Abstract
Dual diagnoses of psychosis and drug or alcohol problems (DDp) are prevalent, high-cost presentations, reliably associated with poorer outcomes and greater difficulty engaging with mental health services compared to either condition alone or other mental health problems. Engagement difficulties restrict access to recommended care that could improve outcomes, and thus are a priority treatment target. Contingency management (financial incentives for positive health behaviours, CM), peer support, and text messaging each improve engagement for psychosis or drug/alcohol problems. Despite the high potential to improve engagement and thus clinical and economic outcomes in DDp, their combined impact remains unevaluated in this poorly treated and hard-to-reach population. We plan a confirmatory trial to test, for the first time, the impact on engagement in DDp of adding CM to usual care alongside text message reminders and peer support. Methodological innovation is required to ensure adequate rates of recruitment, retention, and outcome completion; as well as standardised delivery of interventions, particularly CM, which will be delivered by peer support workers, and will target attendance, to improve engagement in recommended treatments. We comment here on the protocol for CoMPEERS (ISRCTN90137206), a study to test the feasibility of progressing this novel design to a full trial.
Highlights
We aim to recruit a hard-to-reach population with psychosis and drug/alcohol problems
Trained peer support workers will provide contingency management and text messaging
We will measure attendance as a prerequisite for engagement in recommended care
We will collect other outcomes from routine records to reduce participant burden
Background
Prevalence and Impact of Psychosis and Drug/Alcohol Problems
Psychosis within the schizophrenia spectrum carries one of the highest global mental health burdens, associated with life-limiting functional impairment, physical morbidity, early mortality and high societal costs.1-8 The global lifetime prevalence is around 1%. 9 Comorbid drug and alcohol problems are common, with consistently poorer recovery and socio-economic outcomes for dual diagnosis psychosis presentations (DDp) compared to psychosis or drug/alcohol problems alone, and compared to other mental health presentations.5,10-13 Functioning, physical health and longevity are all further adversely impacted, with higher social and health care, housing and criminal justice costs.10-13
Engagement Difficulties
Key to the poorer outcomes for DDp is the impact of both psychosis and drug/alcohol problems on engagement with healthcare.14-19 People with DDp commonly lack insight into their difficulties, struggle with day-to-day organisation, find motivation hard and have limited trust in other people, often because of histories of interpersonal adversity and trauma. These challenges, combined with social stigma and limited understanding of both psychosis and drug/alcohol problems within mental health services, can make it difficult to establish a shared understanding of problems between workers and service users that can guide care and foster acceptable ways of interacting to promote engagement.14-19
A Treatment Priority
The high burden of psychosis and drug/alcohol problems has made their treatment, separately and as co-morbid conditions, priorities in international guidance. 20 In the United Kingdom National Health Service (NHS), initiatives are underway to increase adherence to guideline recommendations, including integrated multidisciplinary care, with access to specialist psychosocial interventions for psychosis and drug/alcohol problems.21-23 However, improvements in care quality are only meaningful if service users are able to access and attend their care – that is, if engagement can be established.
Improving Engagement
Peer support is recommended by the United Kingdom National Institute for Health and Care Excellence (NICE)6-8 for DDp as a way to bridge the engagement gap, based on evidence suggesting improvements in attendance for psychosis and for drug/alcohol problems. 24 However, peer support continues to be offered inconsistently and with poor governance structures in frontline NHS. 25 Text message reminders have also been shown to improve engagement across a range of conditions, but despite the simplicity, are not yet routinely implemented.26-30
The Evidence Gap
NICE has called for the evaluation of peer support and text message reminders, alongside the provision of financial incentives through contingency management (CM), as combined interventions to improve engagement for people with DDp. 8 Incentivising positive health behaviours through CM has been shown to reduce substance use and promote attendance for users of drug and alcohol services,31-33 and to improve attendance at treatment appointments for people with psychosis.34,35 Evaluation with DDp populations is lacking, 36 and unlike peer support and text message reminders, CM is not routinely available for people with DDp and is poorly understood in services. Moreover, the required structures for CM delivery, such as regular and consistent appointments at a frequency to permit contingency learning, are not in place.
