Abstract
Keywords
Introduction
Venous thromboembolism (VTE) is a serious and potentially deadly event with an estimated 10 million cases per year worldwide. 1 Early initiation of anticoagulation is crucial to reduce morbidity and mortality in patients with VTE.2–4 Unfractionated heparin (UFH) is a mainstay of anticoagulation treatment in the inpatient setting for acute VTE.2,3,5,6 A study by Raschke and colleagues compared a fixed-dose regimen (5000 unit bolus followed by a 1000 unit/h continuous infusion) and a weight-based dosing nomogram (80 unit/kg initial bolus followed by an 18 unit/kg/h continuous infusion). They found that weight-based dosing had a significantly lower rate of VTE recurrence. 7 Since then, several institutions have published their experiences with a weight-based dosing protocol and showed more rapid achievement of the therapeutic threshold for anticoagulation without compromising patient safety.8–10
Because of the unpredictable pharmacokinetics and variability of the anticoagulant response to heparin among patients, 11 it is standard practice to monitor heparin and adjust the dose based on the results of anticoagulation tests. In the past, the activated partial thromboplastin time (aPTT) was the only test used to adjust the UFH dose. 12 Later, heparin anti-Xa level monitoring became more desirable since biological factors (e.g., kidney function, liver function, and age) and pre-analytical variables (e.g., time of sample collection and delay in sample analysis) have less impact on anti-Xa levels than on aPTTs. 12 Anti-Xa level monitoring has also been linked to less time to achieve therapeutic anticoagulation, fewer monitoring tests, and fewer dosage adjustments.12–14 Anti-Xa level monitoring is utilized at this study’s institution with a therapeutic range of 0.3–0.7 units/mL.12,15
A recent study from 2019 by Prucnal and colleagues evaluated the proportion of patients with pulmonary embolism (PE) treated with UFH who achieved therapeutic aPTT levels during the first 48 h of therapy. 3 The study found that the majority of patients treated with UFH for acute PE did not remain therapeutic during the first 48 h. To date, there are no published studies investigating the time to therapeutic anticoagulation for patients with VTE treated with UFH while utilizing anti-Xa level monitoring under the study institution’s dosing protocol. The purpose of this study was to evaluate the ability of one facility’s UFH VTE protocol to achieve and maintain therapeutic anti-Xa levels within the first 48 h of therapy.
Methods
Design
This was a single-center study that was conducted at Avera McKennan Hospital and University Health Center via retrospective chart review. The study institution’s data repository system in conjunction with the electronic medical record was used to identify patients from May 2016 through September 2020 who were started on an UFH infusion for either a deep venous thrombosis (DVT) and/or PE. This study was approved by the Avera McKennan Hospital Institutional Review Board with approval for waiving informed consent and ethical approval because the study was no more than minimal risk (waiver number 2020.063-100833).
Participants and setting
Patients 18 years and older who were initiated on the institution’s VTE heparin dosing protocol for a consecutive 48 h with at least one anti-Xa level were included in the study. Exclusion criteria included the following: lack of confirmed VTE on duplex ultrasound or chest imaging, superficial venous thrombosis, and initiation of heparin at an outside hospital. Patients with an underlying coagulopathy (defined as factor VII deficiency, hyperhomocysteinemia, factor II deficiency, activated protein C resistance, factor V Leiden, von Willebrand disease, hemophilia, antiphospholipid syndrome, or sickle cell disease) were also excluded, as well as those who received systemic or catheter-directed thrombolysis, or those on an anticoagulant prior to heparin drip initiation (oral anticoagulants for 1 week, UFH and low-molecular-weight heparins for 8 h, and fondaparinux for 3 days).16–18 While data are described as a whole cohort, patients were further divided into two groups based on whether they received an initial bolus of UFH at the start of the infusion (bolus plus infusion) or started the infusion without an initial bolus (infusion only) to determine whether this affected any end points.
