Abstract
Background
Central venous catheter malposition and pneumothorax remain clinically important complications after insertion. Many emergency departments still rely on post-procedural chest radiography, which may delay catheter use. We evaluated the diagnostic accuracy and workflow impact of point-of-care ultrasound for confirming central venous catheter position and detecting complications in the emergency department.
Methods
We conducted a systematic review and diagnostic test accuracy meta-analysis (International Prospective Register of Systematic Reviews (PROSPERO): CRD420251065638) following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses of Diagnostic Test Accuracy Studies (PRISMA-DTA) and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. The PubMed, Embase, Cochrane Library, Web of Science Core Collection, and Scopus databases were searched from inception to 1 December 2025. Studies were considered eligible for inclusion if they enrolled adults undergoing internal jugular or subclavian central venous catheter insertion in emergency department settings and compared bedside point-of-care ultrasound (cardiac/contrast-enhanced “swirl/bubble” techniques and/or lung ultrasound) with chest radiography and/or computed tomography. For catheter malposition, pooled sensitivity and specificity were estimated using a bivariate random-effects model; pneumothorax accuracy and time outcomes were summarized descriptively due to sparse events.
Results
A total of eight prospective studies (801 central venous catheter insertions) were included. Seven studies contributed estimable 2 × 2 data for malposition. Point-of-care ultrasound showed pooled sensitivity of 0.81 (95% confidence interval: 0.50–0.95) and specificity of 0.99 (95% confidence interval: 0.96–1.00), with hierarchical summary receiver operating characteristic–area under the curve of 0.99 (95% confidence interval: 0.97–0.99), positive likelihood ratio of 137.83 (95% confidence interval: 22.04–862.02), negative likelihood ratio of 0.19 (95% confidence interval: 0.06–0.62), and diagnostic odds ratio of 737.34. Heterogeneity was substantial (I2: sensitivity = 73.85% and specificity = 83.37%). Pneumothorax specificity was consistently high (97.2%–100%), while sensitivity varied and was often nonestimable in zero-event studies. Pneumothorax severity was rarely reported in the primary studies. Reported time savings versus chest radiography ranged from ∼5–284 min (commonly ∼20–60 min). Deeks’ test suggested no significant publication bias (p = 0.86), although power was limited.
Conclusions
In emergency department settings, point-of-care ultrasound provides highly specific and rapid confirmation for detecting central venous catheter malposition, supporting prompt rule-in decisions and potential workflow acceleration. Given heterogeneous sensitivity and limitations of reference standards, negative results should be interpreted cautiously, with confirmatory imaging reserved for equivocal examinations or high-risk scenarios.
Keywords
Introduction
Central venous catheter (CVC) insertion is routinely performed in emergency and critical care settings to deliver vasoactive agents, enable hemodynamic monitoring, and facilitate rapid administration of fluids or blood products. 1 Although ultrasound guidance has improved cannulation success, mechanical complications still occur. 2 A radiography-based study has reported catheter malposition in approximately 3.3% of insertions, and in this cohort, malposition was not associated with severe events such as perforation and thrombosis. 3 Pneumothorax remains a clinically important complication, with reported incidences of ∼1%–6%, depending on the setting and technique. 4 Accordingly, many institutions continue to perform post-procedural chest radiography (CXR) to confirm tip position and exclude pneumothorax, consistent with the recommended guidelines. 5
However, CXR may delay catheter use, consumes resources, and offers limited sensitivity for precise tip localization and small pneumothoraces. 6 Point-of-care ultrasound (POCUS) can provide immediate bedside confirmation by combining transthoracic echocardiography with a rapid saline flush or contrast-enhanced “swirl/bubble” sign to verify central venous drainage, along with lung ultrasound to assess pleural sliding.7,8 Prospective studies in emergency department (ED) and intensive care unit (ICU) settings have reported high specificity and shorter time to catheter clearance compared with CXR.8,9
Despite its increasing adoption, the diagnostic performance of POCUS, specifically in the ED, has not been comprehensively synthesized. Because ED care is time-sensitive, delays while awaiting confirmatory CXR can delay the initiation of urgent therapies. Given the high-acuity, time-pressured ED environment and the need for rapid catheter clearance, it is important to determine whether POCUS provides sufficient accuracy to replace routine radiography. In this systematic review and meta-analysis, we evaluated the diagnostic accuracy of POCUS for detecting CVC malposition and pneumothorax in ED patients.
