Abstract
Health disparities exist in incidence, treatment, and survival of esophageal and gastric cancer based on socioeconomic and minority racial/ethnic group status. How socioeconomic and minority status affect access and utilization of precision oncology in foregut malignancies is not well understood. Based on other cancer types, multiple intrinsic and extrinsic factors influence utilization of cancer genetic testing, participation in precision oncology clinical trials, and access to molecularly targeted therapies. As precision oncology becomes more common in the treatment of foregut malignancies, health equity will require ongoing identification of sources of disparities and proposal of practical solutions.
Introduction
Overall survival of patients with esophageal and gastric cancers has improved since the 1970s, due in no small part to innovations in therapy.1-3 For localized esophageal cancer, modern curative intent treatment includes neoadjuvant chemoradiation, surgery, and adjuvant immunotherapy in the appropriately selected patients. 4 For localized gastric cancer, curative intent treatment includes perioperative chemotherapy and surgery. 5 In advanced, unresectable or metastatic gastroesophageal cancer the addition of molecularly targeted therapies and immunotherapy drugs has increased survival.6-12 Despite this progress, not all patients have benefited equally. Those of low socioeconomic status and members of certain minority racial/ethnic groups have less access to multi-modality curative intent treatment and newly approved drugs than those who are more affluent or from majority racial/ethnic groups. Similar to other aspects of cancer care, as detailed below, significant inequalities in the use of precision oncology testing and access to molecularly targeted therapies affect certain populations, widening pre-existing health disparities. 13
Precision oncology, using genetic testing to identify molecular alterations that predict a tumor’s susceptibility to a specific drug and subsequent administration of that drug to the patient, has dramatically changed the cancer treatment landscape. Examples from gastrointestinal (GI) cancers include HER2 targeted drugs in gastroesophageal cancer, PARP inhibitors in pancreatic cancer, FGFR and IDH inhibitors in cholangiocarcinoma, and immunotherapy in MSI-high colorectal cancer. Within precision oncology, genetic testing can examine the patient’s germline DNA, somatic molecular alterations within the patient’s tumor, or both. For many cancer patients, including those with foregut malignancies, precision oncology testing results directly impact treatment and inform upon prognosis.
Factors Influencing Disparities in Cancer Care
Upstream Determinants
