Abstract
Background
Plasma-derived human C1-esterase inhibitor (C1-INH) concentrates are indicated for the treatment of acute episodes of hereditary angioedema. Intravenous C1-INH concentrate provides rapid relief to patients with acute hereditary angioedema symptoms.
Objective
To develop a safe and efficient purification process for C1-INH from human plasma.
Design
A combination of anion exchange and hydrophobic interaction chromatography was used to purify C1-INH from human plasma.
Method
The cold insoluble fraction (cryoprecipitate) was separated via centrifugation; then, the cryoprecipitate-poor plasma was subjected to different separation techniques to purify C1-INH. First, separation was performed using diethylaminoethyl (DEAE) anion exchange chromatography to capture coagulation factors. Then, C1-INH was purified from the remaining plasma using quaternary amine (a strong anion exchanger) chromatography, followed by DEAE (a weak anion exchanger) chromatography. The final product was purified via hydrophobic interaction chromatography, followed by ultrafiltration to concentrate the purified C1-INH. This process is cost-effective because coagulation factors were captured before C1-INH purification and the waste fraction after the capture stage was diverted to Cohn fractionation to isolate other potential plasma products.
Results
The final C1-INH preparation had a specific activity of >12 IU/mg protein, which is sufficient for its intended purpose. The purified preparation was characterized via sodium dodecyl sulfate–polyacrylamide gel electrophoresis, and coagulation factors were measured in the purified preparation. Furthermore, viral activity decreased via solvent detergent treatment and virus filtration.
Conclusion
We successfully developed a purification process for C1-INH from human plasma. This process is easily adaptable and has been successfully scaled up to approximately 15-fold. It can be further scaled up for the large-scale production of C1-INH.
Introduction
C1 esterase inhibitor (C1-INH), a protein derived from human plasma, is a member of the serine proteinase inhibitor (serpin) superfamily.1,2 It is a single polypeptide chain containing 478 amino acid residues, with a molecular weight of approximately 90–105 kDa under non-reducing conditions and 85–93 kDa under reducing conditions.
C1-INH, generally activated during inflammation, inactivates its substrate by covalently binding to the reactive site. C1-INH is a major regulator of the classical complement, contact activation, and fibrinolytic pathways. Furthermore, it is the only inhibitor of the classical C1r and C1s pathways.1–3 In addition, C1-INH is the main inhibitor of coagulation factor XIa in the intrinsic coagulation cascade. 4
In many countries, C1-INH concentrate is considered the standard of care for treating hereditary angioedema (HAE). Studies have revealed the high effectiveness of this treatment modality, with improvements in symptoms within 30 min to 1 h. In addition to its role in treating acute HAE attacks, C1-INH may also be beneficial as a chronic prophylactic agent. 5
Over the years, C1-INH concentrates have been successfully prepared from human plasma using a combination of conventional precipitation reagents, including ammonium sulfate and polyethylene glycol 4000, and various chromatography techniques, including ion exchange chromatography and hydrophobic interaction chromatography. 6
Chromatography techniques, such as ion-exchange, affinity, or size-exclusion chromatography, allow specific separation of proteins based on distinct properties like charge, size, or specific binding affinities. This selectivity reduces impurities and increases yield. 7 Unlike precipitation, which may involve harsh conditions (eg, high salt concentrations, extreme pH), chromatography generally operates under milder conditions. This reduces the risk of denaturing proteins, thereby preserving their biological activity and structure. 8 Chromatographic methods can achieve very high recovery rates and purity levels due to their precision in separating proteins based on distinct properties. Precipitation may result in lower purity due to co-precipitation of unwanted proteins. 9 Chromatography is highly adaptable for automated systems. This makes it more efficient and scalable for industrial applications compared to precipitation. 10 In summary, chromatography's specificity, gentle processing conditions, and ability to handle complex mixtures make it highly advantageous over precipitation for protein purification, especially in cases requiring high purity and active protein.
Manufacturers of C1-INH use multiple virus reduction procedures to ensure product safety. These procedures include pasteurization, hydrophobic interaction chromatography, and virus filtration. 6
In this study, we present a three-step purification process encompassing a combination of ion exchange chromatography and hydrophobic interaction chromatography. This process utilizes cutting-edge purification technology of a quaternary amine resin, a strong anion exchanger, offering a high dynamic binding capacity. The developed process does not include any precipitation or plasma reconditioning (ie, changes in pH and conductivity) steps, making the plasma available for Cohn fractionation and possibly facilitating the isolation of all plasma products. For buffer exchange and concentration, tangential flow filtration was performed. Furthermore, to decrease the risk of virus transmission via the purified product, two orthogonal virus clearance steps were included in the purification process.
