Abstract
The use of wastes in the development of useful material is a key aspect of solid waste management. The aim of this study was to utilize biochar from elephant grass (Pennisetum purpureum) as fillers in polystyrene composites with focus on the morphological and thermal properties of the developed composites. The composites were prepared by hand lay-up technique. The prepared resin and final composite were characterized using scanning electron microscopy, differential scanning calorimetry and Fourier-transform infrared spectroscopy. The biochar also did not contain any reactive hydrophilic groups, thus, it is compatible with polystyrene. Estimated heat capacities of the composites from the thermal test results showed that to obtain a composite of heat capacity range between 600 J/kg·K and 760 J/kg·K, a 10–30 wt% range of biochar composition should be maintained. At 30 wt% biochar composition, heat capacity was at a maximum value of 757.66 J/kg·K.
Introduction
Waste generation in sub-Saharan Africa is approximately 62 million tons per year. 1 Per capita waste generation is generally low in this region, but spans a wide range, from 0.09 kg per person per day to 3.0 kg per person per day, with an average of 0.65 kg/capita/day. 1 The rapid increase in the generation of waste plastic in the world is attributable to economic growth, changing consumption and production patterns of plastic products. In the United States, for example, municipal waste generation rose from 88.1 million tons to 250.9 million tons in the past five decades, 2 at a 5% yearly increase. 3 This increase in generation has led to plastics waste becoming a key stream in solid waste, following food and paper wastes. 4 One of the major constituents of this plastic waste stream is polystyrene. Polystyrene commonly called Styrofoam is a synthetic aromatic hydrocarbon polymer developed from the polymerization of the styrene monomer (shown in Figure 1). It can be as expanded polystyrene foam or extruded polystyrene foam. General-purpose polystyrene is hard, clear, brittle and inexpensive.
Polymerization of polystyrene.
Polystyrene is one of the most widely used plastics in the world. 5 Polystyrene is transparent but can be coloured with colourants. Because polystyrene is a thermoplastic, it exists in a solid (glassy) state at room temperature but flows when heated above a temperature of about 100°C, which is its glass transition temperature, and solidifies when cooled. Other basic properties are described in Table 1. The decomposition time for polystyrene is unknown but some experts have estimated it to be about 500 years. This makes it a non-biodegradable environmental pollutant.
Properties of polystyrene.

Reinforced polystyrene composites consist of reinforcing fibres and a polymer matrix, which acts as a binder for the fibres.6,7 There is budding interest in the use of natural fibres as reinforcing components for both thermoplastic and thermoset matrices, because of their renewability, biodegradability and environmental affability.8–11 The natural fibres have the prospect of being used as a replacement for glass or other traditional reinforcement constituents in composites. These fibres are abundant, cheap and renewable. 12 Currently, locomotive and construction industries have been interested in composites reinforced with natural plant fibres as alternative materials for glass fibre-reinforced composites in structural applications with modest demands on strength reliability. 6
Interest in biochar development and application has increased recently,13–16 because of the possibility of its suitability in the growing field of biocomposites, which are materials where at least one of the parts is organic in origin. This can mean the filler or polymer is made of sustainable sources, for example, plant fibre, recycled material or waste yields and oils.17,18 Consolidating biochar into composites could help improve the properties of the resulting composites19,20 as well as lessen the utilization of engineered fillers in these materials, for example, carbon black. 21 Carbon black is a non-renewable filler, created from the treatment and preparing of hydrocarbons from the oil and gas industry. 22
Elephant grass biochar is obtained from pyrolysis and gasification of elephant grass which has higher biomass yield than other energy grasses.23,24 Biochar from elephant grass is reported to be suitable for soil amendment.13,25,26 Several studies have investigated the use of biochar in plastic composites. Behazin et al. 27 studied the mechanical, chemical and physical properties of biochar from Miscanthus, switchgrass and softwood chips for possible application as reinforcement in plastic composites. The recommendations from the study were positive. Cuthbertson 28 studied the incorporation of pyrolytic biochar in concrete composite. Das et al. 21 studied the use of biochar from Pinus radiata in polypropylene composite. Das et al. 29 evaluated the mechanical, thermal, chemical and morphological properties of polypropylene composites obtained with a variety of waste-derived biochar as fillers. Das et al. 30 observed that biochar from landfill wood was better in polypropylene composite compared to rice husk, coffee husk and coarse wool. Biochar fillers have also been studied for other polymer composites from high-density polyethylene, 31 polyvinyl alcohol (PVA), 32 carboxylated styrene–butadiene, 33 epoxy34,35 and a host of others.
Within the scope of the authors’ exhaustive search, there are no reports of the use of biomass biochar fillers in polystyrene composites. The aim of this study is to utilize biochar from elephant grass (Pennisetum purpureum) as fillers in polystyrene composites. The focus of this study is the morphological and thermal properties of the developed composites. The prepared resin and final composite were characterized using scanning electron microscopy (SEM), differential scanning calorimetry (DSC) and Fourier-transform infrared (FTIR) spectroscopy.
