Abstract
Background:
Delayed gastric conduit emptying (DGCE) is a recognized complication of esophagectomy which can lead to prolonged vomiting, aspiration, and reduced oral intake postoperatively. To minimize the risk of DGCE, some advocate the use of pyloroplasty, however the practice is varied and controversial. The aim of this study is to investigate the effect of pyloroplasty on DGCE in esophagectomy patients.
Methods:
Consecutive patients that underwent an esophagectomy for esophageal cancer from September 2011 to December 2020 were identified from a prospectively maintained departmental cancer database at our institution. The primary outcome measured was the need for pyloric intervention following esophagectomy. Secondary outcomes included cardiac complications, pulmonary complications, anastomotic leaks, and chyle leaks. For those who had contrast swallow tests done prior to discharge from the index admission, dilated conduit and delayed gastric emptying were also investigated.
Results:
Of 458 patients included in the study, 77(17%) underwent pyloroplasty. Of the 381 patients who underwent esophagectomy without pyloroplasty, 44 (12%) of these patients later required at least one pyloric dilatation. None of the patients who underwent pyloroplasty later required pyloric dilatation. On multivariate analysis, younger age, respiratory complications after esophagectomy and pyloroplasty were significant predictors of the need for pyloric dilatation. Pyloroplasty was not significantly related to dilated conduit or delayed emptying on contrast swallow with P value of 0.979 and 0.147, respectively.
Conclusions:
Pyloroplasty reduces the need for pyloric dilatation postoperatively. However, further research is required to investigate the nature of this relationship, specifically in terms of contrast swallow.
Introduction
Esophageal cancer is the 14th most common cancer in the UK, comprising 2% of total cancer cases (2016-2018), with this number expected to increase rapidly over the next decade. 1 The survival rate is a mere 12% for 10 or more years (2013-2017) in England and Wales, making esophageal cancer the 7th most common cause of cancer deaths. One reason for such high mortality is the low proportion of patients presenting with disease amenable to curative treatment; only 19% of cases are suitable for oeseophagectomy. 2
As with any other major surgery, esophagectomy is a complex procedure that can lead to a myriad of postoperative complications. Denervation of the pylorus during the esophagectomy can lead to obstruction of gastric outflow, manifesting as symptoms of delayed gastric conduit emptying (DGCE). 3 The symptoms of DGCE can have distressing impacts on a patient’s quality of life and are strongly associated with adverse outcomes such as thoracic food retention, nutritional problems, anastomotic leak, pneumonia, and longer hospital stays.4-6
Some surgeons opt to perform pyloroplasty as an adjunct procedure at the same time as esophagectomy to minimize the effects of DGCE. Performing a pyloroplasty acts to widen the gastric outlet, allowing the gastric contents to pass through more easily and thus in theory remove any pyloric obstruction that may occur as a result of the esophagectomy. 7 However, there is ongoing controversy as to whether pyloric drainage procedures are necessary, especially when DGCE can be managed with Botox injection or corrective pyloroplasty post-operatively.5,8,9 There is also further debate as to whether the adverse effects of pyloroplasty itself outweigh any clinical benefit. Various long-term complications of performing pyloroplasty related to the absence of an intact pylorus have been reported, including dumping syndrome and bile reflux.3,7,10
There is a distinct lack of both global and national guidelines on the role of pyloroplasty, leaving the choice of whether to perform pyloric drainage procedures largely up to the preference of the surgeon. The aim of this study was to investigate the outcomes of pyloroplasty following esophagectomy for cancer.
Methods
Consecutive patients that underwent an esophagectomy for cancer at the Queen Elizabeth Hospital, Birmingham from September 2011 to December 2020 were identified retrospectively from a prospectively maintained departmental dataset. Patients were then followed up using the electronic patient record to determine whether these patients then later underwent pyloric dilatation, indicating presence of DGCE. The measured primary outcome was the need for pyloric dilatation. Secondary outcomes were cardiac complications, pulmonary complications, anastomotic leaks, and chyle leaks.
