Abstract
Dynamic left ventricular outflow tract obstruction (dLVOTO) is an important cause of cardiogenic shock to consider as its management differs from garden-variety cardiogenic shock from systolic dysfunction. It is best suited for serial evaluation by point-of-care ultrasound (POCUS) as its dynamic nature may cause it to be missed on routine echocardiograms. We present a case of a 95-year-old male who presented for constipation and went into cardiac arrest after manual disimpaction. After intubation and advanced cardiovascular life support with eventual return of spontaneous circulation, the patient developed cardiogenic shock. He developed persistent lactic acidosis, increasing ventilator requirements, and worsening renal function despite diuresis and escalating epinephrine and norepinephrine requirements. POCUS revealed left ventricular systolic dysfunction with apical hypokinesis and basal hyperkinesis suspicious for stress-induced cardiomyopathy. Moreover, systolic anterior motion of the mitral valve on M-mode ultrasound and a left ventricular outflow tract pressure gradient of 46 mm Hg on spectral Doppler were noted, consistent with dLVOTO. On recognition of dLVOTO, epinephrine and norepinephrine were weaned after adding vasopressin and phenylephrine. The patient's clinical status drastically improved thereafter and he was weaned off vasopressors and the ventilator within 48 hours. POCUS techniques that are often excluded from basic critical care ultrasound exams are required to identify the characteristic features of hemodynamically significant dLVOTO. These features are important to recognize in cardiogenic shock as inotropes and diuresis can lead to paradoxical worsening of the dLVOTO. Instead, these patients should be managed with pure vasopressors and measures to increase left ventricular preload (beta blockers and intravenous fluids).
Introduction
Dynamic left ventricular outflow tract obstruction (dLVOTO) is a phenomenon that is often underdiagnosed and is a result of anatomic and physiological factors that lead to tightening and reduction of blood flow across the left ventricular outflow tract (LVOT). 1 Given its transient nature, it is more suitable for identification and monitoring by point-of-care ultrasound (POCUS) as traditional echocardiograms only capture a snapshot of the patient's cardiac function. While dLVOTO is most commonly associated with hypertrophic cardiomyopathy, it can be found in patients with other structural abnormalities that predispose them to the phenomenon including stress cardiomyopathy (SCM), acute myocardial infarction (MI), and mitral valve repairs. 2 It is more common in the setting of low systemic vascular resistance associated with hyperdynamic circulation (eg, sepsis, anesthesia, and cirrhosis) and can be seen even in structurally normal hearts in the presence of these physiological factors. 2
Hemodynamically significant dLVOTO can lead to cardiogenic shock and can be identified by POCUS using M-mode ultrasound and spectral Doppler techniques that are not commonly included in routine critical care POCUS exams. It is important to rule out dLVOTO as the mechanism for cardiogenic shock as its management is the opposite of traditional cardiogenic shock from systolic dysfunction.1,2 To emphasize early recognition of the phenomenon, we present a case of cardiogenic shock secondary to dLVOTO that was identified by POCUS and drastically changed our management strategy.
This report was prepared in accordance with the CARE guidelines. 3
Case
A 95-year-old man with coronary artery disease, peripheral artery disease, and hypertension presented to the emergency department for constipation. Computed tomography of the abdomen and pelvis revealed stercoral colitis with extensive stool burden. On presentation, the patient's vitals and labs were unremarkable. Immediately after manual disimpaction, he developed ventricular fibrillation cardiac arrest. He underwent intubation without sedation or paralytics and 3 cycles of advanced cardiovascular life support (ACLS) before obtaining return of spontaneous circulation (ROSC). Postarrest electrocardiogram showed diffuse ST depressions. The patient was initially normotensive after ACLS and obtaining ROSC but quickly became hypotensive thereafter with a blood pressure of 70/40 mm Hg. Serum lactate was 6.0 mmol/L and internal jugular central line oxygen saturation was 46%. The patient remained hypotensive despite norepinephrine 0.5 mcg/kg/min and epinephrine 0.5 mcg/kg/min without improvement in blood pressure with further dose escalations. Lactate remained at 6.0 mmol/L on subsequent trends and creatinine trended upwards with reduced urine output. High-sensitivity Troponin I rose exponentially to a peak of 19349 ng/L (Upper Limit of Normal 15 ng/L). The patient was persistently hypoxic with SpO2 84% and arterial blood gas with PaO2 53 mm Hg despite FiO2 of 100%, peak end expiratory pressure of 10, and complete ventilator synchrony on sedation. Chest X-ray revealed evolving pulmonary edema. The infectious workup was unrevealing. The clinical picture was suggestive of refractory cardiogenic shock complicated by pulmonary edema.
