Abstract

Calcific aortic valve disease (CAVD) is now recognised as an active process involving multiple signaling pathways that ultimately lead to fibro-calcic remodeling of the aortic valve leaflets and subsequent obstruction of the blood flow through the valve. 1 While surgical and percutaneous interventions remain effective, they carry the risk of complications and often yield suboptimal long-term outcomes, as there are no medical treatments available. The pathophysiology of CAVD is complex and multifactorial, involving gene modulation, environmental risk factors, and various cellular and molecular mechanisms. 2 Developing a valid in vitro CAVD model is critically important for several scientific, clinical, and translational purposes. Given the pressing need to improve our understanding of CAVD pathogenesis and discover viable therapeutic options, a range of models has been developed in recent years to investigate the underlying mechanisms driving the disease. Decades of extensive research have led to the development of both animal and in vitro cell models, enabling a more comprehensive understanding of the pathophysiological progression and underlying mechanisms of CAVD.
Current Cellular Platforms and Inductive Conditions in Valvular Calcification Studies
In the literature, a variety of cellular sources have been used to study valvular cell calcification. 3 Many in vitro models use human aortic valvular interstitial cells (VICs) isolated from patients undergoing valve replacement or transplant surgeries. However, access to human VICs is limited for many research groups, prompting the development of alternative in vitro models. 4 Some studies use VICs isolated from animal aortic valves (eg, sheep,5,6 porcine,7,8 bovine, 9 rat, 10 and mouse 11 ) while an increasing number of studies utilize immortalized cell lines (eg, immortalized VICs),12,13 human-induced pluripotent stem cell-derived cells, 3 engineered 3D tissue models, and organoids. 14 Emerging approaches incorporate multiple cell types—such as VICs with valvular endothelial cells or immune cells—to study cell–cell interactions.15,16
In all of these experiments, mineralization is typically induced using osteogenic differentiation medium or procalcifying medium. Osteogenic media (OM) generally contains: (a) β-glycerophosphate, which serves as a phosphate source for bone mineralization and promotes osteogenic gene expression through extracellular kinase phosphorylation 17 ; (b) ascorbic acid, which enhances type I collagen secretion 18 ; and (c) dexamethasone, which induces runt-related transcription factor 2 (RUNX2) expression. Procalcifying media, on the other hand, induce inflammation and mineralization by adding NaH2PO4 and ascorbic acid. 19 The exact mechanisms of these media and their effects on calcification capacity, cell proliferation, and viability remain poorly characterized.
Critical Evaluation of Cell Type and Media in in Vitro Calcification
This study by Daria Evensen et al 20 provides timely and valuable insight by comparing the characteristics, gene expression profiles, and calcific and differentiation potentials of 2 cell types: human VICs and mesenchymal stem cells (MSCs). The differentiation potentials of human VICs and MSCs, along with the expression profile of osteogenic markers, were assessed using the following media: 3 designed to induce osteoblastic differentiation and maturation of bone-derived MSCs, 2 designed for dental-derived MSCs, and 1 designed for adipose tissue-derived MSCs. The authors conclude that “there were major differences between the media tested and MSCs and VICs in response to induced calcification. MSCs are inferior for studying aortic valve calcification in vitro.”
This work represents one of the most comprehensive head-to-head comparisons of osteogenic conditions used to model valvular calcification. Its findings are technically informative and biologically meaningful, demonstrating that VICs display substantial variability in their response to osteogenic stimuli. Notably, 2 of the 6 media were found to be toxic to VICs by inducing calcification through likely passive, noncellular chemical mechanisms rather than biologically mediated differentiation. This distinction is important as it underscores the need for rigorous validation of in vitro calcification models before drawing mechanistic conclusions.
A key strength of this study lies in its comparative approach, extending beyond VICs to include MSCs. The findings are unequivocal: MSCs differ significantly from VICs in both their calcification capacities and gene expression patterns. While MSCs maintained morphological stability and survival, their capacity to replicate the osteogenic behavior of VICs was limited and inconsistent. This challenges the assumption that MSCs can serve as surrogates for VICs and reinforces the importance of using disease-relevant primary cell types when studying CAVD. As stated by the authors, the plasticity of MSCs and the eventual differentiation into fibroblasts or VICs-like were not investigated. MSCs are multipotent cells with the ability to differentiate into many lineages (eg, osteoblasts, chondrocytes, and fibroblasts). If the MSCs were not first guided toward a VIC-like or fibroblast-like phenotype, such as in this study, their response to osteogenic induction may not be physiologically comparable to VICs. This limits the validity of the comparison and represents a missed opportunity to more accurately model cellular transitions involved in CAVD.
