Abstract
Background
Although reduced doses of enoxaparin have been studied in low body weight patients, no study has directly compared enoxaparin to unfractionated heparin for venous thromboembolism (VTE) prophylaxis in this population.
Objective
To determine the most safe and efficacious parenteral venous thromboembolism prophylaxis regimen for low body weight patients.
Design
Retrospective cohort.
Methods
Hospitalized patients weighing < 55 kg receiving either enoxaparin or unfractionated heparin for VTE prophylaxis over a period of two and a half years were evaluated. Primary outcomes included rates of VTE and overall bleeding events. Secondary outcomes included rates of major and clinically relevant minor bleeding. Dosing regimens were analyzed to determine associations between dose, weight, creatinine clearance, VTE, and bleeding events. Primary and secondary outcomes were evaluated using Chi squared and Fischer's Exact tests.
Results
Three hundred and sixty-one patients contributed 400 episodes of VTE prophylaxis. Most patients were female (88%), had a Padua score ≥ 4 (82%) and were admitted to medical/surgical units (76%). VTE rates were 0.4% in patients receiving enoxaparin and 2.5% in patients receiving unfractionated heparin (P = 0.334). Bleeding rates were 12% and 11% in the enoxaparin and unfractionated heparin groups, respectively (P = 1). Patients receiving enoxaparin 30 mg daily had bleeding rates of 7.6% compared to 14.7% in patients receiving 40 mg daily (P = 0.134). Patients receiving heparin twice daily had no bleeding events compared to 12.9% of patients receiving heparin three times daily (P = 0.222).
Conclusion
Overall, no difference in VTE or bleeding rates were found between patients weighing < 55 kg receiving enoxaparin versus unfractionated heparin, and there was no difference in bleeding or VTE events in the patients receiving enoxaparin 30 mg daily. Due to this, empirically reducing the dose of enoxaparin for VTE prophylaxis appears reasonable and safe in patients weighing < 55 kg.
Introduction
Venous thromboembolism (VTE) is a disease that is associated with significant short-term and long-term morbidity and mortality. 1 Hospitalization is associated with an increased risk for VTE, and in high risk hospitalized patients the rate of VTE is 11%.2,3 Preventable, hospital-acquired VTE is also associated with significant financial costs to the health care system. 4 For these reasons, it is important for hospitalized patients to continuously be evaluated for VTE prophylaxis candidacy and receive pharmacologic prophylaxis, if indicated.
The 2012 Chest guidelines recommend that acutely ill hospitalized medical patients at increased risk for thrombosis should receive pharmacologic prophylaxis. 5 Though no specific anticoagulant is recommended, subcutaneous unfractionated heparin (UFH) and low-molecular weight heparins, such as enoxaparin, are frequently used. Standard dosing of enoxaparin and UFH for VTE prophylaxis in the general medical population is 40 mg daily and 5000 units every 8 to 12 h, respectively.6,7 In the trauma population, twice daily dosing is recommended for enoxaparin. 8 Standard dosing schemes for enoxaparin and UFH utilize flat doses, meaning patients at lower weights receive a relatively higher weight-based dose. Though UFH and enoxaparin have been studied in obese patients and dosing recommendations exist based on body mass index, recommendations for low body weight are still lacking.9–13
Despite patients with low body weight being at an increased risk of bleeding, they are underrepresented in the literature when compared to obese patients and patients of average body weight. 14 Enoxaparin prophylaxis has been evaluated in low body weight patients; however, many of these studies did not evaluate clinical outcomes and focused on anti-factor Xa activity.15–17 Low body weight patients receiving enoxaparin 40 mg daily were more likely to have supratherapeutic anti-factor Xa concentrations, and patients receiving enoxaparin 30 mg daily were more likely to be within the target prophylaxis range.15,16 Two studies primarily evaluated clinical outcomes of VTE and bleeding. Of those, one did not find statistically significant differences between enoxaparin 40 and 30 mg daily whereas the other found that reduced doses of anticoagulation were associated with less bleeding.18,19 Both studies lacked a comparison between enoxaparin and UFH.18,19 All studies to date have excluded patients with a creatinine clearance (CrCl) less than 30 milliliters/minute (mL/min), a key group of patients in which UFH may be preferentially used.15–19
Due to concern of increased bleed risk with standard doses, especially in the presence of renal dysfunction, empiric dose reductions of enoxaparin without a true indication (CrCl < 30 mL/min) or switching to UFH is sometimes done for low body weight patients. Ultimately, there is limited evidence for this practice. Whether dose reductions or changes in anticoagulants are necessary is unclear. At the time of this writing, there is no study directly comparing enoxaparin to UFH in low body weight patients. The purpose of this study was to determine the most safe and efficacious parenteral VTE prophylaxis regimen for low-body weight patients.
