Abstract
Background
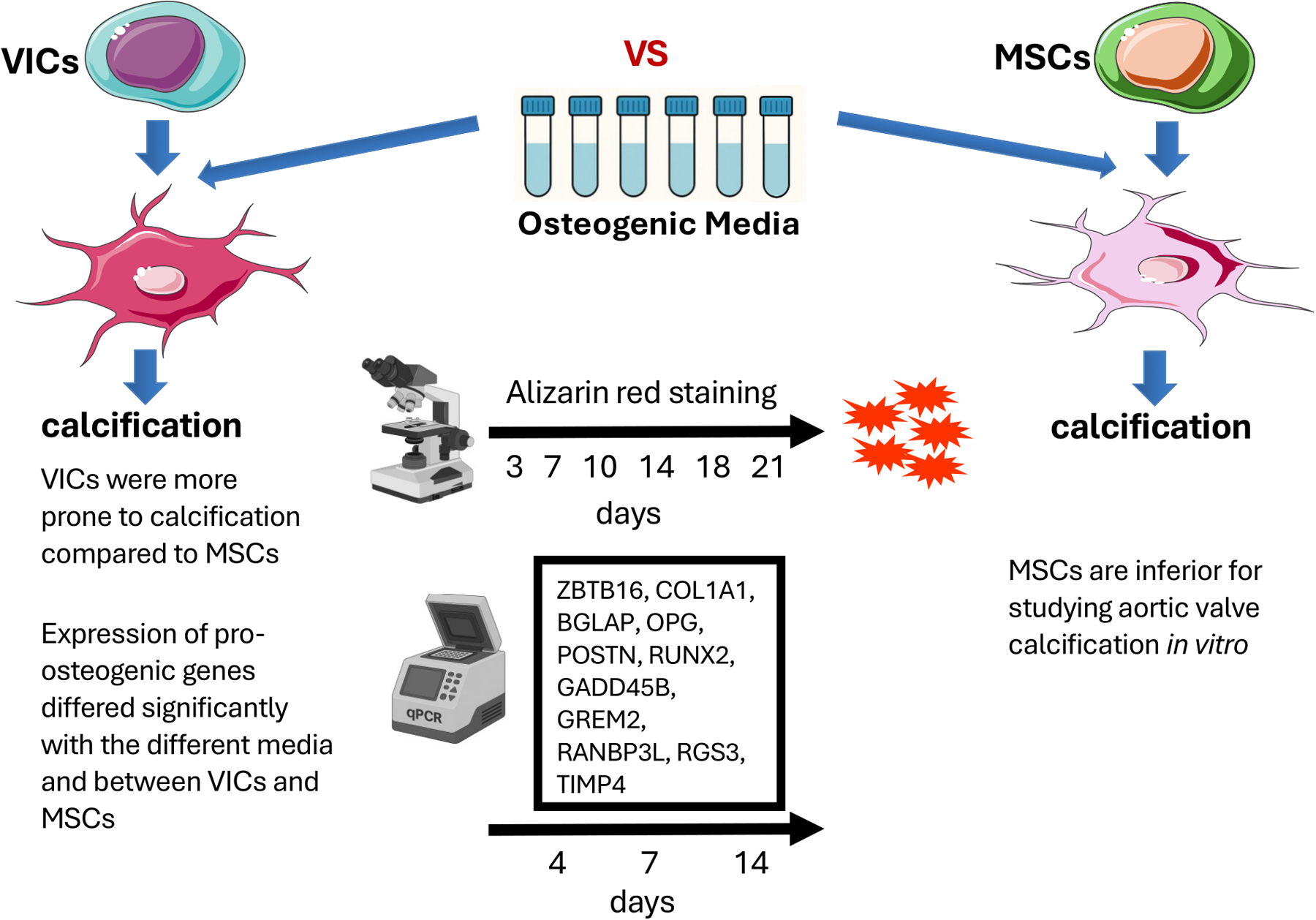
Experimentally induced calcification in primary human aortic valve interstitial cells (VICs) is a good in vitro model for studying calcific aortic valve disease. Mesenchymal stem cells (MSCs) may be used as a substitute for VICs. The aims of the present study were: 1. To compare six commonly used osteogenic media for inducing calcification in human VICs; 2. To assess any difference in calcification between VICs and MSCs caused by the different media; 3. To study the expression of pro-osteogenic genes in both cell types within the same experimental framework.
Methods
Cultures of VICs (n = 6) and MSCs (n = 5) were exposed to six different osteogenic media. Calcification was measured spectrophotometrically after Alizarin Red staining on the 3rd, 7th, 10th, 14th, 18th, and 21st day. Expression of pro-calcification genes was determined by qPCR on the 4th, 7th, and 14th day.
Results
VICs were more prone to calcification compared to MSCs. Two of the media were toxic for VICs. MSCs maintained their morphology and functionality in all osteogenic media. Expression of pro-osteogenic genes differed significantly with the different media and between VICs and MSCs.
Conclusion
There were major differences between the media tested and between MSCs and VICs in response to induced calcification. MSCs are inferior for studying aortic valve calcification in vitro.
Keywords
Key Points
The study highlights significant differences in calcification responses between aortic valve interstitial cells (VICs) and mesenchymal stem cells (MSCs). VICs are more sensitive and prone to calcification.
Different osteogenic media had highly different effects on calcification in cultured aortic valve interstitial cells (VICs) and mesenchymal stem cells (MSCs). Some osteogenic media were toxic for VICs, but not for MSCs. This emphasizes the importance of carefully selecting osteogenic media for studies involving VICs.
The expression of pro-osteogenic genes differed both between the two cell types and with the specific osteogenic media used, suggesting that monitoring gene expression alone may not reliably indicate the occurrence of osteogenic differentiation.
Introduction
Aortic valve stenosis, caused by calcific aortic valve disease (CAVD), is the most prevalent heart valve disorder globally. If left untreated, symptomatic patients have a 2-year mortality rate exceeding 50%,1,2 and its prevalence increases with age. 3 Progression of CAVD follows a continuum from early, subtle changes to a life-threatening condition. The process begins with aortic valve sclerosis, characterized by the first detectable macroscopic alterations in the valve leaflets, while valve function remains normal. For many years, CAVD was believed to be a passive, age-related process in which the valve simply degenerates over time due to calcium accumulation. Instead, it is now understood to be an active molecular and cellular process.4–6 The only available treatments are Surgical Aortic Valve Replacement (SAVR) or Transcatheter Aortic Valve Replacement (TAVR). 7 Understanding the mechanisms driving this disease is critical for developing pharmaceutical and non-surgical treatment options.
