Abstract
Introduction:
People who use anabolic-androgenic steroids (AAS) face health risks from illicit markets and limited tailored harm reduction services. This study aimed to examine whether laboratory testing results from submitted AAS products (ie, compound presence and purity) influence AAS consumers’ future behaviours.
Methods:
As part of Wave 3 of a Steroid Testing Trial (2024), community members in Queensland, Australia, submitted AAS samples for laboratory analysis. Using advanced forensic techniques, we tested these samples for compound presence and dosage accuracy. Participants who accessed their results completed a follow-up survey on demographics, AAS use, healthcare access, and response to results. Behaviour change was measured by self-reported modifications to use. Logistic regression examined predictors including perceived helpfulness, confidence to change, quality concerns, prior health effects, and healthcare access barriers.
Results:
Of 105 AAS samples analysed, 22% were mislabelled and 50% were underdosed. Most participants who engaged with testing results (N = 117) were men (97.4%), with a mean age of 32, and about half resided in Australia. Nearly all (95.7%) found results helpful, and 45.3% reported behaviour changes, including adjusting dosage, source, or testing compounds before use. Although the logistic regression model was not significant overall, concern about AAS quality predicted behaviour change (odds ratio [OR] = 1.57, P = .026), while perceived helpfulness, confidence, prior health effects, and healthcare barriers were not significant.
Conclusions:
Concern about product quality appears to drive harm reduction behaviours more than perceived helpfulness or confidence. These findings support the value of AAS in drug testing data dissemination that highlights product quality issues.
Keywords
Highlights
Illicit anabolic-androgenic steroid markets expose consumers to mislabelled and inaccurately dosed products.
Community members submitted anabolic-androgenic steroid samples for laboratory testing and received aggregated testing results.
Exposure to anabolic-androgenic steroid testing results prompted many consumers to consider modifying their use.
Concern about product quality appears to drive health behavior intentions among anabolic-androgenic steroid consumers.
Introduction
People who use image and performance-enhancing drugs (IPEDs), including anabolic-androgenic steroids (AAS), peptides, insulin, and other diverted pharmaceuticals, represent a growing and under-recognised public health population.1,2 These substances are often used to enhance appearance, performance, or wellbeing, with more than 70% administered via injection, though oral and subcutaneous routes are also common.3,4 Global evidence suggests more than two-thirds of IPED products are adulterated or mislabelled, 5 exposing consumers to potentially serious health risks, including bacterial infections, endocrine disruption, cardiovascular complications, and toxic effects from unknown ingredients.3,6 Illicit AAS markets operate on a global scale, with some “underground brands” shared across international borders.7-9 While certain brands and products are specific to Australia, 10 the landscape is evolving rapidly, with increasing internationalisation resulting in greater overlap between local and global markets. 11 Given this interconnected supply, people who use AAS worldwide often face similar uncertainties regarding product authenticity and purity.5,12-14 Many consumers have been deprived of objective, evidence-based information about the substances they use, creating a strong demand for reliable data.6,15-18 Access to any form of chemical analysis or testing information, whether locally or internationally sourced, is highly valued by consumers seeking to reduce harm and make informed choices. Yet despite this demand for trustworthy information and the global scale of use, tailored harm reduction responses remain limited.