Interventions
The study institution uses a UFH dosing protocol which provides a weight-based initial rate of approximately 18 units/kg/h rounded to the nearest 1000 units (a maximum initial rate of 1600 units/h) with an initial bolus of approximately 80 units/kg rounded to the nearest 500 units (maximum initial bolus of 8000 units) (Table 1). Providers have the option of omitting the initial bolus dose based on patient factors (e.g., risk of bleeding). Anti-Xa levels are obtained every 6 h until two consecutive therapeutic anti-Xa levels are achieved, at which point anti-Xa levels are ordered daily. Dosing is adjusted by the bedside nurse based on the anti-Xa level as shown in Table 1. If the nurse increases, decreases, or holds the drip, a titration verification order is automatically reflexed in the pharmacist order verification queue. A pharmacist then double-checks that the heparin drip was correctly adjusted based on the protocol. If the heparin drip was not adjusted appropriately, the pharmacist contacts the nurse to guide correcting the rate adjustment. The heparin titration protocol has different dosing based on a patient weight cutoff of 70 kg. This weight cutoff was chosen by the institution to balance providing weight-based dose alterations with the improved ease of dose titration using round numbers. For this study, the initiation of UFH was defined as the time of the first documented administration of the heparin drip in the electronic medical record. Anti-Xa values were grouped by 6-h time intervals after the initiation of UFH (e.g., the “18-hour” anti-Xa group included anti-Xa values drawn from 15 to 21 h after initiation of UFH).
UFH infusion protocol for VTE.

Outcome
The primary end point was to determine the number of patients who achieved at least one therapeutic heparin anti-Xa level within the first 48 h of UFH infusion therapy. Secondary outcomes included the percentage of therapeutic levels at each 6-h interval during the first 48 h of therapy, time to first therapeutic level, in-hospital mortality, and hospital length of stay. Other secondary outcomes included the percentage of patients who had subtherapeutic and supratherapeutic anti-Xa levels within the first 24 and 48 h as well as identified potential patient-specific factors that affected anti-Xa levels.
Statistical analysis
A difference in the median values of normally distributed groups was evaluated with a student’s t-test or with a Wilcoxon rank-sum test for non-normal distribution. For assessing the relationship between categorical variables, Fisher’s exact test was used. In all statistical tests, an effect was considered statistically significant if the p value of its corresponding statistical test was not greater than 5%. All statistical computations were performed using R version 3.6.3 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Between May 2016 and September 2020, a total of 8376 patients were identified as having an active heparin infusion order (Figure 1). A total of 8171 patients were excluded mainly for being on a UFH drip for reasons other than VTE. Overall, 205 patients were included in the study with 89 patients being a part of the bolus plus infusion group and 116 patients in the infusion-only group.

Flow diagram for inclusion and exclusion criteria.
Baseline characteristics were similar between the bolus plus infusion and the infusion-only cohorts (Table 2). The median age in the overall cohort was 63 years, with a statistically significant age difference between the bolus plus infusion cohort and infusion-only cohort (61 versus 65 years, respectively, p = 0.033). Baseline hemoglobin values were statistically significantly higher in the bolus plus infusion cohort compared to the infusion-only cohort (10.5 versus 9.7 g/dL, respectively, p = 0.039). All other values were not statistically significantly different between the two groups.
Baseline demographics and clinical variables overall and by study cohort.a,b

Abbreviations: BMI, body mass index; COVID-19, coronavirus disease 2019; CrCl, creatinine clearance; DVT, deep venous thrombosis; ICU, intensive care unit; INR, international normalized ratio; PE, pulmonary embolism; SCr, serum creatinine; VTE, venous thromboembolism.
Many patients did not have all the listed demographic information in their chart from that particular stay leading to varying group numbers.
p Values indicate differences between the bolus plus infusion and the infusion-only cohorts.
Of the 205 patients in the study, 97.6% had at least one therapeutic anti-Xa level within the first 48 h of heparin therapy with no statistically significant difference between the bolus plus infusion and the infusion-only groups (Table 3).
Primary outcome, in-hospital mortality, length of stay, and time to achieve a therapeutic level.