Material and Methods
Equipment and Reagent
The chromatography system and columns were procured from Cytiva (Sweden) and BioRad (USA). The chemicals required for buffer preparation and solvent detergent (SD) reagent were provided by Merck (Darmstadt, Germany). The TECHNOCHROM C1-INH kit was used to determine C1-INH activity using a chromogenic assay on the ACL TOP automatic coagulometer from Instrumentation Laboratory. C1-esterase inhibitor first WHO standard (NIBSC-08/256) was used for calibration. A nephelometer (Atellica Neph 630, Siemens) was used to determine the antigenic content of the C1-INH protein. A specific C1-INH antiserum (Siemens Healthcare Diagnostics GmBH, Germany) was used for the analysis. A standard protein molecular weight marker for sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) was obtained from BioRad. Total protein was estimated using the Quick Start Bradford 1X dye reagent from Biorad (USA).
An automatic coagulometer (ACL Elite Pro) and reagents from Instrumentation Laboratory Company were used as per the manufacturer's instructions to evaluate coagulation factors (factors II, VII, and X). For the assay calibration, the activities of F-II, F-VII, and F-X were determined in calibration plasma using WHO standard (09/172). Undiluted samples were analyzed to obtain the final result.
Figure 1 provides a schematic of the C1-INH purification process. The initial process steps are part of the regular manufacturing process for commercial prothrombin complex concentrate and factor IX products. Table 1 provides the operation parameters for all chromatographic steps in C1-INH preparation.

Schematic of the C1-INH preparation procedure
Operation parameters for the chromatographic steps in the C1-INH preparation procedure.

Abbreviations: NA, not applicable; WFI, Water for injection.
Removal of Coagulation Factors from the Cold Insoluble Fraction
First, plasma was thawed at 2 °C–8 °C; then, the cold insoluble fraction (or cryoprecipitate) of plasma was separated via centrifugation at 5000 × g for 15 min. The remaining plasma, called cryoprecipitate-poor plasma, was passed through a pre-equilibrated weak anion exchange chromatography column (diethylaminoethyl resin, DEAE-1) to capture the prothrombin complex concentrate. The flowthrough after prothrombin complex capture was used as the starting material for C1-INH purification.
Purification of C1-INH
The flowthrough obtained after prothrombin complex capture was passed through a strong anion exchange column (quaternary amine resin). C1-INH and some contaminating proteins were observed to be in bound form. Therefore, the flowthrough was subjected to Cohn fractionation to isolate other potential plasma-derived medicinal products such as immunoglobulin products, albumin, and alpha-1 proteinase inhibitor. Elution was performed by increasing the concentration of sodium chloride to obtain C1-INH. However, before elution, a few contaminating proteins were removed by decreasing the pH of the column. The elute was treated with 1% v/v Tween 80 and 0.3% v/v tri-n-butyl phosphate for 6 h at 24 °C–27 °C (SD treatment) to inactivate any potential enveloped viruses.
Thereafter, the elute was purified using a weak anion exchanger (diethylaminoethyl resin, DEAE-2) for further purification. Chromatographic conditions (Table 1) were designed such that the contaminating proteins were kept in an unbound fraction, whereas C1-INH was bound to the column. Elution was performed using 250 mM sodium chloride. The C1-INH eluted from the weak anion exchange column was further subjected to hydrophobic interaction chromatography (phenyl resin). The conditions for hydrophobic interaction chromatography were optimized to maintain the C1-INH in an unbound form, with the contaminating proteins being bound to the column.
The flowthrough obtained after hydrophobic interaction chromatography was concentrated and exchanged with a buffer. Virus filtration was performed to remove any potential viruses. Finally, the filtrate was concentrated to match the desired potency of the drug product. The drug substance was prepared by filtering the concentrated bulk through a 0.2 µ filter.
The process was conducted at two different scales to evaluate the effect of scaleup. The process was scaled up to approximately 15X.
Analytical Methods Used to Characterize C1-INH
The Bradford assay was used to determine protein content, 11 with UV measurements at 280 mM. Nephelometry was used to determine antigenic C1-INH. 12 The TECHNOCHROM C1-INH kit was used to measure C1-INH activity. 13 SDS-PAGE 14 was performed in reducing conditions using 10% gels. Each well was loaded with 1 µg of protein/well. Silver staining was performed.
An automatic coagulometer (ACL Elite Pro) was used to estimate coagulation factors such as factors II, VII, and X in the preparation.