Methodology
Materials
Dry elephant grasses were gathered from uncultivated lands within the premises of the University of Ilorin, Kwara State, Nigeria. Polystyrene commonly used for packaging materials such as electronic gadgets to prevent damages during movement was obtained from the stream of electronic shops within Ilorin town, Kwara State, after the use or sale of the gadget. The solid polystyrene was solvated in the chosen solvent to produce the resin and have it in a more suitable form for the composite moulding. The process is described in the study of Abdulkareem and Adeniyi. 36
Biochar preparation
Firstly, the leaves of the dried elephant grasses were removed so the stalks are left. The gathered stalks were then crushed into smaller sizes at the mill to increase surface area. It was then fed into a carbonization unit to undergo pyrolysis at a peak temperature of 300°C for 120 min to yield 14.29 wt% char. Further elaborations of the reactor design, operation and experimental methodology can be found elsewhere.13,15 More details of the morphology, functional groups, elemental composition and other properties of the elephant grass biochar utilized in this study can be found in Adeniyi et al. 13 The biochar was further crushed and sieved into a particle size of 100 µm.
Preparation of composite
Manual mixing method and hand lay-up technique were used for the production of the composite. The polymer material (polystyrene) was dissolved in gasoline at room temperature to form polystyrene resin. Biochar (10, 20, 30 and 40 wt%) was introduced into the respective resin and mixed thoroughly until uniformity was obtained. Then the mixtures were poured into moulds and left to cure at room temperature for 7 days.
Characterization of polystyrene resin and reinforced composite
The prepared resin and final composite were characterized using SEM, DSC and FTIR spectroscopy. The SEM was used to identify the morphology of the resin samples. The surfaces of the resin specimen were examined directly by scanning electron microscope. The sample was coated with 100 Å thick platinum in JEOL (Akishima, Tokyo, Japan)sputter ion coater (to avoid sample charging) and then observed at 15 kV at a magnification of 50, 80, 100 and 200 µm. The model of equipment used was S-3400N. FTIR spectroscopy was carried out on both resin and composite samples to qualitatively identify its constituent functional groups. The samples were ground with potassium bromide to form a paste which was then analysed. The analysis was carried out using Thermo Scientific Nicolet iS5-iD1 machine (Waltham, Massachusetts, USA) to determine the absorption band with a wavelength from 4000 cm−1 to 650 cm−1 with 16 scans at a resolution of 8 cm−1. DSC was performed to determine the thermal stability of the polystyrene resin and the composite. Measurements were carried out under dynamic nitrogen atmosphere (50 ml min−1) using a heating rate of 10°C min−1 between 25°C and 125°C using DSC PT1600 (Festool, Australia).
Results and discussion
Thermal characteristics of the polystyrene resin
The thermal effects of polystyrene resin–biochar composites at different biochar loadings were analysed. Thermal property analysis can be used to detect any changes brought in polystyrene resin with the addition of biochar. The thermal property spectrum of unfilled polystyrene resin is shown in Figure 2. The measured thermal property of resin sample in Figure 2 shows that thermal property increases steeply as temperature increases but decreases sharply at the range 35–45°C and increases again then decreases through a wide range. This may be due to the homogeneity of the resin mixture tested. The calculated heat capacity of the resin is 625.56 J/kg·K.
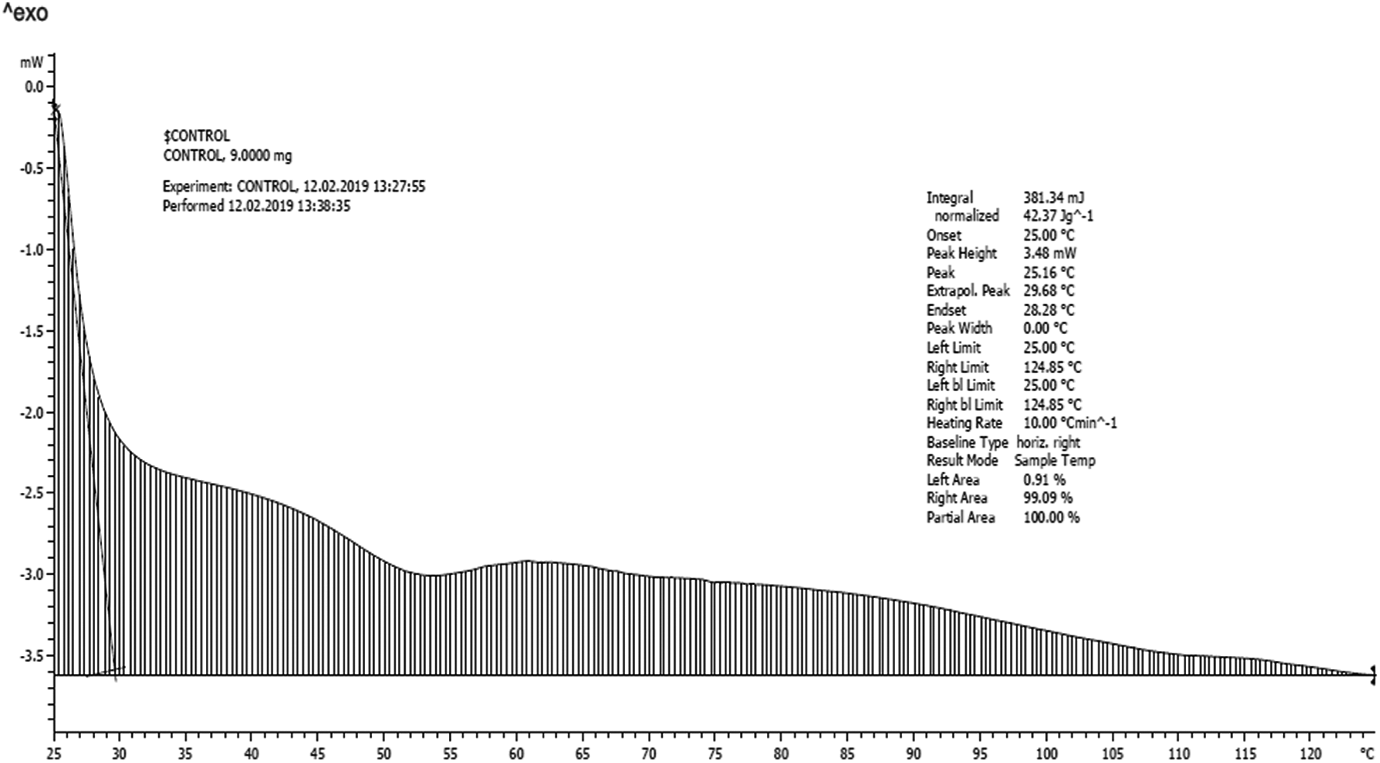
Thermal conductivity of resin with temperature.
