Abstract
Cervical esophageal cancers are rare and generally treated with definitive chemoradiation rather than trimodality therapy due to the need for pharyngolaryngectomy and esophagectomy. However, surgery remains indicated to salvage residual or recurrent disease. We review the management of cervical esophageal cancer and present a case series of complex surgical resection.
Introduction
Cervical esophageal cancers are rare neoplasia, making up approximately 5% of all esophageal cancers. 1 The cervical esophagus is short, comprising the length of esophagus extending from the cricoid cartilage to the thoracic inlet; starting at approximately 18 cm from the incisors when measured endoscopically. 2 While the majority (over 95%) are squamous cell carcinomas (SCC), other rare histopathological subtypes also occur, including adenocarcinomas (AC) thought to arise from an “inlet patch” of heterotopic columnar epithelium in the proximal esophagus.
From the surgical perspective, the proximity of cervical esophageal cancers to the upper esophageal sphincter and larynx frequently necessitates pharyngo-laryngectomy with esophagectomy (PLE) and lymphadenectomy to obtain clear margins and adequate lymph node dissection.3 -5 As this results in significant loss of quality-of-life, current treatment strategies focus on definitive chemoradiotherapy (dCRT) where possible.3 -6 However, surgery remains an option to salvage recurrent or residual disease and for situations where chemotherapy and radiation are contraindicated. The aim of this review is to discuss the current paradigms regarding management of these proximal tumors and provide some real-world examples of complex resections and reconstructions undertaken by our multidisciplinary surgical team.
Current Management of Cervical Esophageal Tumors
As cervical esophageal tumors remain a relatively rare entity, there is a lack of high-quality randomized trials comparing treatment outcomes in this area.6,7 Historically, these cancers were treated with up-front PLE and reconstruction with an intestinal conduit. This approach necessitates a permanent tracheostomy and loss of laryngeal function resulting in significant impairment of quality of life. Consequently, the current approach for tumors where surgery would necessitate a laryngectomy, as per guidelines from NCCN and ESMO, is for dCRT with surgery reserved as a salvage procedure, especially in tumors where surgery would necessitate laryngectomy.8,9 This organ-sparing approach offers good locoregional control with oncological results similar to neoadjuvant chemoradiation followed by surgery and leaves the option of salvage surgery available in future. 6
Most studies, including case series and RCTs examining the efficacy of dCRT in treating esophageal malignancies, are either in mixed populations of SCC and AC or, in studies dedicated to SCCs, a mixed population of cervical and thoracic SCC. 10 Therefore, the optimal dCRT approach for cervical SCC is open to debate. Many centers favor using cisplatin and 5-FU for systemic treatment in higher doses than typically used as neoadjuvant therapy for resectable thoracic esophageal cancer, with 5-year overall survival rates of 35.5% (29.7-41.9). 10 Other regimens used include FOLFOX, and other combination platinum-based therapies, such as carboplatin/docetaxel.11,12 These show acceptable short- and long-term survival and may have more favorable side-effect profiles when compared with cisplatin/5FU. Unfortunately, the evidence for superiority of one approach over others is lacking. With proximal disease remaining a rare entity, it is unlikely that large RCTs will be designed to determine the optimal approach to systemic therapy.