From the initial cohort of 458 patients, 134 patients were further identified who had a contrast swallow study with 10 days of the index esophagectomy. These patients were further assessed for presence of dilated conduit and delayed gastric emptying. As contrast swallow studies are not routinely carried out in this center to diagnose DGCE in the outpatient setting, no data was available regarding swallow studies and conduit dilation more than 10 days post-op.
Pyloric Dilatation and DGCE
The diagnostic modalities used to diagnose DGCE are diverse, with no gold standard radiological modality for either diagnosis or severity grading at present. 11 Water soluble contrast swallows, although widely available, are not routinely used in clinical practice as radiological evidence in diagnosis of DGCE. Solid-phase gastric emptying studies have also been used in DGCE diagnosis, however this is not as widely utilized in post-gastric surgery patients as water soluble contrast studies.5,12
Therefore, endoscopic balloon pyloric dilatation, which are more representative of the degree of DGCE affecting patients’ swallowing and quality of life, was used in this study as a surrogate measure. 13
If a contrast swallow was performed, the presence of dilated gastric conduit and DGCE was determined by a radiologist and the series was also examined by an upper GI surgeon (MSS) for concurrence. Gastric conduits were deemed to be dilated in the presence of functional radiological gastric emptying patterns.
Statistical Analyses
All analyses were performed using IBM SPSS Statistics 26 (IBM Corp. Armonk, NY), with P < .05 deemed to be indicative of statistical significance.
For univariate analysis, Chi-Squared test was used for categorical variables such as histology and Mann-Whitney U test was used for continuous variables such as age. When more than one factor was identified to be statistically significant, multivariate analysis using multivariable cox regression modelling with a forward stepwise approach was then performed to identify independent predictors.