Surface morphology of the polystyrene resin
SEM was used to study the morphology of the polystyrene resin and the resulting image at different magnification ranges is shown in Figure 3(a) to (c). The analysis carried out at increasing resolution gives better detail of the morphology of the resin. Figure 3(a) viewed at 1500× magnification has a plane surface with a clear visual of undissolved polystyrene foams on the surface. The unfilled polystyrene resin was observed to have flat and smooth surfaces, which also indicate the flexibility of polystyrene resin that allows for high elongation at break. This observation is common to Figure 3(b) and (c), respectively. Besides the smooth surface, there is a presence of microparticles in unfilled polystyrene resin. The appearance of these microparticles could be because of undissolved polystyrene in solvent entrapped in polystyrene resin. These particles visible at Figure 3(a) grow smaller at decreasing resolution over Figure 3(b) and (c), respectively. These particles, a product of incomplete dilution of polystyrene foam, have no chemical effect on the polystyrene resin and cannot cause any significant change to the composite formed from it.

SEM micrograph of unfilled polystyrene resin: (a) ×1500, (b) ×1000 and (c) ×500.
Functional groups of the polystyrene resin
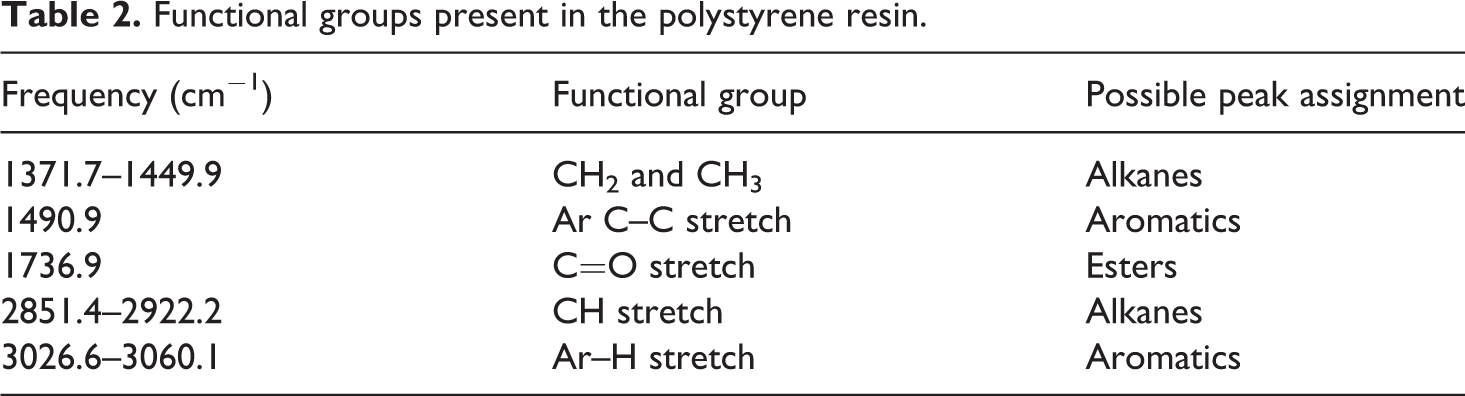
The functional groups of unfilled polystyrene resin were analysed by FTIR. FTIR analysis used to detect any changes brought in polystyrene with the addition of biochar, indicated by the appearance or disappearance of certain peaks of either polystyrene or biochar, which signifies the presence of chemical interaction between polystyrene and biochar. The FTIR spectra of unfilled polystyrene are shown in Figure 4. The absorbance peaks are located in a wide range of spectra scale. The first two peaks at 3060.1 and 3026.6 cm−1 have an aromatic C–H stretching vibration. The peaks at 2922.2 and 2851.4 cm−1 have identical vibrations with the presence of C–H stretch of alkane group. Spectra range 1449.9, 1490.9 and 1371.7 cm−1 indicate the presence of aromatic C–C stretch vibrations and alkane group. Some of the peaks in IR spectra of unfilled polystyrene indicated the presence of O–H stretching probably because of the presence of moisture in the sample. 31 The functional groups present in the polystyrene matrix are summarized in Table 2.