Aortic valve interstitial cells (VICs) are considered to play a key role in the pathogenesis of CAVD. 8 A variety of biochemical and biomechanical stimuli, including mechanical stress and inflammation, may cause VICs to differentiate into osteoblast-like cells and calcify.7,8 The exact cellular and molecular mechanisms causing osteogenic differentiation and calcification are not fully elucidated. The calcification process occurring in CAVD has some similarities to bone formation. 8
There is no good animal model that mimics the development of aortic stenosis in humans. Experimentally induced calcification in cultured human VICs is considered to be a good and probably the best in vitro model of CAVD.8,9 VICs are the predominant cell type in valve leaflets and have a mesenchymal origin. In healthy valves, they maintain a quiescent phenotype. During CAVD, VICs differentiate into myofibroblast-like or osteoblast-like cells, leading to fibrosis and calcification. The differentiation of VICs into osteoblast-like cells is a key event in the calcification process. These osteoblast-like VICs express bone matrix proteins and promote the deposition of calcium and the formation of bone-like nodules within the valve leaflets. The osteogenic differentiation of VICs is regulated by various signaling pathways and factors, including bone morphogenetic proteins (BMPs), Wnt/β-catenin, oxidized lipids, inflammatory cytokines, and mechanical forces.8,10–13
VICs from human aortic valves may not be available to all research groups. Potentially, mesenchymal stem cells (MSCs) could be an alternative to VICs to study the basic biological processes causing CAVD. Multiple in vitro experiments have demonstrated the capacity of MSCs to differentiate into osteoblast-like cells, followed by calcification.14–16 Because MSCs have high proliferative and differentiation potentials, they have some advantages compared to primary heart valve cells such as VICs. MSCs derived from the bone marrow are widely used to study various differentiation mechanisms. 17 In fact, most studies in this field have used MSCs. 18 Comparing calcification properties of VICs and MSCs under different osteoinductive stimuli may improve our understanding of the differences and similarities of these cell types. It might also provide insights into the mechanisms of soft tissue calcification in general.
Several osteogenic media with different chemical compositions have been used to induce osteodifferentiation and calcification in cell cultures.17–24 Most of these media were initially developed for use in MSCs. It is unknown if one medium is superior to another. They may induce calcification at different speeds and involve different genes and signaling pathways. We hypothesize that there are important differences between the different media to induce calcification in cultures of VICs. Furthermore, the calcification process may differ in MSCs and VICs. The aims of present investigation were: 1. To compare six commonly used osteogenic media for inducing calcification in human VICs; 2. To assess any difference in calcification between VICs and MSCs caused by the different media; 3. To study the expression of pro-osteogenic genes in both cell types within the same experimental framework.
The various osteogenic media were applied to VICs and MSCs with observations over a period of 21 days. The purpose was not to investigate the role of different compounds of the media or their concentrations, but only the known media per se.
Materials and Methods
Ethical Considerations
VICs were obtained from trileaflet aortic valves with serious calcified stenosis (n = 6) harvested during aortic valve replacement at the Department of Cardiothoracic Surgery, Oslo University Hospital, Oslo, Norway. All aortic valves were from male donors with an age range from 65 to 75 years old. No specific genetic disorders or diabetes were reported for the chosen aortic valves’ donors. The study was performed in line with the principles of the Declaration of Helsinki. Written informed consent was obtained from all patients. The project was approved by the Regional Ethics Committee Southeast Norway (REK approval no. 2015/1003 and no. 171397).
Human MSCs were obtained by bone marrow aspirates from healthy volunteers (n = 5) after ethical approvals were received from the Regional Committee for Medical and Health Research Ethics Southeast Norway (REK approval no. S-07043a). All data from the volunteers providing MSCs are anonymous.
Isolation and Cultivation of Primary Human Cells
VICs were isolated from the aortic valves as previously described.25,26 Briefly, right after excision during surgery aortic valve leaflets were placed in ice-cold phosphate-buffered saline (PBS, Gibco, Thermo Fisher Scientific, USA) and transported to the lab within 12 h. Then, the whole excised aortic valve leaflets, including both calcified and fibrotic areas, were subjected to digestion with 2 mg/mL collagenase II (Worthington Biochemical Corporation, USA) for 12–18 h at 37 °C. The cell suspension was homogenized by pipetting and centrifuged at 300g for 5 min. The supernatant was discarded, and the pellet was washed and spun down with PBS. Those parts of the valve that calcified into bone-like tissue mostly stay intact after collagenase treatment. Some small hydroxyapatite particles may get into the cultures when the cells are transferred from digested valve tissue. However, large particles are washed away during PBS washing and media change. Small particles, however, remain on the culture plastic during passaging of the cells. After the first passage most remaining particles are eliminated from cell cultures.
VICs were cultured for 3–5 passages in standard growth medium (DMEM (41966-052, Gibco, Thermo Fisher Scientific, USA) supplemented with 15% FBS (10% FBS was used in osteoinductive media) (HyClone, SH30070.03, Cytiva, USA) and 50 µg/mL of gentamicin (15750-037, Gibco, Thermo Fisher Scientific, USA) at 37 °C in 5% CO2 until confluence of 90% before passaging.
Primary human MSCs from bone marrow were prepared according to modifications of a previously described protocol. 27 In brief, a total of 30 to 60 mL of bone marrow was aspirated from the iliac crest under local anesthesia. The cells were characterized and assessed after isolation using different bone marrow processing conditions and expansion culture conditions. Phenotypical characterization as well as differentiation capability of MSCs is described in detail previously. 28 MSCs were cultured for 3–5 passages. MSCs were maintained in tissue culture grade uncoated Petri dishes (Corning Incorporated, Corning, USA) in alpha-MEM medium (11900016, Gibco, Thermo Fisher Scientific, USA), supplemented with 10% FBS (HyClone, SH30070.03, Cytiva, USA), 50 µg/mL of gentamicin (15750–037, Gibco, Thermo Fisher Scientific, USA) at 37 °C and 5% CO2 until confluence of 80–90% before passaging.
Based on considerable experience working with cells, all cell cultures looked normal with their characteristic morphology. Passaging was performed based on cell density and the knowledge of the optimal density for either VICs (approx. 90–95% confluence) or MSCs (approx. 80–85% confluence) cultures.
Throughout the experiments, both MSCs and VICs were examined microscopically every other day to assess any morphological alterations of the cells.
Osteogenic Induction
VICs or MSCs were seeded in 96- and 24-well plates, respectively. Cell density was 9×103 cells per cm2 (for VICs) or 5×103 cells per cm2 (for MSCs). After 24 h, one of the osteogenic media shown in Table 1 was replaced with the cultivation medium. The confluence of the cells was 90% when differentiation started. Control cultures (untreated) did not receive osteogenic medium but remained cultivated in standard growth media. The culture medium was changed twice a week. Each osteogenic medium was freshly prepared for every medium exchange step.
Composition of Osteogenic Media.

Alizarin Red Staining and Quantification of Calcium
Alizarin Red staining was performed to quantify calcification 29 on the 3rd, 7th, 10th, 14th, 18th, and 21st day after osteogenic differentiation induction (VICs n = 6; MSCs n = 5). The cell medium was removed; the cells were washed with PBS and fixed with 70% ethanol for one hour at room temperature. Then the cells were washed with Milli-Q water and stained with Alizarin Red (A5533, Sigma-Aldrich, USA) according to the manufacturer's instructions. Then the Alizarin Red staining was extracted and measured spectrophotometrically. Briefly, for 24-well plates, 200 μL 10% acetic acid was added to each well and incubated for 30 min at room temperature with gentle agitation. The cells were detached with a scraper, and the suspension was transferred to a 1.5 mL microcentrifuge tube and vortexed vigorously for 30 s. The cells were then heated to 85 °C for 10 min before being chilled in ice for 5 min followed by centrifugation (15000g) for 15 min. The supernatant was transferred to a new 1.5 mL microcentrifuge tube, 75 μL of 1 M NaOH was added to each tube to achieve a pH between 4.1 and 4.5. Finally, 50 μL from the tube was transferred to a clear bottom 96-well plate and the absorbance at 405 nm was measured on a plate reader (Molecular Devices, Emax, USA).