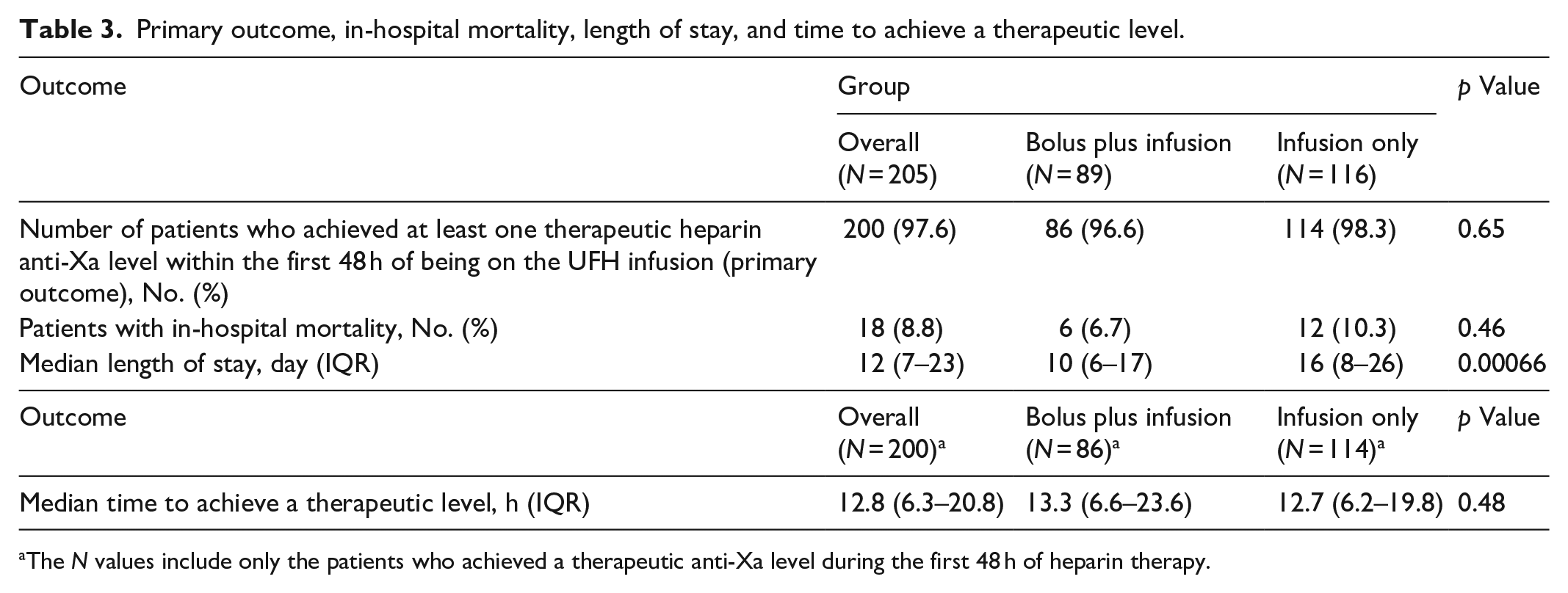
The N values include only the patients who achieved a therapeutic anti-Xa level during the first 48 h of heparin therapy.
Table 4 shows complete percentages and number of patients at each measured time point in both dosing cohorts. No more than 60% of patients achieved a therapeutic level at any of the 6-h intervals throughout the 48 h of the heparin infusion. In addition, comparing the subtherapeutic, therapeutic, and supratherapeutic results, there were high rates of crossover between each 6-h time point within each dosing cohort.
Percentage of patients who achieved subtherapeutic, therapeutic, and supratherapeutic anti-Xa levels within each 6-h time point in the bolus plus infusion and infusion-only cohorts.

The median time to achieve a therapeutic anti-Xa level in the overall group was 12.8 h (Table 3) with no statistically significant difference between the bolus plus infusion and infusion-only cohorts. The percentage of patients who had only subtherapeutic, therapeutic, and supratherapeutic levels for the initial 48 h are listed in Table 5 with no statistical differences seen between cohorts. Overall, 76.1%, 71.9%, and 79.3% of patients had two consecutive therapeutic anti-Xa levels within 48 h in the overall, bolus plus infusion, and infusion-only cohorts, respectively, with no statistical difference between the bolus plus infusion and infusion-only cohorts (p = 0.25).
Secondary outcomes at 48 and 24 h.