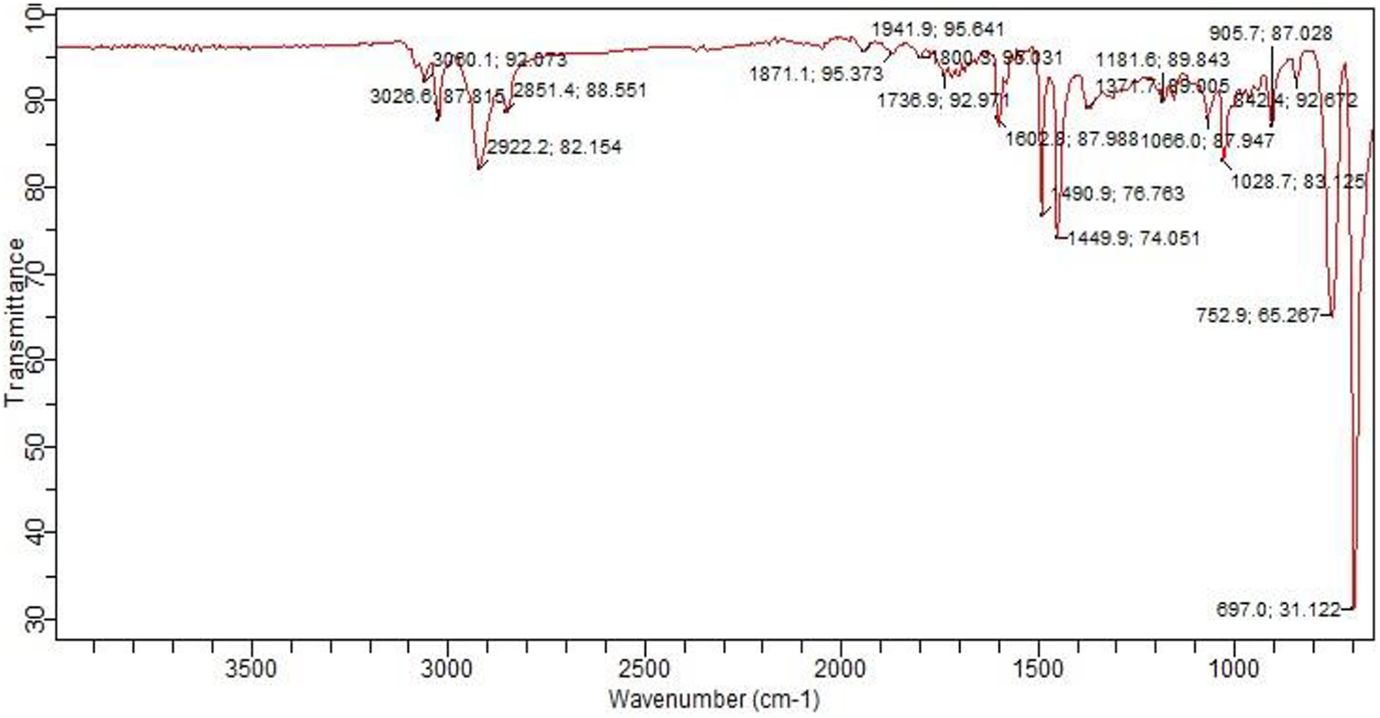
FTIR spectra of unfilled polystyrene resin. FTIR: Fourier-transform infrared spectroscopy.
Functional groups present in the polystyrene resin.
Thermal characteristics of the composite
The thermal conductivity of the composite at 10, 20, 30 and 40 wt% biochar composition was determined over a temperature range of 25–125°C and the values represented in Figure 5(a) to (d). The thermal conductivity decreases steeply then steadily and then steeply again as the temperature increases over time. The thermal conductivity at 20, 30 and 40 wt% decreased steadily then sharply and then increased steadily as the temperature increased over time. The behaviour of the heat capacity of different composites can now be explained using thermal conductivity values of the biochar. With the addition of 10, 20, 30 and 40 wt% of elephant grass biochar in the polystyrene resin, the heat capacity resulted to 657.03, 670.24, 757.66 and 601.92 J/kg·K, respectively, as shown in Figure 6. It is observed that the heat capacity increased at an increasing percentage fraction but decreased sharply at 40 wt%. It is detected that 30 wt% biochar composition possessed the highest amount of heat capacity with 757.66 