Abstract
Formaldehyde is ubiquitous and harmful to human body. In this study, biochar graphene and composites are prepared by hydrothermal method. According to the material properties of formaldehyde, biochar composites are used to catalyze the two ends of electrode to promote the activation reaction of formaldehyde. By analyzing the characteristics of Fourier transform infrared spectrum image, it is found that the biochar composite Cu(OH)2/C is more conducive to activate formaldehyde, which effectively improves the conductivity of the material and provides a reference for studying the effects of biochar composites on formaldehyde.
Introduction
Formaldehyde has been listed as one of the most directly carcinogenic harmful substance by the National Environmental Protection Agency in China and the World Health Organization. 1 Because formaldehyde covers a wide range of areas, it affects all aspects of our daily life, such as food, clothing, home decoration, and office furniture. The presence of formaldehyde has a strong concealment which brings potential harm to people’s lives. Therefore, the establishment of an effective detection method for formaldehyde has become a research hot spot and it is of high commercial value. At present, it is difficult to activate formaldehyde through electrolysis. However, it is shown that the rapid and effective detection of formaldehyde can be achieved by adding chemicals at both ends of the electrode for catalytic activity. 2 Biochar is the material with large pore density, diverse structure, and spatial composition that can be adjusted according to the needs of use. It has a wide range of uses in harmful gas adsorption, material activity reaction, and catalytic properties. 3 By catalyzing the both ends of the biochar composite electrode, harmful substances such as formaldehyde can be activated. According to experimental calculations, the effective area of the catalytic electrode of the composite is much higher than that of the bare electrode. The biochar composite has a high application value in the study for the activation of formaldehyde.
Experimental instruments and methods
Experimental instruments
The main equipment required for the experiment of biochar composites is shown in Table 1.
Experimental equipment for biochar composites.
Preparation of biochar composites by hydrothermal method
Preparation of biochar graphene
The culture medium solution was prepared by dissolving biochar graphene in 200-mL purified water according to the ratio of 1:3. Then it was heated to meet the experimental requirements and was transformed into dispersion by ultrasonic sensor. The dispersed solution was placed in a constant temperature water bath at 60°C with 50 mL as a unit, 3 mL of catalyst was added to each unit medium, and stand for 5 h. The purified water and formaldehyde were filtered respectively with cold water, and the filtered solution was put into a vacuum closed box at 60°C until it was completely dried.
Al-Cu/C biochar composite and Al-CuO grading material
Graphene (0.8 g), CuO (1.5 g), and Al2O (31.3 g) were weighed by a precise balance. These weighed materials were distributed according to the quantitative proportion, dissolved in 80 mL of dimethylformamide and stirred evenly. Graphite powder (1.2 g) was added when the stable state of the medium solution was formed. After the graphite powder was fully contacted with the solution, it was put into an electrochemical generator and completely reacted at 150°C. The mixed solution was cooled to 25°C after 24 h and the solid desiccant was placed in the electrochemical generator and maintained at a constant temperature for 24 h to obtain the biochar composite Al-Cu/C. Al-CuO can be prepared according to the same experimental procedures and instruments without the addition of graphite powder.
Cu(OH)2/C biochar composite
Weigh 1.8 g Al-CuO of the above-prepared composite, put it into purified water and mix it evenly, transfer the mixed solution to a temperature-controlled water heater by 50°C, add 30-mL NaOH solution containing 0.6 g and appropriate amount of graphite powder, stir it continuously, stand for 1 h and cool it to a constant temperature, centrifuge and put it in a vacuum airtight box for drying, then Cu(OH)2/C composites are obtained.
Activating formaldehyde by Cu(OH)2/C biochar composites
Take 0.8 g NaOH experimental material, dissolve it in purified water according to the ratio of 1:3 and stir it evenly, fully fuse the composite Cu(OH)2/C with NaOH solution, place it in the chemical generator for 24 h, add equal proportion concentration formaldehyde solution to this above solution with 5 mL as the standard in sequence, use the scanning electron microscope (SEM) to compare at the rate of 0.5m/s.
Result analysis
Characterization and analysis of Al-Cu/C and Cu(OH)2/C composites
Figure 1 shows the SEM diffraction patterns of Al-Cu/C and Cu(OH)2/C composites, respectively. The flower-like texture of Al-Cu/C composites can be clearly observed as shown in Figure 1(a). Using Fourier transform infrared diffractometer (FTIR), Al-Cu/C biochar composites can be transformed into Cu(OH)2/C petal structure with clear layered structure. From the SEM diffraction image, it can be seen that there is no change in the overall structure during the conversion process for these two composites. The gap of structure texture map of Cu(OH)2/C composite is larger which is conducive to the larger reaction contact area of the composite. At the same time, Cu(OH)2/C composite is more conducive to the mixed solution into the active material, which effectively improves the electrical conductivity of the composite.