Methods
Study design and registration
This systematic review and diagnostic test accuracy meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses of Diagnostic Test Accuracy Studies (PRISMA-DTA) statement. 10 The review protocol was prospectively registered in International Prospective Register of Systematic Reviews (PROSPERO, CRD420251065638). Reporting was further aligned with the updated Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement to ensure comprehensive and transparent reporting of methodology and results. 11
Search strategy
We searched the PubMed, Embase, the Cochrane Library, Web of Science Core Collection, and Scopus databases from inception to 1 December 2025. The strategy combined controlled vocabulary and free-text terms for emergency care, central venous catheterization, POCUS, malposition, and pneumothorax. Full search strategies and run details are provided in Supplementary Material 1. We also screened reference lists of the included studies and relevant reviews to identify additional eligible reports.
Eligibility criteria
We included studies that fulfilled the prespecified criteria for participants, index test, reference standard, outcomes, and study design. Participants were adults (≥18 years) undergoing internal jugular or subclavian CVC insertion in the ED. The index test was bedside POCUS (vascular, cardiac, contrast-enhanced, and/or lung ultrasound) to confirm catheter position and/or detect pneumothorax. The reference standard was post-procedural CXR and/or computed tomography (CT). To be eligible, a study needed to have reported sufficient data to reconstruct 2 × 2 tables (true positive (TP), false positive (FP), false negative (FN), and true negative (TN)). Eligible designs included prospective or retrospective observational studies and randomized controlled trials. We excluded case reports, animal studies, and studies of peripherally inserted central catheters or femoral venous catheters.
Data extraction and quality assessment
Two reviewers independently extracted study characteristics and 2 × 2 diagnostic data using a standardized form. Risk of bias and applicability were assessed using the Quality Assessment of Diagnostic Accuracy Studies (QUADAS)-2 checklist across four domains (patient selection, index test, reference standard, and flow and timing). Disagreements were resolved via consensus. Detailed QUADAS-2 signaling questions, study-level judgments, and rationales are provided in Supplementary Material 2.
Statistical analyses
Analyses were conducted using Stata 17.0 (StataCorp, College Station, TX, USA) using the MIDAS and METANDI commands. For catheter malposition, we pooled sensitivity and specificity using a bivariate random-effects model and derived pooled positive likelihood ratio (PLR), negative likelihood ratio (NLR), and diagnostic odds ratio (DOR). Overall performance was summarized using the hierarchical summary receiver operating characteristic (HSROC) curve and the area under the curve (AUC). Between-study heterogeneity was evaluated using Cochran’s Q and the I2 statistic. For pneumothorax and time-related outcomes, we reported study-level estimates and did not pool accuracy measures because events were sparse and several studies had zero cells, which can destabilize hierarchical models. Robustness was assessed with leave-one-out analyses, and pooled estimates were not materially changed. Studies with no malposition events (nonestimable sensitivity) or with malposition in all participants (nonestimable specificity) were excluded from quantitative synthesis and meta-regression/subgroup analyses; however, they were retained in descriptive tables. Prespecified univariable meta-regression and subgroup analyses explored heterogeneity using ultrasound protocolization and operator experience. Clinical utility was assessed using likelihood-ratio scattergrams and Fagan’s nomograms. Publication bias was evaluated using Deeks’ funnel plot asymmetry test (p < 0.10 indicated statistical significance).
Results
Study selection and characteristics
We identified 5960 records. After removing 1039 duplicates, 4921 records were screened by title and abstract. Eighty-six full-text articles were assessed for eligibility, and 78 were excluded (reviews/commentaries, n = 28; wrong procedure, n = 23; case reports, n = 21; wrong outcomes, n = 4; and insufficient data, n = 2). Eight studies (801 CVC insertions) were included in the qualitative synthesis.7–9,12–16 Seven studies provided estimable 2 × 2 data for the bivariate meta-analysis of catheter malposition;7–9,12,13,15,16 one study with zero malposition events was summarized descriptively (Figure 1; Additional file 3). 14

PRISMA flow diagram of study selection. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
All included studies were prospective and conducted primarily in EDs. Index tests included multi-view B-mode protocols and agitated-saline–enhanced techniques (e.g. the rapid atrial swirl sign); one study evaluated a protocolized pathway (CVC position confirmation and exclusion of pneumothorax (PCEP)). 9 Both internal jugular and subclavian access sites were represented, and CXR served as the reference standard in all studies (Table 1). Across studies reporting enrollment dates, study periods ranged from January 2009 to June 2017; several studies did not report conduct dates, and this information is summarized in Table 1.
