardial BPs demonstrating significantly larger GOAs than porcine BPs (P < .001), and a strong relationship with the stroke volume (P < .001) but no significant relationship with increased heart rate or mean aortic pressure (Tables 2 and 3, Supplemental Tables 1, 2 and Figure 7). Bovine pericardial BPs demonstrated GOAs that were on average 12% larger than porcine BPs across all tested conditions (P < .001).
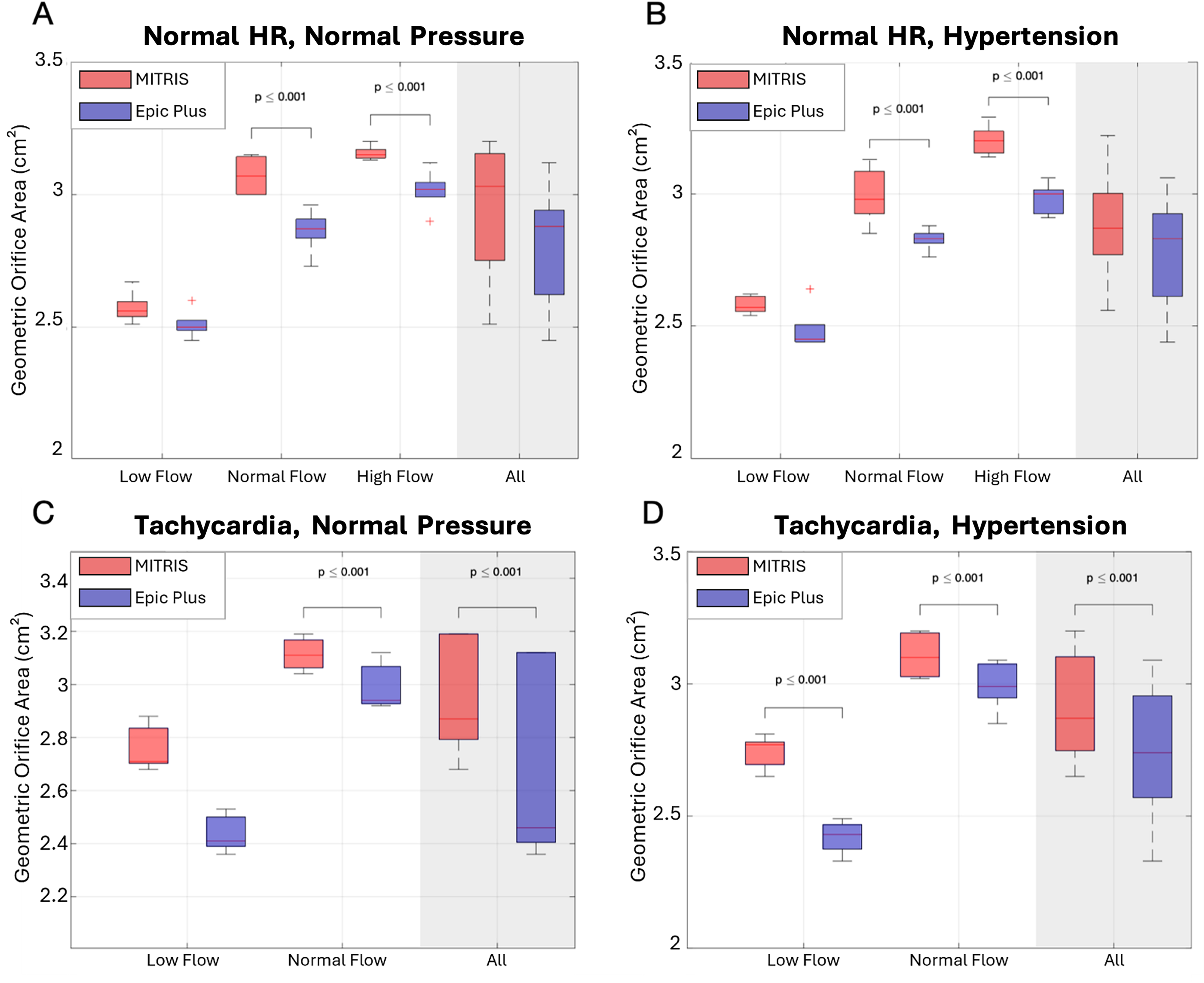
Geometric Orifice Area in MITRIS (red) Versus EPIC Plus (blue) Valves for all Tested Hemodynamic Conditions. Panel A: Normal heart rate (70 bpm), Normal mean aortic pressure (100 mm Hg), Panel B: Normal heart rate (70 bpm), Hypertension (160 mm Hg), Panel C: Tachycardia (120 bpm), Normal mean aortic pressure (100 mm Hg), Panel D: Tachycardia (120 bpm), Hypertension (160 mm Hg).