Aims of CoMPEERS
We aim to conduct the first trial to meet the NICE call by evaluating the impact of CM, combined with peer support and text message reminders, on engagement for people with DDp. We plan to incentivise attendance at peer support worker (PSW) appointments, in order to increase attendance at mental health services, and thereby, opportunities to engage. Our overarching goal is to improve outcomes for people with DDp by facilitating engagement in the full range of treatment options available.
In order to do this, a number of methodological challenges need to be addressed, namely:
Development of strategies to reliably identify, recruit and retain an exceptionally hard-to-reach group.
Establishment of structures for the standardised, consistent delivery of peer support and text message reminders.
Supporting PSWs to deliver CM and adhere to a CM schedule.
Identification and reliable completion of measures that capture key outcomes of interest, without undue participant burden.
The current study therefore was designed to evaluate feasibility and establish innovative methodology for a larger, confirmatory trial of CM to improve engagement for DDp.
Feasibility Study Design
Design
We will conduct a cluster randomised controlled study of the feasibility of a trial to assess the impact of CM on attendance at PSW appointments. Clinical teams will form the clusters to minimise intra-team cross-contamination across trial arms. Clusters will be allocated to 1 of 2 arms: (a) routine care supplemented with peer support and text message reminders or (b) the same package plus the addition of CM.
All participants will therefore receive routine care with peer support and text message reminders. As such, we will not be evaluating the individual impact of these components. This decision is supported by existing guidance for DDp already recommending both interventions,6-8 as well as evidence that they are currently offered inconsistently in routine services.25-27 Our goal is to ensure that both are delivered consistently and in line with best practice. Participants in the intervention arm will also receive CM for attending weekly appointments; the additional impact of CM will be the focus of evaluation in the future trial.
Feasibility outcomes for the study and progression criteria are shown in Table 1. We will test the feasibility of measuring attendance at PSW appointments (the proposed primary outcome), engagement, adverse events, quality of life, severity of drug/alcohol problems and service use/cost-effectiveness of the intervention.
Feasibility Outcomes and Progression Criteria.
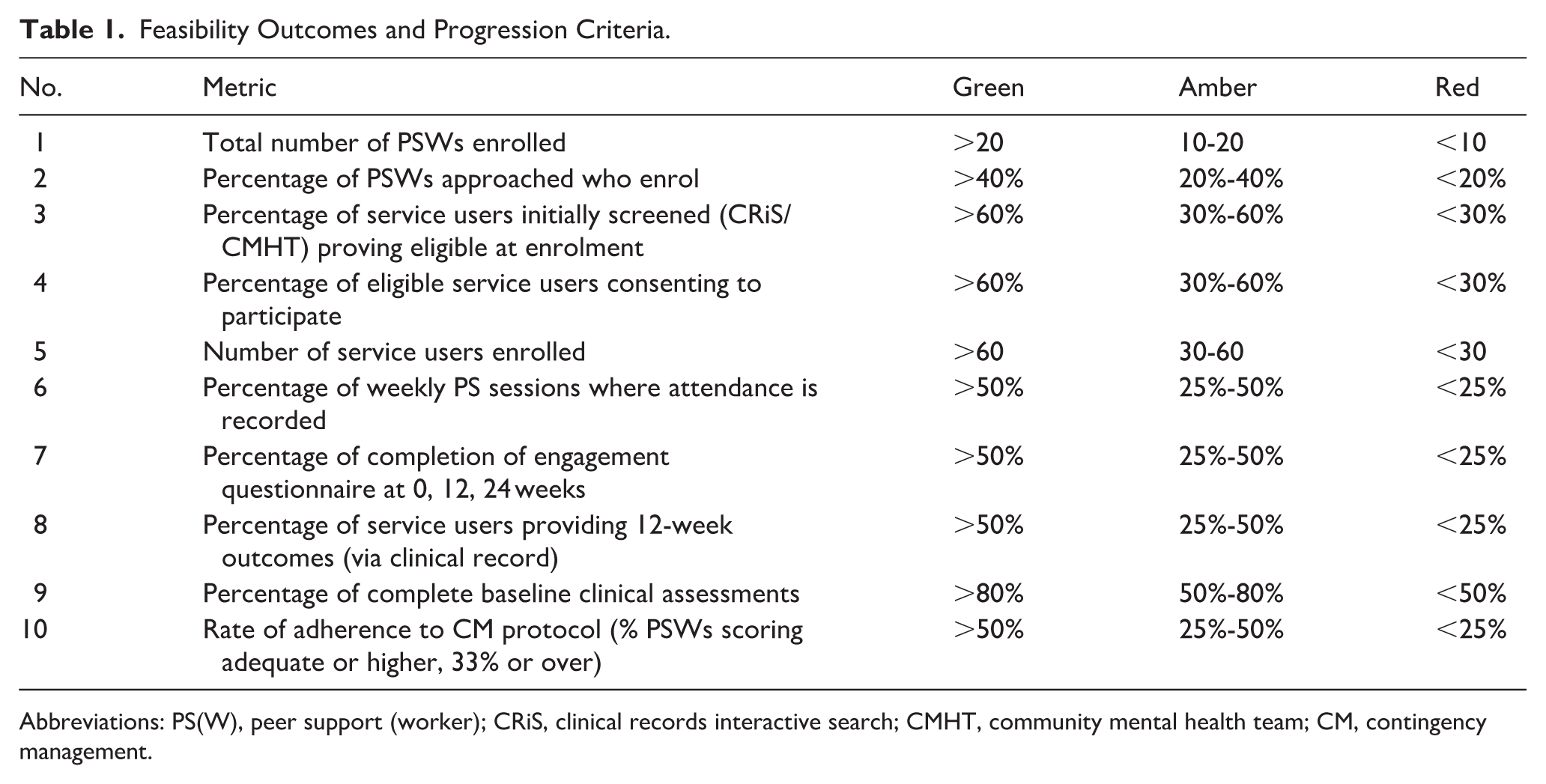
Abbreviations: PS(W), peer support (worker); CRiS, clinical records interactive search; CMHT, community mental health team; CM, contingency management.
Analysis for the current study will employ descriptive and count data to determine the feasibility of the future trial. We will conduct a thematic analysis of interviews with a selection of PSWs, participants, and clinical team members, whose experience of the study will highlight issues to address for the future trial.