Characteristics of the included studies.

Only the ED subgroup data (n = 75) were extracted for Ablordeppey et al. 9
POCUS: point-of-care ultrasound; ED: emergency department; ICU: intensive care unit; CXR: chest radiography; TEE: transesophageal echocardiography; RASS: rapid atrial swirl sign; IJV: internal jugular vein; SCV: subclavian vein; PCEP: position confirmation and exclusion of pneumothorax; US: ultrasound; VC: venous catheter; NR: not reported; PACS: picture archiving and communication system.
Methodological quality
QUADAS-2 assessment indicated generally lesser concerns regarding applicability. Risk of bias was most commonly rated high in the reference standard domain, reflecting limitations of post-procedural imaging for classifying malposition and pneumothorax, and in patient selection due to convenience sampling (Figure 2).

QUADAS-2 assessment of risk of bias and applicability concerns across included studies. Green indicates low-risk/concern, yellow unclear, and red high. QUADAS: Quality Assessment of Diagnostic Accuracy Studies.
Diagnostic accuracy for catheter malposition
Across seven studies in the bivariate model, POCUS yielded a pooled sensitivity of 0.81 (95% confidence interval (CI): 0.50–0.95) and specificity of 0.99 (95% CI: 0.96–1.00) for detecting catheter malposition (Figure 3(a)). Overall performance was high, with an HSROC AUC of 0.99 (95% CI: 0.97–0.99) (Figure 3(b)) and a pooled DOR of 737.34 (Figure 3(c)). The pooled PLR was 137.83 (95% CI: 22.04–862.02), and the pooled NLR was 0.19 (95% CI: 0.06–0.62) (Figure 3(d)). The likelihood-ratio scattergram suggested strong rule-in capability (Additional file 4).