The second component of a definitive non-surgical approach is external beam radiation. Again, the optimal dose has not been established, and evidence is often based on a mixed population of cervical and thoracic malignancies. The usual radiation dose of dCRT for esophageal SCC is 50.4 Gy, although in cervical malignancy there has been a tendency to increase the radiation dose to 66 to 70 Gy.11,13 -15 The ARTDECO study examining dose escalation of radiation therapy in locally advanced, unresectable esophageal cancer did not show improved locoregional control with radiotherapy dose escalation from 50.4 to 61.6 Gy in patients treated with carboplatin/paclitaxel. 16 However, ARTDECO featured both SCC and adenocarcinomas and included predominantly tumors of the mid- and distal esophagus. Evidence for dose escalation in cervical tumors is primarily from case series and although some of these appear to show a dose-response relationship with higher radiation dosing, this finding has not been clearly replicated in clinical trials.17,18 When considering radiation as part of a planned neoadjuvant approach, 41.4 Gy may be administered based on dosing from the CROSS trial, although many centers tend to favor using 50.4 Gy. 6
The final component that may improve outcomes in non-operative management of cervical tumors is the developing field of immune checkpoint inhibitors (ICI) and novel monoclonal antibodies and tyrosine kinase inhibitors. Small case series supported the targeting of the epidermal growth factor receptor with cetuximab as a radiosensitizer to improve locoregional control for cervical tumors, but larger trials have shown no demonstrable benefit and increased treatment-related morbidity.19 -21 ICIs promote an immune response against tumors, by targeting tumor specific antigens such as cytotoxic T-lymphocyte antigen 4 or programed death-ligand 1. ICIs are likely to significantly improve outcomes for cervical tumors in the near future and are already approved for use in recurrent or metastatic esophageal SCC based on ATTRACTION-3, KEYNOTE-181, KEYNOTE-590 and CheckMate-648.22 -25 ATTRACTION-3 and KEYNOTE-181 examined the efficacy of second line nivolumab and pembrolizumab, respectively, as second line monotherapy in metastatic esophageal cancer, and showed improved overall survival compared to chemotherapy, with a response rate of approximately 20%.22,23 CheckMate-648 and KEYNOTE-590 studied nivolumab and pembrolizumab, respectively, as first-line combination therapies in advanced, unresectable, or metastatic cancer. There was improved overall survival when ICIs were used in combination with first line chemotherapy, suggesting a synergistic effect of using chemotherapy and ICIs in combination.24,25 In resectable disease, CheckMate-577 showed improved outcomes when nivolumab was used as adjuvant monotherapy for resected esophageal cancer treated with the CROSS protocol, where the pathological specimen showed residual disease post-operatively (in a mixed SCC and AC population). 26 Although not targeted at proximal cancers, the treatment effect was greater in SCCs, and a treatment effect for cervical SCCs can be extrapolated from this trial. There are ongoing trials that will provide better information regarding the efficacy and timing of ICIs in esophageal SCCs. Results are pending from the CRUCIAL trial, a phase II trial for patients with locally advanced esophageal cancer not amenable to surgical resection. This trial aimed to recruit 130 patients and compared definitive CRT with FOLFOX plus nivolumab followed by sequential nivolumab, and CRT plus nivolumab and ipilimumab followed by sequential nivolumab and ipilimumab; radiation dosing was 50 Gy. 27 The JCOG1804E (FRONTiER) study is a Japanese, multi-cohort study evaluating neoadjuvant nivolumab in combination with either CF, DCF or FLOT. 28 Although this is a phase I study evaluating safety and early efficacy, it will provide evidence to allow further trial design. Evidence in cervical tumors is again limited by focused trials specifically examining outcomes in proximal cancers, but evidence of efficacy in thoracic SCC may be extrapolated to proximal cancers.
Surgical Resection for Proximal Esophageal Cancer
Although surgical resection was historically the mainstay of treatment of these cancers, it was disfiguring, challenging to reconstruct, and led to poor quality of life outcomes. While dCRT is now first treatment of choice, situations of persistent or recurrent disease post-dCRT or SCC arising in a previous radiated field remain common and amenable to surgical therapy. 29 The principles of resection for tumors of the proximal esophagus remain consistent with other esophageal resections, namely to resect the primary tumor with adequate longitudinal and circumferential margins, perform a thorough lymphadenectomy of the draining lymph node basin, and reconstruct the enteric tract to provide intestinal continuity. For radical resections, reconstruction can use pedicled native conduits, such as a gastric pull-up or a colonic interposition, grafts with microvascular reconstruction such as a free jejunal interposition or a “super-charged” jejunal interposition, or local resection and flap reconstruction with free flaps such as forearm skin flaps, or local pedicled flaps such as delto-pectoral or pectoralis major myocutaneous flaps.30 -34 Each of these reconstructive options can be technically challenging with significant morbidity, both from operating in a previously irradiated field, and from advanced tumors. These operations are best undertaken by multidisciplinary teams with expertise in both esophageal resection and reconstruction and free-flap and microvascular surgery. Given the frequent need for laryngeal resection and permanent tracheostomy and the significant risk of perioperative morbidity, patient selection and counseling are of utmost importance. In our multidisciplinary practice, it is standard for patients to have multiple outpatient discussions with various members of the multidisciplinary team before the decision is made to proceed to surgical resection and reconstruction. A number of representative cases from our practice are outlined below.
