Abstract
Background:
Stillbirths are a major global health concern. Improving intrapartum quality of care could reduce stillbirths, but there is no synthesized evidence on the impact of different intrapartum interventions on stillbirths.
Objectives:
The broad aim was to assess whether intrapartum quality improvement packages and related facility-based clinical, technological and health system interventions at maternity units of health facilities led to a reduction of stillbirths.
Design:
A systematic review and meta-analysis of experimental and analytical studies.
Data sources and methods:
This systematic review is reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. PubMed, Web of Science, Scopus, ProQuest, Cochrane, China National Knowledge Infrastructure (CNKI) databases were searched during July–August 2024. A comprehensive search strategy was developed using Medical Subject Headings (MeSH) terms and keywords. We conducted database scanning, article screening and data extraction and included experimental, cohort or case-control designs. Risk of bias was assessed using Critical Appraisal Skills Programme (CASP), ROBINS-I and RoB 2 checklists. A random-effects model was used to account for between-study variability. Heterogeneity was assessed using Cochran’s Q test, tau-squared (τ2) and quantified using the I2 statistic followed by exploration through sensitivity analysis, subgroup analysis and meta-regression.
Results:
The final selection included 24 articles involving 4,647,555 intrapartum women for the systematic review and 17 studies for the meta-analysis. Quality improvement intrapartum interventions of foetal heart rate monitoring, capacity building and mentoring of providers and clinical practice improvement seemed to be beneficial in reducing stillbirths. The pooled estimate on meta-analysis yielded an odds ratio of 0.25 (95% CI: 0.06–1.10), suggesting a 75% reduction in the odds of stillbirth with intrapartum interventions, but not statistically significant. Diverse study settings enhanced the generalizability of findings, but the marked heterogeneity and variable methodological quality of the studies call for caution in making conclusions.
Conclusion:
Comprehensive quality improvement programmes during intrapartum period suggest potential benefits in reduction of stillbirths and can impact intrapartum care practices globally, particularly in low-resource settings. Heterogeneity in study design, study quality and contextual factors underscored the necessity for further research.
Review registration:
PROSPERO CRD42024503854.
Plain language summary
What did we do: We sought to find out whether stillbirths can be reduced by improving quality of care given to women during labour and childbirth. We used a methodology known as systematic review and meta-analysis to address this question. We collected published research studies from large scientific databases through July-August 2024 and selected those studies which assessed the effect of quality improvement programmes implemented during labor on occurrence of stillbirths. The quality improvement programmes included: (1) monitoring the baby’s heartbeat while the mother is in labor, (2) training and mentoring of health providers or birth attendants, (3) improving clinical practices and standards among others. What did we find: Through search strategies, we selected 1356 articles, which came down to 181 after duplicate removals and exclusions. We did full text review of these articles and selected 24 for systematic review. Seventeen articles were selected for meta-analysis; we found that women who delivered in facilities where quality improvement interventions were implemented during labor had lower chances of stillbirths, although statistically, the result was not strong enough to be called conclusive. The studies were from a wide range of settings; hence the findings can be regarded as applicable to diverse backgrounds. However, the studies were not uniform in terms of study design, quality and local contexts. What does this mean: Improving the quality of care of interventions during labor such as monitoring baby’s heart rate as per a protocol and performing immediate caesarean section if needed, capacity building and mentoring of birth attendants, improving standards of care and clinical practices of labour have been found to help reduce the chances of stillbirths (however the result was not strong enough to be confident). While more research is needed, it is noteworthy that improving quality of intrapartum care is worth investing, especially in low resources.
Introduction
Stillbirths continue to be a major global health concern, despite sustained healthcare investments and medical advancements. Stillbirths refer to foetuses who are born with no signs of life, with the deaths occurring before or during labour. For purposes of international comparison, stillbirths are defined as third-trimester foetal deaths (⩾1000 g or ⩾28 weeks). 1
Over the last two decades, approximately 48 million babies were born dead. In 2021, worldwide, about 1.9 million babies were stillborn at 28 weeks or more of gestation. The low-income countries of the world, especially those of Sub-Saharan Africa and Asia account for more than 80% of the global stillbirth burden but only 62% of livebirths. 1 Most of the stillbirths occur from preventable causes; about 40% of them occur during labour.1–3 Despite the high burden, stillbirths were missing from the global health agenda and discourse, even during the Millennium Development Goals (MDG) era. 4 The Lancet series of 2011 and 2016 called out stillbirths as a neglected tragedy and pronounced that about half of stillbirths being alive at the start of labour and losing their lives before or during delivery was nothing less than a health scandal of international proportions. The series discussed risk factors of stillbirths, their devastating impact on families and health providers, presented epidemiological evidence and interventions to address the issue, and called for concerted global action.5,6 The Every Newborn Action Plan 2014, recognizing stillbirths as a health priority, set a global target of less than 12 stillbirths per 1000 births by 2030. 7 The Sustainable Development Goals, 2015, address the issue indirectly in Goal 3 through target 2 of ending preventable deaths of newborns and under 5 children 8 .