Demographic comparison was initially made between pyloroplasty and no pyloroplasty groups using univariate analysis. Multivariate analysis was then conducted with statistically significant factors.
Two univariate subgroup analyses of those who had contrast swallow were also conducted, firstly for factors affecting dilated conduit after esophagectomy and subsequently for factors affecting delayed emptying after esophagectomy.
Results
Of 458 patients included in the study, 77 (17%) underwent pyloroplasty. The demographics of patients who underwent pyloroplasty versus no pyloroplasty are presented in Table 1.
Study Demographics.
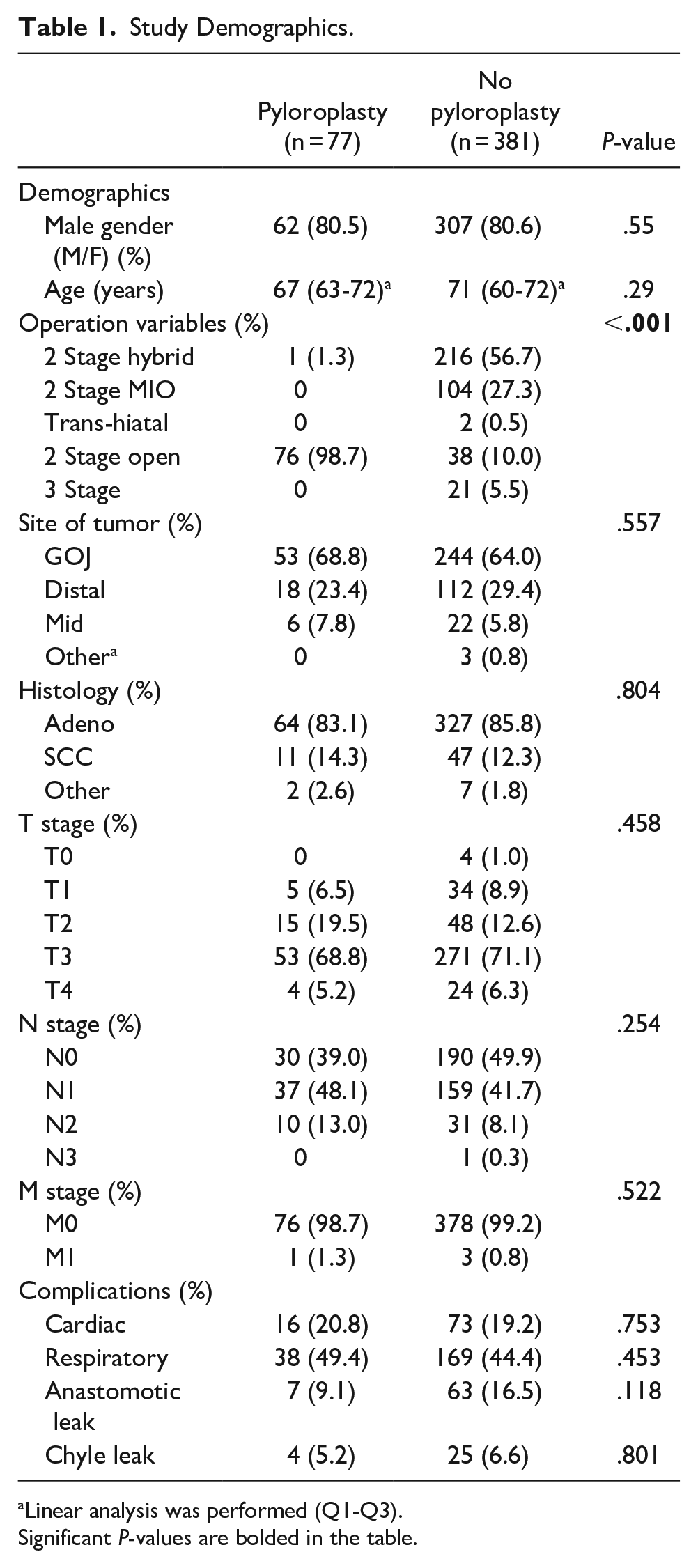
Linear analysis was performed (Q1-Q3). Significant P-values are bolded in the table.
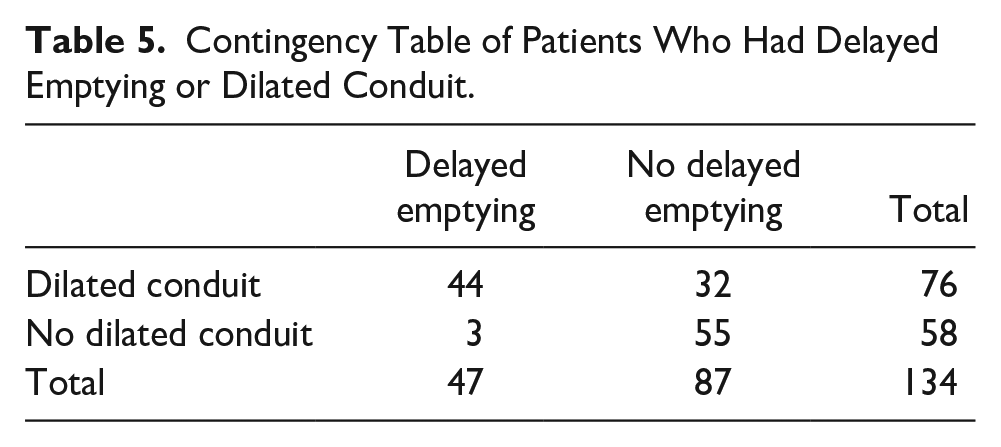
No patients who underwent pyloroplasty later required pyloric dilatation (Table 2). Multivariate analyses showed that age, respiratory complications, and no pyloroplasty were significant factors in terms of need for later pyloric dilatation. The most common respiratory complications were hospital acquired pneumonia (n = 145, 70%), followed by pleural effusion (n = 62, 29.9%). More severe complications such as acute respiratory distress syndrome (n = 8, 3.9%) or respiratory failure (n = 9, 4.3%) were also seen. Only 134 patients of 458 patients had a contrast swallow within 10 days of operation (29%), with Table 3 showing that the only statistically significant factor in presence of dilated conduit was site of tumor, with GOJ tumors experiencing the greatest rate of dilated conduit. Table 4 shows that the only significant factor affecting delayed emptying was presence of an anastomotic leak post-esophagectomy. Table 5 shows the number of patients who had dilated conduit or delayed emptying of those who required contrast swallow (n = 134). The relationship between these was explored with Fisher’s exact test and showed a statistically significant relationship between dilated conduit and delayed emptying (P < .001).