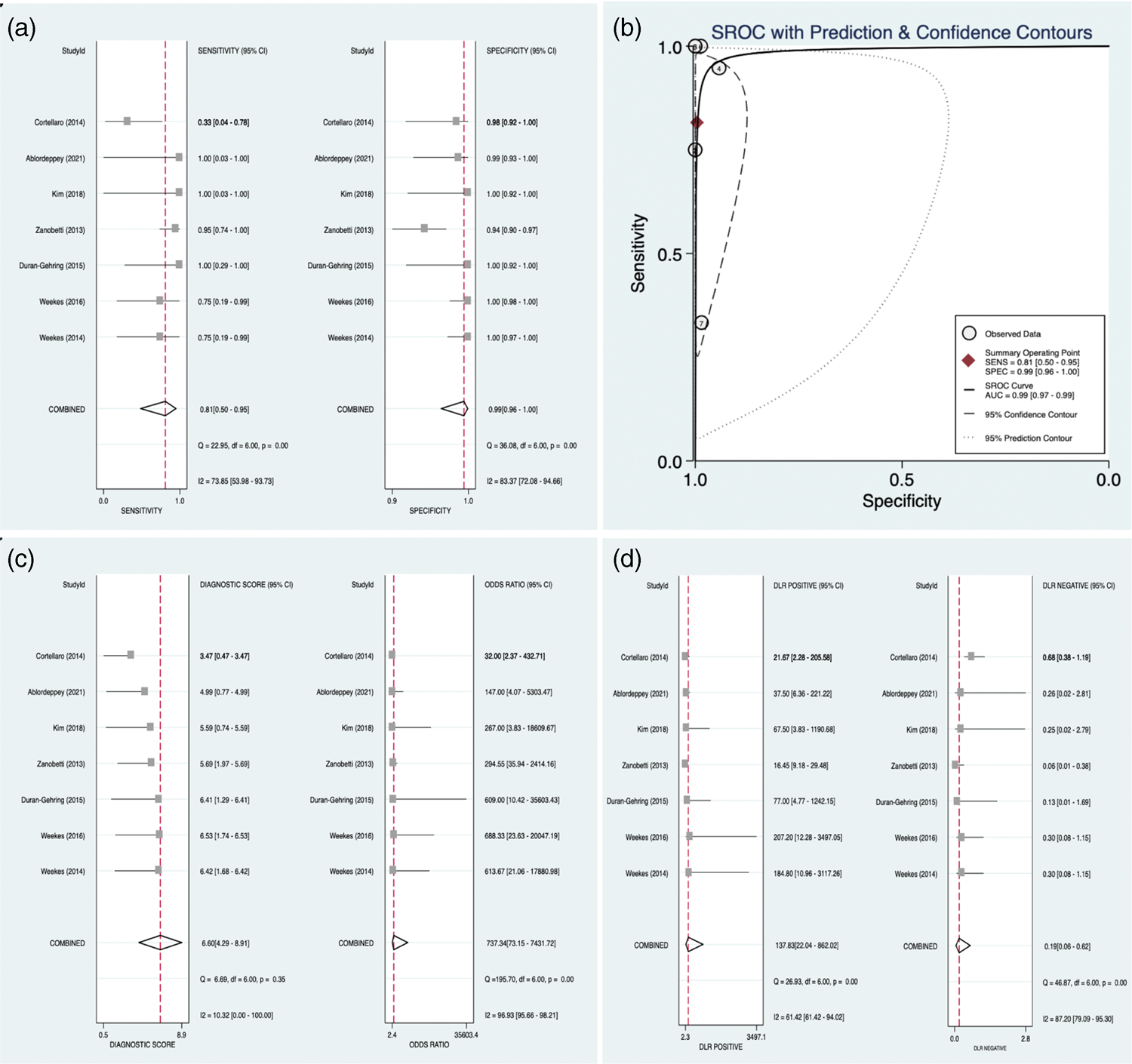
Forest plot of pooled sensitivity and specificity for CVC malposition. (a) Forest plots of sensitivity (top) and specificity (bottom) of point-of-care ultrasound (POCUS) for detecting central venous catheter (CVC) malposition. Squares represent individual studies, and horizontal lines indicate 95% confidence intervals (CIs); diamonds indicate pooled estimates. (b) Hierarchical summary receiver operating characteristic (HSROC) curve with summary operating point and 95% confidence and prediction regions for POCUS detection of CVC malposition. (c) Forest plot of diagnostic odds ratio (DOR) for POCUS detection of CVC malposition. The diagnostic score corresponds to log (DOR). (d) Forest plots of positive likelihood ratio (PLR) and negative likelihood ratio (NLR) for POCUS detection of CVC malposition, with pooled estimates.
Heterogeneity and subgroup analysis
Heterogeneity was substantial (sensitivity I2 = 73.85%; specificity I2 = 83.37%). In prespecified univariable meta-regression and subgroup analyses (restricted to studies with estimable 2 × 2 data), protocolized approaches and explicit reporting of operator experience/specification were associated with higher pooled specificity, whereas pooled sensitivity did not differ materially between subgroups (Table 2, Figure 4). These findings are exploratory and should be interpreted cautiously given the small number of studies per subgroup.
Univariable meta-regression and subgroup analyses to identify potential sources of heterogeneity.