Methods
This retrospective cohort study included hospitalized patients weighing less than 55 kg who received at least three days of enoxaparin or UFH for VTE prophylaxis between January 1, 2019 to June 30, 2021. This study occurred within UofL Health, a health system in Louisville, Kentucky consisting of four hospitals totaling 1234 beds. One of these hospitals, UofL Hospital, is an academic teaching hospital with a level I trauma center and cancer center. Data were collected from patient charts in the electronic health record. Patients were excluded if they had an active VTE or bleed documented prior to VTE prophylaxis, received enoxaparin 30 mg twice daily (BID), oral anticoagulants or fondaparinux, had a history of heparin-induced thrombocytopenia, diagnosis of a thrombophilic condition or bleeding disorder, platelet count < 50,000 platelets/microliter (mcL), end-stage renal disease requiring hemo- or peritoneal dialysis. Pharmacologic VTE prophylaxis was initiated at the discretion of each provider. An episode of prophylaxis was defined as an individual order for enoxaparin or UFH prophylaxis from initiation of the order to discontinuation of the order. Patients were eligible to have multiple episodes of VTE prophylaxis included throughout their admission, and a patient could have received both enoxaparin and UFH during their admission. Episodes of VTE prophylaxis with enoxaparin were compared with UFH, then analyses were subdivided into dosing regimens of each anticoagulant. Per UofL Health policy, pharmacists could dose adjust enoxaparin for CrCl < 30 mL/min. However, no dose adjustments could be made automatically based on BMI or body weight. Padua scores were calculated based on patient characteristics at initiation of VTE prophylaxis. Because all patients weighed less than their ideal body weight, CrCl was calculated using the Cockcroft-Gault equation and total body weight. The study protocol was approved by the University of Louisville Institutional Review Board (21.0735).
The primary efficacy outcome was the rate of VTE, and the primary safety outcome was the rate of bleeding. VTE and bleeding events were attributed to anticoagulation if they occurred within 24 h from the last dose of enoxaparin or UFH. Secondary outcomes included the rates of major and clinically relevant minor bleeding (CRMB) assessed via chart review and as defined by the International Society of Thrombosis and Haemostasis. 20 (Table 1). Additionally, sub-analyses of VTE and bleeding events by individual dosing regimens within each group, and associations between dose, weight, CrCl cut-off of 30 mL/min, and VTE and bleeding events were conducted. An exploratory analysis was performed regarding bleeding and the risk factor of trauma/surgery.
ISTH Criteria for Bleeding.

Data were analyzed using R software (R Foundation for Statistical Computing; Vienna, Austria). Unpaired t-tests and chi-squared tests were performed for normally distributed continuous and categorical data. The Mann-Whitney U test was used for non-parametric data. Point biserials were used to evaluate associations with respect to bleeding with weight and dose. Bleeding rates in prior literature range from 5.7%−21.4%. Based on an estimated bleeding rate of 14%, 1510 episodes of VTE prophylaxis would be necessary to detect a difference of 5% with 80% power. A P-value < 0.05 was considered statistically significant. P-values were adjusted due to multiple comparisons using a Benjamini–Hochberg method.
Results
Six hundred and fifty-eight patients were evaluated for inclusion. After exclusion criteria, 361 patients contributed 400 episodes of VTE prophylaxis for analysis. Of these episodes, 242 were included in the enoxaparin arm and 158 in the UFH arm. Baseline demographics can be found in Table 2. Compared to the enoxaparin group, more patients in the heparin group were male (18%), had acute or chronic kidney impairment (59% and 16%), lower platelet counts (237 ± 124 platelets/mcL), and were originally admitted to the intensive care unit (ICU) before transferring to a progressive or medical-surgical unit (32%). Most patients did not have trauma/surgery in the preceding 30 days (62.5%). One-hundred and five of the 400 VTE prophylaxis episodes utilized enoxaparin 30 mg daily, with the majority receiving this dose (93; 39%) with a CrCl ≥ 30 mL/min. Conversely, 13 episodes of VTE prophylaxis utilized 30 mg daily with a CrCl < 30 mL/min. The mean ± standard deviation (SD) weight-based dose of enoxaparin was 0.8 ± 0.2 mg/kg/dose. The mean ± SD weight-based dose of UFH was 108.3 ± 17.7 units/kg/dose.
Baseline Demographics.

BID = two times daily; BMI = body mass index; CrCl = creatinine clearance; Hgb = hemoglobin; ICU = intensive care unit; NSAID = non-steroidal anti-inflammatory drug; SNRI = serotonin-norepinephrine reuptake inhibitor; SSRI = selective serotonin reuptake inhibitor; SCD = sequential compression device; TBW = total body weight; TID = three times daily.