Univariate and Multivariate Analysis of Factors Affecting Need for Pyloric Dilatation After esophagectomy.
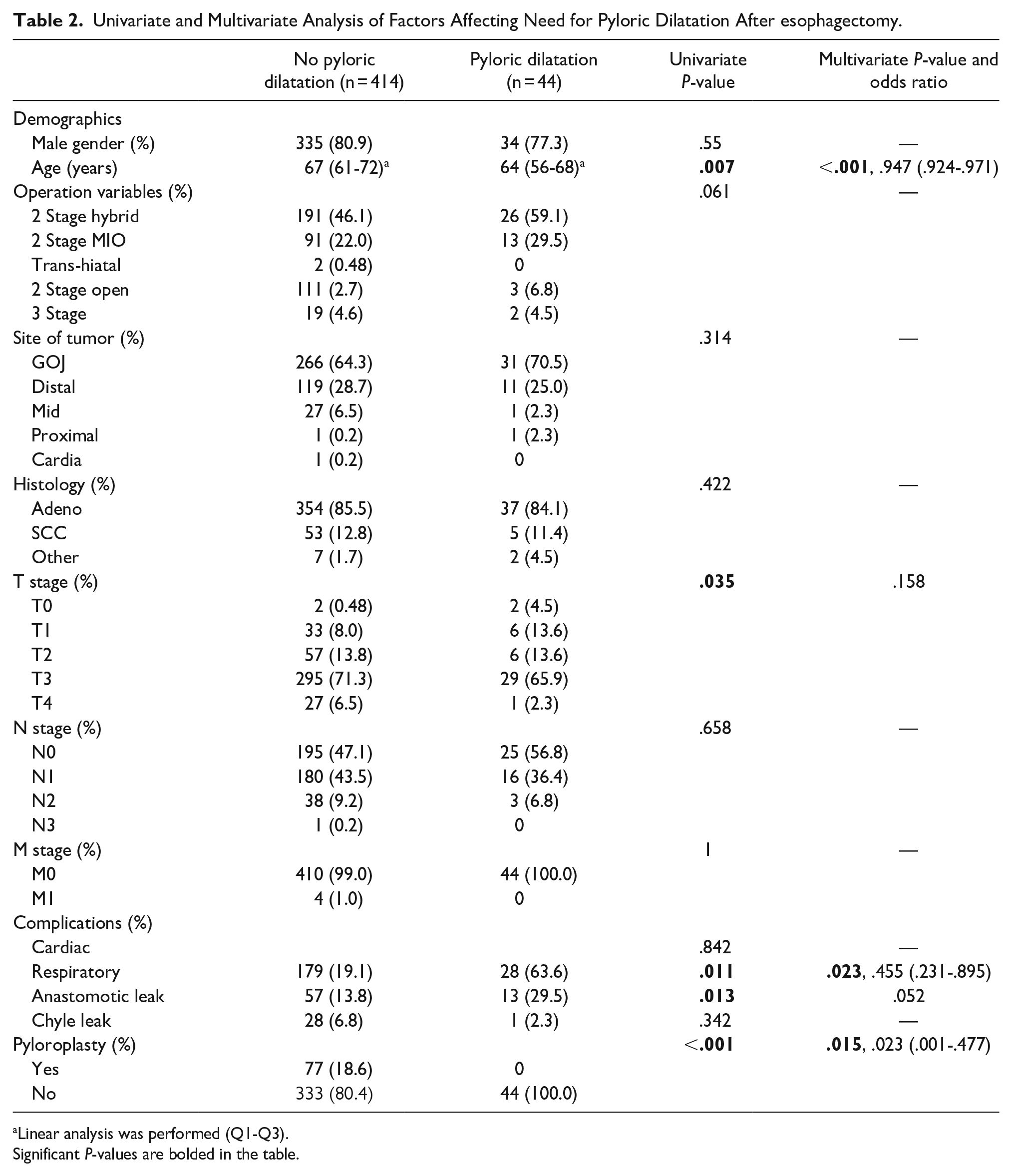
Linear analysis was performed (Q1-Q3). Significant P-values are bolded in the table.