PCR Real Time
Total RNA from the cultured cells (VICs n = 6; MSCs n = 3) was isolated using Trizol Reagent (15596026, Thermo Fisher Scientific, USA). RNA (1 μg) was reverse transcribed with an MMLV RT kit (Quantabio, USA). Real-time PCR was performed with 1 μL cDNA and SYBRGreen PCR Mastermix (Applied Biosystems, USA) in the Light Cycler system using specific forward and reverse primers for the target genes shown in Table 2. ZBTB16, COL1A1, BGLAP, OPG, POSTN, RUNX2, GADD45B, GREM2, RANBP3L, RGS3, TIMP4). RUNX2, COL1A1, OPG, POSTN, and BGLAP were considered well-known, from the literature, as the most relevant markers of osteogenic processes. ZBTB16, GADD45B, GREM2, RANBP3L, RGS3, and TIMP4 are additionally studied osteogenic genes. These genes may also be involved in osteogenic differentiation of VICs. 19 The thermocycling conditions were as follows: 95 °C for 5 min, followed by 40 cycles at 95 °C for 15 s and 60 °C for 1 min. A final heating step of 65 °C to 95 °C was performed to obtain the melting curves of the final PCR products. Changes in the expression of the target genes were calculated as fold differences using the comparative ΔΔCT method. The mRNA levels were normalized to GAPDH mRNA.
Pro-Osteogenic Markers.

The information and description of the genes were cited from NCBI Gene (https://www.ncbi.nlm.nih.gov/).
Statistics
GraphPad Prism 9.5.1 software (GraphPad Software, La Jolla, CA, USA) was used. Differences between groups were evaluated by the Kruskal–Wallis test, followed by Dunn's post-hoc test. P < 0.05 was regarded as significant.
Results
Different Osteogenic Media Caused Different Levels of Calcification in VICs
The different osteogenic media showed major differences. There was no calcification on day three (therefore not included in the Figures 1 and 2). The earliest significant signs of calcification were observed on day seven using media 3, 4, and 6 (Figures 1 and 2). Untreated cells were not included in Figure 1 because there was no calcification at all.
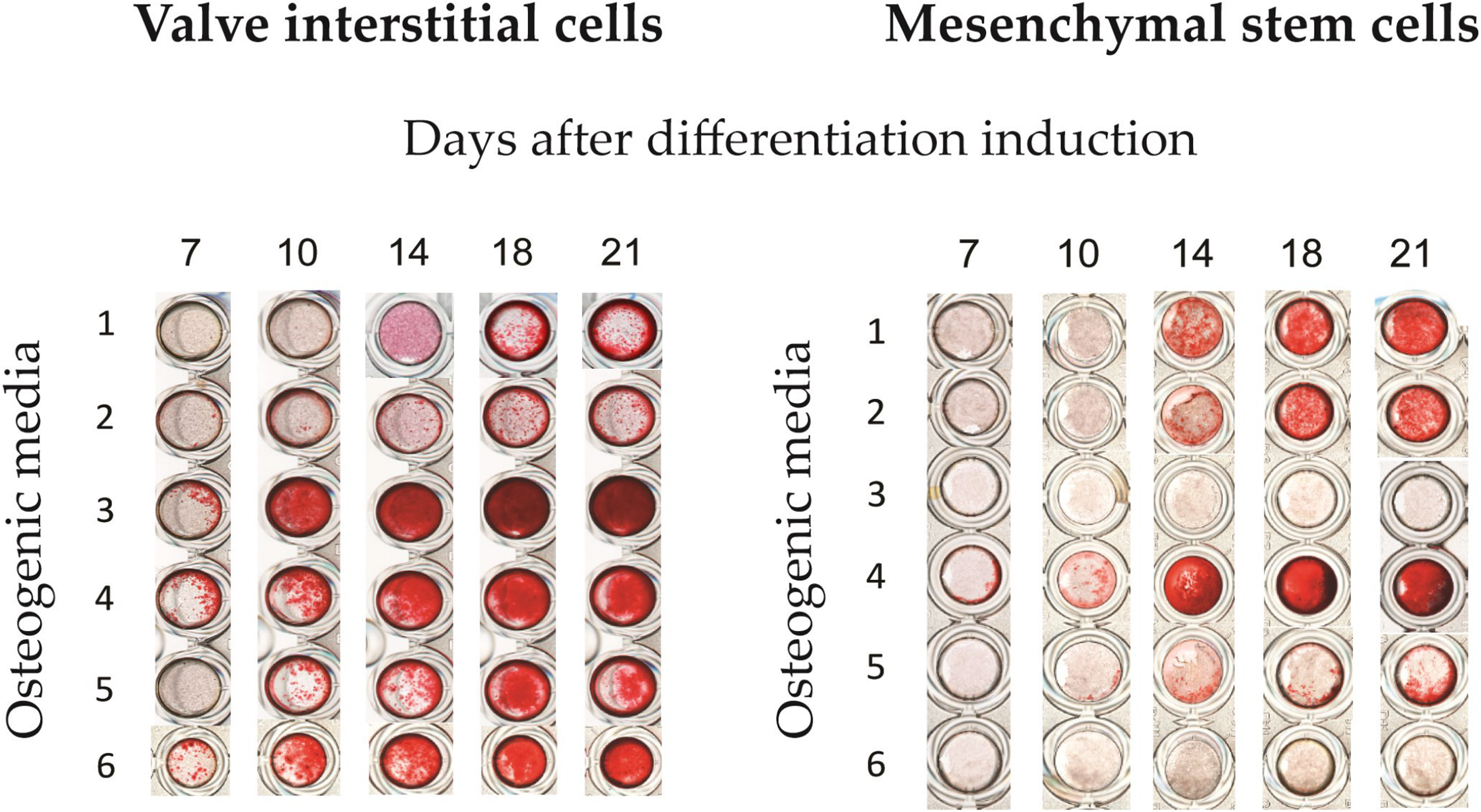
Alizarin red staining of calcified aortic valve interstitial cells and mesenchymal stem cells. Calcification induced by six different osteogenic media is shown at different time points (7th, 10th, 14th, 18th, and 21st day). Calcium deposits were stained by the Alizarin Red Method. Six different osteogenic media were used: 1. 50 µM ascorbic acid, 0.1 µM dexamethasone, and 10 mM β-glycerophosphate; 2. 50 µM L-ascorbic acid 2-phosphate, 0.1 µM dexamethasone, and 10 mM β-glycerophosphate; 3. 7.5 mM CaCl2; 4. 100 µM L-ascorbic acid 2-phosphate, 10 mM KH2PO4, 20 mM HEPES, 50 ng/mL BMP2; 5. 50 µM L-ascorbic acid 2-phosphate, 10 mM β-glycerophosphate, 500 nM 1a, 25-dihydroxycholecalciferol; 6. 50 µM L-ascorbic acid 2-phosphate, 3 mM NaxH3-xPO4 (pH 7.4), 20 mM HEPES, 0.1 µM dexamethasone. Untreated cells were not included because there was no calcification.