A cardiac POCUS exam was performed. Moderately reduced left ventricular systolic dysfunction with apical ballooning, hypokinesis extending from apical septum to mid inferolateral wall, and basal hyperkinesis were noted. This was concerning for SCM versus MI involving the left anterior descending artery (LAD). Moreover, systolic anterior motion (SAM) of the mitral valve was noted on M-mode (Figure 1) with moderate mitral regurgitation on color Doppler. The LVOT peak instantaneous gradient (PG) was 46 mm Hg on continuous wave (CW) Doppler (Figure 2). Pulse wave (PW) Doppler localized the site of obstruction at the mid-LVOT, demonstrating a high-velocity, late-peaking LVOT tracing (Figure 3). These findings were consistent with hemodynamically significant dLVOTO.

M-mode imaging through the mitral valve on parasternal short axis view demonstrating moderate SAM (arrow) given the presence of septal contact but the duration of contact <1/3rd of the systolic period. Abbreviation: SAM, systolic anterior motion.

CW Doppler through the LVOT on apical 5-chamber view demonstrating an LVOT PG of 46 mm Hg. Abbreviations: CW, continuous wave; LVOT, left ventricular outflow tract; PF, peak instantaneous gradient.

PW Doppler at the site of obstruction (mid-LVOT) on apical 5-chamber view demonstrating a late systolic peak noted in the LVOT VTI tracing (arrow), typical of dLVOTO. PW Doppler just proximal to the aortic valve leaflets demonstrating an LVOT VTI of 22 cm. Of note, the narrowed LVOT is expected to increase the LVOT VTI. Thus, the ability to apply LVOT VTI to estimate cardiac output is limited in this case. Abbreviations: LVOT, left ventricular outflow tract; PW, pulse wave; VTI, velocity time integral.
Before the POCUS exam, both norepinephrine and epinephrine were utilized to tackle the presumed mixed shock attributed to vasoplegia and left ventricular systolic dysfunction. The patient worsened with refractory shock and hypoxemia despite vasopressor escalation and maximal ventilatory support including FiO2 of 100%. After dLVOTO was identified on POCUS, it was decided to add vasopressin and phenylephrine and wean epinephrine while closely monitoring central venous O2 saturations. With this strategy, epinephrine and norepinephrine were eventually discontinued, accompanied by an improvement in cardiac output as evidenced by an increase in central venous O2 saturation (46% to 82%) and the development of warm extremities. Perfusion improved dramatically as evidenced by improvement in capillary refill time, urine output, creatinine, and lactate (7.3 to 1.3 mmol/L). Additionally, the hypoxemia resolved, and ventilator support was weaned, including FiO2 (100% to 40%). Cardiology was consulted to consider invasive strategies, but it was felt that conservative management (without coronary angiography or mechanical circulatory support) would have a better risk-benefit profile. The patient was extubated and weaned off vasopressors within 48 hours of epinephrine wean. The patient was eventually transferred out of the intensive care unit to the floor. However, due to complications that arose later during the hospitalization unrelated to the initial presentation, the patient eventually chose a comfort-care-based approach and expired in the palliative care unit.