Ethical Approval
Ethical approval was granted by the London-Bloomsbury Research Ethics Committee of the United Kingdom National Research Ethics Service (24/LO/0264). The study was prospectively registered (ISRCTN90137206).
Patient and Public Involvement
Consultation with people who have lived experience of DDp, or of informal caregiving for somebody with DDp highlighted the importance of lack of understanding as an obstacle to engagement. Mental health workers recognised the difficulties engaging. All consulted groups supported research into strategies to enhance engagement, including using CM.
Study Context
The study is taking place across a single large NHS mental health trust, taking care to adopt procedures that could be generalised to a multisite design for the future full trial. Recruitment is based in community secondary mental healthcare teams in 2 inner-city boroughs. We have selected teams with high proportions of DDp service users: 4 serving people with established psychosis (onset ≥2 years ago) and 2 specialist homelessness teams.
Identification, Recruitment and Retention of a Hard-to-Reach Population
The target population for this study, managing the difficulties of psychosis and drug/alcohol problems, often struggles to attend clinical services and, as a result, is less likely to engage in research, particularly when recruitment relies on contact with mental health professionals. Moreover, professionals’ beliefs about the conditions may lead them to pre-emptively exclude people from research, by deciding, without asking them, that they will not be interested or would not wish to engage. 37 To assess recruitment rates, we will use a clinical record interactive search to support the clinical team with generating a list of all those eligible to participate and ask staff to record whether they were asked if they agreed to contact with the research team, whether they agreed, and reasons for not being asked, or not agreeing to contact. Anybody who has given a priori consent to research contact will be approached directly by the research team. We will ask clinical teams to share aggregated demographic data (eg, age, NHS self-reported ethnicity category) with the research team. This will allow us to identify how many were not approached or declined contact, their demographic profile and reasons for non-recruitment, helping us assess sample representativeness and improve reach in the future trial.
Clinical teams will be asked not to describe the study and ask consent to participate, because of the high likelihood of getting into arguments with potential participants about whether or not they have psychosis and drug/alcohol problems. 38 Study research workers will do this, and will be specifically trained in managing both different perspectives on mental health and/or drug/alcohol problems and the impact of other difficulties of the condition. To support retention, contact will be maintained with participants via PSWs, the clinical team, and directly with the research worker (eg, to reschedule peer support meetings, check in following periods of non-attendance).