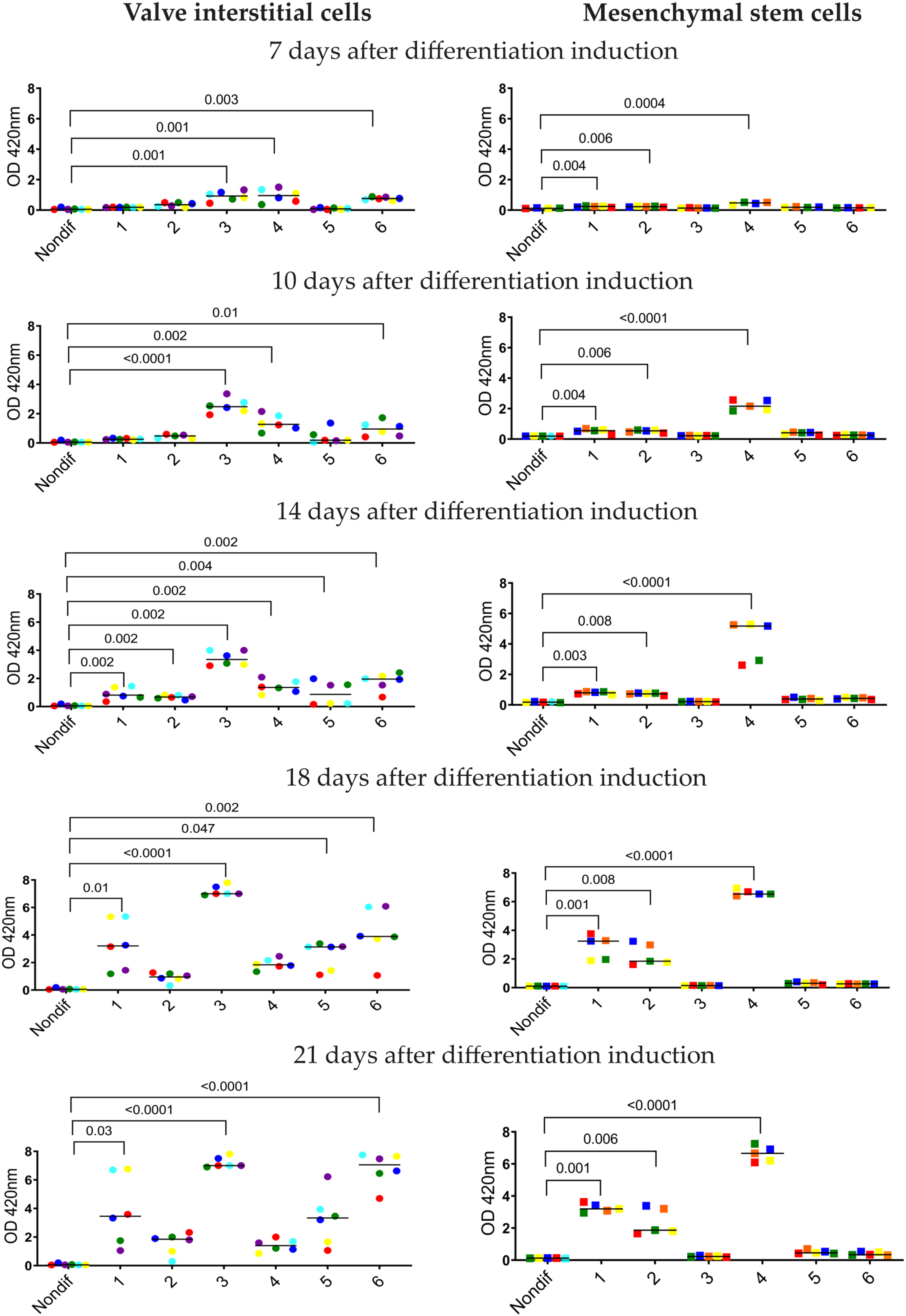
Calcification of cultured aortic valve interstitial cells (n = 6) and mesenchymal stem cells (n = 5). Calcification was induced by six different osteogenic media shown at different time points (7th, 10th, 14th, 18th, and 21st day) and measured by the Alizarin Red method. Six different osteogenic media were used with the composition as described in Figure 1. The lines represent the median. Different colors show different cell donors. Each dot represents a row number of optical density readouts. Numbers on the x-axis show different osteogenic media. Nondif are untreated cells grown without an osteogenic medium. Statistics by Kruskal–Wallis test followed by Dunn's post-hoc test. P < 0.05 was regarded as significant.
Microscopy examination showed that after exposure to medium 4, VICs changed their morphology and acquired a round shape. They lost their adhesive properties and were washed away during a change of medium (by day seven). Despite no cells, calcification continued to increase on day 10.
From day 14 to 18, osteogenic media 1, 5, and 6 increased noticeably the calcification rate (Figures 1 and 2). On day 14, microscopy examination showed that VICs calcified by osteogenic medium 3 formed separate conglomerates of cells with disturbed monolayers. After that, the cells detached and were washed away during change of medium. Using media 3 and 4, a calcified salt precipitation with no cells left on the growth surface was observed on the bottom of the wells. At the end of observation (21 days), osteogenic media 1, 5, and 6 had induced significant calcification of VICs. Medium 2 did not cause significant calcification (Figures 1 and 2).
Different Osteogenic Media have Different Effects on MSCs Compared to VICs
Contrary to VICs, MSCs maintained their normal morphology assed by microscopy examination and did not detach during exposure to any osteogenic media, including media 3 and 4. MSCs treated with medium 3 did not calcify at all (Figures 1 and 2). Medium 4, which was toxic to VICs, gave the strongest calcification in cultured MSCs. MSCs treated by medium 1 were calcified in a similar way as VICs. Medium 2 caused minor calcification in VICs, but induced strong calcification in MSCs. Media 5 and 6 caused calcification in VICs, but not in MSCs (Figures 1 and 2).
Osteogenic Stimuli Induce Different Gene Expression in MSCs and VICs
Classic osteogenic markers
The gene expression profiles were markedly different in MSCs and VICs (Figure 3). In VICs, we did not study gene expressions in cells treated by medium 4 after the 4th day, because all cells were detached from the surface of the wells by day 7. There were no cells left for analysis. In VICs, the key pro-osteogenic marker RUNX2 increased significantly by osteogenic media 1 and 2 already after four days. During the differentiation process from the 4th day to the 14th day, the expression of RUNX2 in VICs continued to increase by media 1 and 2. However, media 5 and 6, which caused strong calcification in VICs, did not increase RUNX2 at all (Figure 3).