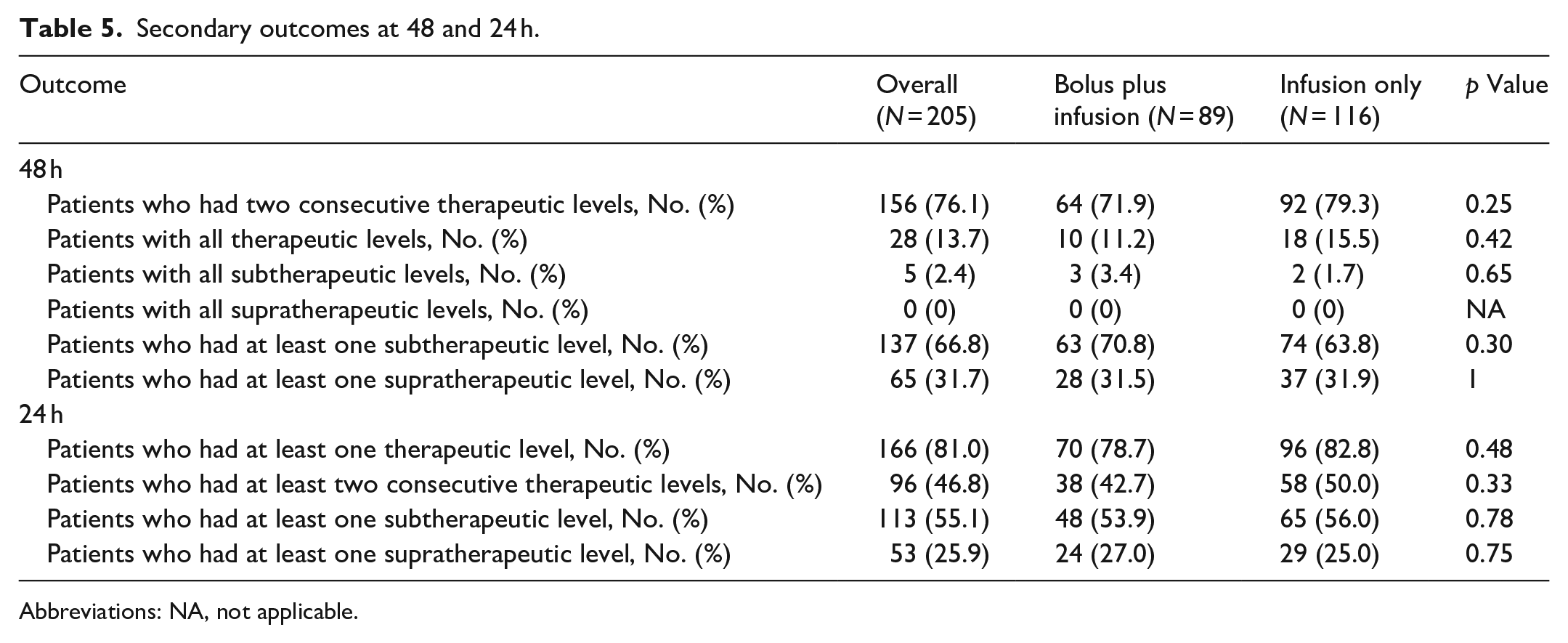
Abbreviations: NA, not applicable.
There was no statistical difference seen for in-hospital mortality when comparing results between the two dosing cohorts (Table 3). However, the bolus plus infusion group did have a statistically significant shorter length of stay when compared with the infusion-only group (10 days versus 16 days, respectively, p = 0.00066).
Once the above results were reviewed, it was determined that a post hoc analysis would be beneficial by reviewing the same end points, except within a 24-h period rather than the original 48-h period. Table 5 shows 81%, 78.7%, and 82.8% of patients had at least one therapeutic anti-Xa level within 24 h in the overall, bolus plus infusion, and infusion-only group, respectively, with no statistical difference between the two dosing cohorts (p = 0.48). There were no statistically significant differences in the percentage of patients who had at least one subtherapeutic or supratherapeutic level within 24 h (Table 5). 46.8%, 42.7%, and 50% of patients had at least two consecutive therapeutic levels within 24 h in the overall, bolus plus infusion, and infusion-only group, respectively, with no statistical difference between the two dosing cohorts (p = 0.33).