Establishing Standardised, Consistent Delivery of Interventions
Routine Care
All participants in the study will continue to receive their usual (‘routine’) care. Routine care typically includes monthly meetings with a care coordinator, medical reviews every 6 to 12 months and access to specialist dual diagnosis interventions, NICE-recommended psychological therapies, and social, occupational and vocational support. An individualised care plan that identifies recovery priorities, such as establishing medication adherence, improving day-to-day functioning and wellbeing and supporting community engagement should be routinely in place. We are not intervening in routine care in any way, but will monitor what study participants are offered and accept via the clinical record.
Peer Support
All participants in the study will receive peer support, delivered in 16 sessions of up to an hour, weekly for weeks 1 to 12 of the study with 4 ‘tapered’ follow-up sessions at 14, 16, 20 and 24 weeks, delivered face-to-face at the team base. To ensure consistency, PSWs will be asked to limit their contact with participants to these sessions, and not offer any additional telephone calls or contact outside the team base.
We will recruit PSWs who are already employed to deliver peer support in the NHS trust hosting the study, ensuring their managers agree to their participation. This means they have workplace support and reimbursement structures already in place. PSWs will be reimbursed for the hours they spend on the study, and their involvement treated as research participation, allowing them to manage their time in the study flexibly with no impact on their current or future employment.
The peer support intervention offered in the study will be based on a publicly available NHS-based competence framework, 39 adapted for the particular difficulties of DDp.7,8
To train in this intervention, all PSWs will join a one-day interactive workshop with 2 of the study leads, including role plays and demonstrations, covering an understanding of the experiences of psychosis and substance use; strategies for engaging and supporting, drawing on cognitive therapy and motivational interviewing; and the key components of peer support required for the study; as well as the study logistics.
Peer support sessions will be audio-recorded if the PSW and the participant are both agreeable, with an audio-recording for each case to be listened to by supervisors to monitor adherence.
PSWs will be allocated to a trial arm, and attend weekly supervision groups with local study leads, for all PSWs in that arm, as part of their participation in the study. Supervisors will check that peer support sessions are being delivered to protocol. Supervision will afford opportunities to discuss cases and listen to audio-recordings to maintain adherence.
PSWs, and the research worker, will maintain contact with clinical teams, so that any concerns about the wellbeing of the participant can be communicated. The research worker will also manage any concerns raised by participants, PSWs, or the team about participation, including any difficulties in the PSW/participant relationship, when an alternative PSW will be organised as needed.
Text Message Reminders
All participants in the study will receive text message reminders for their peer support sessions. Three reminders will be sent: 1 week before the session, the day before, and on the day. Text message reminders will be sent by PSWs from a confidential web-based service, designed for NHS, using standard templates, personalised with names and appointment times. Text messages take the form: ‘COMPEERS Research NHS: Hello ((first name)), I am texting to confirm our meeting ((on the Date in full e.g. Monday 24th November/tomorrow/today)) at ((time e.g. 2.00pm)) at ((place and address of teambase))’. I hope you can make it and look forward to seeing you then. With best wishes, ((PSW name)). Any appointment changes will be notified to the study research worker, who will call participants and PSWs to arrange an alternative, or if a PSW is away, cover by another PSW.
Supporting PSWs to Deliver CM
Participants in the intervention arm will also receive CM, in addition to routine care, peer support, and text message reminders. CM will be delivered by PSWs, during the first 5 minutes of the peer support session, during weeks 1 to 12. CM will not be delivered during the tapering period, weeks 13 to 24. For the 12 weekly sessions, a £10 shopping voucher will be given for every appointment attended within 15 minutes of the scheduled time, and an additional £10 voucher for 4 consecutive on-time attendances. We chose to incentivise attendance at PSW meetings, since CM requires a regular reward schedule to establish learning, and frequent monitoring of the target behaviour to provide opportunities for the reinforcer to be delivered frequently, and routine clinical contacts are too infrequent or unreliable. Providing the reinforcer immediately upon achieving the target behaviour helps to establish a clear contingent relationship between achieving the target behaviour and receiving the reinforcer (financial reward).40,41
PSWs allocated to the CM arm will receive an additional half-day interactive training in the delivery of CM, including role-plays.