Despite the scale of use, estimated at 6.4% of men 19 and 4% of women 20 globally, people who use IPEDs remain underserved by mainstream harm reduction and healthcare systems. Existing strategies are largely adapted from models designed for people who use opioids or stimulants, and often fail to reflect the distinct motivations, patterns of use, and risks associated with IPEDs.21-23 For example, while blood-borne virus prevention remains important, consumers are equally concerned with optimising injection technique, enhancing drug absorption, and avoiding counterfeit substances. However, engagement with formal health services remains limited. People who use IPEDs frequently report feeling judged, misunderstood, or deprioritised in care settings. Stigma, misinformation, and a lack of tailored services contribute to disengagement from care, 24 particularly among women 20 and regional populations.25,26 In this context, peer networks play a vital role in harm reduction, providing informal education, distributing equipment and AAS products, and supporting decision-making. 27 However, these networks often operate without access to verified or evidence-based information, leaving people vulnerable to harm despite their proactive efforts.10-12,28-30
One promising harm reduction intervention is drug checking, which provides analytical feedback on the content and composition of illicit substances. 31 While drug checking is increasingly available for substances such as MDMA and methamphetamine, AAS have rarely been included in such programmes.13,32 In response, a pilot steroid checking initiative was launched in Queensland, Australia, offering the first formal AAS-specific drug checking trial internationally. The initial phase (Wave 1) provided grouped chemical analysis of consumer-submitted AAS samples. Though well-received, it had limitations: consumers were unable to link results to brand names or interpret variability across batches, reducing the usefulness of findings for individual risk management. Wave 2 addressed some of these concerns, introducing more precise reporting and exploring how purity data influenced behaviour. This study investigates how people who use AAS engage with AAS testing results to inform their harm reduction practices. It further examines the extent to which collective information on product purity, alongside healthcare access barriers and prior adverse health experiences, predicts behavioural adaptations among this cohort.
Method
Study Design and Ethics
This study forms the third wave (“Wave 3”) of the World’s First Steroid Checking Trial and builds on previous waves by evaluating how consumers responded to the full dissemination of community testing results. Unlike Waves 1 and 2, which relied on qualitative interviews, Wave 3 employed a two-phase sequential observational design via a cross-sectional online survey to capture broader, international perspectives on how people who use AAS interpreted and responded to the released data. While no qualitative data were collected in this wave, previous qualitative findings helped shape survey items and behavioural indicators (see below). This survey also allowed for exploration of behaviour change intentions and health-related attitudes following the availability of AAS testing results. Ethical approval was granted by the respective Human Research Ethics Committees (Griffith University HREC: 2023/784; The University of Queensland HREC: 2025/HE001496). All procedures followed the principles of the Declaration of Helsinki and complied with national standards for research involving human participants. Reporting follows the STROBE guidelines 33 for observational research.
Phase 1: AAS Testing and Community Dissemination
AAS products were voluntarily submitted to CheQpoint drug checking services located in Brisbane and the Gold Coast, Queensland. Participants submitted used and unopened vials, oral tablets, and capsules, which were securely logged and stored before transport to the University chemistry laboratory. Initial screening employed the Radian-Advanced Spectral Analysis Platform or Fourier-Transform Infrared spectroscopy to detect active compounds. Confirmatory testing was conducted using Orbitrap Liquid Chromatography–Mass Spectrometry, a validated and reproducible method employed previously,13,14 ensuring accuracy of compound identification and dosage quantification. Results were reviewed by senior chemists and cross-checked against national and international standards. Following the analysis, results were disseminated to the community through posters at submission sites, peer-led outreach, and targeted online channels. 34 The specifics of this process can be found in previous “Waves” of this programme of work.13,14 These findings became the basis for Phase 2 of the study.
Phase 2: Online Survey
Recruitment and Sampling
Participants over the age of 18 who have used AAS in their lifetime and engaged with the AAS checking report were invited to participate in an online survey assessing their response to the data released to the community from the World’s First Steroid Checking Trial.13,34 All participants were required to have engaged with the Wave 3 steroid checking data report prior to following a link, embedded in the report to the survey. Recruitment occurred between February and May 2025 via social media, community events, and fitness influencer digital networks. Members of the research team with lived-living experience also promoted the study through relevant peer networks. A total of 468 participants completed the survey. Recruitment materials directed participants to an online consent form, and participation was entirely voluntary.