J/kg·K. It can then be said that to obtain a composite of heat capacity range between 600 J/kg·K and 760 J/kg·K, a percentage range of biochar should be maintained at 10–30 wt%. The thermal conductivity study of composites helps to predict the composite response to heat.11,37 Researchers have studied the influence of biochar loading on the thermal properties of the composite. 32 Among these works, there have not been observed any potential conflict to the fact that an increasing amount of biochar loading increases the heat or thermal capacity of the composite. Das et al. 30 compared four biomass wastes (rice husk, coffee husk, coarse wool and landfill wood) with biochar filled composite. It was observed that the biochar did not undertake ignition under the radiative heat, whereas all the biomasses ignited rapidly. The conclusion drawn hence was the addition of thermally stable biochar in biomass added composite is beneficial when the thermal properties are concerned. 30
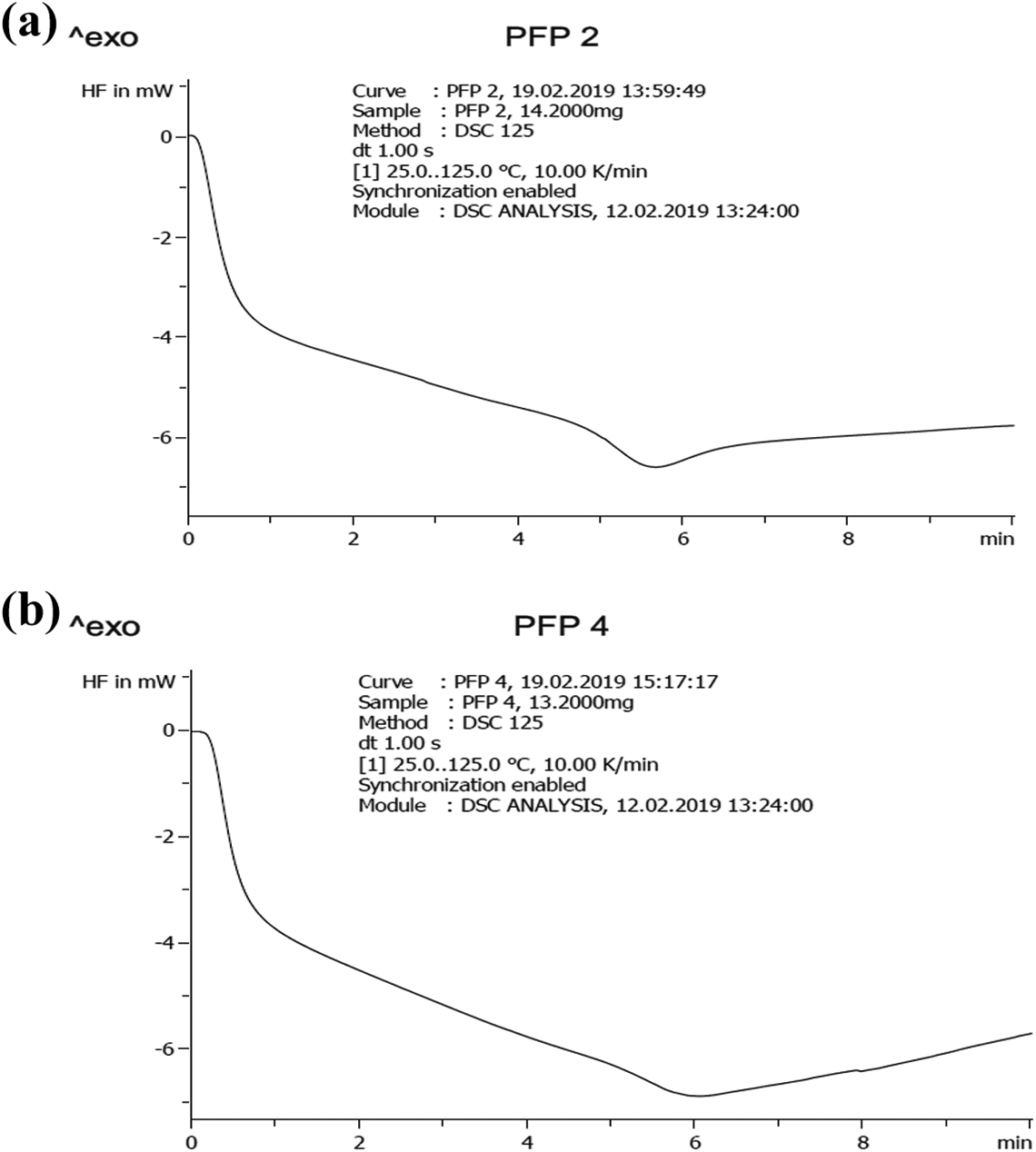
Thermal conductivity of composite with temperature over time: (a) 10%, (b) 20%, (c) 30% and (d) 40% biochar.
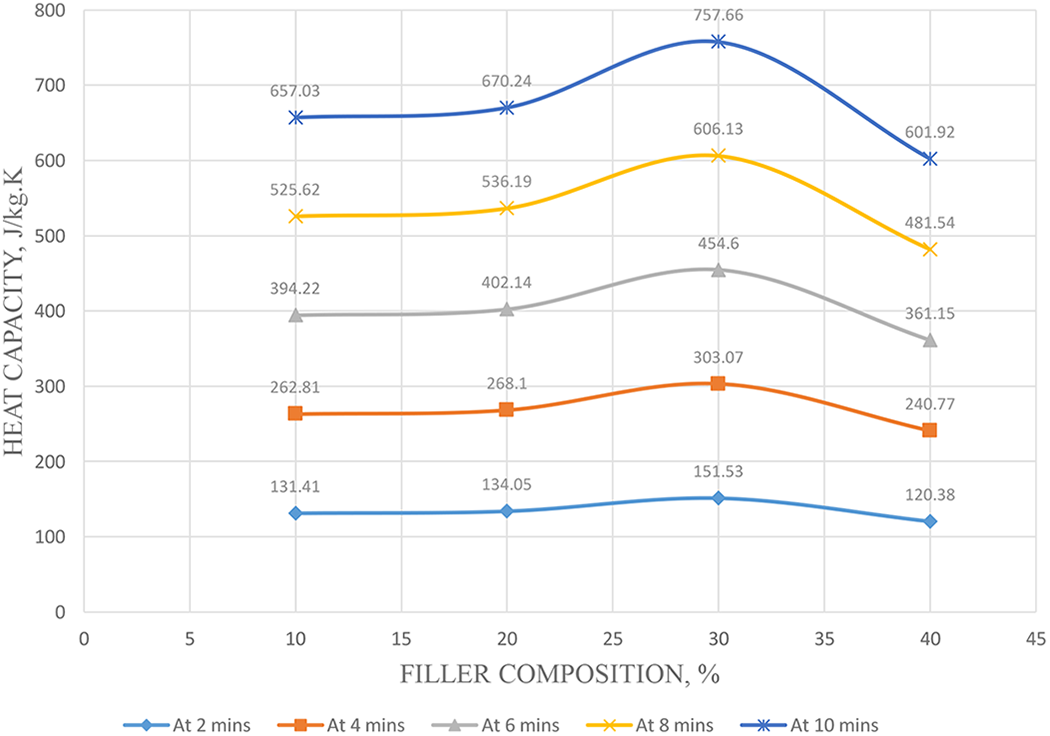
Heat capacity against composition at different times.