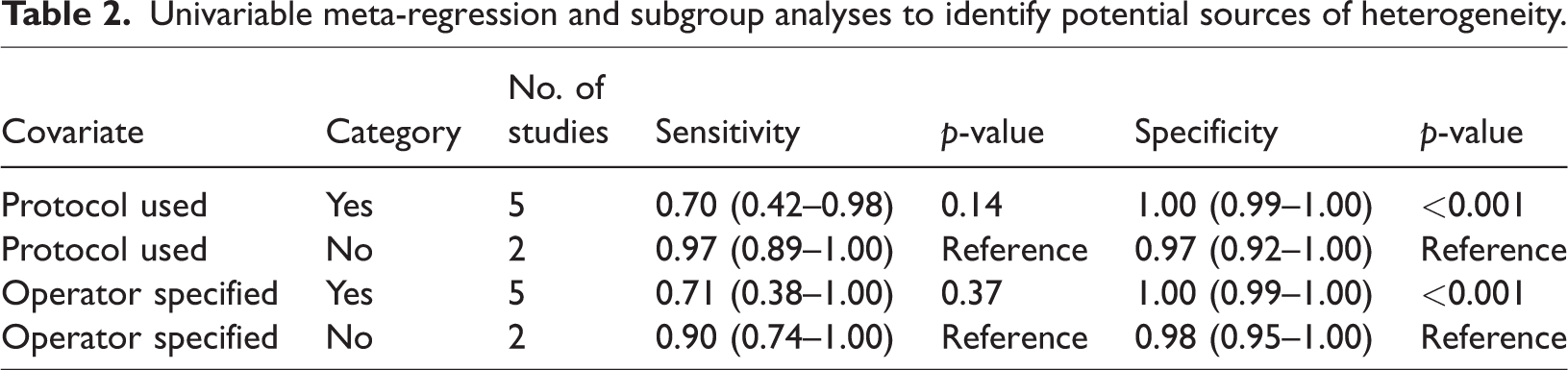

Univariable meta-regression and subgroup analyses exploring heterogeneity in sensitivity and specificity according to protocol use and operator factors.
Clinical utility
At a pretest probability of 50%, a positive POCUS result increased the post-test probability of malposition to 99%, whereas a negative result reduced it to 16% (Additional file 5).
Secondary outcomes
For pneumothorax detection after CVC insertion, study-level specificity was consistently high (97.2%–100%). Sensitivity was 100% in studies with events, nonestimable in zero-event studies, and 25% in one study (Table 3). Across studies reporting workflow metrics, POCUS was faster than CXR, with time savings ranging from approximately 5 to 284 min (most commonly, 20–60 min) (Table 4). Pneumothorax events were uncommon, and severity (e.g. size, need for tube thoracostomy, and tension physiology) was inconsistently reported; when pneumothorax occurred, it was described as iatrogenic after CVC insertion (e.g. 3 cases in Zanobetti et al.). 16
Diagnostic accuracy of POCUS for detecting pneumothorax after CVC insertion (study-level 2 × 2 data).

TP: true positive; FP: false positive; FN: false negative; TN: true negative; POCUS: point-of-care ultrasound; CVC: central venous catheter; ID: identification; CI: confidence interval.
Values are study-level diagnostic accuracy estimates for pneumothorax; 95% confidence intervals were calculated using the exact (Clopper–Pearson) method to ensure consistency with the forest plots. Studies with zero target events have nonestimable sensitivity.
Time efficiency of POCUS versus chest radiography (CXR) for post-insertion confirmation and complication screening.

Weekes et al. 12 reported ultrasound “swirl” onset time only and did not report a comparable CXR confirmation time; therefore, it is presented descriptively and was not used to describe time savings.
Only the ED subgroup data (n = 75) were extracted for Ablordeppey et al. 9
ID: identification; POCUS: point-of-care ultrasound; US: ultrasound; ED: emergency department.
Publication bias
Deeks’ funnel plot asymmetry test was not significant (p = 0.86); however, the small number of studies limited the power to detect publication bias (Additional file 6).
Discussion
In this diagnostic test accuracy meta-analysis of eight prospective studies (801 CVC insertions), POCUS showed high specificity (0.99) and overall discrimination ability (AUC 0.99) for detecting post-procedural CVC malposition. The large, pooled PLR and DOR support strong rule-in performance. Sensitivity was 0.81 with substantial s, and the pooled NLR of 0.19 suggests that a negative examination reduces—yet does not eliminate—the probability of malposition.