Procedure
Participants were presented with Wave 3 of the trial results, containing all trial data (See Supplemental Materials – Appendix A), and were notified of relevant support services should they experience distress throughout completion of the survey, and informed that confidentiality would be safeguarded, unless the research team were legally required to present specific information under subpoena. Participants were informed that should they agree to participate they were free to withdraw consent at any time without penalty. Demographic questions were presented first, followed by a block of questions that aimed to understand participants’ responses to the Wave 3 data presented to the community, estimated to take 10 minutes to complete. These included questions assessing perceived usefulness of the results (eg, “Did you find the testing results for AAS helpful?”), quality-related concerns (eg, “How concerned are you about the quality of AAS in circulation?”), and behavioural intentions following exposure to the results (eg, “Did the results prompt you to reconsider your AAS usage in any way?”, with follow-up items relating to dosage, compound selection, or sourcing). The survey was distributed between February and May 2025 and promoted through community events, conferences, and social media. Given the hard-to-reach nature of the study population, members of the research team with lived-living experience also actively promoted the programme online.
Statistical Analysis
All analyses were conducted using IBM SPSS Statistics Version 30. Responses with missing data or no data were removed, leaving only complete cases (N = 117). Data demonstrated that 478 cases of the survey were commenced. Data was removed for 5 tests and/or previews, 3 participants did not consent (and the survey subsequently ended), and 238 surveys were commenced but had no response. Response information for 107 participants, who only completed demographic information and/or Question 1, was removed. Of those remaining, additional responses were removed due to participants aged < 18 (5), not ever used AAS. A total of 117 participants remained and were used in the analysis.
Descriptive statistics were calculated to summarise participant responses regarding the perceived helpfulness of AAS testing results, subsequent confidence in behaviour change, and reported concerns about the quality and safety of AAS. A composite binary outcome variable was created to capture whether participants reported any behaviour change in response to the testing results. Participants who selected any of the following were coded as having changed their behaviour: reconsidering the number of compounds used, adjusting dosage, changing source, or testing compounds prior to use, consistent with behavioural indicators identified in previous research.13,14 Those who indicated no change were coded accordingly. A binary logistic regression model was then conducted to examine whether perceptions of test helpfulness, confidence in the ability to make changes, concern about AAS quality, and previous experience of negative AAS-related health effects predicted behaviour change. Odds ratios (ORs) with 95% confidence intervals (CIs) were reported. Model fit was assessed using the Hosmer–Lemeshow test and pseudo-R2 values. Cross-tabulations with chi-square tests were used to explore bivariate relationships between perceived helpfulness and behaviour change.
Results
Phase 1
Wave 3 comprised a total of 128 samples, which were submitted for analysis, of which 105 were suitable for chemical analysis. Of the 105 samples successfully analysed, 78% (n = 82) contained the expected AAS compound, while 22% (n = 23) contained an unexpected or mislabelled substance. Purity data, that is, dosage accuracy, were available for 83 samples. Among these, 50% (n = 29) were underdosed, 32% (n = 26) were correctly dosed, and 8% (n = 7) were overdosed. A further 25% (n = 21) were unable to be quantified due to containing different or unexpected substances. See Table 1 for further details.
Cumulative Summary of AAS Testing Results Over 1 Year Trial.