Surface morphology of the composite
The SEM analysis used to study the morphology of the composite was carried out and shown in Figure 7(a) to (c). In contrast to unfilled polystyrene resin that possessed a flat and smooth surface, the image displays a rougher surface due to the presence of biochar in the resin. The biochar took long cylindrical shapes closely packed together. The biochar when prepared was screened to microparticles which enhanced the interaction between the resin and biochar in the composite as more penetration of polystyrene resin was allowed into the highly porous and cracked structure of the biochar. Mechanical properties of reinforced polymeric composite material mainly depend on dispersion and distribution of filler, adhesion between filler and polymer resin, filler aspect ratio and orientation of filler. 32 In a study by Nan et al. 32 on the PVA/biochar composite, it was reported that due to the biochar filler’s relatively wide particle size distribution and apparent lower adhesion within the polymer resin, the increase in biochar content resulted in a significant decline in tensile strength. Figure 7(a) and (b) reveals the compactness of the infused biochar in the resin. As observed, there is no coagulation of biochar to a particular area but an even distribution of biochar particles. However, at a reduced resolution in Figure 7(c), the presence of gaps is observed in the composite. These gaps allow for elongation break in the composite when stress or load is applied or acts on it, therefore, enhancing the durability of the composite. However, these gaps would allow for transport of oxygen between the ambient environment and the underlying materials which might give the composite a good reaction to fire properties. 30

SEM micrograph of biochar composite: (a) ×1500, (b) ×1000 and (c) ×500. SEM: scanning electron microscopy.
Functional groups of the composite
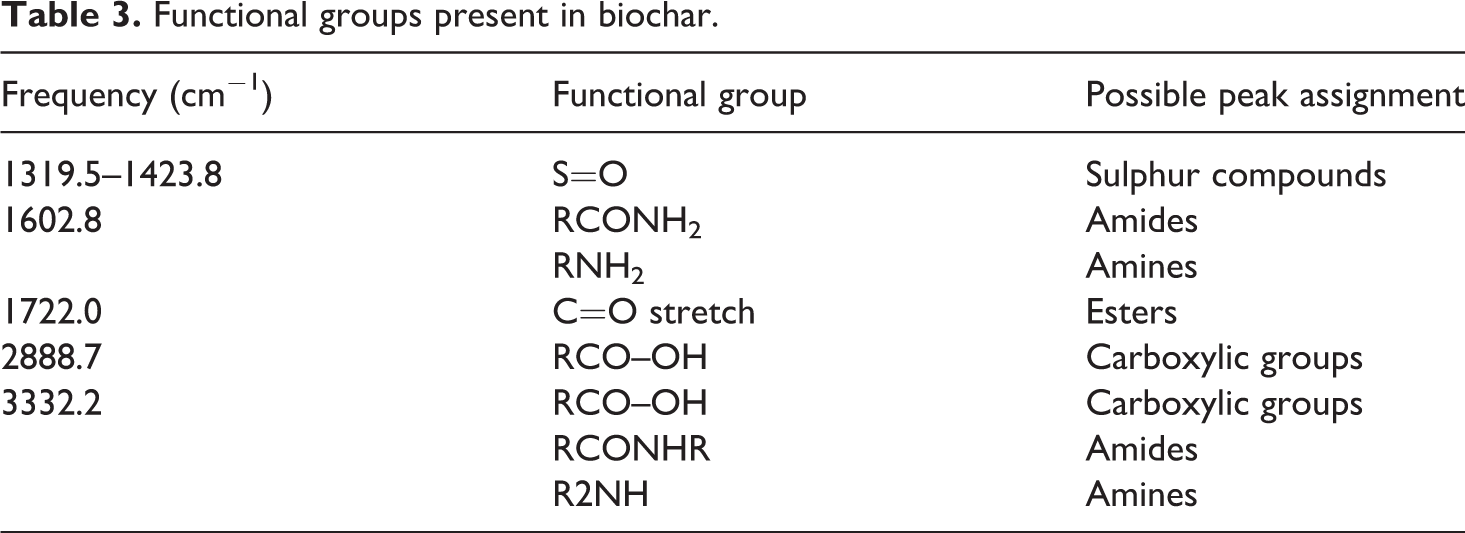
The functional groups of elephant grass biochar and biochar composite were also analysed using FTIR. The functional groups of biochar analysed using FTIR as shown in Figure 8 illustrate the peaks of biochar. Carboxylic groups were largely present at different frequencies, however predominant at 3332.2 and 2888.7 cm−1. C=O stretching was observed at 1722.0 cm−1 frequency indicating the presence of esters and aldehydes. Amide and amine groups were also observed in biochar at frequencies 3332.2 and 1602.8 cm−1 as well as sulphur compounds detected at 1423.8, 1367.9 and 1319.5 cm−1 due to the presence of S=O stretching. Furthermore, the absorption band identified at 1423.8 cm−1 indicated the presence of an aromatic compound in biochar, which corroborated with the findings of Ahmetli et al., 38 where aromatic compounds are found in chars derived from different materials. Likewise, as reported by Das et al., 29 wastes such as sewage sludge and poultry litter-derived biochar showed aromatic mode.