The SEM diagram of composite. SEM: scanning electron microscope.
The X-ray diffraction (XRD) spectrogram of biochar composite is shown in Figure 2. It can be seen from Figure 2 that the characteristic diffraction peaks (as shown by counts per second (cps)) at 19.31°, 31.21°, 41.33°, 45.41°, 58.05°, and 60.19° are corresponding to (001), (100), (101), (102), and (110), respectively, which match the diffraction peaks of crystal structure. The characteristic value of graphene 4,5 is not shown in XRD spectrogram, indicating that the biochar composite has a low content of graphene.

The XRD spectrogram of biochar composites.
Study the effect of Cu(OH)2/C biochar composites on formaldehyde
The effect of Cu(OH)2/C biochar composites with different concentrations on formaldehyde is shown in Figure 3. It is shown that the wave oscillation appears after adding NaOH, and the detected value changed regularly in the range of concentration. As the concentration of formaldehyde gradually increases, the redox reaction between the mixed solution of Cu(OH)2/C and NaOH and formaldehyde also increases. The peak of the redox reaction appears between the concentrations. After adding the formaldehyde solution with the same concentration, the current of the oxidation electrode and the reduction electrode also increases, indicating that the biochar composite has a significant electrocatalytic effect on formaldehyde.
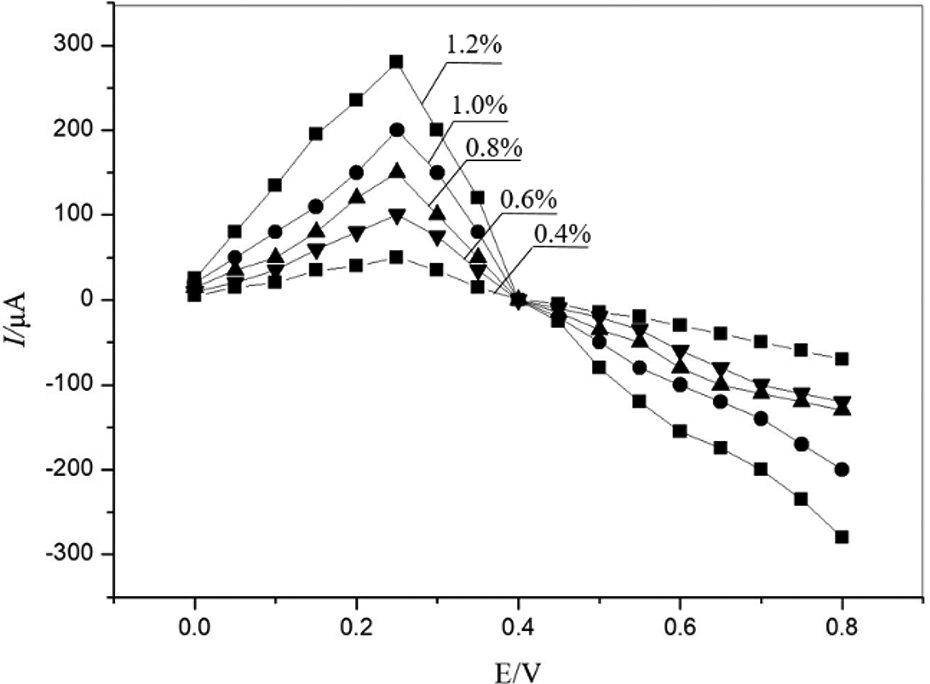
The variation of formaldehyde with different concentrations.
Electrocatalysis of Cu(OH)2/C biochar composites to formaldehyde
It can be observed from the above experiments that the biochar composite has a significant electrocatalytic effect on formaldehyde. To improve the accuracy of the composite Cu(OH)2/C in determining formaldehyde, the standing time during redox process is analyzed. The relationship between the standing time of the solution and the redox peak value is shown in Figure 4. It can be seen from the curve that when the standing time is 2 s, the peak current of oxidation–reduction reaction appears. As the standing time increases, the redox peak current also increases significantly. When the standing time is 12 s, the peak current reaches the maximum value. The peak current does not change as standing time increases when the standing time is longer than 12 s, indicating that the Cu content of biochar composite is stable. The chemical reaction between Cu(OH)2/C and formaldehyde tends to be stable and saturated, and the critical point of standing time is 12 s.

The relationship between composite and standing time.
Conclusion
Both ends of the biochar composite electrode have an activating effect on formaldehyde. In this article, the biochar composites Al-Cu/C and Cu(OH)2/C, and graded materials Al-CuO were prepared by hydrothermal method. By FTIR, it is found that the Cu(OH)2/C composite more effectively improves the electrical conductivity at both ends of the electrode, and the formaldehyde solution of different concentrations fully reacts with the composite. The experimental results show that biochar composite has obvious electrocatalytic effect on formaldehyde, and the critical point of standing time is 12 s, which provides technical support for the preparation of biochar composite and the activation reaction of formaldehyde.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the fund of the State Key Laboratory of Solidification Processing in NWPU (Grant No. SKLSP201725) and the fund from National Innovation (Qingdao) High speed Train Material Research Institute Co. LTD (Grant No. HCGC2021001).