Case 1
Patient 1 highlights the need for combined expertise from esophageal surgery with minimally-invasive expertise and head and neck surgery with expertise in flaps. A 54-year-old woman underwent definitive chemoradiotherapy for SCC of the upper esophagus 10 years prior. She had since required serial dilation for radiation induced stricture of the esophagus. On a recent dilation, a tracheoesophageal fistula was identified. Biopsy demonstrated recurrent SCC. A G-tube was placed for enteral nutrition and pre-surgical discussion and planning ensued. The tumor was at the inlet of the thorax (Figure 1a) invading the membranous trachea distally. The patient gave consent for a PLE with gastric pull up and anterolateral thigh (ALT) flap reconstruction of the posterior tracheal defect. Figure 1b shows the resulting tracheal defect following PLE. Figure 1c shows the ALT flap reconstruction prior to parachute down into the chest. The final specimen demonstrating the fistula is shown in Figure 1d. Figure 1e shows the pink gastric conduit in place under the thigh flap undergoing anastomosis to the pharynx. Figure 1f shows the final post-operative appearance of the reconstruction. The patient recovered well and was discharged home on day 14 tolerating an oral diet supplemented by jejunal tube feeds.
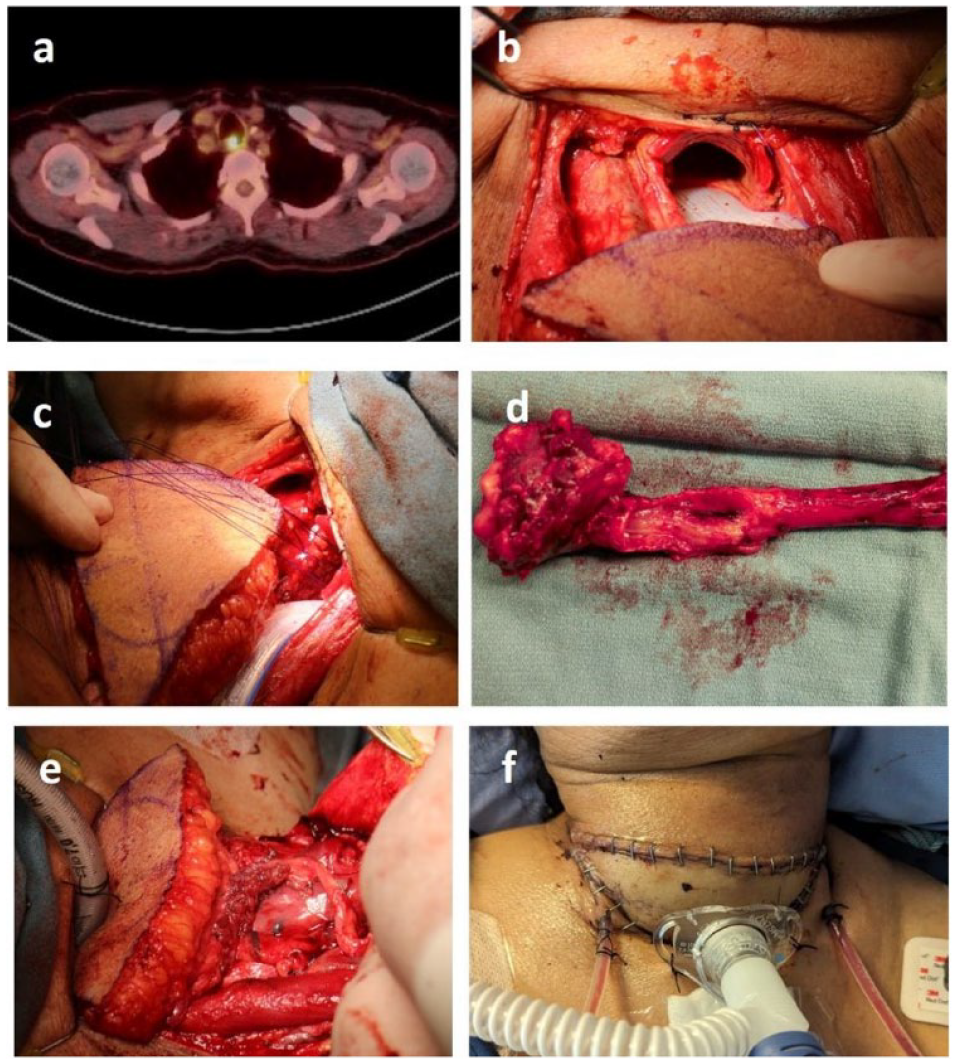
(a) PET scan showing proximal location of tumor, (b) demonstration of defect following surgical resection, (c) ALT flap in position to “parachute” onto defect, (d) surgical specimen demonstrating PLE, (e) ALT flap in final position with anastomosis being fashioned, and (f) final post-operative appearance with permanent tracheostomy and bilateral Jackson-Pratt drains in situ.