Results
Coagulation factors were removed using the DEAE-1 column (data not shown) and the obtained cryoprecipitate-poor plasma (after capturing prothrombin complex concentrate proteins) was used to isolate C1-INH using a strong anion exchange column (Figure 2). The elute contained ∼70% of C1-INH with respect to the content of C1-INH loaded onto the strong anion exchange column. The specific activity increased from 0.02 IU/mg to >4 IU/mg (Table 2 Parts A and B). The data presented is the average of three batches at each scale.
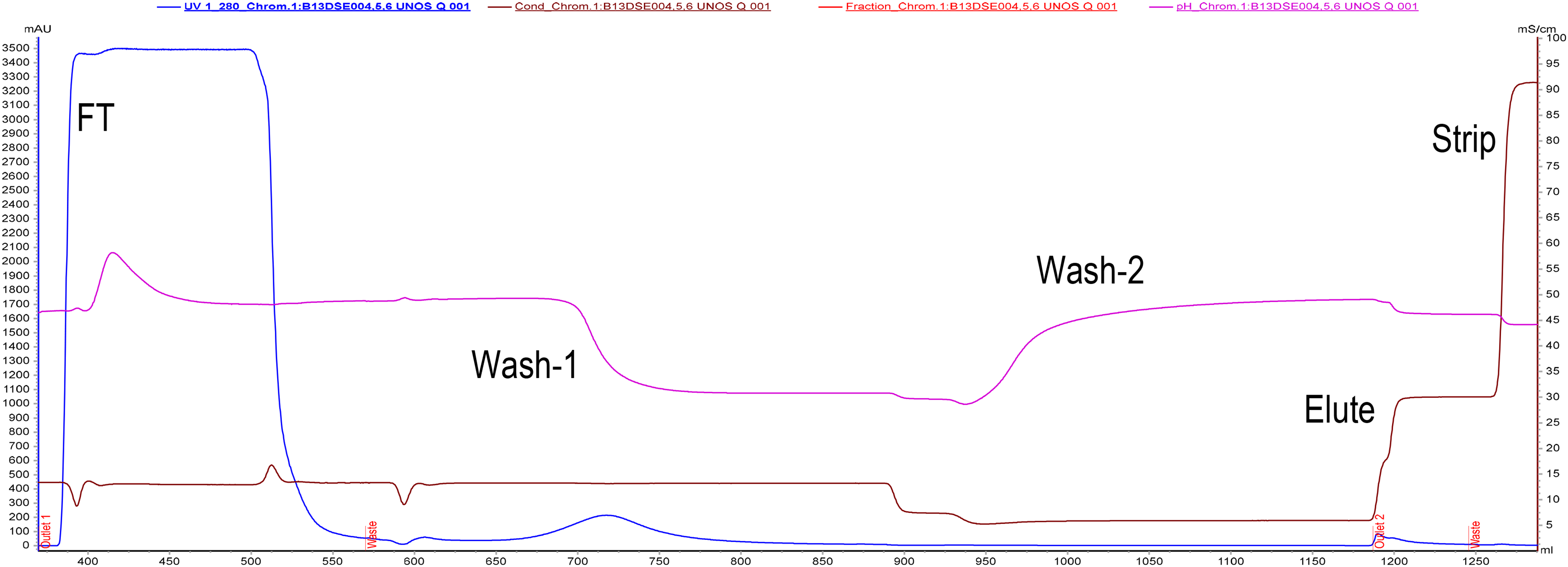
Purification of C1-INH on the quaternary amine resin. The maximum absorbance in the flowthrough indicated the presence of contaminating proteins in the unbound fraction. In wash 1, a few contaminating proteins were eluted. C1-INH was eluted as a small peak in the elution buffer.
Results of the C1-INH purification batches.


This elute was subjected to SD treatment for 6 h; no activity was lost in this stage (Table 3).
Recovery of C1-INH in the SD treatment step.

The reagents used in the SD step were removed during the subsequent purification step (DEAE-3) (Figure 3), in which the step recovery was >90% and the specific activity increased to >9 IU/mg. Hydrophobic interaction chromatography also yielded a step recovery of approximately 90% and the specific activity increased to >12 IU/mg (Figure 4).

Purification of C1-INH on weak anion exchange column (DEAE-2) where C1-INH eluted with maximum absorbance. Contaminating proteins were either in the unbound fraction or were tightly bound; therefore, they appeared in the column strip.