Discussion
dLVOTO is characterized by its variability throughout the cardiac cycle and is modified by changes in preload, afterload, and the heart's contractility. 4 The static structural substrate of the LVOT obstruction is dictated by preexisting septal hypertrophy (eg, hypertrophic obstructive cardiomyopathy, chronic hypertension with sigmoid septum, and infiltrative cardiomyopathy) and mitral valve apparatus abnormalities.5,6 These ultimately predispose patients to the main mechanism of dLVOTO: SAM of the anterior mitral leaflet. 2 Other components that contribute to the dynamic substrate include basal hyperkinesis (eg, SCM, low preload states, and inotrope use) and septal bowing (eg, right ventricular failure).5,7
Identification of dLVOTO by POCUS can be done systematically using techniques beyond those commonly used in basic critical care ultrasound exams. Color Doppler can be employed to initially screen for obstruction by revealing turbulent LVOT flow and may reveal mitral regurgitation with posterior jet secondary to SAM. 6 M-mode aids in identifying and grading SAM based on the presence and duration of septal contact. It can also be leveraged to detect early aortic valve closure, supporting the diagnosis of SAM. 5 CW Doppler of the LVOT is used to identify hemodynamically significant dLVOTO by an LVOT PG ≥ 30 mm Hg. Subsequently, PW Doppler is utilized to pinpoint the site of obstruction along the LVOT, characterized by a high-velocity, late-peaking signal.4,7 The need for multiple techniques to identify dLVOTO highlights that intentional training and a certain level of expertise are required to identify it. In particular, accurate determination of the level of obstruction is challenging but of paramount importance as LVOT obstruction can be caused by multiple pathologies including aortic stenosis.
dLVOTO may lead to shock and is exacerbated by culprits of reduced LV filling, including increased heart rate, increased contractility, and low preload states (Figure 4). 4 Hence, positive inotropes/chronotropes, afterload reduction, and diuretics should be avoided. Instead, the typical strategy should focus on vasopressors without inotropic properties (eg, vasopressin and phenylephrine) to increase afterload in combination with volume administration and heart rate reduction to increase preload, effectively stenting the LVOT.4,5 Mechanical circulatory support can be considered if the risk-benefit profile is favorable. However, in cases with cardiogenic shock and acute pulmonary edema due to combined systolic dysfunction and dLVOTO, management is challenging due to competing management priorities. Thus, management should be individualized.
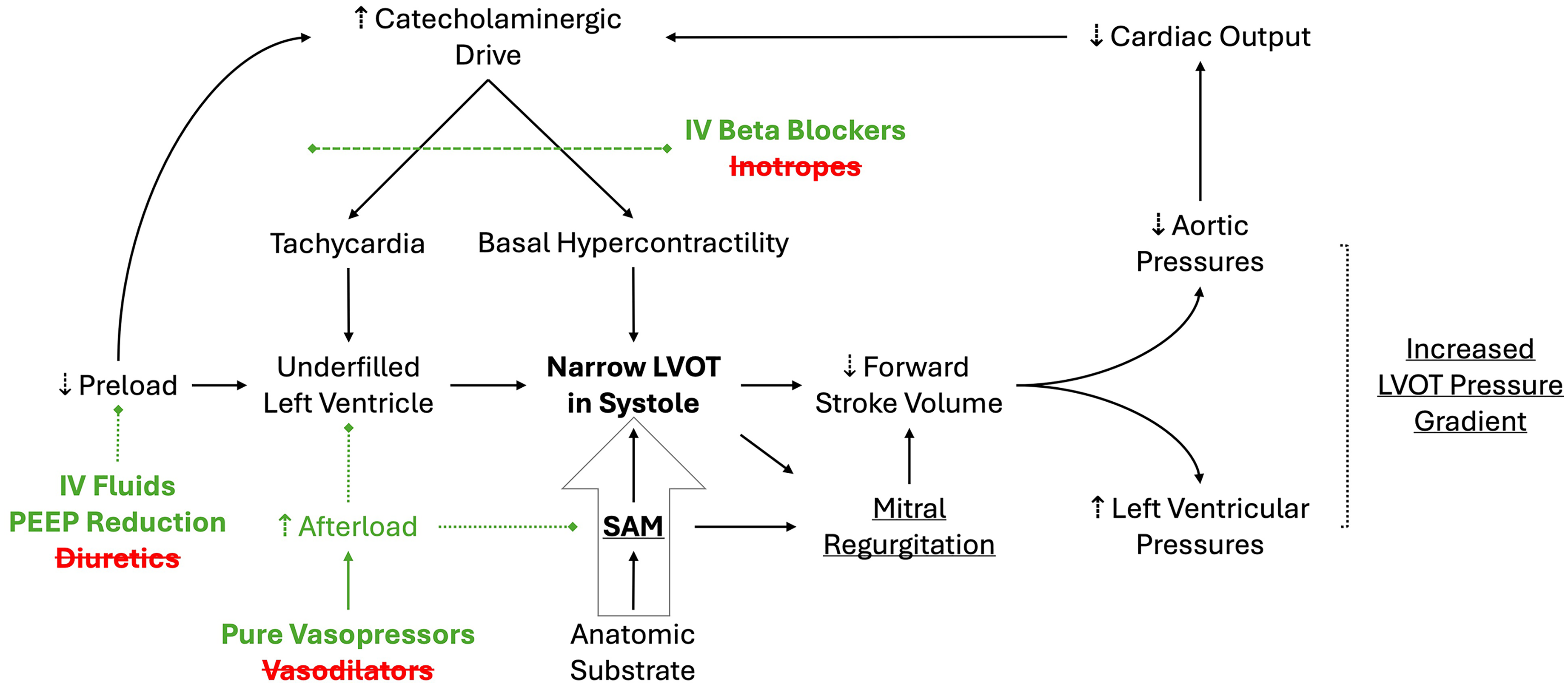
Pathophysiology of dLVOTO and treatment targets. Green entities represent treatment strategies with targets. Red entities represent strategies to discontinue that are typically utilized for cardiogenic shock to prevent the progression of shock from dLVOTO. Underlined entities represent the echocardiographic triad of findings in dLVOTO. Static anatomic substrate leads to the narrowing of the outflow tract directly and indirectly via predisposition to SAM. Abbreviations: dLVOTO, dynamic left ventricular outflow tract obstruction; IV, intravenous; PEEP, peak end expiratory pressure; SAM, systolic anterior motion.