Flow Contraction Coefficient
The ratio of valve EOA to valve GOA, the so-called flow contraction coefficient, was significantly larger (ie, less flow contraction) for the bovine pericardial BP versus the porcine BP (P < .001). The Cc increased with increasing stroke volume and heart rate (P < .001) for both BP models (Tables 2 and 3 and Supplemental Tables 1, 2).
Prosthesis-Patient Mismatch
EOAi was calculated for a “simulated” BSA between 1.40 and 2.30 m2 (Figure 8). Only normal and high flow conditions with normal heart rates were considered when studying the PPM. The porcine BP presented the first cases of moderate PPM (EOAi < 1.2 cm2/m2) at a BSA of 1.6 m2 and severe PPM (EOAi < 0.9 cm2/m2) at 2.1 m2 while the bovine BP presented the first case of moderate PPM at BSA of 2.2 m2. No cases of severe PPM were observed in the bovine BP.
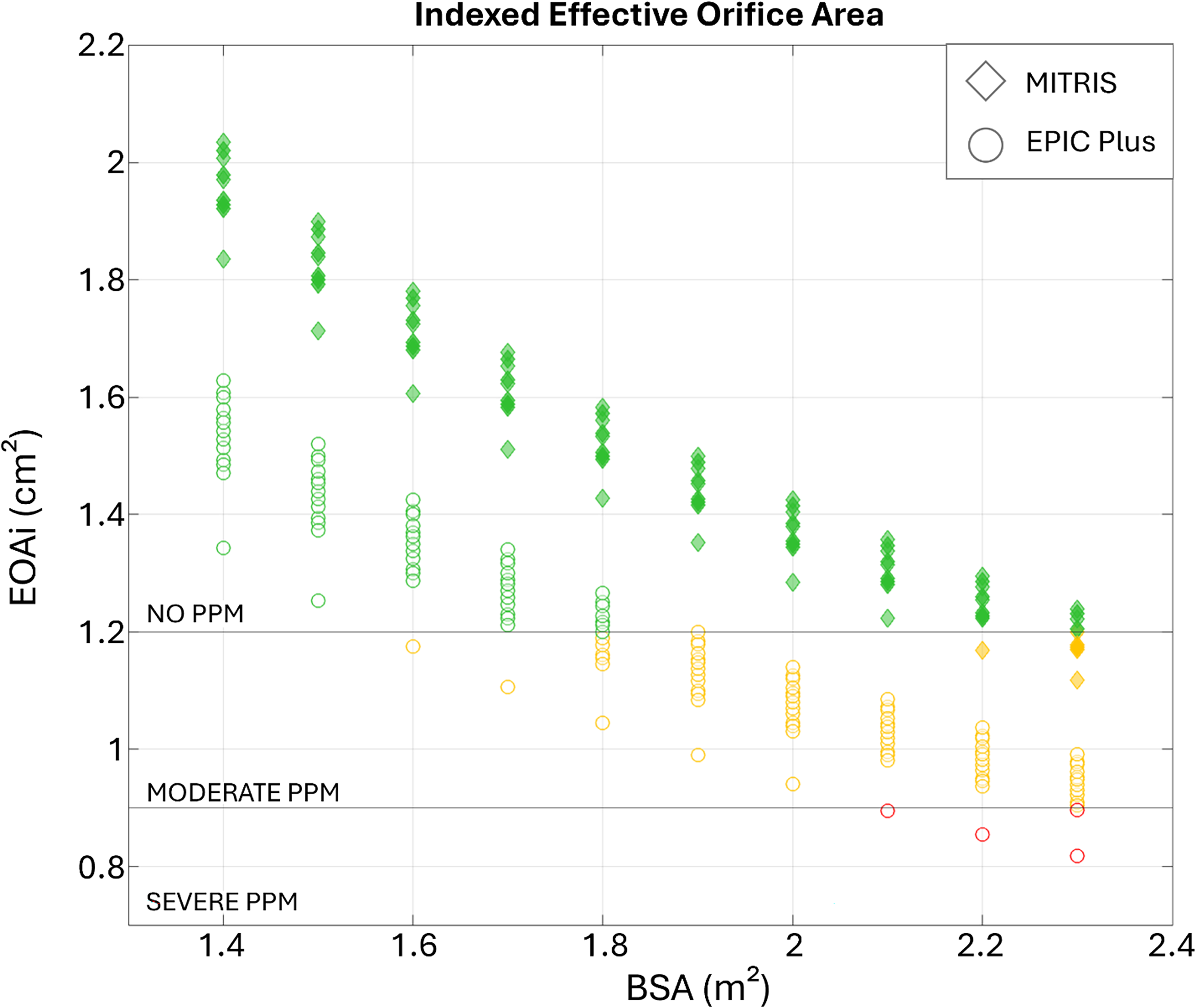
Patient Prosthesis Mismatch in MITRIS Versus EPIC Plus Valves for Normal Heart Rate and Normal or High Flow Conditions. EOAi was calculated assuming a “simulated” body surface area between 1.40 and 2.30 m2.
Assuming the average adult BSA of 1.7 m2, normal and high flow conditions and normal heart rate, for the porcine BP, PPM of moderate severity occurred in 16% of the tested conditions compared to 0% for the bovine BP.
Discussion
The main findings of this study are: (i) at normal heart rate, mTPGs are on average 25% lower for the bovine pericardial versus the porcine BPs; (ii) EOAs are on average 15% higher for the bovine pericardial versus porcine BPs; (iii) under normal and high flow conditions with normal heart rate, for BSA of 1.7 m2, only the porcine BPs presented with PPM (16% of moderate PPM).
Unlike the aortic BPs, there have been very few studies comparing the valve hemodynamic performance of the different types of BP MVs.18,21,22 However, the optimization of BP valve hemodynamic performance is essential in the mitral position to prevent PPM and high residual transprosthetic gradients, which have been shown to have a negative impact on clinical outcomes following MV interventions.23–30 Furthermore, with the aging population and the ongoing development of potential transcatheter MV replacement, BPs are increasingly being considered when establishing meaningful strategies for lifetime management of younger patients.