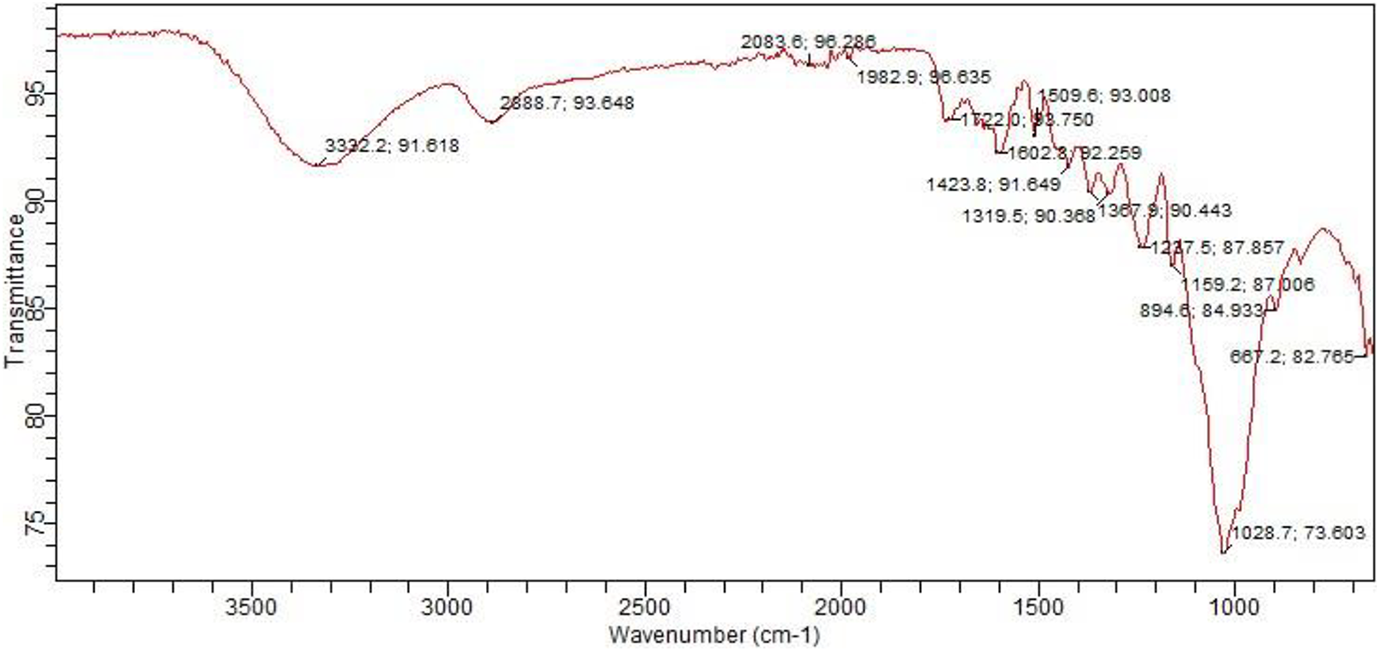
FTIR spectra of biochar.
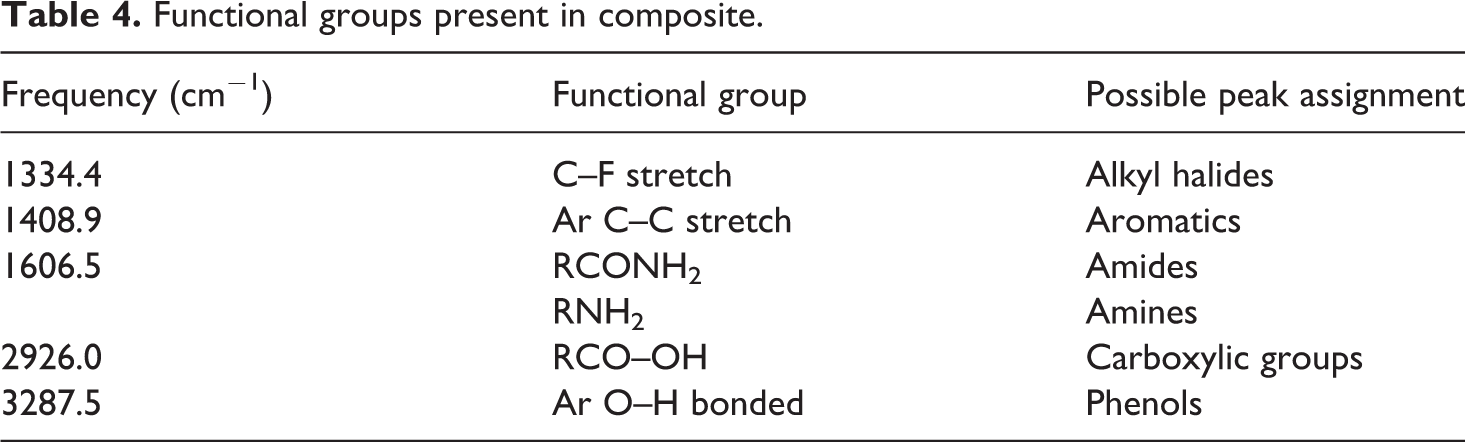
In reference to that, aromatic compounds in biochar composites shown in Figure 9 were identified due to the presence of absorption band indicating phenol at 3287.5 cm−1 and aromatics group at 1408.9 cm−1. C=C stretching at 1606.5 cm−1 was also observed indicating the presence of alkenes. Furthermore, amide and amine planes were also detected in biochar composites at 1606.5 cm−1 are presented in FTIR spectra along with the C–F stretching at a frequency around 1334.4 cm−1. In comparison, polystyrene possesses high peaks intensity from 1371.7 cm−1 to 3060 cm−1 frequency compared to biochar and biochar composite. Furthermore, biochar composite also possessed wider bands around 3287.5 cm−1 and fewer frequency compared to that of biochar and polystyrene resin due to the chemical reaction between the biochar and the resin leading to the appearance of new compounds such as phenols and disappearance of others such as esters. Most of the peaks shown in IR spectra of unfilled polystyrene indicate the presence of C–H bending of alkane groups. Functional groups present in biochar and composite are presented in Tables 3 and 4, respectively.