Univariate Subgroup Analysis of Those That Had Contrast Swallow (n = 134) of Factors Affecting Dilated Conduit After Esophagectomy.

Linear analysis was performed (Q1-Q3). Significant P-values are bolded in the table.
Univariate Subgroup Analysis of Those That Had Contrast Swallow (n = 134) of Factors Affecting Delayed Emptying After Esophagectomy.
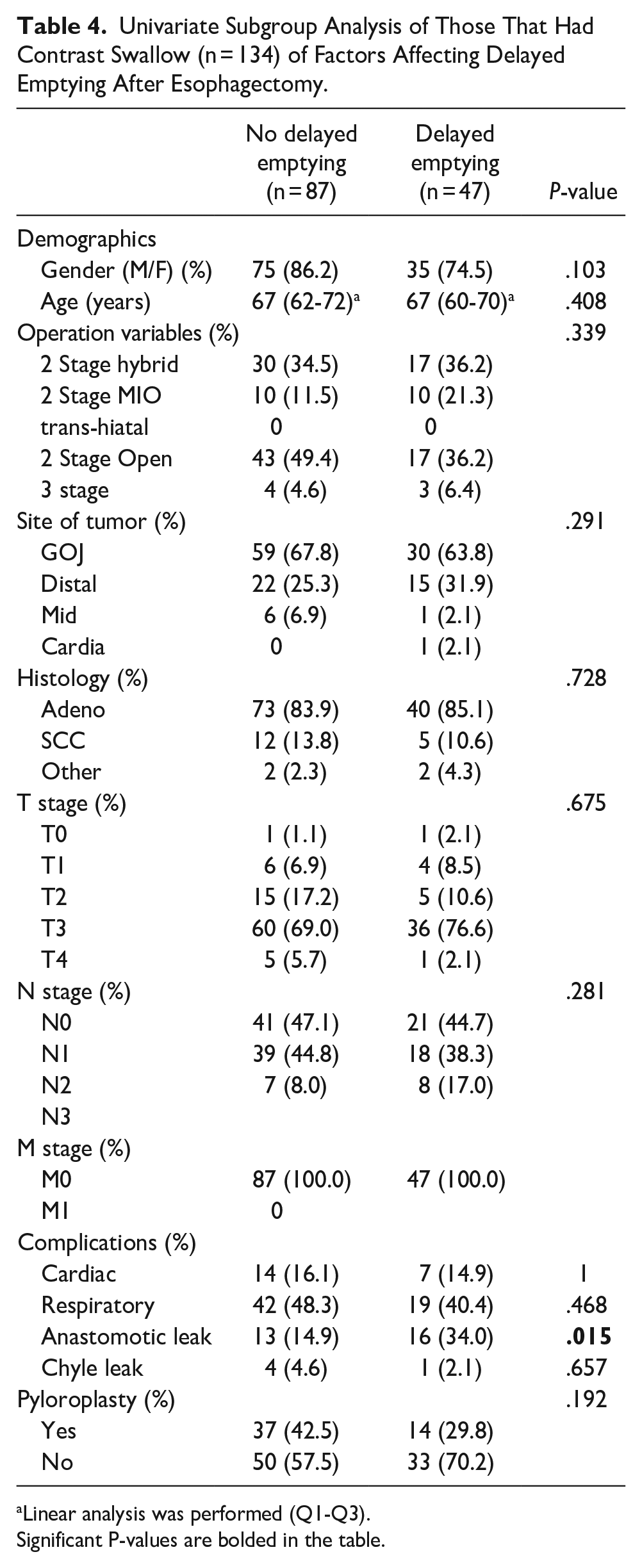
Linear analysis was performed (Q1-Q3). Significant P-values are bolded in the table.