The stillbirth rate (SBR) is one of WHO’s core health indicators and measures the proportion of stillbirths among 1000 births, dead and live. 9 SBRs have reduced over the MDG period, but they could not match the more definitive decline of maternal and child mortality.10,11 Stillbirths were notably absent or underrepresented in global trackers till the 2015 inclusion in the Global Burden of Disease (GBD) tracking. The GBD created estimates of stillbirths and rates across geographies in its 2015 iteration for stillbirths of equal to or more than 28 weeks gestation. 12 The GBD 2021 inducted stillbirths into the global disease burden estimation framework for the first ever time, using stillbirths at 20 weeks or more as reference and stillbirths at 28 weeks gestation or more as comparison. GBD estimated the global SBR at 23 per 1000 births at 20 weeks gestation or longer, in comparison to 16.1 per 1000 births at 28 weeks gestation or longer.13,14
A meta-analysis of risk factors in high-income countries reported higher odds of stillbirth for women with pregnancy-induced hypertension, pre-eclampsia and eclampsia. 15 A study conducted in nine states in India reported that pregnancy complications, including anaemia, eclampsia, other hypertensive disorders, antepartum haemorrhage, intrapartum haemorrhage, abnormal **fetal position, breech presentation and obstructed labour significantly increased the odds of stillbirth. 16 Stillbirths due to congenital anomalies amount to less than 10% of the total in most of the countries, except for some high-income countries including Australia, Canada, Ireland, Scotland, etc. 11 About 70% of intrapartum stillbirths are preventable by provision of quality intrapartum care and early identification of high-risk pregnancy cases.17,18 Intrapartum stillbirth is a sensitive marker reflecting the delayed, low quality of care which leads to reduced or absent intrapartum monitoring and emergency obstetric care to hasten delivery of a distressed foetus.18,19
Quality of intrapartum care can be viewed through the lens of the Donabedian framework as an interplay between structure (health infrastructure, availability of workforce, equipment, etc.), processes (clinical protocols, standard operating procedures, audit, skilling of workforce, etc.) and outcome (e.g. stillbirth). 20 Quality improvement (QI) focusses on ‘change’ (improvement), a ‘method’ (a systematic approach aided by appropriate tools) and a ‘context’ (the environment in which the improvement is made) to achieve better outcomes. Delivering in healthcare institutions cannot guarantee prevention of stillbirths; it is also contingent on the availability of adequate infrastructure, skilled human resources, implementing evidence-based interventions, not subjecting women to ineffective, potentially harmful medical interventions, provision of emotional support through labour companionship, effective communication and respectful care as well as birthing options.21,22 Good-quality care thus requires appropriate use of effective clinical and non-clinical interventions, strengthened health infrastructure, optimum skills and a positive attitude of health providers. These will provide women, families and health providers a positive experience, besides improving health outcomes. 21
Countries report different experiences in the quality-of-care parameters. In a study conducted in a low-resource setting in Tanzania, it was seen that neither international nor locally adapted standards of intrapartum routine care were optimally achieved, most likely due to a grossly inadequate capacity of birth attendants. 23 In India, for instance, the number of health facilities has increased but workforce shortages are universal across the three tier public health delivery system. 24 A study from Italy reported that more than 90% of intrapartum mothers were always or often treated with dignity and respect, had always or often their privacy and confidentiality preserved and 89.1% of mothers declared that always or often had an efficient communication with hospital staff. 25
There are studies investigating the impact of individual components of intrapartum care such as foetal heart rate monitoring (FHRM), capacity building of intrapartum care providers or other quality improvement interventions in isolation on maternal and foetal outcomes (mainly neonatal), as well as on process indicators such as compliance to protocols and timely caesarean sections.26–28 Stillbirth, when reported, is part of the perinatal outcome or is not adequately disaggregated, thus limiting the ability to draw conclusions. Many studies present inconclusive evidence of individual intrapartum interventions on stillbirths, and there is a clear dearth of these interventions being synthesized with system-level interventions. Our paper would focus on stillbirths as an outcome and synthesize evidence across a broad range of intrapartum interventions by integrating clinical and health system interventions within a single analytical framework. This is expected to provide a comprehensive assessment of which intrapartum quality improvement interventions are most likely to contribute to stillbirth reduction, thereby strengthening evidence base for programmatic and clinical decision making.
Objectives
The broad objective of this systematic review and meta-analysis was to assess whether intrapartum quality improvement packages and related facility-based clinical, technological and health system interventions at maternity units of the health facilities led to a reduction of stillbirths.
The systematic review and meta-analysis attempted to answer the following questions:
Was there a reduction in the risk of stillbirths when quality improvement packages and related facility-based clinical, technological and health system interventions pertaining to intrapartum care were implemented?
Did the risk of stillbirths with intrapartum interventions vary between settings?
What methodological characteristics would explain heterogeneity in results?
Methodology
Study design
To address these research questions, a systematic review and meta-analysis was done. The protocol for this review was registered in the PROSPERO database on 30 April 2024 and was published as per the Preferred Reporting Items for Systematic Reviews and Meta-Analyses–Protocol (PRISMA-P) guidelines. 29 This report followed the PRISMA guidelines (Supplemental Appendix 1). 30
Eligibility criteria
Inclusion criteria
Inclusion criteria were fixed to select the studies with the ability to assess whether intrapartum quality improvement programmes at intrapartum points of care could have an impact on the risk of stillbirths.
