Abstract
Introduction:
Unregulated anabolic-androgenic steroid (AAS) markets present major health risks due to frequent mislabelling and contamination, especially among oral compounds as compared to injectable compounds. Despite growing harm reduction interest, AAS checking remains rare. This study aimed to (1) describe Australia’s unregulated AAS market using community-submitted reports, and (2) compare oral and injectable compounds to identify differences in product legitimacy.
Methods:
We analysed 2068 AAS testing reports submitted to PEDTest, Australia’s only reagent-based AAS checking service, from 2022 to 2024. Participants used community-distributed test kits to assess the presence of AAS in oral or injectable compounds and reported results to the PEDTest database. Each sample was categorised as confirmed, swapped, bunk (no active ingredients), or inconclusive. Chi-square tests examined associations between formulation type (oral vs injectable) and legitimacy outcomes.
Results:
Overall, community-submitted results identified that products were predominantly legitimate (68.8%), swapped (12.9%), inconclusive (11.4%), and a smaller proportion were bunk (7.0%). Oral compounds were significantly more likely to be bunk (10.3%) compared to injectable products (5.6%), while injectable products had a higher chance to be confirmed as legitimate (70.2%) than oral compounds (65.3%). These patterns were supported by a significant overall association between formulation type and legitimacy outcome, χ 2 (3) = 21.29, p < .001.
Conclusions:
Oral AAS are reportedly more prone to quality issues than injectable compounds, with elevated rates of results indicating no active compound presence. These findings underscore the importance of responding to unregulated AAS markets with programmes to empower objective data-driven decision-making among people who use AAS.
Keywords
Highlights
Community-submitted anabolic-androgenic steroid checking reports provide insight into unregulated markets.
A substantial proportion of anabolic-androgenic steroids did not match as expected.
Injectable products were more likely to match expected contents than oral products.
Community drug checking reports can help monitor emerging risks in anabolic-androgenic steroid markets.
Introduction
Anabolic-androgenic steroids (AAS) are used by 3.3% of people worldwide 1 and are consumed among diverse groups, including competitive bodybuilders, powerlifters, and recreational athletes seeking to enhance their physique or athletic performance.1,2 AAS are predominantly used through intramuscular injection, orally (via tablet or capsule), or via gel and patch.3,4 Despite rising use, AAS-related risks are frequently overlooked in public health responses, 5 particularly in relation to unregulated products and access to harm reduction. 2 The unregulated nature of the AAS market exposes people who use AAS to significant risk, with products frequently mislabelled, contaminated, or lacking the claimed active ingredient.2,6 For instance, international studies estimate that between one-third and two-thirds of AAS on the market are counterfeit or substandard6,7, with some products entirely inert, substituted with alternative compounds, or adulterated with potentially harmful substances.2,8,9 These unregulated products sit atop the well-documented physical and psychosocial harms associated with AAS use itself, including dyslipidaemia, hypertension, cardiomyopathy, mood disturbance, and infertility.10-12 When compound and purity are unknown, people may escalate doses, combine multiple products or consume highly hepatotoxic formulations, further increasing the risk of long-term organ damage.2,10,13 Taken together, these uncertainties are believed to markedly heighten the risk of serious, sometimes irreversible health consequences.
People who use AAS rarely have access to formal quality control measures when sourcing products, often relying instead on informal peer networks and anecdotal reputation. 14 This is concerning, given that research elucidates there are discrepancies in the legitimacy of non-medically sourced AAS compounds,2,8,9 defined here as the extent to which a product’s actual contents match its labelled claims. For instance, it is believed oral AAS formulations may be more likely to be misrepresented than injectable products,10,15 yet few people who use AAS seek to verify compound legitimacy prior to use.2,9 Several factors help explain this low engagement with testing. Qualitative work shows that people who use AAS frequently rely on trusted suppliers and peer recommendations, which can diminish the perceived need for formal verification.8,16 Limited awareness of drug checking, perceived inaccessibility and the small number of available services further reduce testing uptake.2,9
In Australia, AAS are classified as controlled or prohibited substances under the Poisons Standard, with criminal penalties enforced across federal and state legislation.17,18 Possession, use, or importation without a prescription is illegal, with penalties ranging from minor possession charges to lengthy imprisonment for commercial trafficking.18,19 Because these products are obtained almost exclusively through illicit, non-medical markets, they are not subject to pharmaceutical manufacturing standards. Packaging may appear professionally branded, but labels are unregulated, often inaccurate, and easily counterfeited, meaning stated ingredients frequently do not match actual contents.2,8,9 Many products circulate in unlabelled vials, sachets, or repackaged formulations made by ‘underground labs’, further increasing uncertainty.2,8,16 PEDTest reagent kits can be used on both labelled and unlabelled products, making them one of the few practical mechanisms available for people who use AAS to evaluate the authenticity of compounds obtained in an unregulated market.