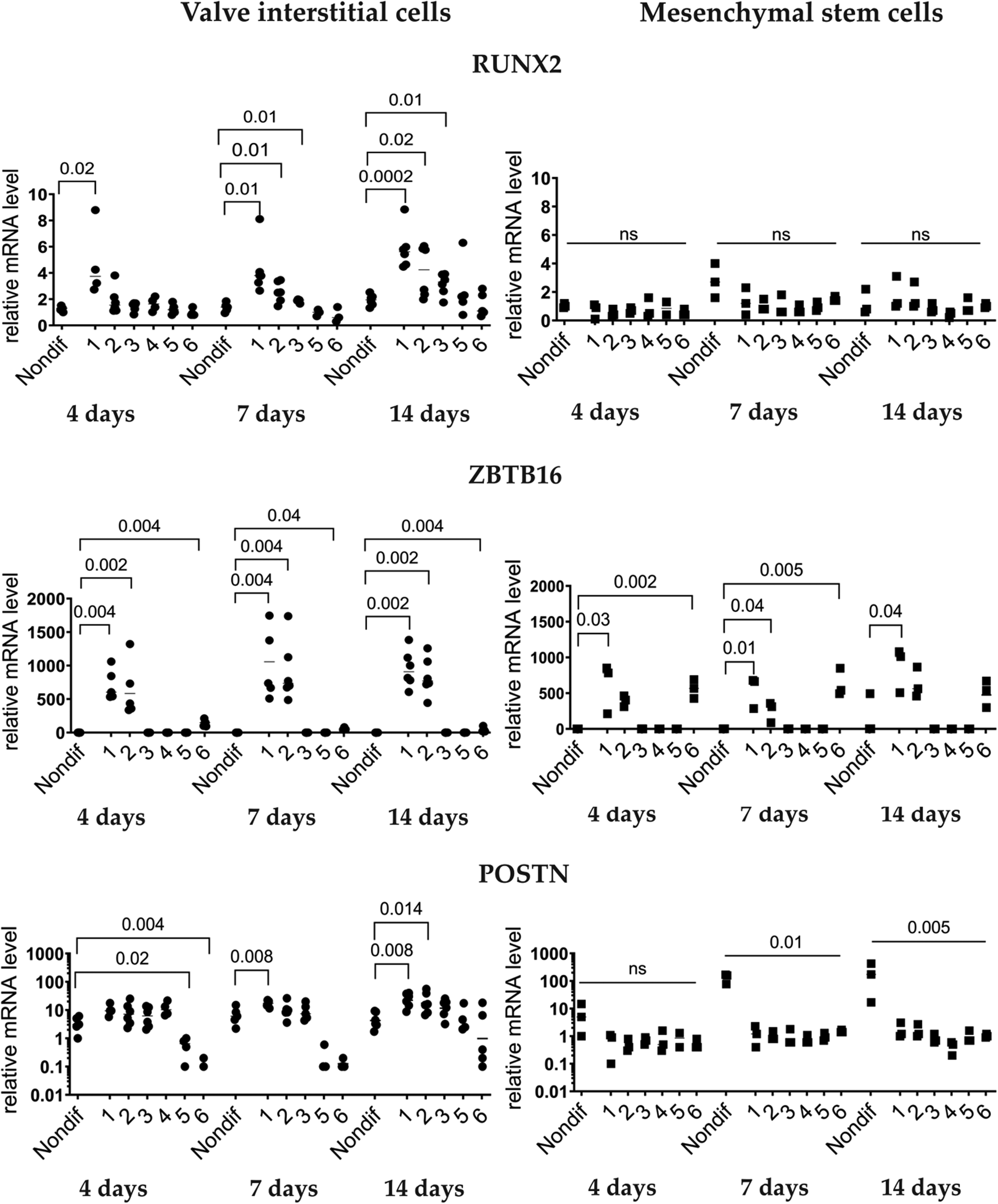
Expressions of pro-osteogenic genes during induced osteogenic differentiation in aortic valve interstitial cells (n = 6) and mesenchymal stem cells (n = 3). Analysis by qPCR After 4, 7, and 14 days of induced osteogenic differentiation. Six different osteogenic media were used with the composition as described in Figure 1. No measurements for osteogenic media 4 after four days of differentiation induction, because cells detached and were washed away. Numbers on the x-axis show different osteogenic media. Nondif are cells grown without an osteogenic medium. Statistics by Kruskal–Wallis test followed by Dunn's post-hoc test. P < 0.05 was regarded as significant.
In contrast, none of the media increased RUNX2 in MSCs at any time point studied (Figure 3). Expression of ZBTB16 had some similarities in VICs and MSCs. However, medium 6 upregulated levels of ZBTB16 in MSCs, but not in VICs. Different from VICs, POSTN was downregulated in MSCs by all media (Figure 3).
Additional osteogenic markers
We studied the expression of some additional genes, which may be connected to osteogenic processes (Figures 4 and 5). 19 Expression of OPG did not differ significantly (Figure 4). Only in VICs, BGLAP was upregulated 14 days after exposure to media 3 and 5. COL1A1 expression was downregulated in MSCs by media 2, 3, and 4 on the 7th and 14th days after the induction. In VICs, no medium increased COL1A1 expression. Patterns of TIMP4 expression were similar in VICs and MSCs (Figure 4).
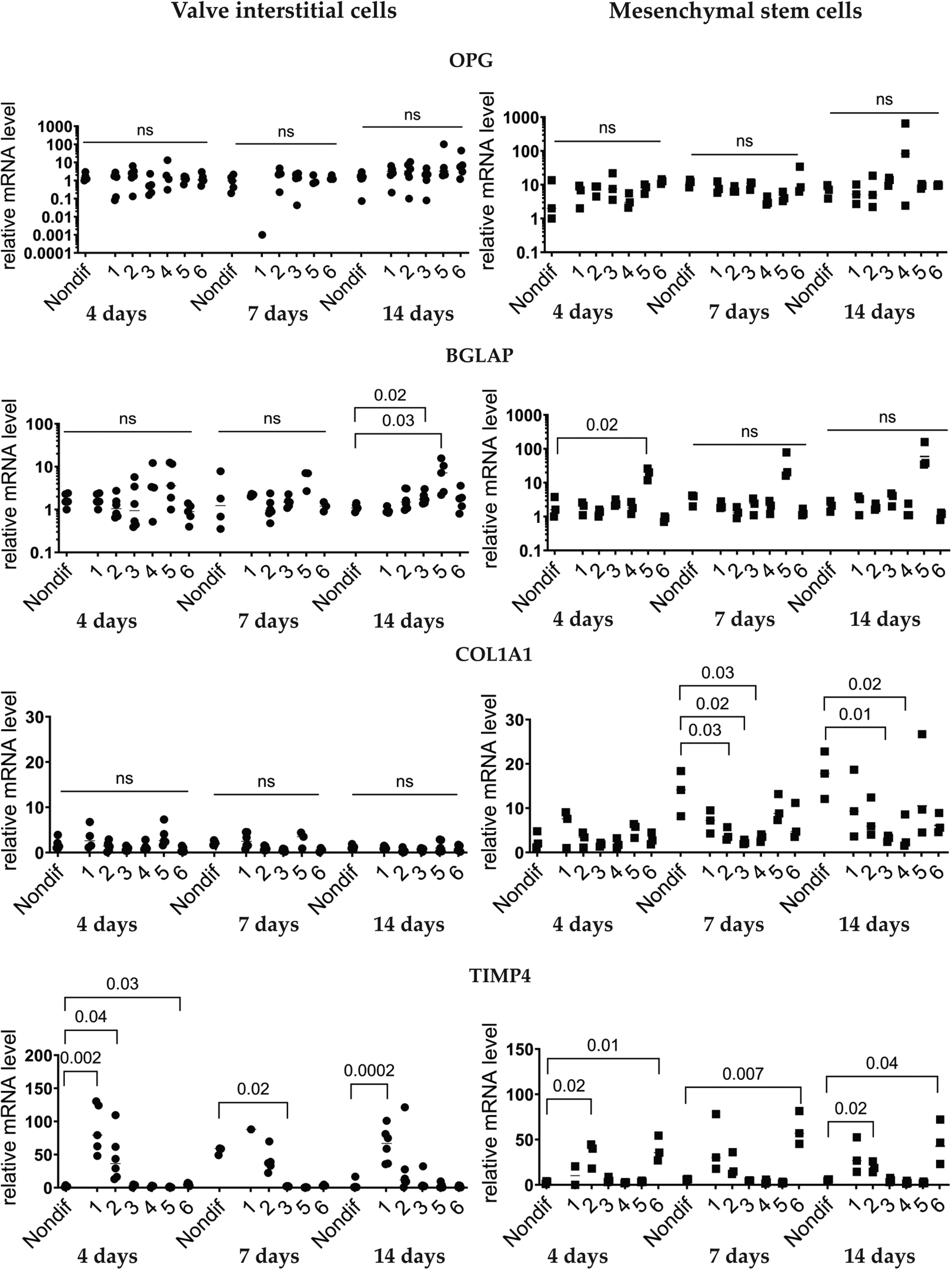
Expressions of pro-osteogenic genes during induced osteogenic differentiation in aortic valve interstitial cells (n = 6) and mesenchymal stem cells (n = 3). Analysis by qPCR after 4, 7, and 14 days of induced osteogenic differentiation. Six different osteogenic media were used with the composition as described in Figure 1. No measurements for osteogenic medium 4 after four days of differentiation induction, because cells detached and were washed away. Numbers on the x-axis show different osteogenic media. Nondif are cells grown without an osteogenic medium. Statistics by Kruskal–Wallis test followed by Dunn's post-hoc test. P < 0.05 was regarded as significant.