Rethinking Markers: Beyond RUNX2 in Valvular Calcification
The study also raises concerns about the reliability of traditional gene expression markers such as RUNX2 in assessing in vitro calcification. The authors compellingly show that significant calcium deposition can occur without concomitant upregulation of classical osteogenic genes, particularly in VICs. This disconnect between calcification and transcriptional signatures calls for a reassessment of the biomarkers used to define osteodifferentiation in valve cells and emphasizes the need for functional endpoints, such as mineralization assays, to complement molecular analyses. For a thorough analysis of calcification, a combination of markers of early to mid-stage (eg, alkaline phosphatase and osterix), late-stage markers (eg, osteocalcin, osteopontin, bone sialoprotein, and BMP2), and mineral deposition assays (eg, OsteoSense staining and Annexin V) is recommended to ensure a comprehensive and biologically meaningful evaluation.
Maintaining VIC Identity: The Role of Cell Passage
The differentiation capacity and calcification behavior of VICs are also influenced by passage number, making it a critical parameter for consistency in in vitro CAVD models. 21 VICs should ideally be used for osteogenic induction at early passages, typically between passages 2 and 5, to balance the need for cell expansion with the preservation of their native phenotype and responsiveness. In the study by Daria Evensen et al, 20 VICs and MSCs were used from passages 3 to 5. At later passages (>P5), VICs can undergo phenotypic drift toward a myofibroblast-like or senescent state, which alters their response to osteogenic stimulation.
Accounting for Sex and Complexity in CAVD Modeling
Despite its strengths, the study has several limitations, thoughtfully acknowledged by the authors. The exclusive use of male-derived VICs limits the generalizability of the findings. A growing body of evidence shows that CAVD is a sexually dimorphic disease, differing significantly between men and women in terms of the diagnosis, presentation, progression, treatment, and underlying mechanisms leading to the development of the disease.22–26 For the same severity of the disease, men tend to develop more calcification, whereas women are more prone to fibrotic remodeling of the aortic valve.27,28 Different cellular and molecular pathways are activated in each sex, highlighting the importance of conducting experiments that present and interpret observations in both sexes separately and equally. Moreover, although VICs are more pathophysiologically relevant than MSCs, their culture in 2D monolayers inevitably lacks the mechanical and spatial complexity of the disease. The absence of protein-level validation is another limitation, although the study's gene expression and morphological assessments are thorough.
Toward Standardized Protocols in CAVD Research
Importantly, this work highlights a major unmet need in the field: the standardization and validation of in vitro models of valve calcification (Figure 1). This includes standardized protocols for cell isolation, culture conditions, OM composition, and analytical techniques. Without such standardization, cross-study comparisons remain difficult, and the translational relevance of in vitro findings is compromised. By identifying an OM composition as the most reliable condition for inducing VICs calcification, this study provides a practical foundation for future investigations aiming to elucidate the cellular mechanisms and identify pharmacological targets in CAVD.