Recent Australian harm reduction initiatives have begun to respond to these concerns by offering people who use AAS opportunities to verify product content. This shift includes the world’s first AAS checking trial, which combined laboratory analysis, results dissemination, and community feedback and documented substantial issues with the presence and purity of AAS products in circulation. 20 Complementing this, a recent brief report by Craven et al. 21 described independent forensic analysis of community-donated AAS samples facilitated and funded by PEDTest Australia, a privately owned, lived-experience-led harm reduction business that manufactures reagent-based testing kits. That study found that more than half of the products were mislabelled and that all product types contained detectable heavy metal contamination. Harm reduction resources focused on the Australian AAS market now highlight at-home AAS testing kits, including PEDTest, as a pragmatic option for people unable to access fixed-site drug checking services. 20 However, although these initiatives demonstrate growing interest in AAS testing, there remains no population-level analysis of the large volumes of community-generated reagent-testing data now available.
To address this gap, the current study sought to examine self-report data from a community-led service which allows people who use AAS to verify their products via reagent testing kits. We sought to (1) describe Australia’s unregulated AAS market using community-submitted reports, and (2) compare oral and injectable compounds to identify differences in product legitimacy.
Methods
Sampling and Recruitment
Data were drawn from community-submitted AAS product test results collected by PEDTest Australia between 2022 and 2024. PEDTest is a community-led harm reduction service that provides people who use AAS with reagent-based testing kits to assess product legitimacy. 22 Participants voluntarily submitted results from testing their AAS products via PEDTest’s online submission portal. As a community-led service, no active recruitment was conducted by the research team. An ethics exemption was granted for this research by Griffith University Research Ethics Committee. All data were de-identified prior to analysis.
PEDTest is a privately owned, commercially operated harm reduction service. One co-author (A.C.) is affiliated with PEDTest; T.P. collaborates with PEDTest to undertake community health service harm reduction ‘pop-ups’ (eg, at Queensland Injectors for Health Network) and social media dissemination of safer use messaging (eg, via podcasts). No other authors have any financial or professional involvement with PEDTest, and the service did not fund this study.
Data Collection
PEDTest kits are branded, reagent-based testing kits provided through a community-led harm reduction initiative. PEDTest was launched in 2022 as a privately owned, lived-experience-led harm reduction initiative operating independently of government or academic institutions. Kits are distributed nationally through the PEDTest website and social media channels, where people who use AAS can purchase reagent-based testing kits at cost. The service is self-funded through kit sales and does not receive external financial support. Marketing is limited to online harm reduction communities, with dissemination occurring primarily via word-of-mouth among people who consume AAS. At the time of writing, no peer-reviewed validation or historical documentation of the service exists, and available information is drawn from publicly accessible programme materials.
These kits enable people who use AAS to assess the legitimacy of oral or injectable compounds through reagent-based testing. A small amount of the product is placed into a test vial, followed by the addition of a reagent solution. The resulting colour change is compared against an in-house reference guide developed by the PEDTest service, which outlines whether the compound aligns with what was expected. People are encouraged to submit their results anonymously to the PEDTest database to support community knowledge and safer market monitoring. For each product, people who use AAS reported the formulation type (eg, oral tablet or injectable oil), the specific AAS compound (eg, testosterone, methenolone), the branded label under which the product was marketed, and the presence result related to active compounds.
Based on reagent testing outcomes, each submitted product was initially categorised into 1 of 4 result types: bunk (no active compound detected), swap (a different active compound detected than the one indicated on the label), confirm (the compound matched the label) and inconclusive (the result could not be clearly classified). In this study, we use the term legitimacy specifically to refer to these reagent-based indicators of compound presence. Because reagent testing does not currently assess purity, concentration or contaminants, legitimacy should be interpreted as a preliminary indicator rather than a definitive analytical determination.