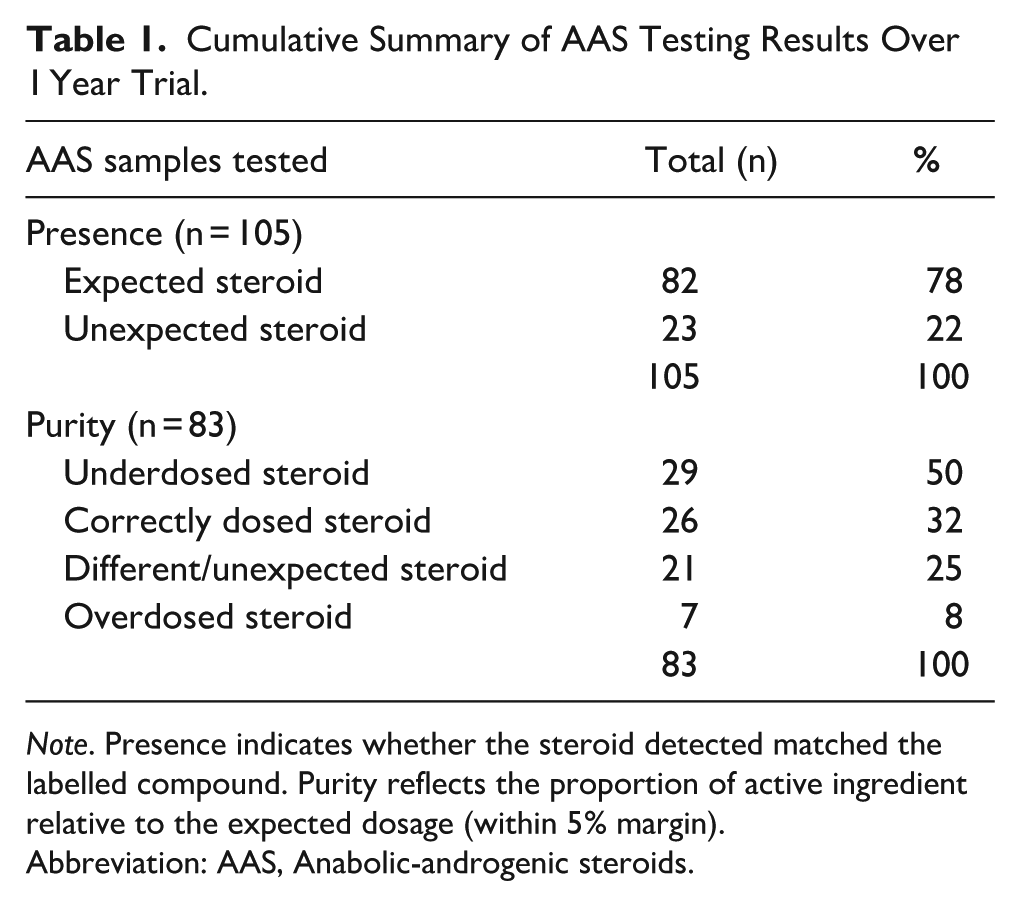
Note. Presence indicates whether the steroid detected matched the labelled compound. Purity reflects the proportion of active ingredient relative to the expected dosage (within 5% margin).
Abbreviation: AAS, Anabolic-androgenic steroids.
Phase 2
Participant Characteristics and Usage Patterns
Participants (N = 117), aged 18 and over (n = 101, Mage = 32, SD = 8), completed an online survey if they had used AAS in their lifetime. Most participants were males (n = 114, 97.4%; female n = 3, 2.6%), and 59% resided outside of Australia (n = 69; USA = 34, Europe = 23), and of those in Australia, 20.5% were from Queensland (n = 24), 7.7% were from Victoria (n = 9), 3.4% were from Western Australia (n = 4), and 1.7% each from South Australia (n = 2) or Australian Capital Territory (n = 2). Participants were either currently using AAS (n = 104) or had used it in the past 12 months but were on an extended break (n = 13). Most (n = 62) had a combination of 3 types of usage strategy (ie, associated with dose, cycle/cruise, compound/s). Participants used 2 to 3 compounds most frequently (n = 83), followed by 4 to 5 (n = 18), then 0 to 1 (n = 15), and only 1 participants reported using more than 6 compounds, in terms of dosage, most individuals used between 251 to 500 mg (n = 29) and 501 to 750 mg (n = 29), followed by 1000+ (n = 20), then 741 to 1000 mg (n = 19), 156 to 250 mg (n = 13), and 0 to 125 mg. Which is reflected in the usage strategies, where most individuals would use a “blast-cruise” method (n = 67) or cycle (n = 21), which is typically done at higher doses, compared to those who permanently cruise (n = 19).