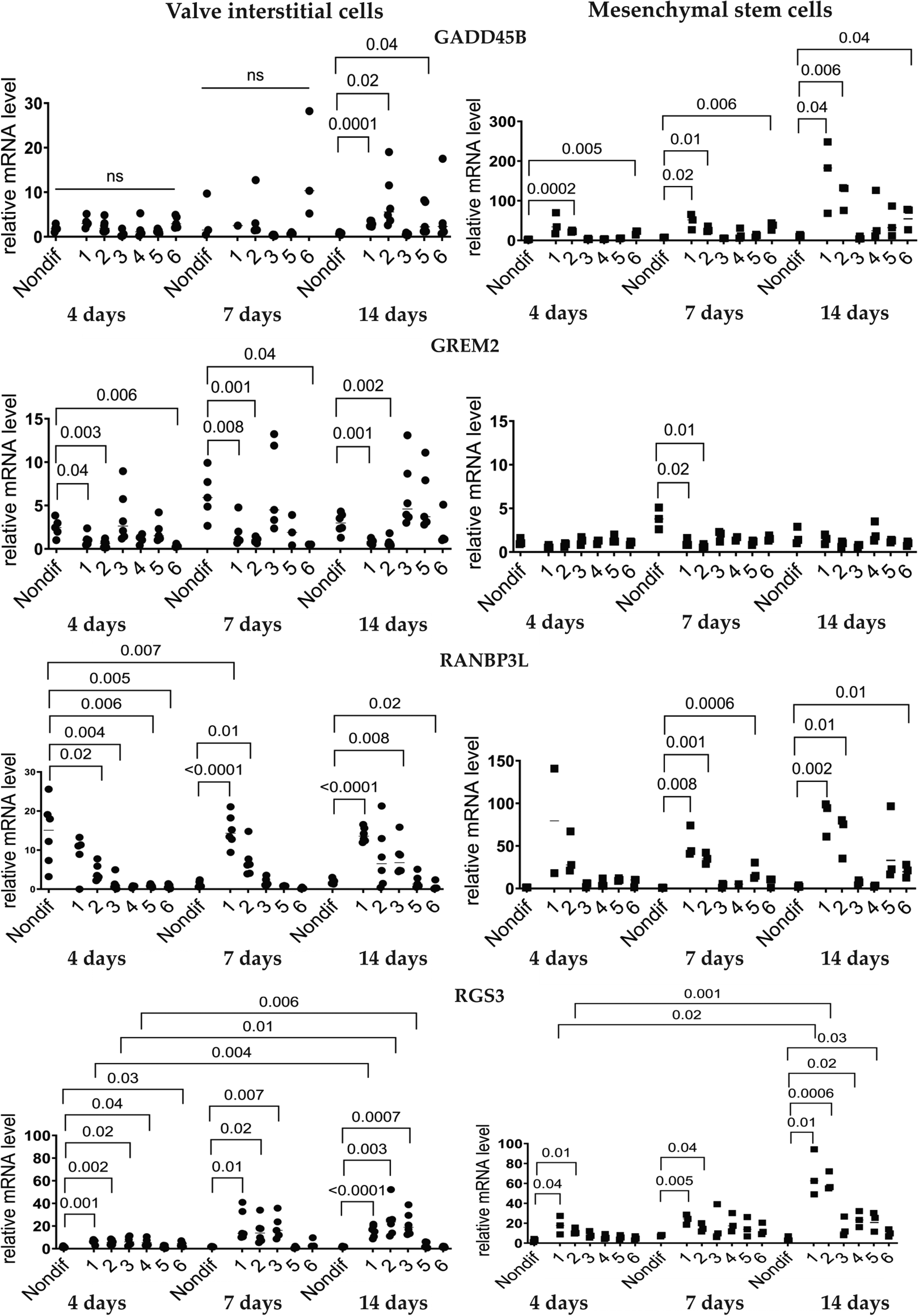
Expressions of pro-osteogenic genes during induced osteogenic differentiation in aortic valve interstitial cells (n = 6) and mesenchymal stem cells (n = 3). Analysis by qPCR after 4, 7, and 14 days of osteogenic differentiation. Six different osteogenic media were used with the composition as described in Figure 1. No measurements for osteogenic media 4 after 4 days of differentiation induction, because cells detached and were washed away. Numbers on the x-axis show different osteogenic media. Nondif are cells grown without an osteogenic medium. Statistics by Kruskal–Wallis test followed by Dunn's post-hoc test. P < 0.05 was regarded as significant.
GADD45B expression was more pronounced in MSCs than in VICs. The upregulation was observed already four days after osteogenic induction (Figure 5). Media 1 and 2 downregulated GREM2 expression more strongly in VICs than in MSCs. Expression was higher in untreated VICs compared to untreated MSCs. Changes in RANBPL3 expression started after 4 days of osteogenic induction in VICs and after seven days in MSCs (Figure 5). RGS3 expression increased earlier in VICs than in MSCs (Figure 5).
Discussion
Six commonly used osteogenic media were used to induce calcification in VICs and MSCs. There were major differences among these media regarding speed and efficiency to induce calcification. Media 3 and 4 were toxic for VICs. Different media influenced gene expression as well as the morphology and functionality of primary VICs differently. It is important to emphasize that the present study did not test the role of specific components of the osteogenic media. We investigated the six osteogenic media per se that are commonly used.