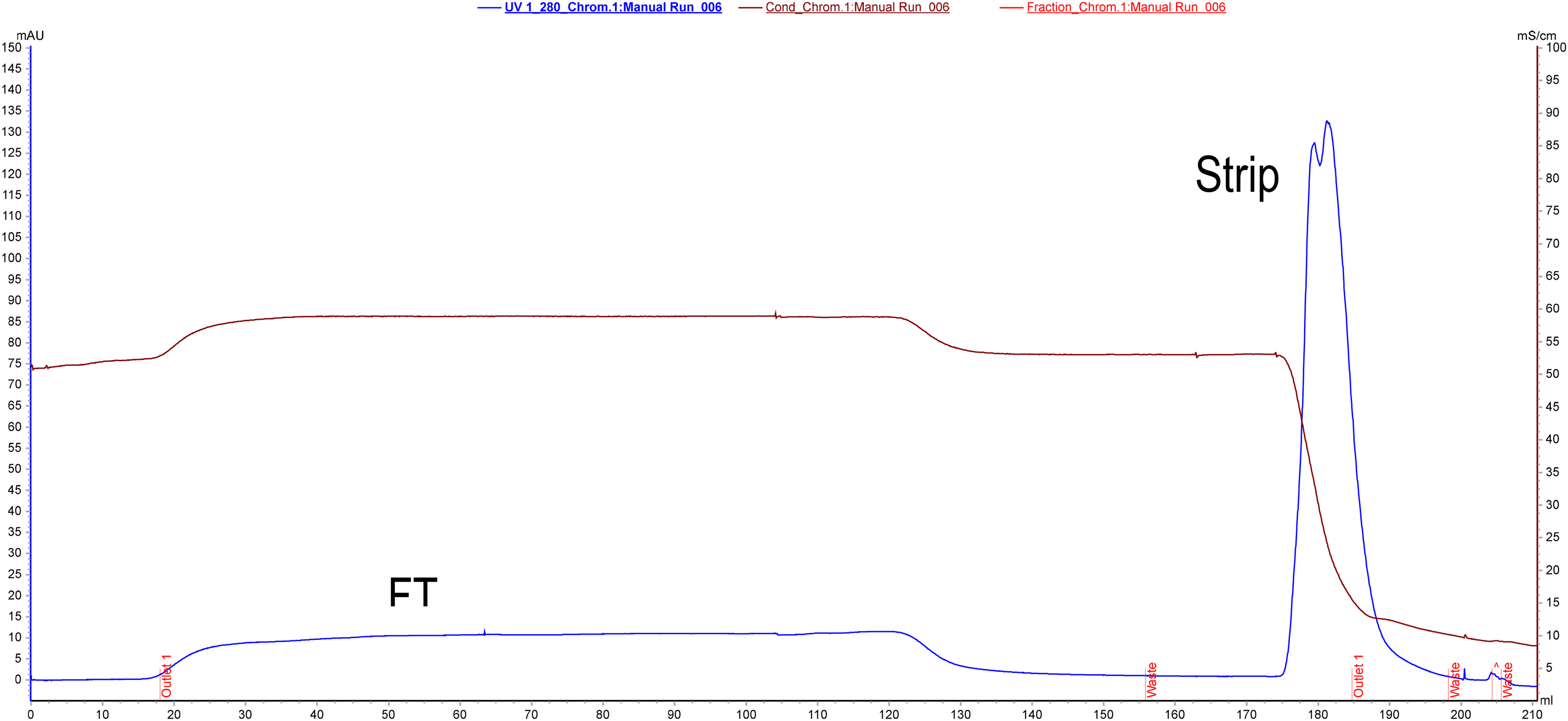
Purification of C1-INH on hydrophobic interaction chromatography. In this column, C1-INH remained in an unbound fraction (negative purification step). Contaminating proteins
Table 2 Part A and Part B presents the results from the C1-INH preparation steps, with step recoveries and the specific activity values, which clearly indicate the increase in specific activity during the purification process.
Furthermore, SDS–PAGE (Figure 5) revealed that the contaminating proteins were successfully removed during the purification process and the final purified protein had high purity.

Reducing SDS–PAGE. Electrophoresis was performed using 10% Mini-PROTEAN® TGX™ precast gel from BioRad. Each lane was loaded with 1 µg of total protein. Lane 1: DEAE-1 load, Lane 2: quaternary amine column load, Lane 3: quaternary amine column eluate, Lane 4: DEAE-2 eluate, Lane 5: molecular weight marker, Lane 6: Reference marketed product, and Lane 7: HIC flowthrough.
Negligible amounts of coagulation factors were measured in the preparation (factor II: 0.004 IU/mL, factor VII: 0.003 IU/mL, and factor X: 0.0061 IU/mL).