Two-thirds (67.5%, n = 79) reported experiencing negative side effects from AAS use, while 32.5% (n = 38) had not experienced adverse health effects. Regarding access to healthcare, 62.4% (n = 73) reported no difficulty accessing health services. Among the 35% (n = 41) who did experience difficulties, most reported barriers to a single service type (17.9%), followed by 2 services (8.5%, n = 10), 3 services (6.0%, n = 7), and 4 or more services (2.6%, n = 3). The most common service access issues involved general practitioners (29.9%, n = 35), followed by pharmacies (13.7%, n = 16), needle and syringe programmes (10.3%, n = 12), and hospitals (9.4%, n = 11).
When asked about the types of information or support they would like, participants reported wanting: safer usage guidelines (69.2%, n = 81), information on health risks (52.1%, n = 61), and peer-led education (57.3%, n = 67).
Behavioural Responses to AAS Testing Results and Predictors of Change
Of the 117 participants, the vast majority (95.7%, n = 111) reported that they found the AAS testing results helpful. Nearly half (45.3%, n = 53) indicated that they changed at least one aspect of their AAS use following receipt of the testing results. These behaviour changes included reducing the number of compounds used (19.7%), adjusting dosage (14.5%), changing source (12.0%), or testing compounds prior to use (22.2%) (see Table 2). The logistic regression model predicting behaviour change was not statistically significant overall, χ 2 (5) = 9.20, P = .101, but demonstrated acceptable goodness-of-fit (Hosmer–Lemeshow test: χ 2 (8) = 11.47, P = .177). The model accounted for approximately 10.4% of the variance in behaviour change (Nagelkerke R2 = 0.104).
Descriptive Frequencies of Behaviour Change Variables.
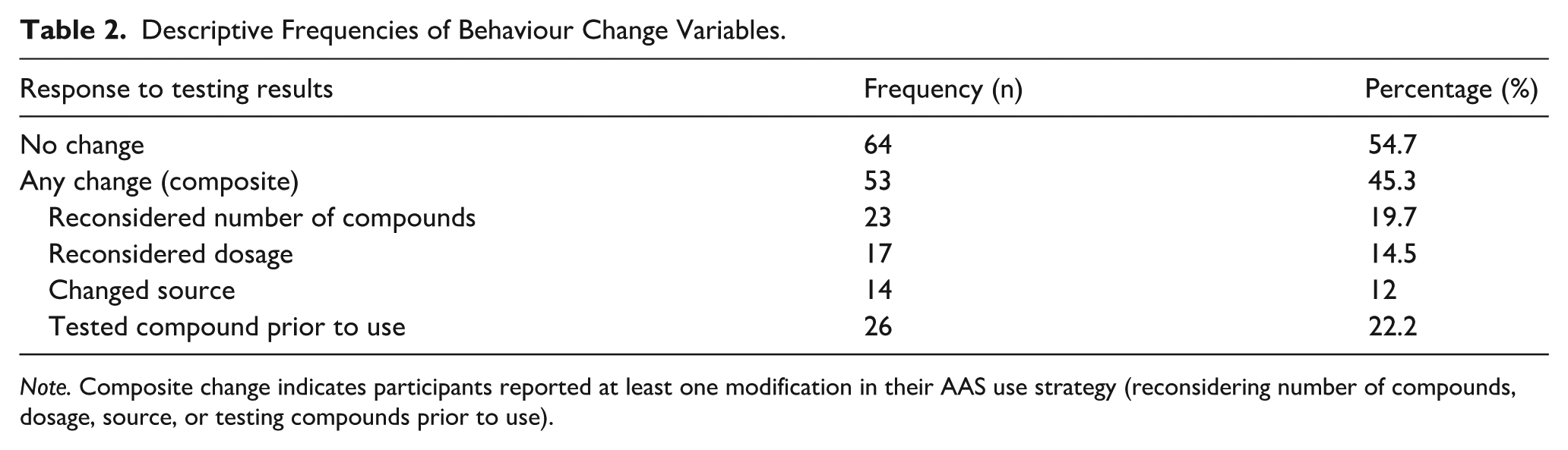
Note. Composite change indicates participants reported at least one modification in their AAS use strategy (reconsidering number of compounds, dosage, source, or testing compounds prior to use).