Although equity in cancer care has been a policy goal in the United States for many years, marked disparities in cancer incidence, morbidity, and mortality remain. 4 A 2014 study indicated a reversal and worsening of these trends since the 1950s, with increased incidence and mortality of all cancer types associated with socioeconomic deprivation and membership in certain minority racial/ethnic groups. 14 There are several reasons for this (Figure 1). First, there is a higher prevalence of risk factors for cancer among those of low socioeconomic status and within certain minority racial/ethnic groups. These risk factors include high calorie and high fat diets, increased rates of smoking and alcohol use, decreased rates of physical activity, and increased rates of obesity.15,16 Second, these populations have lower rates of cancer screening and prevention, which increase cancer incidence and delay diagnosis until cancer stage is more advanced. Decreased cancer screening rates are highly correlated with lower levels of income and education. 14 Since stage of cancer at diagnosis is most predictive of survival, reducing prevalence of risk factors and augmenting cancer prevention and screening are important targets for addressing disparities in cancer incidence, morbidity, and survival.17,18
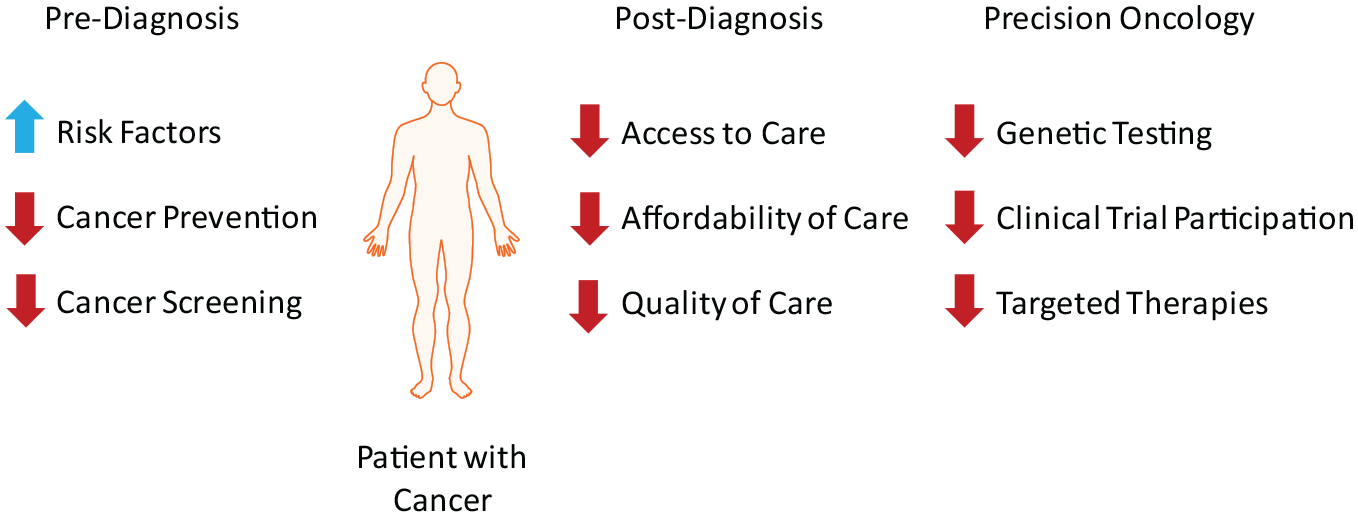
Sources of Health Disparities in Patients Diagnosed with Cancer. Disparities exist for patients with low socioeconomic status and members of certain minority racial/ethnic groups before and after a cancer diagnosis and in utilization of precision oncology.
Access to Cancer Care
Unfortunately, health care disparities also occur after a cancer diagnosis, specifically in access, affordability, and quality of oncologic care (Figure 1). Socioeconomic status and geographic location are 2 primary determinants of access to medical care in the United States. A 2022 study demonstrates that members of certain minority racial/ethnic groups and those of low socioeconomic status often drive farther distances to receive the same quality of care as majority, high socioeconomic status patients. Longer drive times lead to lower treatment adherence rates and place an increased economic burden on patients, who may have to take time off from work to obtain treatment. 19 In addition to lost wages, the cost of transportation and childcare for cancer patients is often prohibitive to treatment. 20 Workplace discrimination is common, and the side effects of cancer therapies often force patients to take reduction in work hours or face job elimination through layoffs. This is especially true for jobs involving manual labor or in the service sector, which are disproportionately held by those of low socioeconomic status, compounding existing disparities. 21
Cost of Cancer Care
