Abstract
This is a visual representation of the abstract.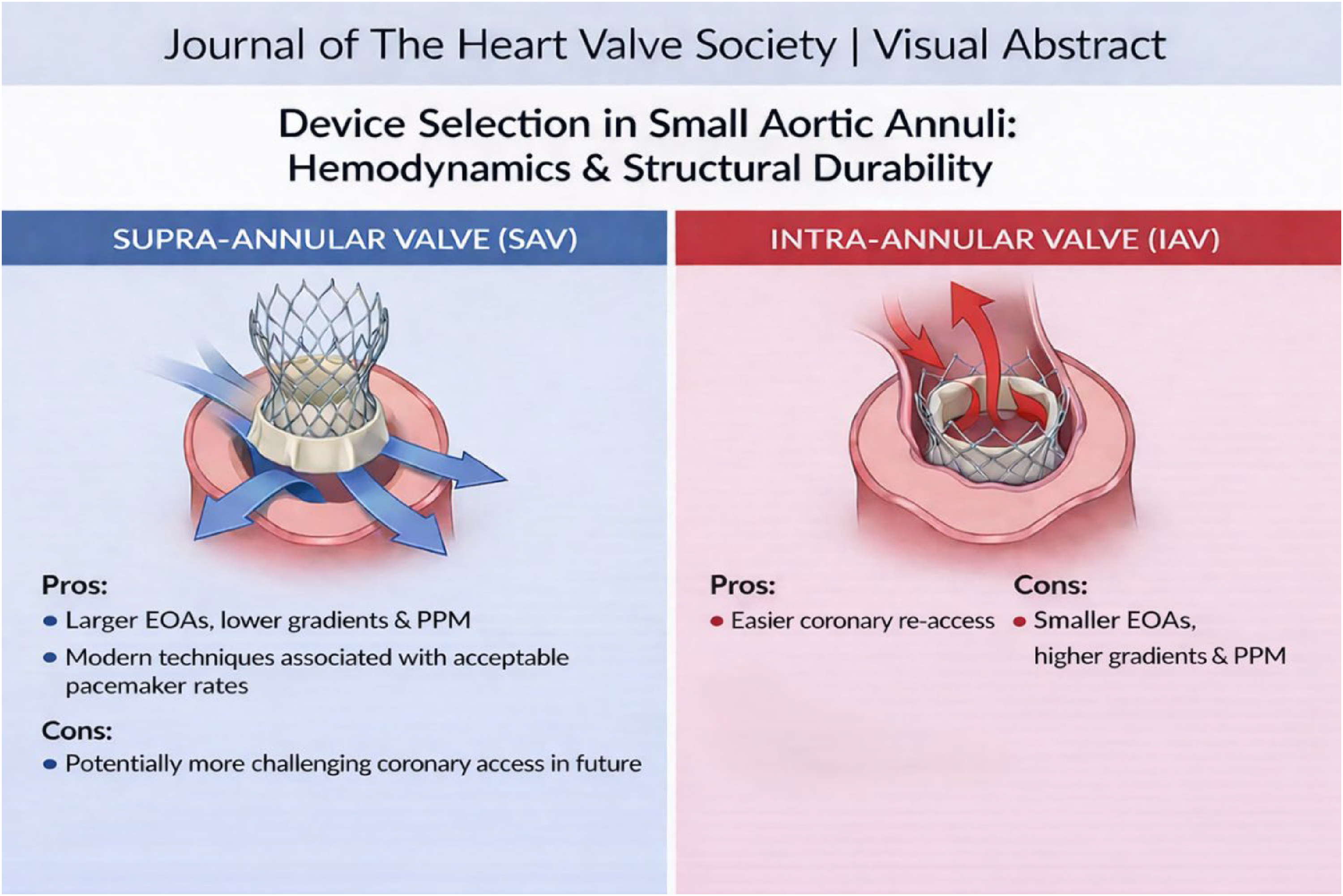
Transcatheter aortic valve implantation (TAVI) has become the standard treatment for patients with severe aortic valve stenosis across the operative spectrum, with a class 1A indication for those ≥70 years of age in European guidelines and ≥65 years in North American guidelines.1,2 The evidence supporting these guidelines stems from randomized data demonstrating that TAVI in low surgical risk patients is non-inferior to surgical aortic valve replacement (SAVR), with comparable performance and durability of transcatheter and surgical valves at 5 years in the Evolut Low Risk trial, 3 7 years in the PARTNER 3 trial, 4 and 10 years in the NOTION trial. 5 As TAVI expands to younger and healthier patients with longer life expectancies, understanding the long-term performance and durability of different transcatheter valve platforms becomes crucial, especially in patients with small aortic annuli, where outcomes may be less favorable. 6 These patients are at increased risk for higher gradients, prosthesis-patient mismatch (PPM), reduced exercise capacity, and increased mortality during follow-up.7–10
Important 10-year follow-up data from the NOTION trial, which randomized low-risk patients between TAVI using supra-annular valves and SAVR, demonstrated sustained lower valve gradients and better effective orifice areas (EOAs) for TAVI valves compared to surgical valves. 5 These findings support the notion that supra-annular transcatheter valve designs offer superior long-term hemodynamics over intra-annular surgical valves. In patients with a small aortic annulus undergoing TAVI, the SMART trial demonstrated superior hemodynamics with the self-expanding supra-annular valve as compared to the balloon-expandable intra-annular valve, although long-term data are not yet available. 11
Whether intra-annular valves mounted in a self-expanding frame come with the concession of less favorable hemodynamics compared to supra-annular self-expanding technology has remained an open question.