Among the predictors included, only concern about the quality of AAS was significantly associated with reported behaviour change (OR = 1.57, 95% CI [1.06, 2.32], P = .026). Specifically, individuals who reported concern about AAS quality were more likely to report modifying their use practices, such as reducing the number of compounds, adjusting dosage, changing source, or testing their products, following receipt of the community testing results. Neither perceived helpfulness of the testing results, confidence in the ability to change, prior negative health effects, nor difficulties accessing health services demonstrated statistically significant associations (see Table 3). Although behaviour change was more commonly reported by those who found the testing results helpful (47%) compared to those who did not (20%), this difference was not statistically significant (χ2(1) = 1.39, P = .238).
Logistic Regression Predicting Reported Behaviour Change.
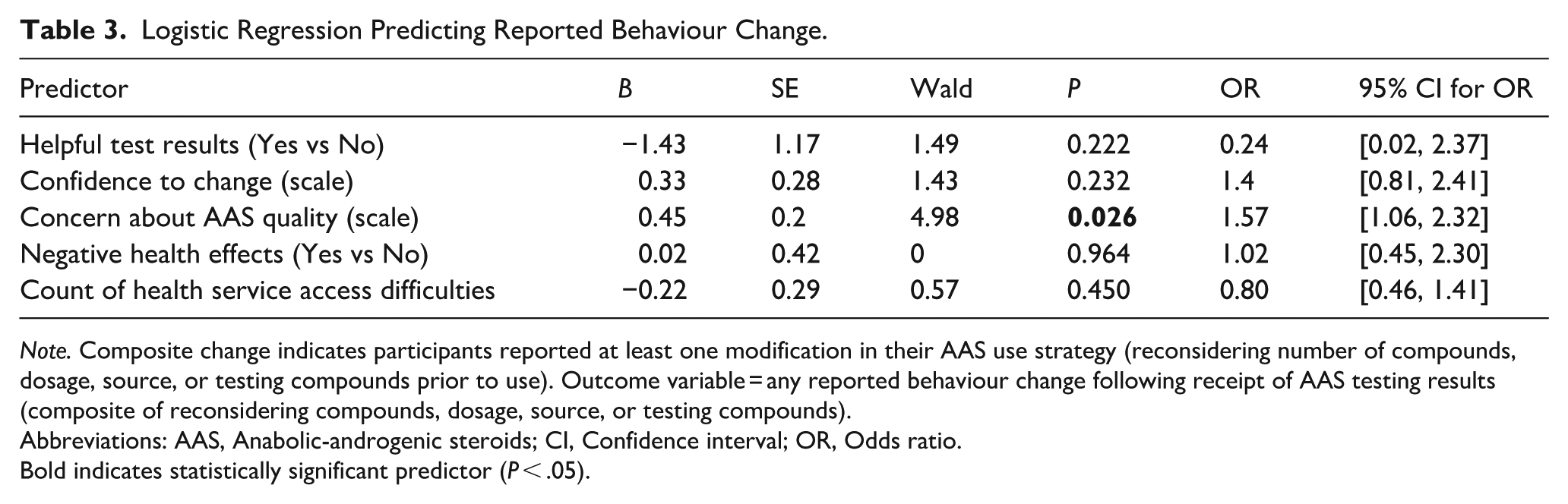
Note. Composite change indicates participants reported at least one modification in their AAS use strategy (reconsidering number of compounds, dosage, source, or testing compounds prior to use). Outcome variable = any reported behaviour change following receipt of AAS testing results (composite of reconsidering compounds, dosage, source, or testing compounds).