To our knowledge, this is the first in vitro head-to-head evaluation, under standardized and controlled anatomic and hemodynamic conditions, of two commercially available platforms of surgical mitral BPs with different design. In this study, we observed that the overall hemodynamic performance of the bovine pericardial MITRIS was superior to the porcine EPIC Plus BP. In a pig model, Wang et al., also found that the GOA measured by 2D or 3D echocardiography were larger and mTPG were lower with the bovine pericardial MITRIS versus the porcine BPs (EPIC Plus and MOSAIC). 18 However, the EOA and Doppler velocity index were larger in the MOSAIC versus MITRIS and EPIC Plus. The discrepancies in the GOA versus EOA findings in this previous animal study were likely related to the fact that the stent and posts of the porcine BPs are bulkier than those of the bovine pericardial BP and thus causes a flow acceleration in the LV outflow tract. This phenomenon, in turn, led to an overestimation of LVOT flow velocity-time integral, and thus EOA and Doppler velocity index. In the present study, the results of GOA and EOA were concordant, and both showed a superiority of the bovine BP over the porcine BP. This advantage may be due to the distinct design differences between the tested BP models; porcine BPs have a sloped strut design characterized by a narrowed outflow relative to inflow diameter, whereas bovine pericardial BP utilizes a column configuration that maintains uniform inflow and outflow diameters, thus potentially favoring larger EOAs for the same annular size. The tested bovine pericardial BPs demonstrated also significantly higher flow contraction coefficients compared to the tested porcine BPs, which could reflect their uniform inflow geometry. Since the contraction coefficient is highly dependent on valve inflow shape and aperture angle—with fluid jets continuing to constrict through sharp-edged orifices for a certain length before expanding radially—this parameter ranges from nearly 1.0 in fully open healthy native valves to as low as 0.6 in severely calcified and rigid stenosis. 31
Study Limitations
The present study was limited to two models of mitral BPs: one porcine and one pericardial (one size per model) in three samples each. A total of three porcine and three bovine pericardial BPs were evaluated. The double activation left heart duplicator system was unable to perform the high flow conditions at high heart rate. Furthermore, the displacement of the mitral annulus during a cardiac cycle could not be replicated because the left heart duplicator system requires a rigid mitral holder to create a link between the atrial and ventricular box. Finally, the electromagnetic flowmeter probe was positioned immediately upstream of the mitral BPs, which differs from physiological direct atrial-valve attachment. A minor flow convergence may have occurred, potentially influencing inlet flow conditions and measured hemodynamic parameters. However, identical measurement configurations were maintained for all tested valves, ensuring that any systematic effects would affect both valve types equally and preserving the validity of the comparative findings.
Conclusions
Both bovine pericardial and porcine mitral BPs have good hemodynamic performance. However, the hemodynamic performances of the bovine pericardial MITRIS were overall better than that of porcine EPIC Plus BP, with 25% lower mTPG, 12% larger GOA and 15% larger EOA. Further in vitro studies are recommended to test additional types and sizes of mitral BPs. In vivo studies are needed to determine if these in vitro differences translate into significant differences in long-term valve durability and patients’ outcomes.
Supplemental Material
sj-docx-1-hvs-10.1177_30494826251393909 - Supplemental material for Hemodynamic Comparison of Two Models of Bioprosthetic Mitral Valves Under Standardized In Vitro Conditions
Supplemental material, sj-docx-1-hvs-10.1177_30494826251393909 for Hemodynamic Comparison of Two Models of Bioprosthetic Mitral Valves Under Standardized In Vitro Conditions by Katell Delanoë, Marie-Annick Clavel, Nancy Côté, Philippe Pibarot and Viktória Stanová in Journal of the Heart Valve Society
Supplemental Material
sj-docx-2-hvs-10.1177_30494826251393909 - Supplemental material for Hemodynamic Comparison of Two Models of Bioprosthetic Mitral Valves Under Standardized In Vitro Conditions
Supplemental material, sj-docx-2-hvs-10.1177_30494826251393909 for Hemodynamic Comparison of Two Models of Bioprosthetic Mitral Valves Under Standardized In Vitro Conditions by Katell Delanoë, Marie-Annick Clavel, Nancy Côté, Philippe Pibarot and Viktória Stanová in Journal of the Heart Valve Society
Footnotes
Abbreviations
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Pr. Philippe Pibarot reports research grants from Edwards Lifesciences, Medtronic, and Pi-Cardia. Dr Marie-Annick Clavel reports research grants from Edwards Lifesciences, Medtronic, Novartis, Rednvia, and Pi-Cardia. All other authors have no conflict of interest to disclose.
Supplemental Material
Supplemental material for this article is available online.
Correction (December 2025):
Article has been updated to correct the article type from “Technical Note” to “Original Research Article”.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.