Lastly, only samples identified as oral or injectable were included in the analysis. Raw (powder) compounds were excluded because PEDTest does not currently provide reagent kits validated or marketed for testing raw materials, and the service explicitly advises that testing procedures are intended for oral and injectable formulations. As a result, raw submissions are sparse, methodologically inconsistent and not comparable to tested oral or injectable products.
Data Analysis
All analyses were conducted using IBM SPSS Statistics Version 30. Descriptive statistics were computed to examine frequencies of AAS compound types, formulation types, product brands and product legitimacy. Cross-tabulations were performed to assess the relationship between formulation type (oral vs injectable) and product legitimacy (confirmed, swapped, bunk, or inconclusive). A Pearson’s Chi-Square test of independence was used to assess whether formulation type was significantly associated with test outcomes. Only entries with non-missing data for formulation type and legitimacy outcome were included in the inferential analyses; missing data for these variables were listwise excluded. Valid percentages were reported where appropriate.
Results
Sample Characteristics
Community-submitted reports (N = 2068) were included in the analysis. These represented a subset of the full PEDTest dataset (N = 2614), with exclusions applied to entries that were missing data on formulation type, legitimacy outcome, or were classified as raw/unformulated compounds not relevant to the current study aims. The most frequently reported compound was testosterone (n = 772, 37.3%), followed by oxandrolone (n = 417, 20.2%) and methenolone (n = 260, 12.6%). See Table 1 for full breakdown of the 10 most frequently reported compound types. Additionally, most samples were injectable formulations (n = 1455, 70.4%), with the remainder consisting of oral products (n = 613, 29.6%). Further, only 15.5% of submissions (n = 320) included a reported brand name, of these, with Nexnos (n = 163, 7.9%), Underground Labs (UGLs) Oz (n = 59, 2.9%) and Quantum (n = 54, 2.6%) were the most frequently cited.
The Most Frequently Reported AAS Compound Types (n = 10).
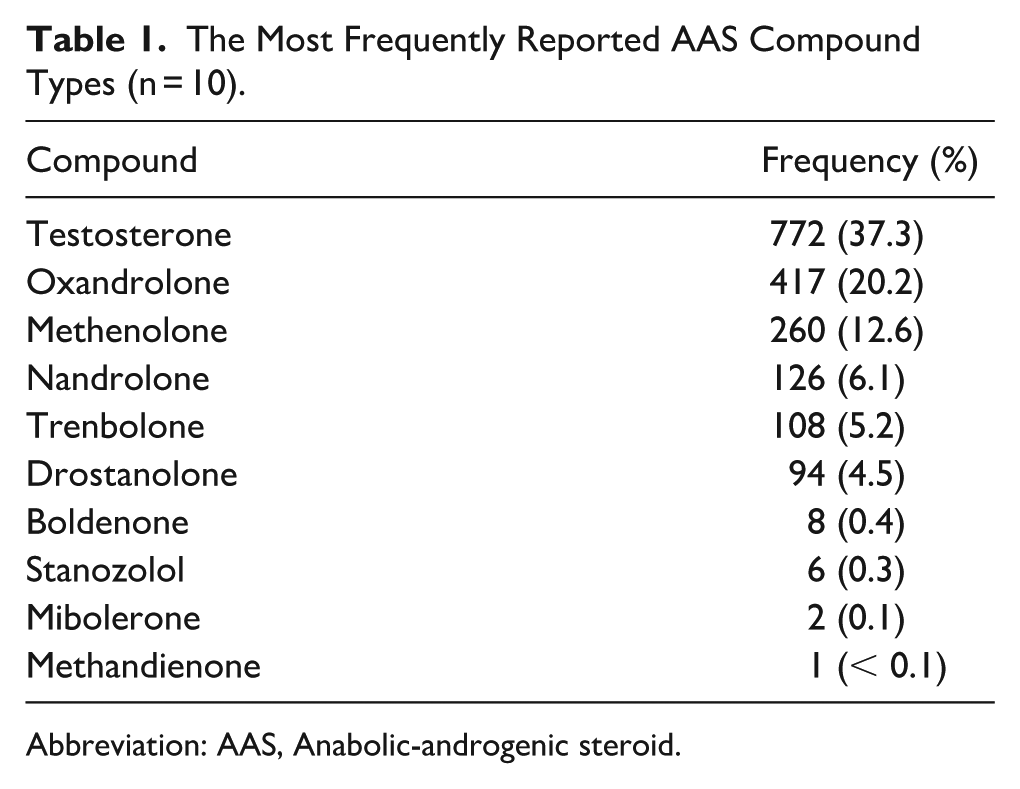
Abbreviation: AAS, Anabolic-androgenic steroid.