Five VTE events occurred, one (0.4%) in the enoxaparin group and four (2.5%) in the UFH group (P = 0.334). Of the four VTE events in the UFH group, three patients experienced a pulmonary embolism, and one had an isolated thrombosis in the left iliac vein. The patient receiving enoxaparin had a catheter-associated thrombosis (Table 3). Twenty-eight (12%) and 18 (11%) patients had a bleeding event in the enoxaparin and UFH groups, respectively (P = 1).
Patients with VTE.
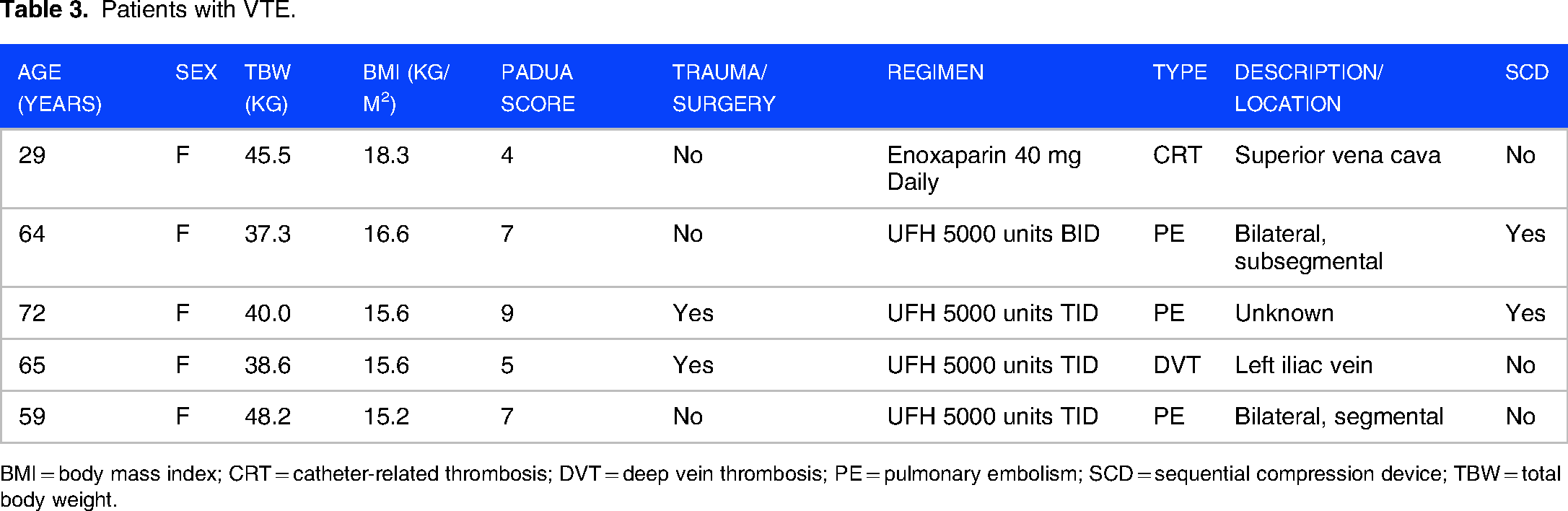
BMI = body mass index; CRT = catheter-related thrombosis; DVT = deep vein thrombosis; PE = pulmonary embolism; SCD = sequential compression device; TBW = total body weight.
Of the patients with bleeding events, rates of major bleeding and CRMB were 44% and 56% in both groups, respectively (P = 1). Overall, no statistically significant differences in rates of bleeding were found between dosing strategies (P = 0.138) (Table 4). There were eight (7.8%) bleeding events in the enoxaparin 30 mg daily group compared to 20 (14.7%) bleeding events in the enoxaparin 40 mg daily group (P = 0.134). There were 18 (12.9%) bleeding events in the heparin 5000 units three times daily (TID) group compared to no bleeding events in the heparin 5000 units BID group (P = 0.222). Bleeding rates did not differ based on having a CrCl < 30 mL/min (9.6% vs 11.9%; P = 0.717). Higher rates of bleeding were found in patients who had trauma/surgery prior to prophylactic anticoagulation (8% vs 17.3%; P = 0.008). No associations between bleeding events and total body weight were found.
Bleeding Rates Per Dosing Regimen.