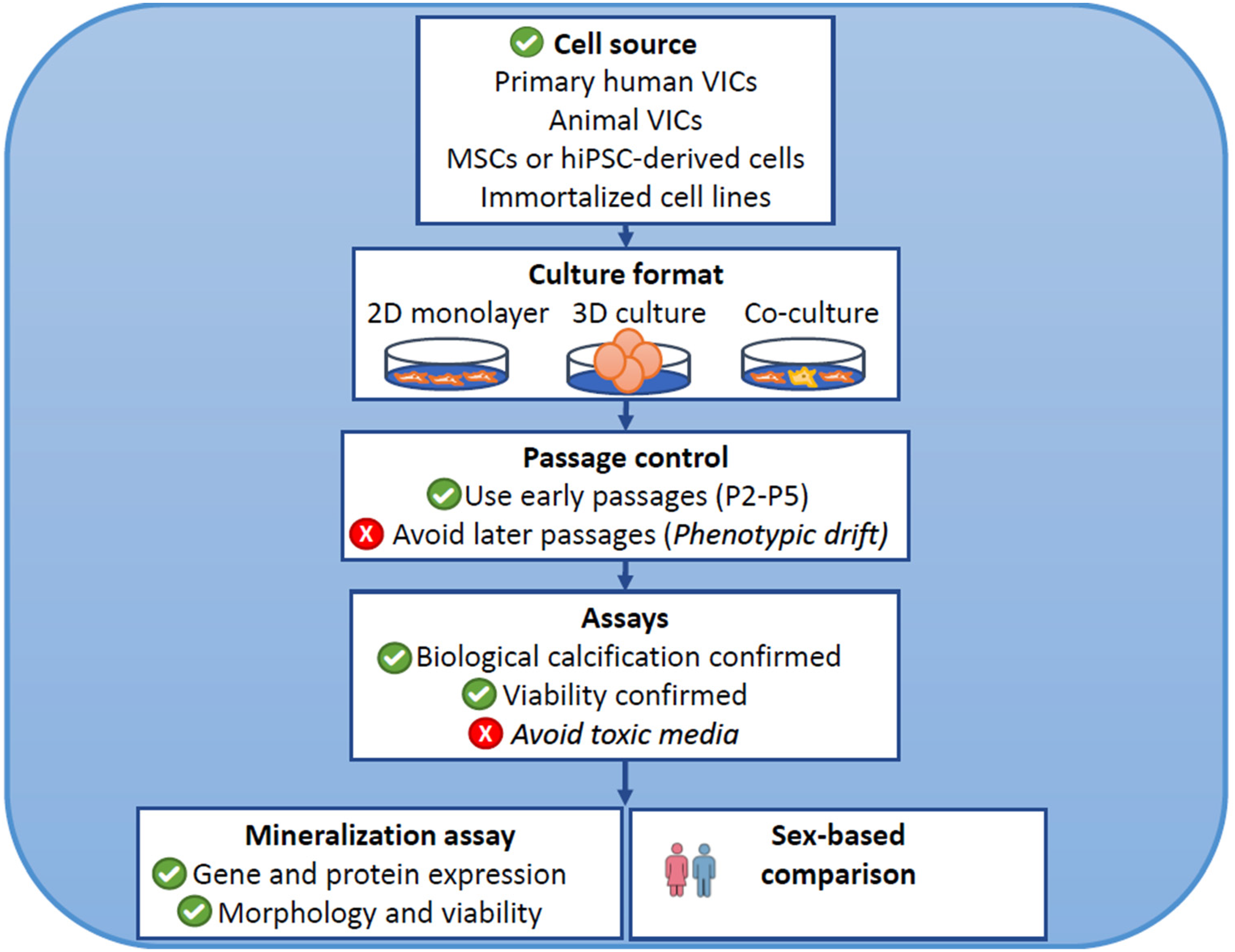
Key Considerations for in Vitro Models of Aortic Valve Calcification.
Conclusion: Refining the Tools to Understand CAVD
In conclusion, Daria Evensen et al 20 present a meticulous and insightful study that advances our understanding of how experimental conditions influence calcification modeling in vitro. These findings have direct implications for experimental design, model selection, and biomarker interpretation in valve research. As the field moves toward more sophisticated models of CAVD-including 3D cultures and sex-specific analyses, this study serves as a valuable reference point and contributes to the broader effort to uncover disease, driving mechanisms, and develop novel therapies for this unmet medical need.
Footnotes
Abbreviations
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: PP is funded by the Canada Research Chair in Valvular Heart Diseases, Canadian Institutes of Health Research (No. FDN-143225, Ottawa, Ontario, Canada). ML and AD are funded by FRQS—Health Data Hub. AD is also funded by the INRIA International Research Chair.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PP holds the Canada Research Chair in Valvular Heart Diseases, Canadian Institutes of Health Research, and has received funding from Edwards Lifesciences and Medtronic for echocardiography CoreLab analyses with no personal compensation. The remaining authors have nothing to disclose.