The findings of Pompeu Sà et al., 12 now presented in the Journal, expand our understanding of this topic by providing a comprehensive overview of the differences in hemodynamic performance and clinical outcomes between supra- versus intra-annular self-expanding transcatheter platforms in the context of small aortic annuli. In their systematic review and meta-analysis of 5 observational studies, they compared the supra-annular self-expanding valve (SAV), which included 2295 CoreValve/Evolut R/PRO/PRO+/FX devices, with the intra-annular SAV (IAV), comprising 1008 Portico/Navitor devices. The authors observed superior hemodynamic performance in the SAV group, characterized by larger EOAs, higher indexed EOAs, lower mean gradients, and lower rates of PPM compared to the IAV group. Early clinical outcomes were similar, except for a higher pacemaker rate in the IAV group, whereas long-term survival favored the SAV group (at 5-year follow-up: hazard ratio 0.770; 95% CI 0.600–0.980; P = 0.034). Despite the inherent limitations related to the inclusion of non-randomized studies in this meta-analysis, the findings suggest an important “hemodynamic imperative” for patients with small annuli where SAV appears to offer better hemodynamics compared to IAV. This may be explained by SAVs featuring a low-profile nitinol frame and sealing fabric positioned within the annulus, with the leaflets functioning above the annular level, whereas IAVs have the frame, leaflets, and sealing fabric contained within the annulus, potentially limiting both geometric and EOAs. The less material placed within a small aortic annulus, the lower the expected gradients and risk of PPM.13,14
However, it cannot be concluded that the favorable hemodynamic characteristics of SAV also account for the observed survival benefit in this group. First, IAV delivery systems are known for their (best-in-class) small profile and hydrophilic coating, making them an excellent choice for patients with challenging vascular access, such as those with severe peripheral arterial disease who often suffer severe comorbidities, shortening their life expectancy. 15 Therefore, it cannot be ruled out that IAV may have been used in a sicker patient subset as compared to those treated with SAV. Second, the hemodynamic assessment was based solely on echocardiography-derived gradient measurements, which are known to overestimate invasive and true gradients, especially in small-sized intra-annular balloon expandable valves. 16 Hatoum et al recently demonstrated that structural differences in supra- versus intra-annular leaflet positioning and tall versus short stent frame design affect blood flow turbulence, pressure recovery efficiency, and net transvalvular gradients.17,18 The extent to which echocardiography overestimates gradients following implantation of a self-expanding intra-annular valve relative to other valve designs remains uninvestigated.
Interestingly, Pompeu Sà et al challenge the historical trend of self-expanding SAVs causing increased pacemaker rates. With modern implantation techniques (eg, cusp overlap and higher deployment), SAVs in this study were associated with acceptable pacemaker rates that were significantly lower compared to the intra-annular group. Despite the latter, however, important considerations for use of IAV, such as Portico or Navitor has traditionally rested on the device's anatomical friendliness. The large open-cell design and intra-annular leaflet mounting offer easier coronary re-access even after future valve-in-valve TAVI as compared to SAV designs. 13
These are crucial considerations for younger patients with longer life expectancies who may require future valve or coronary re-interventions.
In summary, while current data seem to favor the superior hemodynamics of supra-annular valves in patients with small aortic annuli, the balance between procedural safety, device performance, long-term durability, and future (coronary and valve) reintervention needs remains critical, highlighting the importance of personalized approaches and further research to optimize outcomes for all patients undergoing TAVI.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Rutger-Jan Nuis received research grant support from Vifor Pharma and Merill, and consulting fees from Edwards Lifesciences, Abbott, and Boston Scientific.