Abbreviations: AAS, Anabolic-androgenic steroids; CI, Confidence interval; OR, Odds ratio.
Bold indicates statistically significant predictor (P < .05).
Discussion
This study provides new empirical insights into how people who use AAS engage with testing results and the degree to which these data may influence self-reported harm reduction behaviours. While nearly half of the participants reported making changes based on their results, it is important to note that the predictive model did not yield statistically significant findings. Nonetheless, the consistently positive reception of testing suggests that AAS checking may hold value as a harm reduction tool. With further evaluation, particularly in more representative samples, such services could play a role in supporting safer decision-making in this historically underserved population.
Our chemical analysis confirmed substantial product adulteration and dosage variability, with 22% of samples containing unexpected substances and half of the quantified samples being underdosed. This aligns with global research documenting widespread IPED product mislabelling and the resultant health risks.5,12,32 The high prevalence of adulterated or inaccurately dosed products underscores the urgent need for accessible, evidence-based interventions to improve consumer knowledge and safety. The current results also highlighted that concern about AAS quality significantly predicted behaviour change, while perceived helpfulness of the testing results and confidence in the ability to change did not reach statistical significance. It is possible that increased confidence in the product itself could, in some contexts, reinforce use rather than reduce harm, and these effects may therefore be dependent on individual circumstances and usage patterns. This suggests that cognitive appraisal of product safety risks may be a more salient motivator for behavioural adaptation than the mere reception of test results or self-efficacy alone. It also signals that raising awareness about product adulteration may be a crucial target for harm reduction messaging and peer-led education efforts. However, to raise awareness of product alteration, public dissemination of AAS testing data is of critical importance and, therefore, these programmes have an important place across all areas of the world where AAS consumption is prevalent. Notably, the utility of this information appears to extend beyond local contexts. Consumers in other jurisdictions may encounter the same compounds, under similar labels, and face comparable risks. In this way, chemical analysis results, even when geographically specific, may serve broader harm reduction purposes, especially for populations that have long lacked access to any verified data about the substances they use. Participants’ high engagement with the findings, despite differing locations, speaks to the global appetite for actionable, evidence-based information within AAS-using communities.
Prior negative health effects and reported difficulties accessing health services were not significantly associated with behaviour change. This finding may reflect complex barriers within healthcare systems2,35-37 or the nuanced ways individuals interpret health risks in relation to behavioural intentions.37,38 For instance, in our study, while over two-thirds of participants reported experiencing negative side effects, these experiences alone did not translate into increased likelihood of change following receipt of test results. This highlights the potential limitations of individual-level health experiences to drive proactive harm reduction without concurrent structural support or tailored interventions. In a similar vein, the absence of a significant association between healthcare access barriers and behaviour change may also reflect the resilience and self-reliance observed within peer networks in IPED-using communities.8,21,22,39 Peer-driven harm reduction initiatives remain central to disseminating practical knowledge and countering stigma that often limits engagement with formal healthcare. 24 Nonetheless, structural challenges in healthcare access persist11,40 and warrant ongoing attention in policy and practice to ensure equitable support for this population.
This study contributes to the emergent literature on IPED-specific harm reduction by demonstrating the feasibility and potential impact of AAS checking programmes. The findings support the argument that drug checking interventions must move to encompass a broader spectrum of substances and consumer needs, and the AAS consumer community is where this may occur next. Importantly, the approach taken here, combining rigorous chemical analysis with community dissemination and behavioural evaluation, offers a model for future harm reduction initiatives targeting IPED consumers.