Our findings align with the results of a previous meta-analysis by Ablordeppey et al. (pooled sensitivity = 0.82 and specificity = 0.98) 17 and with the critical care synthesis by Smit et al., which similarly reported high specificity and emphasized faster confirmation compared with radiography. 18 Notably, several foundational prospective studies underpinning these prior syntheses have compared multi-view vascular/cardiac ultrasound strategies—often incorporating saline flush or contrast-enhanced “bubble” techniques—with CXR, consistently demonstrating high specificity in both ICU and ED settings.7,16,19 The higher pooled sensitivity observed in our analysis may reflect greater use of standardized “swirl/bubble” confirmation (rapid right-heart opacification after flush) and broader integration of lung ultrasound for immediate pneumothorax screening. Because malposition is typically infrequent, sensitivity estimates can be disproportionately influenced by small changes in FN counts, which may have contributed to the observed heterogeneity across studies and to the imprecision noted in previous reviews. 18
An additional driver of apparent heterogeneity is the reliance on CXR as the reference standard in most included studies. CXR is an imperfect surrogate for true cavo-atrial junction (CAJ) positioning, and inter-reader variability in identifying the CAJ has been documented. 20 Moreover, conventional bedside radiograph interpretation may be suboptimal for the detection of intra-atrial tip positioning. 21 These limitations may bias diagnostic accuracy estimates in either direction and plausibly contributed to the higher risk of bias in the QUADAS-2 reference standard domain. Importantly, clinical acceptability of “ideal” tip location remains debatable, and the clinical implications of marginally deeper placement (e.g. right atrium) are context-dependent.22,23 Accordingly, future studies validating POCUS against more definitive standards (e.g. fluoroscopy, CT, transesophageal echocardiography (TEE), or electrocardiography (ECG)-guided tip confirmation) are needed to strengthen inference.
These findings should also be interpreted in the context of current standards for CVC tip location. Contemporary guidance generally considers the lower third of the superior vena cava, CAJ, or high right atrium as acceptable target positions for upper-body CVC tips while emphasizing avoidance of excessively deep placement and the importance of catheter type and clinical purpose. This issue is directly relevant to our review because several included studies had used CXR as the reference standard; however, radiographic definitions of “correct” tip position are not uniform across studies and may not always reflect current anatomical and practice standards. 24
In addition, the present results should be viewed in the context of published structured ultrasound protocols for tip navigation and tip location. The ECHOTIP protocol was proposed as a standardized framework for ultrasound-based tip navigation and final tip location during placement of central venous access devices in adult patients. 25 More recent work has revisited and refined this approach, reinforcing that ultrasound confirmation should be considered a part of a protocolized workflow that may include intraprocedural navigation, saline–enhanced echocardiographic visualization, and immediate screening for complications. 25 In this context, the high pooled specificity observed in our meta-analysis supports the role of POCUS as a clinically useful component of a standardized post-placement confirmation pathway.25,26
From a bedside decision-making perspective, the consistently high specificity supports the use of a positive POCUS result to trigger prompt corrective action—such as withholding high-risk infusions, repositioning, or recannulation—without waiting for radiography results when time-sensitive therapies are required. Evidence supporting saline flush/bubble strategies indicates that bedside confirmation can be performed rapidly and reproducibly when cardiac windows are adequate.8,12,14,27,28 In parallel, lung ultrasound offers immediate screening for pneumothorax, with classic signs such as lung sliding (to rule out) and the lung point (highly specific to confirm) supporting a bundled post-procedural scan.29–31 Meta-analyses comparing ultrasound and radiography for pneumothorax diagnosis further reinforce ultrasound’s diagnostic advantage, particularly in supine or critically ill patients.31,32 Operationally, these findings support a pragmatic post-insertion “POCUS-first” pathway consistent with published recommendations. 33 CXR may be deferred in clinically stable patients when a standardized protocol demonstrates the following: (a) immediate right-heart opacification/swirl after a rapid saline flush consistent with central venous drainage and (b) no sonographic evidence of pneumothorax (bilateral lung sliding and/or B-lines). Conversely, CXR (and/or CT/TEE) should be obtained when ultrasound is incomplete or equivocal (suboptimal windows, indeterminate swirl, and uncertain laterality), when clinical suspicion remains high (multiple attempts, aberrant anatomy, and unexpected respiratory/hemodynamic changes), or before initiating high-risk infusions when definitive tip localization is required.17,33 This approach is also supported by evidence suggesting that routine CXR is unnecessary after certain low-risk, ultrasound-guided insertions, particularly right internal jugular cannulation, when bedside ultrasound confirmation is adequate. 6 Most primary studies have described CXR as “portable” or “digital mobile” without providing detailed technical specifications or workflow characteristics. Although several studies have reported the use of digital mobile systems, detailed information regarding image acquisition processes or immediate bedside image availability was generally lacking. Because radiography system characteristics were inconsistently reported and not amenable to subgroup analysis, comparisons of CXR turnaround time with POCUS should be interpreted as workflow- and context-dependent.