Socioeconomic status also affects affordability of care. Cancer care is quickly becoming the costliest medical expense for patients, exacerbated by the development of molecularly targeted agents and immunotherapy, to the point that treatments are virtually inaccessible for the average uninsured American.22,23 According to the US Census Bureau, 8.3% of Americans in 2021 were uninsured, with higher uninsured rates among minority racial/ethnic groups and those in the lowest income brackets. 24 Individuals without insurance can spend almost 30% of their income on medical expenses, a percentage that increases for patients with cancer. Even after completing treatment, cancer patients spend more on health care than those without cancer. 25 For patients with insurance, high copays can limit available treatment options and lead physicians to forego the highest quality care in favor of more affordable alternatives. 26 This is especially true for treatments that involve off-label uses, which are not always covered by insurance. Low socioeconomic status and less education also affect quality of care. These patients often seek treatment at safety-net hospitals, where in the case of esophageal cancer, patients are less often offered curative surgery. 27 African-American and low-income patients have less access to minimally invasive gastrectomy for treatment of gastric cancer, which shortens recovery time. 28
Factors Influencing Disparities in Precision Oncology
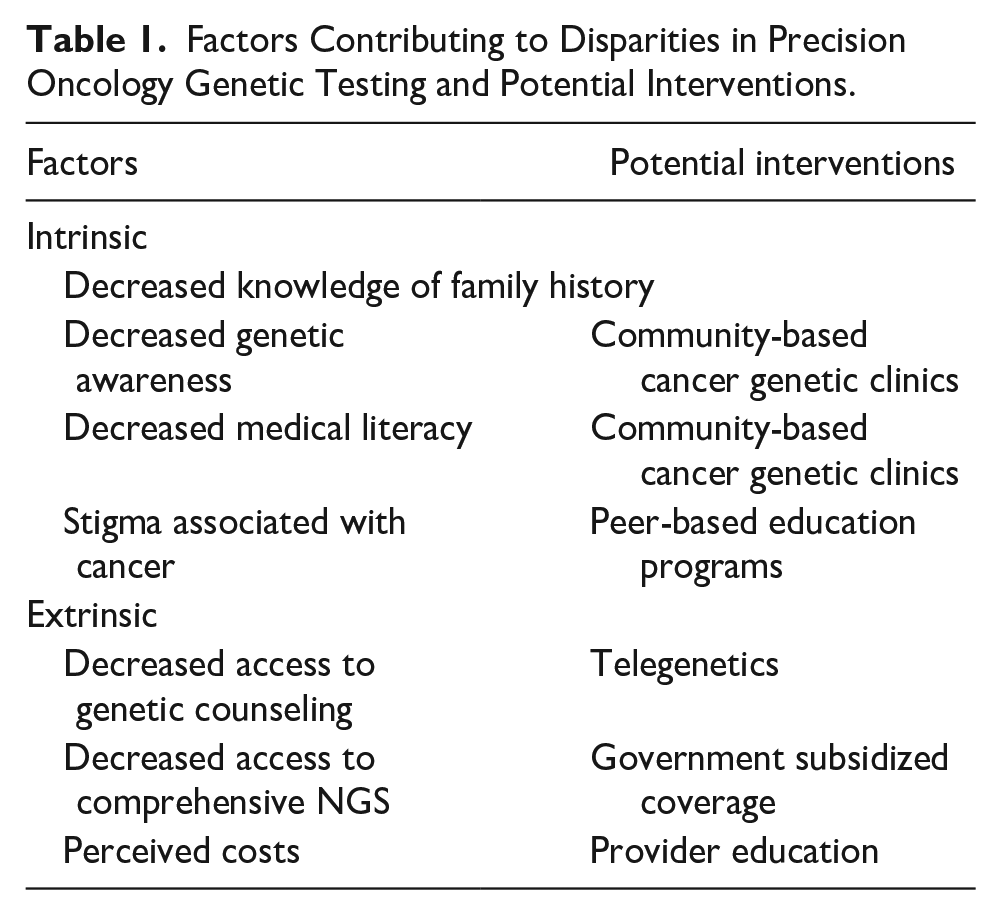
While disparities in cancer incidence and mortality and access to quality cancer care exist, there are additional intrinsic and extrinsic factors specific to precision oncology that further increase disparities (Table 1). Intrinsic factors are from within an individual, while extrinsic factors involve outside influence. Factors intrinsic to patients with low socioeconomic status and minority racial/ethnic background include decreased knowledge of family medical history, decreased genetic awareness, decreased medical literacy, and stigma attached to genetic testing. Extrinsic factors include decreased access to genetic counseling, decreased access to comprehensive somatic tumor testing, and inaccurate perception of costs by providers.
Factors Contributing to Disparities in Precision Oncology Genetic Testing and Potential Interventions.
Intrinsic Factors Influencing Precision Oncology Disparities
Knowledge of family medical history
Members of certain minority racial/ethnic groups and those of low socioeconomic status are less likely to have a thorough, well-defined family medical history. Individuals of low socioeconomic status tend to have decreased access to medical care, leading to missing information and discrepancies in family medical history, under-identification of potential heritable risk factors, and decreased rates of somatic molecular testing. 29 Since eligibility for cancer genetic testing may depend on a positive family history of inherited cancers, this puts these patients at a disadvantage for receiving genetic testing. It also leads to underdiagnosis of inherited cancers. 30 This widens pre-existing disparities in treatment outcome and survival.