Osteogenic Media and Stimuli
It is important to state that Alizarin Red also stains several other minerals in addition to calcium. However, the staining of non-calcium minerals is far weaker than for calcium, and Alizarin Red is regarded as the golden standard of calcium detection and quantification of calcification in cell cultures.43,44
Osteogenic differentiation is a complex process, which has been studied for many years, but primarily on MSCs. Most media aimed to induce osteogenic differentiation were developed and tested on MSCs. Apart from the media used in the present study, there are several other ways to trigger and enhance osteogenic differentiation in MSCs. This include conditioned media from osteoblasts or osteocytes. Osteocyte-conditioned media induced earlier peaks in ALP activity and greater calcium deposition compared to osteoblast-conditioned media. 45 This suggests that factors secreted by mature bone cells, particularly osteocytes, may provide more potent osteogenic signals. The addition of specific growth factors to osteogenic media can significantly influence the differentiation process. For instance, bone morphogenetic protein-2 (BMP-2) has been shown to have varying effects depending on the MSC source. Enhanced osteogenic differentiation in dental and bone-derived MSCs, inhibited osteogenic differentiation in adipose-derived MSCs. 45 These findings highlight the importance of considering the MSC source when optimizing osteogenic differentiation protocols. The overexpression of connective tissue growth factor in MSCs has been shown to improve their ability for osteogenic differentiation. 46 This genetic modification approach opens up new possibilities for enhancing the osteogenic potential of MSCs. Furthermore, the use of 3D culture systems, such as scaffolds in radial-flow bioreactors, has been demonstrated to accelerate both proliferation and osteogenic differentiation of MSCs compared to standard 2D culture conditions. 47
In general, each of the compounds used in the osteogenic media is necessary for the specific molecular processes involved in osteogenic differentiation and calcification. Dexamethasone is used in most media to induce osteogenic differentiation, most probably by activation of Wnt/β-catenin, RUNX2, and BMP2 signaling. 48 How dexamethasone acts on cell proliferation, metabolism, and differentiation depends on its concentration and context. 32 However, dexamethasone might initiate osteogenic and calcification processes different from the development of calcification in the human body. 19 In order to investigate the molecular mechanisms of calcification without the influence of glucocorticoids, dexamethasone was replaced by BMP2 in osteogenic medium 4. Using in vitro models from humans and rodents, BMP2 stimulates the differentiation of preosteoblastic cells. 49 Furthermore, BMP2 enhances the osteogenic differentiation capacity of MSCs derived from both bone marrow and umbilical cord. 50
Ascorbic acid, or its more stable derivative L-ascorbic acid 2-phosphate, was added to the osteoinductive medium to induce collagen type I fibril expression. 20 An external phosphate source, generally inorganic phosphate or hydrolyzable substrates like β-glycerophosphate, facilitates the mineralization of the extracellular matrix (mainly collagen fibrils) produced by the cells. 51 For mineralization processes, NaH2PO4 or a mixture of the sodium phosphate compounds, Na2HPO4 and NaH2PO4, as well as KH2PO4, have been used as inorganic phosphate sources. 17 CaCl2-containing growth media caused strong calcification of MSCs in cultures. 33 25-Hydroxycholecalciferol (25OHD3) (in osteogenic media 5) may be important for bone metabolism. Several studies in vitro show its inductive role in osteogenic processes.52–54
Osteogenic media 3 and 4 were toxic for VICs in culture. These media caused morphological changes and the VICs lost adhesion. Consequently, cells detached from the growth surface and were washed away during medium change. In this situation, media 3 and 4 most probably caused calcium deposits by a chemical reaction and not by an active biological process. Calcium deposits continued to accumulate in the wells even after VICs were not present anymore. This is probably related to the accumulation of salts on the surface of the wells. Dead cells with debris were washed away during multiple rounds of PBS wash before cells were obtained for RNA isolation. Only living cells were used for qPCR.
The primary treatment options for CAVD focus on addressing severe disease stages, as no medical therapies currently exist to prevent or reverse its progression. The mainstay treatments are surgical and transcatheter valve replacement. SAVR involves replacing the diseased valve with a mechanical or bioprosthetic valve with open heart surgery. While effective, mechanical valves require lifelong anticoagulation, and bioprosthetic valves have limited durability, often necessitating reoperation. TAVR is a minimally invasive alternative, particularly suited for patients at high surgical risk where a bioprosthetic valve is implanted. It offers faster recovery and shorter hospital stays but may not be suitable for all patients due to anatomical or procedural limitations. TAVR has inherited limitation of bioprosthetic valve durability as well.55,56 Different mechanisms and processes are involved in the development of CAVD. In the valve tissue, mineral deposition can take place by several different mechanisms, including dystrophic, osteogenic, and endochondral calcification. It involves the activation of VICs.57–63 Different types of calcification may occur simultaneously or independently in human aortic valves. The dystrophic calcification theory suggests that cell damage is a crucial initial step in the calcification process.64,65 The primary event involves the interaction between calcium-rich extracellular fluid and the phosphorus-containing membranes of non-functional cells that cannot regulate calcium ion influx. Dystrophic calcification in the aortic valve is often considered a passive process. This form of calcification typically occurs in damaged or necrotic tissue where calcium phosphate precipitates due to local changes in the tissue environment, such as pH shifts or the presence of necrotic cells. The process is characterized by the deposition of calcium salts in areas of tissue injury or necrosis, rather than being driven by active cellular processes. The cytokine TGF-β1 has been shown to induce rapid dystrophic calcification in vitro through an apoptosis-mediated process.65,66
In CAVD, the mechanical and biochemical factors may contribute to cell stress and calcification by an active biological process in VICs. 8 This theory is associated with various signaling pathways and pro-osteogenic genes. It remains unclear whether these different calcification processes occur independently and simultaneously, if they are interconnected or interdependent, or if they are mutually exclusive. In the present study, we concentrated on osteodifferentiation processes occurring in VICs when the cells acquire an osteoblast-like phenotype with expression of pro-osteogenic proteins. The end result is calcium deposits in the extracellular matrix—calcification.
VICs are a heterogeneous cell population. Depending on their position in the valve tissue, VICs may have different functions and characteristics. Their pro-osteogenic potential is also variable.67–69 This may explain the variability of calcification intensiveness of different cell cultures. Furthermore, cells treated with osteogenic media containing organic phosphate are suggested to be less consistent in their calcification compared to cells treated with calcification media containing inorganic phosphate. 68 However, we did not notice any specific differences in the consistency of calcification from donor to donor between media no. 1, 2, and 6 in VICs.
Gene Expression of Osteogenic Markers in VICs
Calcification itself does not necessarily mean that the cell cultures undergo osteogenic differentiation. Alizarin Red staining reveals only calcium deposits in the cell cultures, which may be caused by both passive calcium deposition and a biological process. In the present study, some osteogenic media did not influence gene expression patterns. This suggests that osteogenic media (3 and 4) did not cause osteogenic differentiation. Most probably, calcium deposits were initiated by a “passive” chemical reaction, not by a biological process. Another explanation is that in addition to the most well-known genes, some osteogenic media may trigger other signaling pathways that have not been studied in connection with osteogenic differentiation. However, only changes in gene expression are not proof of an osteogenic process.
One can question the concepts of “marker of calcification,” “osteogenic biomarker,” and “marker of osteodifferention.” In fact, the effect on these so-called markers was highly variable or even none despite considerable calcification. Calcification is the golden standard and end-product of osteogenic differentiation. Therefore, measurements of gene expression should not be used as a strong marker of ongoing calcification. At least this is the case for VICs in vitro. Calcification may take place even though there is no or little effect on the so-called markers. In the present study, this is especially shown for RUNX2, which has been regarded as a biomarker of aortic valve calcification.70,71 In VICs, media 5 and 6 caused strong calcification without increased expression of RUNX2. Consequently, a word of caution is necessary when using RUNX2 as a marker of calcification.