Product legitimacy outcomes across the full sample showed that the majority of tested products were confirmed as legitimate, with relatively few identified as bunk, swapped, or inconclusive. Notably, injectable products were more likely to be confirmed and less likely to be bunk compared to oral compounds. In contrast, oral products had higher rates of inconclusive and bunk results. Full frequencies and percentages across legitimacy outcomes by formulation type are presented in Table 2.
Frequencies for Each Legitimacy Outcome as a Function of Formulation Type.
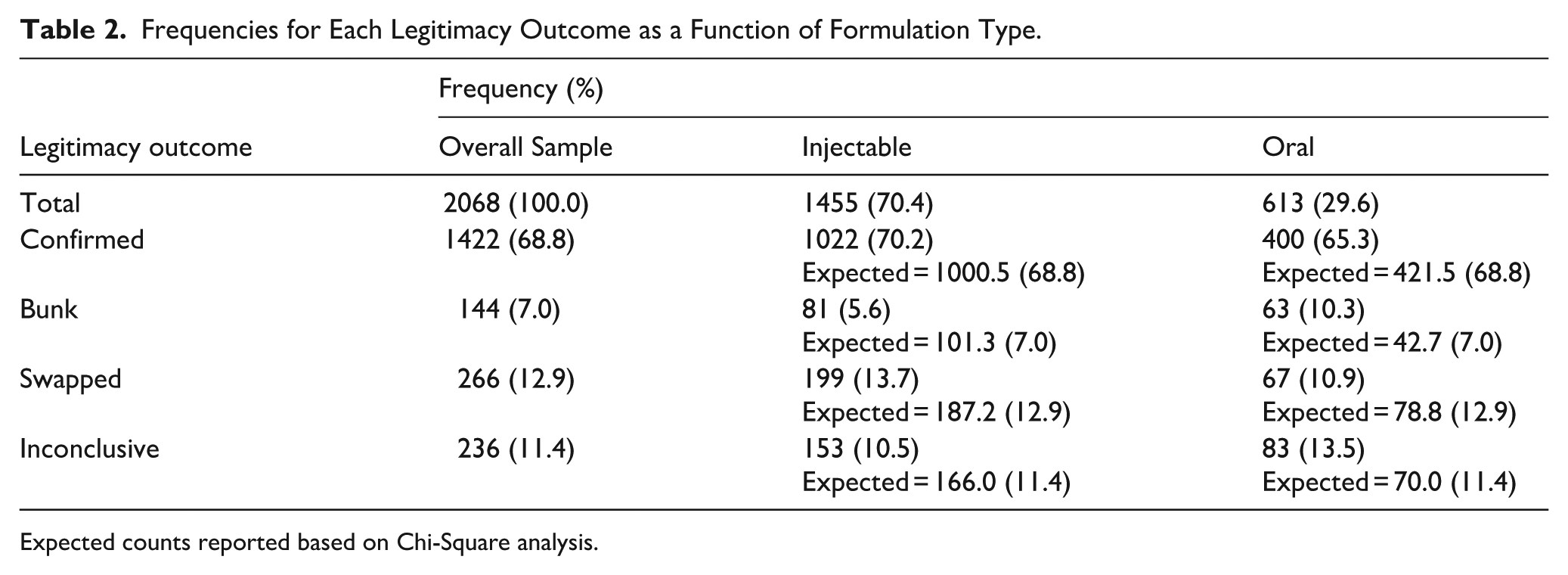
Expected counts reported based on Chi-Square analysis.
Oral versus Injectable Differences
A Chi-Square test revealed a significant association between formulation type and legitimacy outcome, χ 2 (3) = 21.29, p < .001. Adjusted standardised residuals indicated that the association was primarily driven by significantly more confirmed outcomes among injectable submissions (adjusted residual = 2.2) and significantly more bunk outcomes among oral submissions (adjusted residual = 3.8). Other outcome differences, such as swapped and inconclusive rates, did not exceed statistical thresholds for meaningful deviation from expected values. See Table 2 for full observed and expected frequencies.