Discussion
In this cohort of predominantly non-ICU, non-surgical medical patients with low body weight, no difference in VTE or bleeding events was identified between patients receiving enoxaparin or UFH for VTE prophylaxis. Of the patients receiving enoxaparin 30 mg daily, no cases of VTE occurred, but a clinically significant reduction in bleeding rates can be noted when compared to enoxaparin 40 mg daily. Our larger cohort corroborates the findings by Buckheit et al., where reduced dose anticoagulation was associated with less major bleeding without a significant increase in VTE events. 19 It appears that the VTE event rate for patients receiving enoxaparin 30 mg daily is likely not more than enoxaparin 40 mg daily, though a larger cohort of patients would be needed to truly answer this question. Relatively few patients received UFH 5000 units BID, however, no bleeding events occurred compared to the 12.7% of patients with a bleeding event in the UFH 5000 units TID group. A retrospective comparison of UFH 5000 units BID and TID found no difference in rates of bleeding or VTE, however, at least 75% of patients in this study were > 55 kg. 21 Additionally, a meta-analysis of six studies evaluating BID and TID heparin dosing concludes any difference in bleeding or VTE events, if one exists, would be small. 22 One of the 18 patients receiving UFH 5000 units BID in our study developed a pulmonary embolism, raising concerns of this dose providing limited protection from VTE.
The overall rate of VTE in our study at 1.3% is similar to the general population but higher than two previous studies of low body weight patients at 0.3% and 0%.3,18,19 This may be due to our study having a higher portion of trauma/surgical patients who are at an elevated risk of VTE and require higher doses of enoxaparin (8). Over 80% of the patients in our cohort had a Padua score ≥ 4, indicating they were at a higher risk of VTE and warrant pharmacologic VTE prophylaxis. All the patients with a VTE event had a Padua score ≥ 4. No clear trend was identified among the patients with a VTE.
The overall rate of bleeding of 11.5% in our study was considerably higher than the estimated 0.4% reported in the 2012 Chest guidelines. 5 Our bleeding rate is higher than reported in the study by Buckheit et al. but lower than in the study by Dybdahl et al.18,19 As the variable trauma/surgery was found to be significantly associated with bleeding, this could be the reason for the higher rates compared to Buckheit et al.'s findings. 19 Our elevated bleeding rate is consistent with previous studies in low body weight patients which reported overall bleeding rates ranging from 5.7%−21.4%.16–19 Though all retrospective data, together this appears to support the understanding that low body weight patients are at an increased risk of bleeding when compared to average weight patients. 14 Additionally, it raises the importance of judicious anticoagulation in low-body weight patients. In our study, approximately one in five patients were categorized as low risk of VTE based on the Padua score, yet still received pharmacologic VTE prophylaxis. Unfortunately, these patients were not spared from the excessive bleeding rates noted in our study and had bleeding rates similar to high-risk patients.
Limitations of our study include its retrospective design. Accuracy of documentation and charting may have omitted bleeding events. Asymptomatic VTE events may be undetected as imaging may not have been performed to confirm and document the presence of the VTE. Patient selection was not randomized, and uncaptured patient-specific factors may have influenced the choice of pharmacologic agent and dose, and our data does not capture whether decisions were made with foresight by an ordering provider or were intervened upon by a pharmacist at order verification. Though our sample size and event rate for VTE prohibited propensity-matching, our groups were well-balanced except for renal function. It is clear that providers in our study were biased toward using UFH in patients with renal impairment. Our cohort had a significant portion of patients with trauma/surgery, which may limit extrapolation of results to a completely medical population. This, however, also makes our results more generalizable to an overall hospitalized population. Anti-factor Xa levels are not routinely measured for prophylaxis at our institution and as such were not able to be evaluated, so a direct correlation between degree of anticoagulation and bleeding cannot be discerned. A small number of patients contributed multiple episodes of VTE prophylaxis for analysis though of these patients few received both heparin and enoxaparin during their admission. The Padua score used for our predominantly medical population is not validated for patients who underwent surgery and may not be an accurate predictor of their VTE risk. Our study was likely underpowered to detect the clinically significant differences in dose subgroups, especially within the subgroups of UFH. Though our study attempted to evaluate patients with CrCl < 30 mL/min, few patients received enoxaparin with a CrCl < 30 mL/min limiting our comparison to UFH.
In conclusion, no differences in VTE or bleeding rates were found between patients weighing < 55 kg receiving enoxaparin versus UFH. Together, the rates of bleeding and VTE events in the patients receiving enoxaparin 30 mg daily suggest empirically reducing the dose of enoxaparin for VTE prophylaxis is reasonable and safe in patients weighing < 55 kg.
Footnotes
Acknowledgments
Thank you to Michael W. Daniels, MS for his expertise with statistics.
Author contribution(s)
Ethics Approval and Consent to Participate
The University of Louisville Institutional Review Board exempted this study from approval. The IRB number is 21.0735. Informed consent was waived by the institutional review board as this study was retrospective in nature.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Not applicable
Availability of Data and Material
Not applicable.