Based on cell morphology and behavior as well as stability and consistency of calcification processes and gene expression, only osteogenic medium 1 works well for studying osteogenic differentiation and calcification processes in VICs.
Comparing Valve Interstitial Cells and Mesenchymal Stem Cells
Both regarding calcification and expression of genes, the six osteogenic media acted differently on the two cell types. The mechanisms of osteogenic differentiation and calcification were different in VICs and MSCs. MSCs were much more homogeneous and consistent in their calcification capability. All taken together, VICs seemed to be more prone to calcification, being more sensitive to the different osteogenic media. Some osteogenic media were obviously injurious for VICs. However, in all osteogenic media, MSCs maintained their morphology, adhesive capability, and survived.
Osteogenic media 3 and 4 seem to have the most distinguished differences between MSCs and VICs differentiation. VICs cultures calcified intensively, but the calcification was most probably due to a non-biological chemical reaction. This matrix is most probably produced and mediated by two different mechanisms–secretion by cultured cells before dying and a pro-calcific substrate made of cells undergone apoptosis. In MSCs, osteogenic medium 3 did not induce calcification at all. We can speculate that MSCs did not produce the same amount of extracellular matrix to act as a core or “scaffold” for calcification by a chemical reaction.
Medium 4 was highly toxic for VICs, but MSCs showed strong calcification. VICs lost their adhesive capability and got a roundish shape under the influence of osteogenic media 3 and 4. A possible cause might be the concentrations of inorganic salts in these media.
Media 5 and 6 had different effects on VICs and MSCs as well. Both media induced calcification in VICs; however, they did not have any effect on MSCs. The observed differences between VICs and MSCs indicate that MSCs are not the best choice to study CAVD in vitro.
Limitations of the Present Study
Studies based on cell cultures will always have limitations and must be interpreted with caution. Even though human primary cell cultures are a good model for studying cardiovascular calcification in vitro, there are plenty of challenges and limitations. An important limitation is that it is not easy to get a sufficient number of samples for the study. Furthermore, primary cells usually proliferate quite slowly. Consequently, for extensive studies different analyses and methods, it is a challenge to get enough cells in each cell culture to conduct all investigations.
The impact of cell passage number on VICs differentiation and calcification is a critical factor in standardizing in vitro models of CAVD. VICs are stable for approximately 8 passages, but the majority of studies are performed between passages 2 and 5 due to the changing biology of the VICs.8,72,73 In our study, we utilized VICs and MSCs from passages 3 to 5. This range was selected to ensure consistency in experimental conditions and minimize variability between replicates. Maintaining similar passage numbers allows to make reliable comparisons across different cultural conditions. To ensure sufficient cell numbers for the experiments while maintaining reliable differentiation and calcification properties, passages 3–5 were chosen as an optimal range. We acknowledge that the use of passages beyond passage 5 could introduce variability in differentiation or calcification potential, which represents a limitation of the study. However, this limitation was carefully mitigated by consistently using cells within the defined passage range across all experiments.
Osteogenic differentiation with calcification may be different in cell cultures compared to in valve leaflets. Calcification induced by osteogenic media over 21 days in a cell culture is different from calcification occurring in an extracellular matrix over several years. Cultured VICs lack the influence of the microenvironment of the valve leaflets in vivo. 74 Consequently, the results from in vitro experiments cannot directly be extrapolated to processes in the human body. 9 The selection of primers in this study was specifically chosen to investigate osteogenic differentiation in cultured VICs under the experimental conditions. While our primer selection was based on previous studies and chosen to capture the most relevant and responsive genes under these specific conditions, it inherently reflects the constraints of studying isolated cell cultures. 19 Future studies could expand primer selection to include genes associated with extracellular matrix remodeling and other pathways influenced by the native valve environment, thereby providing a more comprehensive understanding of calcification mechanisms.
Current study is focused only on VICs derived from male patients with CAVD. Sex differences in aortic valve calcification, particularly in VICs, reveal distinct molecular and cellular mechanisms driving disease progression. Males exhibit significantly higher calcification rates in aortic valves, while females show a more fibrotic remodeling pattern. These differences are rooted in sex-specific cellular responses and hormonal influences. Male VICs demonstrate twice higher CAVD incidence than females, with greater expression of osteogenic markers such as RUNX2 and myofibroblastic activity, while female VICs showed greater metabolic activity and collagen production.75,76 The higher aortic valve calcification density scores were reported in men compared to women, even when both had a similar degree of stenosis. At the same time, there were significantly higher levels of fibrosis in the stenotic valves from women compared to those from men.77,78 Male porcine VICs cultured in osteogenic medium produce larger and denser calcium nodules compared to female VICs. Male VICs show upregulated RANK/RANKL/OPG pathway activity, which promotes osteogenic differentiation and calcification. Female stenosed aortic valves have more fibrosis, however, they still exhibit some calcifications, 79 so the study of calcification in female aortic stenosis is still of interest. 76 Though, based on the differences in the mechanisms of CAVD development, calcification processes in VICs from females and males should be studied separately.
Concerning the differences between VICs and MSCs, the present study did not exploit the plasticity of MSCs and the possibility to make MSCs more fibroblast- or VIC-like in their phenotype before osteodifferentiation was induced.
All existing osteogenic media are artificial regarding their components and the concentration of the substances. Nonetheless, experimentally induced calcification in primary cell cultures is an easy and affordable model of osteogenic differentiation processes. With the correct osteogenic medium, VICs may be suitable for studying the basic molecular and cellular mechanisms involved in osteogenic differentiation and calcification. VICs in culture can also be used for screening potential pharmacological inhibitors of calcification. It would be an advantage to analyze changes on protein level. However, for this study, we concentrated on the calcification assessment and genes changes.
In addition to the six investigated osteogenic media, there are other media that have been reported to induce or enhance osteogenic differentiation in investigated cell cultures.80–82 The present study included the six media most commonly used.
Conclusion
Six different, commonly used osteogenic media had major differences in their effects. Some of them were even toxic for cultured VICs, but not for MSCs. Due to the differences from VICs, MSCs appear to be suboptimal for studies of aortic valve calcification in vitro. It is vital to select the best osteogenic medium to study processes and mechanisms of calcification and osteodifferentiation. Based on a combination of calcification and gene expression, medium 1 appears to be the best one in VICs.
Footnotes
Abbreviations
Acknowledgements
Professor Leiv Arne Rosseland, University of Oslo and Oslo University Hospital, is gratefully acknowledged for administrative support. Faiza Fayyaz Moghal is gratefully acknowledged for administrative and secretarial assistance. Professor Jan E. Brinchmann, University of Oslo, kindly provided MSCs.
ORCID iDs
Author contributions
Conceptualization: D.E., J.V., and A.Z.; methodology: D.E.; validation: D.E, J.V., and A.Z.; investigation: D.E.; resources: T.A.K, ML.K, and JP.E.K.; data curation: J.V. and A.Z.; writing—original draft preparation: D.E.; writing—review and editing: J.V.; visualization: X.X.; supervision: J.V. and KO.S; project administration: J.V. and A.Z.; funding acquisition: J.V. and A.Z. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Helse SørØst, University of Oslo, Digital Life Norway, and Nasjonalforeningen for Folkehelsen.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