Discussion
This study represents a rare empirical examination of AAS product legitimacy using real-world data submitted by people who use AAS. With regard to the first aim, the data demonstrate issues with AAS presence legitimacy which fit with Australia’s AAS checking trial 2 as well as international AAS checking programmes. 6 Second, our analyses reinforce long-standing concerns that substantial variability exists among oral formulations.8,9 Reports of oral AAS demonstrated significantly higher rates of having no active compound, and inconclusive test results, compared to injectable products. These discrepancies may reflect differences in manufacturing practices, storage conditions, or distribution pathways, factors that disproportionately affect oral preparations, which are easier and cheaper to counterfeit or adulterate.14,15
Oral products are also more commonly produced and distributed through informal, small-scale operations with limited quality control, increasing the likelihood of filler substitution, incorrect active ingredients or degradation during transport.14,15,23 In contrast, injectable preparations typically require more specialised equipment and sterility, which may reduce, but not eliminate, counterfeiting.24,25 Oral products also circulate more widely through online suppliers and peer-to-peer markets, creating additional opportunities for misrepresentation.14,15,23 These structural features of the illicit market likely contribute to the higher proportion of bunk and inconclusive results observed among oral compounds.
The broader implications of these findings are twofold. First, they highlight the persistent risks associated with the unregulated AAS market in Australia. For instance, inaccurate labelling, absence of active ingredients, or substitution with unknown compounds introduces considerable uncertainty into pharmacological effects for people who use AAS, which may elevate the risk of adverse health outcomes.2,7 Specifically, some of these health harms may be long-lasting or even permanent (eg, virilisation for women, infertility for women and men), 26 meaning that providing objective data regarding AAS composition is an invaluable tool in stemming public health burden. Second, the results underscore the critical role of harm reduction approaches which prefer providing people who use AAS with objective data regarding the substances they are using. 10 We call for increased investment in harm reduction strategies that address the specific needs of people who use AAS, including public access to reliable, low-threshold AAS checking services and integrated market surveillance that combine community reporting with longitudinal cohort monitoring (ROIDTrends). Embedding these services within broader community and health service settings is essential to ensure diverse cohorts have access – especially women 9 – so people who use AAS can make informed decisions.
Limitations
Several limitations should be noted. The reagent-based testing method, while informative, is limited in its ability to determine compound concentration, presence of impurities or multicompound formulations. 22 Although PEDTest is an emerging, privately operated harm reduction service, detailed information on kit validation and internal quality-assurance procedures is not publicly available. As such, PEDTest should be understood as a community- and commercially driven tool rather than a laboratory-accredited analytical service, and findings should be interpreted with this context in mind. Further, PEDTest kits may have potential for ‘end user–error’, depending on an individual’s ability to perform the test correctly. Although PEDTest operates nationally, geographic clustering may exist in submissions, potentially limiting generalisability across the broader Australian market. Compound-level analyses were not feasible because stratifying legitimacy outcomes by individual AAS compounds resulted in sparse cell counts that violated Chi-Square assumptions and produced unstable estimates. Future research should assess longitudinal changes in product quality and evaluate the behaviour of people who use AAS in response to legitimacy data.
Conclusions
Our findings support the notion that oral AAS products are significantly more likely to contain no active ingredient when compared to injectable formulations. These data add weight to the need for AAS testing programmes across Australia and the continued development of combined community-reporting and longitudinal surveillance models. National programmes would provide equitable support to people who use AAS across Australia, who have been traditionally underserved.
Footnotes
Ethical Considerations
Ethical approval was granted via the university prior to conducting this study.
Consent to Participate
All participants provided consent to their data being used for the purpose of this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article:
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: One co-author (A.C.) is affiliated with PEDTest; TP collaborates with PEDTest to undertake community health service harm reduction ‘pop-ups’ (eg, at Queensland Injectors Health Network), and social media dissemination of safer use messaging (eg, via podcasts). No other authors have any financial or professional involvement with PEDTest, and the service did not fund this study.
Data Availability Statement
The dataset used for this study is available upon reasonable request.*