SCM and MI were the leading differentials in this case. Coronary angiography is necessary to distinguish between these 2 diagnoses. 4 POCUS findings in this case, including apical hypokinesis and basal hyperkinesis, were indicative of SCM, which often predisposes to dLVOTO. In fact, up to 25% of cases of SCM have evidence of dLVOTO. 6 Moreover, SCM has been cited to occur after resuscitation of a cardiac arrest due to the physical stress of resuscitation. 8 MI, usually involving mid-LAD, has also been shown to present with dLVOTO. 9 However, the argument for coexisting SCM can be made in many of the published cases. Ultimately, these observations indicate that there may be more of an overlap between the pathophysiology of SCM and some myocardial ischemic syndromes than previously understood. 9 While it is likely that the patient had components of both SCM and MI, rapid clinical improvement without invasive intervention in combination with imaging favors SCM as the diagnosis.
Given its dynamic nature and the importance of rapid identification, dLVOTO is particularly amenable to POCUS. LVOT gradients may fluctuate rapidly depending on hemodynamic conditions, making serial assessments with POCUS valuable in assessing clinical response as therapies are titrated. 10 The value of cardiac POCUS in real-time assessment is emphasized by its reported utility in guiding intra-operative management and anesthesia in patients with dLVOTO. 11
Unfortunately, systematic research and literature on the utility of POCUS in dLVOTO and its related outcomes is limited. This case highlights that early POCUS in cardiogenic shock can help differentiate two very different pathophysiological processes with opposite management strategies. Moreover, it stresses the need to rule out dLVOTO in all patients with cardiogenic shock and the ability of cardiac POCUS to rapidly identify the finding.
Supplemental Material
sj-docx-1-cra-10.1177_29768675251344037 - Supplemental material for A Dynamic Cause of Cardiogenic Shock Identified on Point-of-Care Ultrasound: A Case Report
Supplemental material, sj-docx-1-cra-10.1177_29768675251344037 for A Dynamic Cause of Cardiogenic Shock Identified on Point-of-Care Ultrasound: A Case Report by Akhilesh Gonuguntla, Thomas M Ruli, David N Maynard and Steven Fox in Therapeutic Advances in Pulmonary and Critical Care Medicine
Supplemental Material
Supplemental Material
Footnotes
Ethical approval
Ethical approval is not required for case reports by the Institutional Review Board of the University of Alabama at Birmingham.
Consent for publication
Written informed consent for the publication was provided by the participant's legally authorized representative.
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Footage of POCUS on this patient is available as supplemental material. Data is available upon request from the corresponding author and with permission from the University of Alabama at Birmingham.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