Contingency Table of Patients Who Had Delayed Emptying or Dilated Conduit.
Discussion
This retrospective data analysis study aimed to investigate if pyloroplasty reduces the incidence of DGCE in the cohort operated in our institution. The use of pyloric drainage procedures such as pyloroplasty has had a long history of controversy, with research to support both the advantages and disadvantages of the practice. Our study provides evidence to support the advantages of pyloroplasty; the main finding was that those that underwent adjunctive pyloroplasty had a statistically significantly lower rate of need for later pyloric dilatation, thus indicating a lower rate of clinical DGCE. While the rates of anastomotic leaks were not statistically significant (P = .087), the raw data showed that patients that underwent pyloroplasty had almost half the rate of anastomotic leaks than those that did not (8.9 vs 16.6% respectively). The lack of statistical significance may be attributed to the comparatively lower sample size of pyloroplasty patients compared to the sample of non-pyloroplasty patients, highlighting the need for further research on a larger scale. We also note that those who required adjunct pyloroplasty underwent either 2 stage hybrid or 2 stage open, whereas those who didn’t undergo pyloroplasty had more diverse operation variables. We can thus conclude that adjunctive pyloroplasty reduces the risk of clinical DGCE, without adverse effects in terms of increased complications in the immediate perioperative period. The large total volume sample size of our study increases the precision of these findings.
Of the 44 patients who required pyloric dilatation post-operatively, 10 patients required more than 1 pyloric dilatation in the period between their operation date and time of data collected (September 2021). The mean period was 199.7 days, and the median period was 208.5 days. Their second pyloric dilatation was conducted as late as 1561 days post-operatively to as early as 33 days post-operatively. Performing a pyloroplasty during the initial esophagectomy may have avoided not just one, but multiple corrective surgeries (and their associated complications) in these patients.
Several previous studies have found that pre-existing pulmonary comorbidity and postoperative complications (such as anastomotic leak) were significant risk factors for DGCE.13-15 It has been suggested that dilation of the gastric conduit may cause increased tension at the anastomosis, leading to susceptibility to local ischemia and hence an anastomotic leak. 15 Conversely, it has also been proposed that DGCE may alternatively be a result of an anastomotic leak, due to local inflammation as a result of the leak causing impaired motility of the gastric conduit.
In any case, our study found that rates of anastomotic leaks were not statistically significantly associated with rates of DGCE. Only post-operative respiratory complications (within days to weeks) were statistically significantly associated with increased risk of DGCE. One possible explanation for the relationship found between incidence of respiratory complications and need for pyloric dilatation post-esophagectomy may be that the presence of infection in the thorax leads to reduced motility of the conduit. It is thus difficult to predict the development of DGCE pre-operatively—it can be argued that pyloroplasty should be performed in all appropriate patients, due to the lack of significant pre-operative risk factors. The main finding is supported by previously published research. Urschel et al’s 16 2002 meta-analysis of 9 RCTs (total of 533 patients) comparing use of surgically performed pyloric drainage procedures and no drainage procedures after esophagectomy found that use of pyloric drainage procedures significantly lowers the risk of DGCE at 1-week post-operative contrast study. 15
However, a more recent systematic review of 25 publications (total of 3172 patients) by Arya et al 5 found that while pyloric drainage procedures were associated with a reduced incidence of DGCE compared to no drainage procedures, this was non-significant. Furthermore, use of different definitions and methods of quantifying DGCE (including clinical symptoms, radiological swallows, radionuclide scintigraphy and direct gastropic visualization) among the studies also meant that inter-study comparison was not possible.
It should also be noted that Arya et al’s 5 review found that no RCT on this subject has been published since 2000, while of the 7 RCTs published between 1950 and 2000 only 2 were rated as good quality (according to the Jadad criteria). This clearly highlights the need for robust, high quality RCTs on the impact of pyloroplasty on clinical outcomes and should be considered for further research.