To determine patient factors that might affect a therapeutic anti-Xa level outcome, patients who achieved at least one therapeutic anti-Xa level within 48 h of therapy were compared to patients who did not achieve any therapeutic anti-Xa levels. Due to the low number of patients who did not achieve at least one anti-Xa level, we were unable to perform this analysis. Another post hoc analysis was done in an effort to determine patient factors that might affect the time (hours) to achieve the first therapeutic anti-Xa level in the overall cohort alone (Table 6). Characteristics that were investigated included age, body mass index, ethnicity, creatinine clearance, sex, type of VTE, and weight. Patients who were 65 years or older were more likely to achieve a therapeutic anti-Xa level faster than those who were less than 65 years old (12.1 h versus 14 h, respectively, p = 0.014). All other characteristics listed above were not statistically different.
Effect of patient factors on the time to achieve the first therapeutic Anti-Xa level.
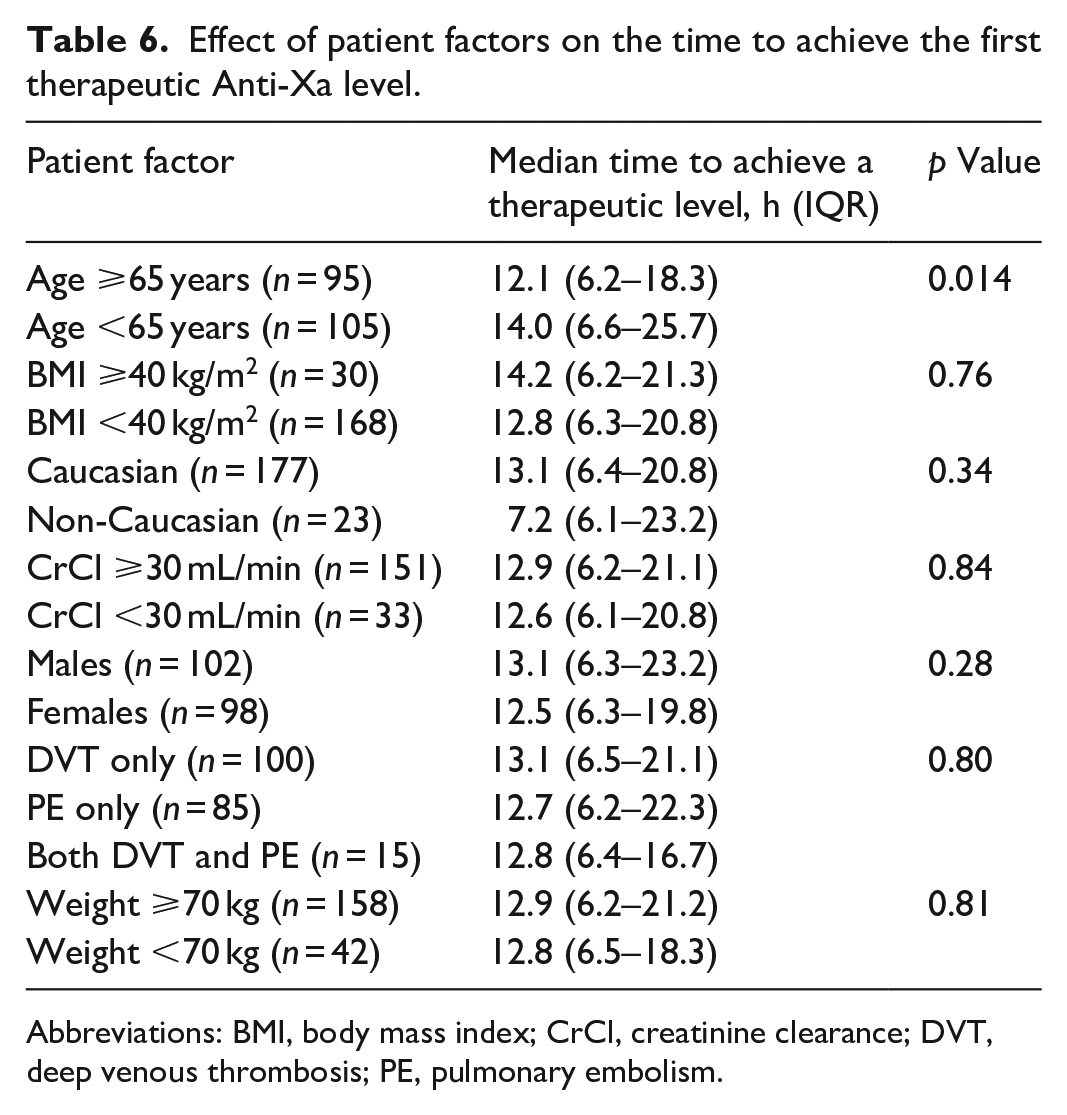
Abbreviations: BMI, body mass index; CrCl, creatinine clearance; DVT, deep venous thrombosis; PE, pulmonary embolism.