Case 2
Patient 2 represents a challenging reconstruction in previously irradiated tissue, with a prior resection and reconstruction. A 68-year-old female had a remote history of bilateral breast cancer treated with bilateral mastectomies, chemotherapy and radiation. Following an interval of approximately 20 years, she was diagnosed with a proximal esophageal cancer beginning just below the upper esophageal sphincter measured at 16 cm endoscopically. Histology showed a squamous cell carcinoma with imaging demonstrating locally advanced disease and local lymphadenopathy, with PET scan in Figure 2a. Following extensive counseling and multidisciplinary discussion, the decision was made to proceed with PLE with free flap reconstruction of the involved esophagus. The patient did not receive dCRT or neoadjuvant therapy due to her prior chemotherapy and radiation.
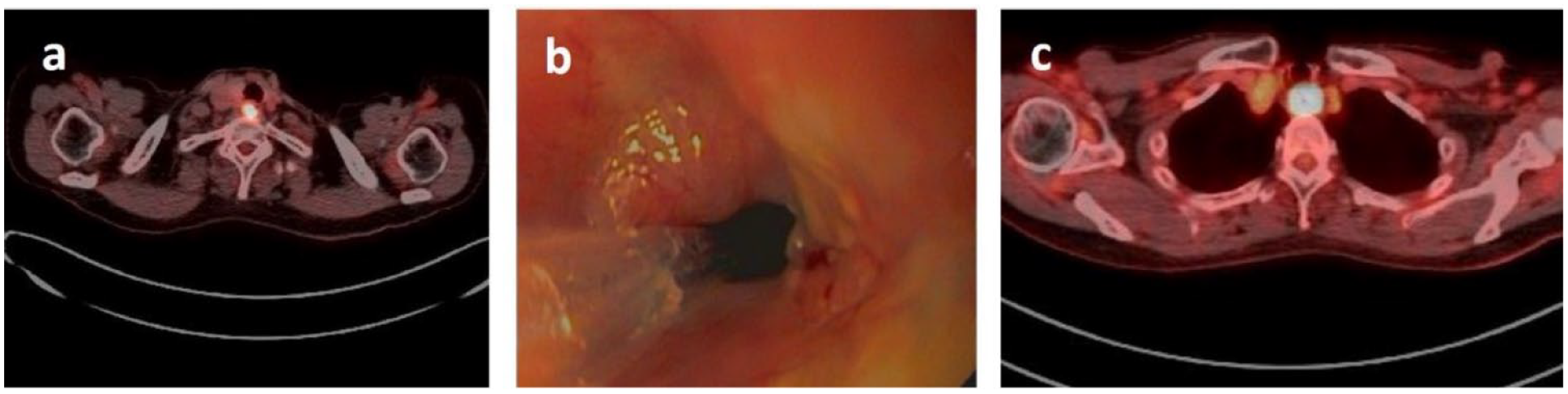
(a) Initial proximal esophageal lesion on PET/CT scan, showing FDG-avid proximal lesion, (b) endoscopic image demonstrating stricture and tumor at anastomosis between flap and esophagus, and (c) PET/CT demonstrating tumor at thoracic inlet, just distal to prior flap reconstruction.