Genetic awareness
A patient’s genetic awareness greatly influences their pursuit of genetic testing and desire for precision oncology treatments. Genetic awareness includes awareness of genetic risk factors for a disease, an understanding of how those genetic risk factors alter the incident risk and interact with other relevant (eg, environmental) factors, and a knowledge of the mechanisms behind the genetic influence on the disease. 31 Patients with decreased genetic awareness participate less frequently in genetic testing, resulting in fewer receiving precision oncology treatments. Although the general US population’s awareness of genetic testing for cancer susceptibility is low, those from minority racial /ethnic groups were even less likely to be aware. 32 A 2021 study by Giri et al 33 found that higher income and educational attainment levels correlate with an increased awareness of hereditary cancer risks, availability of genetic testing for cancer predisposition, and completion rates of genetic testing. A 2017 study by Hann et al indicated that genetic awareness is highly stratified by racial/ethnic group, exacerbating disparities in genetic testing rates. The study found that minority groups were less likely to understand the genetic basis of cancer, which served as a barrier to receiving genetic counseling and genetic testing. 34
Medical literacy
While race is not independently predictive of rates of genetic testing, race does impact beliefs on the genetic basis of cancer and medical literacy, with non-Hispanic Blacks having weaker beliefs and lower levels of literacy than non-Hispanic Whites, even when controlling for socioeconomic status. 31 However, these lower levels of medical literacy are often not recognized by a patient’s provider. A 2016 study by Ciardiello et al demonstrates the gap between patients’ self-reported genetic awareness and the perceived awareness by their physicians. This study found that while only 66% of patients reported a willingness to delay treatment in favor of further genetic testing, physicians perceived that 82% of their patients would be willing to postpone treatment. This discrepancy reflects physicians’ overestimation of their patients’ genetic awareness and is indicative of a communication barrier between physicians and patients. 35 These findings emphasize the need for additional public education about the availability and purpose of genetic testing. The disparities that persist in genetic awareness between racial, ethnic, and socioeconomic groups indicate that this education should be tailored to individual populations to maximize efficacy and equity. This could involve community-based cancer genetic outreach clinics. 36 To address the unique barriers that impede rates of genetic testing in minority populations, physicians should be cognizant of cultural and linguistic differences that lead to disparities in literacy.
Stigma
Even for individuals aware of available genetic testing options, the stigma surrounding cancer can be prohibitive. Cancer stigma decreased health care utilization by patients due to fear of discrimination and social isolation. 37 Since genetic testing can establish the cancer susceptibility of entire families, concerns about discussing results with family can influence patients against undergoing genetic testing. 38 In addition, patients from minority racial/ethnic groups may not participate in genetic testing because of concerns about discrimination and confidentiality. 37 Known as the “Tuskegee Effect,” distrust in the medical field persists in minority racial/ethnic communities as a result of the Tuskegee syphilis experiments. 39 One potential solution is peer-based educational programs that attempt to address stigma, increase medical literacy, and spread awareness of the benefits of genetic testing outside of the medical office. 40
Extrinsic Factors Influencing Precision Oncology Disparities
Access to genetic counselling
Rates of usage of germline genetic testing are also impacted by availability, accessibility, and affordability of genetic counseling. As with trends in cancer incidence, morbidity, and survival, accessibility of genetic counseling is also disparate along socioeconomic gradients and is highly dependent on geography. Although genetic counseling is an integral step in precision oncology treatment, Medicare does not provide coverage for genetic counseling of any type, making the costs of genetic counseling prohibitive for those of low socioeconomic status. 41 There are also associations between geographic areas with low socioeconomic status populations and decreased access to genetic counselors. There is a tendency for genetic counselors to be located in urban areas, especially those with a younger, more affluent population with a higher level of educational attainment. This results in longer drive times for residents of non-urban areas to reach genetic counselors. 42 Long drive times to care centers, especially those over 1 hour, have been associated with worse outcomes and lower rates of treatment compliance for cancer patients. 43 This indicates that patients from non-urban areas, who are more likely to represent minority racial/ethnic groups and low socioeconomic status, are at a disadvantage in regards to obtaining genetic counseling, negatively impacting their ability to receive genetic testing and by extension precision oncology treatments. Although the COVID-19 pandemic expanded telemedicine and telegenetic opportunities, their impact has yet to be fully characterized. Even with telegenetics, access to counseling is still disparate along socioeconomic gradients due to access differences to stable internet. In addition, state licensing policies can complicate telegenetics, especially for individuals in states with insufficient genetic counselors. For example, the majority of genetic counselors in the state of Wyoming practice across state lines via telegenetics. While this increases accessibility of counseling, payment for these out of state counselors is almost entirely out of pocket. 42 Policy changes are needed to address the inequitable distribution of genetic counselors across state lines and between urban and rural areas.