One possible limitation is that pyloroplasty is generally only performed during open surgery, due to the long-established technical challenges associated with performing the maneuvers laparoscopically. As seen in our own single-center study, 98.7% of pyloroplasties were performed as part of 2 stage open esophagectomies, while contrastingly open surgeries only made up 15.5% of non-pyloroplasty esophagectomies (Table 1). However, a 2002 retrospective review found that laparoscopic pyloroplasty was both practically feasible and yielded similar complication and success rates to open operations. Furthermore, a recent 2021 systematic review and meta-analyses found that open esophagectomy is associated with higher risks of mortality, DGCE and pulmonary complications compared to partial MIO. 17 Despite this, the findings from our study have demonstrated that the benefits of pyloroplasty, even in a center that generally only performs 2-stage open surgery esophagectomies, are significant despite the associated risks of open esophagectomy—the potential benefits may be even more pronounced when utilized alongside minimally invasive laparoscopic techniques. 18 It must however be noted that in our single-center study, the overwhelming majority of pyloroplasties were performed as part of open esophagectomies—this disparity between operation demographics in pyloroplasty versus no pyloroplasty patients may lead to a degree of unavoidable selection bias. Further multi-center studies, especially including those that perform laparoscopic pyloroplasty on a more routine basis, is required to support the validity of our findings. An unexpected finding was that age was inversely correlated with need for pyloric dilatation. One explanation may be because older patients tend to delay seeking medical attention unless the symptoms are pronounced. 19 Furthermore, it may be possible that older patients adopt better to eating smaller amounts and more frequently, thus reducing the need of pyloric dilatation even with the same degree of DGCE.
Throughout the literature, there is distinct lack of consensus with regards to the exact definition of DGCE, as demonstrated by the heterogeneity found in Arya et al’s review. We argue that the need for intervention, rather than radiological or visualization of a dilated conduit, is the most clinically relevant (and thus most useful) quantification. Although dilatation of the gastric conduit has conventionally been associated with, and used to support diagnosis of DGCE, not all patients with radiological evidence of dilated conduits require intervention, thus the presence of DGCE is irrelevant if it does not manifest in any significant symptoms.20,21
The pathophysiology behind manifestation of DGCE is multifactorial and complex, with predominating etiology likely to vary from patient to patient. While not fully understood, it has been suggested that relaxation dysfunction of the pylorus, peristalsis dysfunction, and kinking/dilation of the conduit all may contribute to different extents to the development of DGCE. 12 However, the underlying common ground for all patients with DGCE is the delayed passage of gastric contents leading to symptoms. Indeed, as part of recent modified Delphi process conducted by experts with an interest in DGCE after esophagectomy, it was agreed that diagnostic criteria for late DGCE should include both significant symptomatic manifestations of DGCE and evidence of radiological delayed contrast passage. 11
The use of pyloric dilatation (i.e., the need for intervention to remedy significant symptoms) in our research paper thus focuses on the merits of pyloroplasty in terms of improvement on patient experience of DGCE symptoms, rather than arbitrary radiological signs alone.
Limitations
Limitations include the retrospective nature of the study, and the data collected was of a single institution (although it is a high-volume specialist esophago-gastric cancer center). Additionally, fewer patients had pyloroplasty compared to those that did not (77 vs 391 patients respectively). This led to disproportionate sample size, affecting the level of confidence of results.
Conclusions and Future Studies
Performing pyloroplasties as an adjunct could help to reduce the rate of DGCE post-esophagectomy. This has further implications on the cost and patient burden of the need for subsequent surgery (pyloric dilatation) to treat this. Cost-effectiveness analysis (in terms of cost of treating complications vs. cost of pyloroplasty) studies are required to confirm this hypothesis. High-power, high quality RCTs are required to demonstrate a definite cause and effect. Furthermore, there is a need for more homogeneity in definitions and measurements of DGCE between studies to allow inter-study comparisons across different centers. Further collection of data exploring other well-documented measures of complications as a result of pyloroplasty and esophagectomies (such as rates of dumping syndrome and nutritional problems) will also aid in the benefits versus risk discussion of performing pyloroplasty.
Footnotes
Correction (May 2024):
Article updated to add Ethical approval statement.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study is exempt from our institutional review board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