Discussion
In this study of assessing one institution’s UFH protocol for VTE, 97.6% of patients achieved at least one therapeutic anti-Xa level within the first 48 h of therapy. The Prucnal and colleagues study found that the proportion of patients who achieved at least one therapeutic aPTT at 48 h was around 60% in both groups which is numerically lower than what our study found examining anti-Xa levels. 3 Upon evaluation of this end point alone, the institution’s UFH protocol is effective. However, it is also important to consider the dosing protocol’s ability to maintain therapeutic levels. With this regard, only 40%–60% of patients had a therapeutic level during each 6-h period in the initial 48 h of therapy, and the percentage of patients who remained therapeutic throughout the entire first 48 h was low (13.7% in the overall cohort). Patients often crossed between having therapeutic, subtherapeutic, and supratherapeutic levels. Similarly, the Prucnal and colleagues study found that less than half of all patients in either cohort had a therapeutic aPTT level during any 6-h period. Ours and Prucnal and colleagues’ dosing protocols both used the same initial dosing strategy (80 unit/kg bolus with 18 unit/kg/h infusion) but differences in actual dosing may exist based on rounding, as Prucnal and colleagues utilized pure weight-based dosing, whereas ours rounds dosing based on 10-kilogram weight measurements.
Faster achievement of therapeutic levels is associated with better clinical outcomes when using UFH for the treatment of VTE.4,5,7 A study on patients with PE found that patients had lower in-hospital and 30-day mortality rates if they achieved therapeutic aPTT levels within 24 h, which suggests that a goal of achieving therapeutic anti-Xa levels should be 24 h. 4 In our study, the median time to achieve the first therapeutic anti-Xa level was 12.8 h in the overall patient population, 13.3 h in the bolus plus infusion group, and 12.7 h in the infusion-only group. Comparing our results to the 2019 study, Prucnal and colleagues found that patients took an average of nearly 36 h to achieve a therapeutic aPTT level. 3 So while the study institution’s heparin protocol did result in the vast percentage of patients achieving at least one therapeutic anti-Xa level within 24 and 48 h, the impact of the inconsistency of maintaining therapeutic anti-Xa levels on patient outcomes remains in question.
Younger patients and those with higher baseline hemoglobin tended to receive the bolus plus infusion UFH protocol versus infusion only. This may be due to a perceived difference in bleeding risk favoring withholding the initial bolus in older patients and patients with lower hemoglobin levels. At this institution, prescribers are allowed the option of omitting the initial heparin bolus without documenting the reason why. Not having this data is a limitation of this study as it could provide more information on potential confounding variables that contributed to differences seen between groups.
A total of five patients did not achieve any therapeutic anti-Xa levels within the first 48 h. However, due to the small number of patients, it is difficult to draw any conclusions regarding patient factors that may have contributed to suboptimal dosing in those patients. There is evidence suggesting that age is an independent factor for response to heparin.7,19–21 The exact mechanism behind this increased sensitivity is unknown. One retrospective study, assessing an institution’s heparin protocol for VTE treatment, found that elderly patients (age >70 years) had significantly higher initial anti-Xa levels than non-elderly patients, with no difference in time to achieve the target range. 21 Our study found that elderly patients (age ⩾65 years) took statistically significantly less time to achieve a therapeutic anti-Xa level than those patients less than 65 years old (12.1 h versus 14 h, respectively, p = 0.014).