Implementation feasibility depends on protocol standardization and operator competency. International training standards for critical care ultrasonography and validated competency tools provide a framework for structured education and quality assurance, which may explain why subgroup estimates in prior work show trainees can achieve high specificity under supervised protocols.34,35 Embedding a brief, checklist-based multi-view confirmation bundle into ED workflows may therefore be a high-yield target for dissemination.
Furthermore, notably, several studies included in this meta-analysis utilized modern CVCs equipped with echogenic tips (e.g. Blue FlexTip catheters), which may have contributed to enhancing the diagnostic accuracy of POCUS.
Advances in catheter design may also influence sonographic tip visualization. Some studies described the use of echogenic-tip catheters engineered with high-echo materials to enhance ultrasonographic conspicuity.7,15,16 Although such technology may facilitate B-mode identification of catheter tips, catheter characteristics were not consistently reported and were not incorporated into the pooled analyses; therefore, their precise contribution to overall diagnostic performance remains uncertain.
From a systems perspective, the integration of immediate ultrasound confirmation into post–central venous catheterization workflows may streamline ED processes, reduce radiation exposure, and shorten time to catheter utilization. These benefits may be particularly relevant in high-acuity or resource-limited settings. However, successful implementation depends on operator training, adherence to standardized scanning protocols, and institutional workflow integration.
Certain limitations merit emphasis. First, the relatively small number of studies and low event rates limited precision and yielded wide CIs for sensitivity; small changes in FN counts can materially affect pooled estimates. Second, all included studies used CXR as the reference standard; because CXR is not definitive for tip localization or subtle pneumothorax, misclassification may have biased accuracy estimates in either direction. Future studies should validate POCUS against more definitive reference standards (e.g. fluoroscopy, CT, TEE, and ECG-guided tip confirmation). Third, differences in ultrasound protocols, operator expertise, and patient populations likely contributed to heterogeneity; meta-regression was underpowered and exploratory. Fourth, Deeks’ test has limited power when few studies are available. Finally, most data were reported at the procedure level, and repeated catheterizations within the same patient could not be accounted for, which may have affected variance estimates. Despite these limitations, consistently high specificity across studies supports a clinically useful rule-in role for a positive POCUS result. We also noted incomplete reporting of study conduct dates and pneumothorax severity across primary studies, which limited temporal comparisons and precluded stratified analyses by clinically significant pneumothorax.
Conclusions
POCUS provides highly specific, rapid confirmation of CVC malposition, supports rule-in decisions, and may accelerate post-procedural workflow. Given the heterogeneous sensitivity and limitations of available reference standards, negative results should be interpreted cautiously, and confirmatory imaging may remain appropriate only in selected high-risk scenarios.
Footnotes
Acknowledgments
This manuscript was polished for language clarity using Grammarly Premium and ChatGPT (OpenAI).
Author contributions
Conceptualization: Xianhua Zhu, Weiting Chen
Methodology: Xianhua Zhu, Xiaowei Wang, Gang Yang, Weiting Chen
Systematic search & study selection: Yangtian Ye, Yaling Jin, Jiuzhou Lin, Min Tang
Data curation: Yangtian Ye, Yaling Jin, Jiuzhou Lin, Min Tang
Risk of bias/quality assessment: Lihui Chen
Formal analysis: Xiaowei Wang, Yangtian Ye
Visualization (figures/tables): Xiaowei Wang, Yangtian Ye
Writing–original draft: Xiaowei Wang, Xianhua Zhu, Gang Yang
Writing–review & editing: Weiting Chen, Xiaowei Wang; all authors contributed to critical revision of the manuscript.
Supervision: Xiaowei Wang, Gang Yang, Xianhua Zhu, Weiting Chen
Project administration: Xiaowei Wang, Weiting Chen
All authors read and approved the final manuscript and agree to be accountable for all aspects of the work.
Data availability statement
Data are contained within the article.
Declaration of conflicting interests
The authors declare no conflicts of interest.
Funding
This research received no external funding.
Institutional review board statement
Not applicable.
Informed consent statement
Not applicable.
Supplementary information
Additional file 1: Search strategy (full electronic search strategies and search history). Additional file 2: QUADAS-2 risk of bias and applicability assessment (study-level judgments and rationale).