A standard transverse cervical incision was made following induction of general anesthesia. A standard lymphadenectomy at levels 2A, 2B, 3 and 4 was performed. As the larynx was mobilized, the superior portion of the thyroid gland was noted to be involved and was resected en bloc, with preservation of the inferior parathyroid glands. PLE was competed, with specimens sent for frozen section showing clear margins. A permanent tracheostomy was fashioned. The defect for enteric reconstruction was approximately 11 cm and was reconstructed with a free, tubularized anterolateral thigh flap. The flap was anastomosed distally to the remnant of the proximal esophagus and proximally to the pharynx. An arterial anastomosis was performed with the right superior thyroid artery and a venous anastomosis to the right facial vein. A temporary salivary bypass tube was placed to facilitate healing. The patient recovered well and was discharged 3 weeks post procedure. Final histology showed a T3N0 (0/32) SCC with negative margins.
Five years following her resection, she presented with new onset dysphagia and weight loss. Investigations showed a new lesion in the proximal remnant esophagus, just distal to the anastomosis with the prior free thigh flap reconstruction (Figure 2b and c). Staging confirmed localized disease with no evidence of distant metastasis. Following extensive multidisciplinary discussion, the decision was made to proceed with salvage resection. A minimally-invasive 3 phase (McKeown) esophagectomy was performed. The prior free flap was preserved, with the distal end resected en-bloc with the remnant esophagus. A gastric pull-up was performed with a hand-sewn anastomosis to the free thigh flap. The patient had a small leak from the vertical staple line on the gastric conduit post-operatively, but recovered well and was discharged from hospital after 3 weeks tolerating PO diet. Final histology showed a T3N0 (0/15) tumor, with negative margins.
Case 3
Patient 3 illustrates preservation of the larynx in an upper esophageal cancer. A 52-year-old female presented with a history of progressive dysphagia and weight loss. She had a complex history, with a prior tracheal resection for an adenoid cystic carcinoma over 20 years ago. At that time, there was an R1 resection margin at the proximal tracheal edge and the patient underwent adjuvant radiation. She subsequently had a number of metastasectomies, but when referred for dysphagia was disease free and an excellent quality of life. Investigations showed a lesion in the proximal esophagus. This was measured endoscopically as at 20 cm, with the upper esophageal sphincter at 18 cm. Histology showed an SCC, likely induced by prior radiation and therefore precluded dCRT. Initially, a PLE was discussed, but the patient was keen to preserve her larynx, if possible, at the expense of a limited proximal margin.
Due to previous tracheal and lung surgery, the thoracic esophagus was mobilized via redo right thoracotomy. The proximal tumor was abutting the trachea but could be mobilized free without evidence of invasion. A lymphadenectomy was undertaken, within the limits of a prior operative field and previous lymphadenectomy. Following closure of the thoracotomy, the patient was positioned supine and the stomach mobilized to fashion a neo-esophagus in standard fashion using laparoscopy. A transverse cervical incision was made and the proximal esophagus mobilized circumferentially, preserving the recurrent laryngeal nerves. The esophagus was divided proximally, just below the UES and just above the tumor. The specimen was sent for frozen section and showed a negative proximal margin. The esophagus was removed, and the gastric conduit pulled to the neck. A hand-sewn anastomosis was performed between the conduit and the short remnant proximal esophagus. The patient recovered well and was discharged on enteral feeds and some PO intake 10 days post-operatively. Final histology showed a T3N0 (0/12) SCC, with no LVI or PNI and a 5 mm final proximal margin. As the CPS score was elevated, the patient received adjuvant nivolumab.
Conclusion
Proximal esophageal tumors are rare and challenging entities to treat. The treatment of choice should remain dCRT, with surgical resection reserved for cases of local recurrence, or where dCRT is not feasible. Extensive preoperative counseling and planning is necessary, but complex resections and reconstructions can be safely delivered through multidisciplinary operating teams with expertise in minimally-invasive surgery, esophageal cancer, head and neck surgery, and free flaps.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