The CM element of the session (the first few minutes only) will be routinely audio-recorded (consent to do this is a condition of participation). Audio-recordings will be rated for adherence using an established 8-item CM fidelity measure. 41
Supervision for PSWs allocated to deliver CM will include checking that CM as well as peer support is being delivered to protocol, alongside opportunities to discuss cases and listen to audio-recordings to maintain adherence. Supervision will be by study leads with expertise in CM and in peer support.
Collecting Measures Without Participant Burden
The proposed primary clinical outcome for a future confirmatory trial will be attendance at peer support sessions, as recorded by PSWs in the clinical record system. To support accurate data collection and assess the most reliable method, PSWs will also record attendance in a study booklet, and supervisors will maintain attendance logs based on PSW reports and text message reminder checks.
A key secondary clinical outcome will be engagement with the community team and adherence to the care plan. This will be assessed through verbal reports to the PSW by the participant and documented both in the clinical record and the study booklet. The study research worker will extract these data from the clinical record, along with any other attendance-related information.
The same approach – PSW report and case note review – will be used to collect adverse event data. Service use data for economic analyses 42 will also be obtained from the clinical record.
In addition, participants will complete a measure of health-related quality of life (EQ-5D-5L) 43 and of engagement 44 (with the PSW, and separately with their care coordinator) at 0, 12 and 24 weeks. PSWs and care coordinators will also complete engagement assessments.
We have kept self-report measures to a minimum because the difficulties of psychosis and drug/alcohol problems often make such tasks burdensome. We have aligned our measures of quality of life/recovery 45 and drug/alcohol problems46,47 with the UK NHS standardised outcomes, which community mental health services are meant to complete routinely with service users. 48 We will collect these from the clinical record and ascertain the rate of completion.
Discussion and Conclusion
Using a traffic light system (green, amber, red) we will assess whether a future full trial is feasible and if there are any changes we need to make to the protocol.
Our study will be the first to combine CM, peer support and text messaging, and to target attendance, as a pre-requisite to engagement, in DDp, a population that is hard-to-reach and consequently poorly treated and under-researched. The combined interventions hold the potential to improve engagement and thus clinical and economic outcomes for people with DDp, who make up a significant proportion (40%) of mental health service caseloads, with poor outcomes, and high economic costs of care. Our feasibility study will address key challenges to conducting a robust evaluation of the impact of adding CM to usual care, augmented by peer support and text messaging. We will pilot innovative methods of recruitment, intervention delivery and outcome collection, that suit the target population and the service context and will facilitate progression to effectiveness evaluation should CM prove effective in the future confirmatory trial.
Footnotes
Ethical Considerations
This study was approved by the London-Bloomsbury Research Ethics Committee (24/LO/0264) on 5 June, 2024. The research will be conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Consent to Participate
Written informed consent required prior to enrolment in the study.
Author Contributions
All authors
1. Have made a substantial contribution to the concept or design of the article; or the acquisition, analysis, or interpretation of data for the article; AND
2. Drafted the article or revised it critically for important intellectual content; AND
3. Approved the version to be published; AND
4. Agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
SJ conceptualisation, funding acquisition, project administration, supervision, writing – original draft, writing – review and editing. EC conceptualisation, data curation, formal analysis, funding acquisition, methodology, writing – original draft, writing – review and editing. SN data curation, investigation, project administration, writing – review and editing. JS conceptualisation, data curation, formal analysis, funding acquisition, methodology, writing – review and editing. TW conceptualisation, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, supervision, writing – review and editing. JD conceptualisation, funding acquisition, project administration, supervision, writing – review and editing. NM conceptualisation, funding acquisition, methodology, project administration, supervision, writing – original draft, writing – review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study/project is funded by the NIHR Research for Patient Benefit Programme (NIHR204991). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. We gratefully acknowledge support from the South London NIHR Clinical Research Network (CRN).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NM has through her university, worked with pharmaceutical companies and the university receives grants, costs etc. from companies including, in the past 3 years, Mundipharma and Mayne Pharma. SJ conducts publicly funded training and research in psychological interventions for people with psychosis and consults nationally on psychological intervention training curricula through King’s College, London and South London and Maudsley NHS Foundation Trust, and is employed in the NHS in Lambeth (a study site). There are no other conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be made available for data sharing upon reasonable request.