Access to genetic testing
Accessibility of somatic (tumor) molecular testing is another determinant of precision oncology treatment that introduces disparity. Somatic molecular testing, which can range from single-gene sequencing to next-generation sequencing (NGS) of a panel of genes, provides crucial information for selecting appropriate molecularly targeted therapies. While this technology has the potential to address disparities in cancer incidence and mortality, at its current state the distribution of somatic molecular testing in the field of oncology aggravates rather than mitigates disparities. Race is one driver behind disparities in somatic molecular testing, especially when considering rates of NGS (Figure 1). A 2022 study by Bruno et al found that while rates of single-gene sequencing did not differ between Black and White patients with advanced non-small cell lung cancer, Black patients received NGS at much lower rates than their White counterparts, at any time during treatment. As a result, Black patients saw statistically significantly lower rates of participation in precision oncology clinical trials 44 (Figure 1). Similar trends are seen in prostate cancer. 45 It is also important to consider that single-gene testing, while less costly, more accessible, and more often ordered by physicians, may be less effective at identifying molecular targets, especially for diseases in which multiple potential targets have been elucidated. 44
Socioeconomic status also influences rates of somatic molecular testing due to associated actual, perceived, and indirect costs. In 2018, a national coverage determination (NCD) allowed for the coverage of NGS for any solid tumor at the time of diagnosis by the Centers for Medicare and Medicaid Services. 46 However, this NCD came with the caveat that the NGS would be covered only if it used an FDA approved assay, despite no significant differences in efficacy having been elucidated between FDA and non-FDA approved sequencing protocols. 47 Thus, the NCD disadvantaged individuals located near academic laboratories or other small Clinical Laboratory Improvement Amendment (CLIA)–certified laboratories, which tend to be individuals of a low socioeconomic status or minority racial/ethnic groups. Although the full downstream effects of this NCD on private insurance coverage remain to be seen, it is likely that private insurance companies will provide similar coverage in the future. Given historical trends in the adoption of new medical technology, private insurance coverage for somatic molecular testing will exacerbate existing disparities. Individuals of a high socioeconomic status tend to have better insurance coverage, resulting in lower copays and more exhaustive treatment options. This includes earlier adoption of coverage for somatic molecular testing. Lower quality insurance, available to individuals of a low socioeconomic status, tends to lag in adoption of new technologies. 48 Out of pocket costs for somatic molecular testing, especially NGS, are generally prohibitive for individuals of this income bracket, resulting in lower rates of somatic molecular testing and prohibiting these individuals from participation in precision oncology clinical trials.
Perceived testing costs
Recent studies indicate that even when the costs of somatic molecular testing are low, perceived costs by physicians and patients result in lower rates of testing. 30 A study from 2022 found that physicians cited the costs of somatic molecular testing as the most influential factor for not ordering genetic tests. 49 Although costs of NGS remain prohibitive for some populations, in many cases NGS would have been covered. This indicates that misconceptions about insurance coverage of somatic molecular testing remain, which is unsurprising given the current changing landscape of the science and economics of somatic molecular testing. These misconceptions disproportionately affect patients of a low socioeconomic status; providers may conservatively under-treat low socioeconomic status individuals in favor of preserving economic stability. However, even for patients for whom somatic molecular testing is covered by insurance or is affordable out of pocket, the indirect costs of genetic testing can be prohibitive. Especially for rural populations, access to genetic testing is often limited by driving distance. Although direct to consumer somatic molecular testing exists, in which at-home test kits are mailed to patients, studies indicate that very few patients are aware of this option and even fewer make use of it. 50 With many patients unable to access the invaluable information provided by somatic molecular testing, the benefits of precision oncology are diminished. In order to address the disparities in precision oncology, additional measures need to be taken to increase awareness and economic feasibility of genetic testing. In Singapore, government subsidies led to increased uptake of cancer genetic testing. 51
Current State of Disparities in Precision Oncology
Underreporting and Underrepresentation in Clinical Trials
Precision oncology promises cancer patients more individualized and effective treatment options. 52 A study performed in 2018 indicated that only 8% of cancer patients were eligible for molecularly targeted agents, with even fewer (5%) actually benefiting from the drug. 53 These statistics are even worse when compounded with socioeconomic and racial/ethnic disparities. In a 2021 study, Aldrighetti et al compared rates of cancer incidence to precision oncology trial participation for racial/ethnic groups. They found that within precision oncology clinical trials, there was a marked overrepresentation of White and Asian participants and an underrepresentation of Black and Hispanic participants. 54 They also discovered that the majority of trials did not report race/ethnicity. This aggravates existing disparities by inhibiting an understanding of the differences in tumor biology between racial/ethnic groups. A lack of reporting also helps to disguise inequitable access to precision oncology clinical trials for racial/ethnic minorities. The trends described in the Aldrighetti study also hold true in clinical trials involving GI cancers. In the study that demonstrated the benefit of maintenance therapy with the PARP inhibitor olaparib in advanced pancreatic cancer patients with germline BRCA1 and BRCA2 mutations, there is no mention of the race or ethnicity of the patients enrolled. 55 Similarly, in the 2020 trial of pemigatinib in locally advanced or metastatic cholangiocarcinoma patients with FGFR gene alterations, while data was segregated by region (United States, Europe, Asia, and the Middle East), there was no reference to the race or ethnicity of the trial participants. 56 While the 2021 clinical trial of ivosidenib for patients with advanced cholangiocarcinoma with IDH1 mutations did include self-reported race/ethnicity data, this data indicates a significant underrepresentation of racial and ethnic minorities. There was also no race/ethnicity subgroup analysis performed. 57 In order for precision oncology to mitigate rather than exacerbate existing disparities, a thorough understanding of how these treatments function in different populations is necessary, which requires both better recruitment of underrepresented minorities in clinical trials and additional analysis by subgroup.