Discussion
We successfully developed a manufacturing process for C1-INH. In this approach, the separation of the cryopaste from the cryoprecipitate-poor plasma leads to the isolation of factor VIII and fibrinogen products.15,16 The cryoprecipitate-poor plasma is used to capture the prothrombin complex proteins, leading to the isolation of the prothrombin complex concentrate and factor IX product.17,18 The flowthrough of weak anion exchange chromatography can be used to isolate C1-INH using strong anion exchange chromatography. The waste fraction from the C1-INH capture stage can be subjected to fractionation to isolate other plasma-derived medicinal products such as immunoglobulins and albumin. 19 This approach allows the extraction of several different proteins from the same starting material, making the process cost-effective. The capture stage of the C1-INH purification process, involving strong anion exchange chromatography, was optimized efficiently to capture and purify C1-INH, with a step recovery of approximately 70% with respect to the content added into the strong anion exchange chromatography column. Furthermore, a more than 200-fold increase in specific activity was observed. During the weak anion exchange chromatography step (DEAE-2), the reagents used in SD treatment were removed, further increasing the specific activity of the product. As depicted in Figure 5, maximum contaminating proteins were removed during the hydrophobic interaction chromatography step. The final purified product exhibited a specific activity of >10 IU C1-INH/mg of protein, making it suitable for use as a drug. Two virus reduction steps based on different principles were employed to ensure a high degree of virus safety. SD treatment and virus filtration are the most widely used methods for virus inactivation. Using the SD treatment procedure, enveloped viruses such as human immunodeficiency virus, hepatitis B virus, and hepatitis C viruses can be inactivated. 20 Virus filtration is included to increase virus safety by removing both enveloped and non-enveloped viruses. 20 Principally, virus reduction in virus filtration is based on size exclusion; 20 in contrast, virus reduction in SD treatment is achieved by disrupting the lipid bilayer of enveloped viral pathogens. 21 Virus filtration was originally introduced to remove non-lipid enveloped viruses such as parvovirus B 19 and hepatitis A virus and to provide protection from unknown infectious agents. 22 Moreover, coagulation factors such as factors II, VII, and X were identified in trace quantities in the preparation, adding to the purity of the preparation.
In conclusion, we developed a process for purifying C1-INH from human plasma for application in HAE treatment and prophylaxis. 23 The C1-INH preparation has a high degree of virus safety 24 and sufficiently high specific activity for this purpose. Furthermore, the process is cost-effective because it allows the extraction of multiple plasma-derived products from the same plasma.
Limitation
Limited plasma donations lead to a scarcity of such critical care products in India. There is a huge requirement of spreading awareness for blood donation so that more plasma is available to the plasma fractionation industry.
Future Prospects
In this study, we clearly defined the purification process for C1-INH from human plasma that is easily adaptable and scalable. However, for the commercial use of the product, process scale-up and validation studies are warranted.
In addition, to establish the product as a drug, virus clearance, stability, re-usable resin cycle, and detailed characterization studies of the drug product as per Pharmacopeia are warranted.
Conclusion
We successfully developed and scaled up the process for purifying C1-INH from human plasma. The C1-INH preparation has high specific activity, making it suitable for developing a drug for treating HAE. The developed process involves chromatographic steps which have mild conditions compare to the precipitation steps, thus the risk of protein denaturation is minimum. Additionally, the process developed is cost-effective because the cold insoluble fraction separated via centrifugation can be used to isolate fibrinogen and factor VIII products, whereas the cryoprecipitate-poor plasma can be used to isolate coagulation factor products such as prothrombin complex concentrate and factor IX. After removing the cold insoluble fraction and coagulation factors, the remaining plasma was used to purify C1-INH. The waste fraction after C1-INH capture on the quaternary amine resin was subjected to Cohn fractionation to isolate other potential plasma-derived products.
Footnotes
Acknowledgements
We are thankful to the management of Intas Pharmaceuticals Ltd for allowing the work to be conducted at the research and development laboratory of the plasma fractionation unit. We are also thankful to all donors who have contributed to the project by donating their blood/plasma.
Author contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the management of Intas Pharmaceuticals Ltd and did not receive any financial grant from other funding agencies in the public or commercial sectors.
Ethics Approval and Consent to Participate
Indian Blood banks provided freshly frozen recovered plasma in polyvinyl bags. These blood banks had been approved by the Drug Controller General of India (DCGI) -NOC No. X11026/269/2018-BD and registered with the government of India's National Aids Control Organization. Blood was collected after receiving written consent from the blood donors and after verification by medical practitioner.