Limitations
Our study is the first, to the authors’ knowledge, to evaluate the achievement and maintenance of therapeutic anti-Xa levels for a UFH dosing protocol in treating patients with VTE. This was a real-world study with strict inclusion and exclusion criteria applied, attempting to minimize the impact of other concomitant anticoagulants and patient factors. This study has several limitations including its small, retrospective design. Also, it was conducted in a single institution and the sample size was not robust enough to identify patient factors associated with achievement and maintenance of therapeutic anti-Xa levels. A sample size calculation was not performed a priori which leads to a risk for type II error. Another potentially limiting factor is that there were multiple individuals including/excluding patients and collecting data which may have led to inconsistencies. We attempted to mitigate this risk by creating a flowsheet for each process so that each individual evaluated patients and collected data in the same fashion. We also noticed the number of anti-Xa levels drawn decreased over time. This is likely due to the retrospective, pragmatic nature of this study with the potential for protocol errors in obtaining levels and the institution’s protocol allowing for daily monitoring of anti-Xa levels when patients achieve two consistent therapeutic levels. Protocol compliance was not obtained which is a potential limitation as it may have aided in the interpretation of the study. With about 30% of the overall group starting heparin in the intensive care unit, there may have been a selection bias with more complicated patients being put on heparin over other anticoagulants. This could have predisposed a higher variability in anti-Xa levels compared to a less complex patient population, thus potentially leading to more difficult heparin management. In addition, uneven distribution of levels among different time windows may lead to inaccurate interpretation of the results.
Further studies should focus on multiple end points for evaluating UFH dosing. In addition, patient-oriented outcomes need to be assessed to have a clearer picture of the efficacy of a dosing protocol as the monitoring-oriented end points of our study are less useful in clinical decision-making. More specifically, the difference between two dosing strategies, bolus plus infusion versus infusion only, needs to be evaluated further to see whether an initial bolus provides any clinically meaningful difference in the outcome. In our study, the only statistically significant difference we found between the bolus plus infusion and infusion-only cohorts was the median length of stay which was shorter in the bolus plus infusion cohort. This observation could be explained by the fact that patients who were younger with higher initial hemoglobin levels tended to receive the bolus plus infusion and these patients may be more likely to be discharged earlier than older patients with lower initial hemoglobin levels. The length of stay was longer than expected for patients, which may be due to the pragmatic nature of this study allowing for patients who may have developed VTE events during hospitalization in addition to patients who were admitted primarily for VTE.
Conclusion
In evaluating a single institution’s UFH dosing protocol using anti-Xa level monitoring, most patients were able to achieve at least one therapeutic anti-Xa level within the first 48 h of initiation. However, fewer patients were able to consistently maintain therapeutic anti-Xa levels within this timeframe. Further studies are warranted to determine whether there are dosing strategies that could yield more consistent achievement of therapeutic anti-Xa levels and whether alternative dosing strategies would affect patient-oriented outcomes.
Footnotes
Acknowledgements
The authors would like to acknowledge the following individuals who assisted in collecting data for the study: Julia Beethe, PharmD; Shelby Buller, PharmD; and Dustin Moon, PharmD.
Author contributions
Lily Nguyen: Made a substantial contribution in the concept and design of the study, data collection, interpretation of data, drafted the article, revised the article critically, and approved the version to be published. Xiaoxiao Qi: Made a substantial contribution in the concept and design of the study, data collection, interpretation of data, drafted the article, revised the article critically, and approved the version to be published. Abraham Karimi-asl: Made a substantial contribution in the concept and design of the study, data collection, interpretation of data, drafted the article, revised the article critically, and approved the version to be published. Alicia Thole: Made a substantial contribution in the design of the study, revised the article critically, and approved the version to be published. Jodi Wendte: Made a substantial contribution in the design of the study, revised the article critically, and approved the version to be published. Tobias Meissner: Made substantial contribution in the analysis of data, revised the article critically, and approved the version to be published. Bing Xu: Made substantial contribution in the analysis of data, revised the article critically, and approved the version to be published. Kyle Dvoracek: Made a substantial contribution in the concept and design of the study and interpretation of data, revised the article critically, and approved the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was waived by Avera McKennan Hosp IRB #1—DBA Avera IRB #1/IRB00001096 because the study was no more than minimal risk. The waiver number is 2020.063-100833.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was waived by Avera McKennan Hosp IRB #1—DBA Avera IRB #1/IRB00001096 for the present study because the study was no more than minimal risk. The waiver number is 2020.063-100833.
Trial registration
Not applicable.