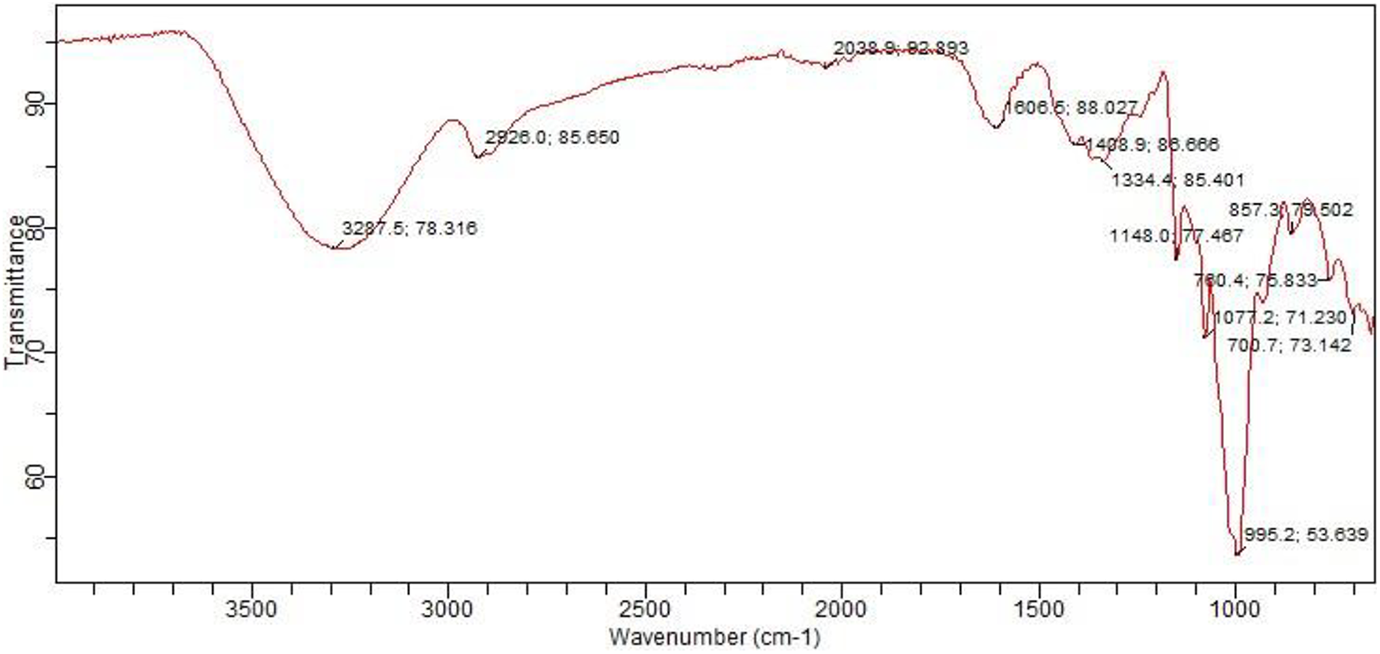
FTIR spectra of composite. FTIR: Fourier-transform infrared spectroscopy.
Functional groups present in biochar.
Functional groups present in composite.
Conclusion
Biochar-filled polystyrene composite was successfully prepared through manual mixing method and hand lay-up technique. Characterization of biochar revealed that biochar has high potential as other commercial fillers due to the presence of surface porosity and cracks which may contribute to physical interaction through polystyrene chain interlocking at biochar surface. Particles of biochar have a small aggregate structure which suggests that it can be easily dispersed in polystyrene resin. Besides, the particle size distribution of biochar falls in the range of other commercial fillers. Furthermore, biochar also did not contain any reactive hydrophilic groups, thus, it is compatible with polystyrene. Estimated heat capacities of the composites from the thermal test results also showed that to obtain a composite of heat capacity range between 600 J/kg·K and 760 J/kg·K, a 10–30 wt% range of biochar composition should be maintained where at 30 wt% biochar composition, heat capacity was at maximum with 757.66 J/kg·K. Furthermore, the addition of thermally stable biochar in the composite is beneficial when the thermal properties are concerned.
Supplemental material
Supplementary_Material - Morphological and thermal properties of polystyrene composite reinforced with biochar from elephant grass (Pennisetum purpureum)
Supplementary_Material for Morphological and thermal properties of polystyrene composite reinforced with biochar from elephant grass (Pennisetum purpureum) by Adewale George Adeniyi, Sulyman Age Abdulkareem, Joshua O Ighalo, Damilola Victoria Onifade, Samson Akorede Adeoye and Abia Elizabeth Sampson in Journal of Thermoplastic Composite Materials
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