Cost of Precision Oncology Drugs
With the price of novel precision oncology drugs climbing into the tens of thousands of dollars, accessibility and affordability of these treatments are practical issues (Figure 1). Patients with private insurance can benefit from coupons distributed by pharmaceutical companies that greatly reduce the cost of medications. However, users of Medicare and Medicaid are unable to use these coupons, leaving out of pocket costs high for this population that is already traditionally underserved.58,59 A 2016 study of specialty pharmaceuticals indicates that these costs affect patients’ ability to adhere to treatment regimens. Prescription abandonment, described as patient refusal of medication after approval by insurance, is common for precision oncology medications, especially when out of pocket costs rise above $500. 60 Prescription adherence also decreases with increased cost of medication. 61 Patients who do not fill their prescriptions or who do not complete therapies will not maximize the benefits of precision oncology treatments. Thus, inhibitory costs of precision medications and lack of sufficient coverage by insurance providers exacerbate existing disparities in oncologic care. Consistent with historical trends in oncologic outcomes, individuals of higher socioeconomic status are more likely to be able to afford and to adhere to precision oncology treatments than those of lower socioeconomic status, leading to better health outcomes and widening existing disparities.
Conclusion and Future Directions
While disparities in cancer incidence, treatment, and mortality have been characterized in esophageal and gastric cancers, disparities in accessibility and effective use of precision oncology in these patients have not been well studied. Studies examining disparities in precision oncology from other tumor types demonstrate that many factors are involved, some intrinsic to the patient and others that are extrinsic. Achieving a goal of health equity in precision oncology in foregut malignancies starts with an awareness of these factors and beginning to apply solutions that have been successful in other cancer types. A 2017 study by Chachula et al. 62 examined how their ACCESS “(awareness and education, community outreach, cultural and linguistic competence, equity and inclusion, strong partnership, and sustainability)” framework, which was based on the strategies identified by Jongen et al, has been successful in mitigating specifically cancer disparities at the Moffitt Cancer Center in Florida. This framework attempts to address disparities at multiple time points, with a primary emphasis on upstream determinants of care, such as with the M-POWER program that provides health education to community members. The authors found that there was a greater reduction in disparities when they were attempted to be resolved at an upstream level, such as with community health risk education and increased access to screening. Additionally, there was a greater reduction in disparities when multiple programs were implemented in tandem. The implications of this study are that (1) there is no one solution to addressing inequality in cancer care and (2) fighting inequity requires a specific knowledge of the population you are serving in order to provide targeted interventions. Although this program was generalized to cancer literacy across all treatment options, this indicates that addressing disparities in precision oncology for foregut malignancies necessitates addressing the upstream extrinsic and intrinsic factors described in this article, with an emphasis on increasing genetic awareness and rates of genetic testing in traditionally underserved communities. Increasing patient education regarding precision oncology, access to genetic testing, and return of results to patients can occur in community-based cancer genetic outreach clinics or through peer-based education programs. Increasing provider education regarding actual costs of genetic testing and available financial resources, increasing diverse patient populations in clinical trial design, and improving provider-patient communication can occur through deliberate efforts by health care systems and individual providers. Reducing the costs of NGS and precision oncology drugs will take national collaborative efforts; one example is LUNGevity’s partnership with the Association of Community Cancer Centers and their Eliminating Precision Oncology Disparities initiative. As we move further into the age of precision oncology, identification of factors contributing to disparities in treatment outcomes in esophageal and gastric cancer need to be ongoing and addressed through practical solutions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