Limitations
Several limitations should be acknowledged. The cross-sectional design precludes causal inferences about the impact of test results on behaviour change and means that outcomes reflect short-term behavioural intentions rather than longer-term changes. The study also cannot account for context-dependent effects, such as how increased confidence in product quality might, in some circumstances, reinforce AAS use rather than reduce harm. The sample, while international and diverse, was recruited through community networks and social media, potentially biasing towards more engaged or informed individuals. The small sample size limited statistical power, reducing the ability to detect significant predictors of behaviour change. In addition, the sample comprised predominantly men, which limits the generalisability of findings to women and other gender groups; for example, gender-specific pathways in response to test results may differ but could not be adequately explored here. The self-reported nature of behaviour change and health outcomes may also be subject to recall bias. While this study represents the largest collection of community-submitted AAS samples to date, concentration may vary slightly between batches or across regions. However, concentrations observed in Queensland samples are broadly consistent with findings from Switzerland, the only other jurisdiction with a comparable AAS testing pilot. 41 Future research should consider longitudinal designs to assess sustained behavioural adaptations, recruit larger and more gender-diverse samples, and integrate objective measures of health outcomes.
Conclusions
This study points towards the potential role that verified, accessible drug testing information can play in empowering people who use AAS to reduce risks associated with illicit AAS markets. Concerns about product quality emerged as a key driver of behavioural change, highlighting the need for harm reduction strategies that emphasise transparency and education around substance authenticity and purity. These findings underscore the need for the expansion and integration of AAS-specific drug checking within broader harm reduction frameworks, alongside sustained efforts to address healthcare access barriers and support peer-led community initiatives.
Supplemental Material
sj-docx-1-saj-10.1177_29767342261427145 – Supplemental material for Behavioural Intentions Associated with Exposure to Anabolic-Androgenic Steroid Testing Results: A Two-Phase, Multi-Wave Intervention Trial
Supplemental material, sj-docx-1-saj-10.1177_29767342261427145 for Behavioural Intentions Associated with Exposure to Anabolic-Androgenic Steroid Testing Results: A Two-Phase, Multi-Wave Intervention Trial by Timothy Piatkowski, Ross Coomber, Cameron Francis, Emma Kill, Geoff Davey, Sarah Cresswell, Alan White, Madeline Harding, Karen Blakey, Steph Reeve, Brooke Walters, Cheneal Puljević, Jason Ferris, Benjamin Bonenti, Luke T. J. Cox and Monica J. Barratt in Substance Use & Addiction Journal
Footnotes
Acknowledgements
We sincerely thank the communities of people who use steroids and other enhancement drugs for their invaluable support in this trial, contributing both samples and expertise through consultation. This work would not be possible without them.
Ethical Considerations
Ethical approval was granted by the respective Human Research Ethics Committees (Griffith University HREC: 2023/784; The University of Queensland HREC: 2025/HE001496).
Author Contributions
T.P. conceptualised the study. T.P., C.F., E.K., G.D., S.C., A.W., M.H. coordinated data collection. M.H. conducted chemical analysis, with A.W. and S.C. providing analytical oversight. T.P. and S.R. led data collection and T.P. and B.B. led analysis. J.F., C.P., and M.B. provided methodological guidance. T.P. drafted the manuscript with input from all authors. T.P., B.B., and J.F. verified the data. All authors reviewed, revised, and approved the final manuscript and had final responsibility for the decision to submit for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Queensland Mental Health Commission. TP is supported by a National Health and Medical Research Centre Investigator Grant (2041822). MJB is supported by a National Health and Medical Research Centre Investigator Grant (2042605).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr’s Piatkowski, Puljević, A/Prof Barratt, Prof Ferris, Emma Kill, Alan White, Madeline Harding, and Karen Blakey are volunteer members of The Loop Australia, which is a national organisation for drug checking and drug checking research. Cameron Francis is the CEO of The Loop Australia. Geoff Davey is the CEO of Queensland Injectors Health Network. Emma Kill is the CEO of Queensland Injectors Voice for Advocacy and Action, and Dr Piatkowski is on the Board of Directors of the organisation.
Data Availability Statement
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