Inclusion criteria included:
(i) study designs such as randomized controlled trials, controlled non-randomized trials, controlled before and after designs, interrupted time series studies with at least three data points before and after the intervention, prospective and retrospective cohort studies, case control and nested case-control studies
(ii) study population of women admitted in health facilities for delivery or case records of women delivered, irrespective of gestation age or risk status
(iii) intervention consisting of a quality improvement action for intrapartum care involving clinical or health system-related
(iv) comparators or controls of pregnant women in labour – full term, or pre-term or high-risk who will be receiving standard care or business as usual and not the intervention in the quality improvement package
(v) outcome of interest is the reduction in stillbirths in the participants and comparators
(vi) studies in all languages.
Exclusion criteria
They included descriptive studies, study protocols, conference presentations, case series and case reports.
Data sources
A literature review was conducted after finalizing the Medical Subject Headings (MeSH) and terms related to the main study variables according to PICO (Population, Intervention, Control and Outcomes). We searched databases, namely PubMed, Web of Science, Scopus, ProQuest, Cochrane and China National Knowledge Infrastructure (CNKI) databases. Additionally, grey literature was searched to identify unpublished research studies, dissertations, ongoing trials, etc. This was carried out using Google search and referring to website and library guides such as Shodhganga. The reference lists of studies identified during the search process were also scanned. Data sources were searched through July−August 2024.
Search methods
Search strategy
A comprehensive search strategy was launched to identify studies reporting stillbirths and other adverse pregnancy outcomes following the implementation of intrapartum quality improvement interventions. The study period was limited to 20 years between 2004 and 2024 to ensure adherence to the latest developments in the field. An initial string was developed for PubMed, which was modified for other databases. The search string was developed using Boolean operators in conjunction with a mix of MeSH terms and keywords such as ‘quality improvement’, ‘intrapartum’, ‘stillbirth’, ‘neonatal mortality’, ‘maternal mortality’, ‘fetal monitoring’ and ‘training’ to maximize the chances of selection of relevant articles. The details of the search methodology, database wise, are provided in (Supplemental Annexure 1). The search strategy was re-executed up to 30 August 2024, the point of final article selection, to ensure comprehensive coverage.
Selection process
Two team members carried out a pilot screening of titles and abstracts from an initial set of 50 studies to ensure compatibility of the prepared checklist with eligibility criteria. Subsequently, all the titles and abstracts were screened by four team members, with each title and abstract assessed by two individuals independently (Supplemental Annexure 2). The disagreements were addressed in consultation with the adjudicator. The selected articles were then subjected to full-text screening (Supplemental Annexure 3). Checklists had already been developed for full-text screening, by the team through multiple deliberations, for different study designs (Supplemental Annexures 4). Two pairs of team members did full-text screening independently with no blinding within a pair. The third pair of two team members validated the selection; the teams dealt with disagreements through discussions, before consulting the adjudicator for a final consensus. The selected articles were later categorized based on outcomes – stillbirths, neonatal mortality and maternal mortality. For this paper, we selected only articles that had stillbirths as outcomes, with or without maternal mortality or neonatal mortality, for systematic review and studies with stillbirths only as outcomes for the meta-analysis.
Data collection process
Data extraction was carried out from articles with stillbirth as outcome, using two data sheets preprepared separately for experimental designs and for analytical designs. Two teams extracted data independently and discussed the findings in the presence of an adjudicator who helped in reaching a consensus. Data was extracted on study characteristics, facility settings, intrapartum interventions and outcomes.
Risk-of-bias appraisal
The case-control studies and cohort studies were appraised using the Critical Appraisal Skills Programme (CASP) checklists.31,32 The quasi-experimental and randomized controlled trial (RCT) studies were assessed with ROBINS-I and RoB 2, respectively. 33,34
The CASP checklist for case-control studies and cohort studies covers 11–12 domains about study validity, results and applicability. For both designs, each criterion was rated as ‘Yes’, ‘No’ or ‘Can’t tell’. The quasi-experimental studies were assessed with ROBINS-I tool, which assesses bias across seven domains. The first two domains covered issues of confounding and selection of participants, which belong to the pre-intervention period. The third domain was about the intervention itself, and the next four domains addressed issues after the start of interventions: biases due to deviations from intended interventions, missing data, measurement of outcomes and selection of the reported result. Based on these parameters, studies were assessed as low, moderate, serious or critical risk or insufficient information. The RCTs was appraised using Version 2 of the Cochrane risk-of-bias tool for randomized trials RoB 2, which is the recommended tool to assess the risk of bias in randomized trials. RoB 2 was structured into a fixed set of bias domains, focusing on different aspects of trial design, conduct and reporting. Within each domain, a series of questions (‘signalling questions’) asked for information about features of the trial that were relevant to risk of bias. Judgement was expressed as ‘Low’ or ‘High’ risk of bias, or ‘Some concerns’.
Statistical analysis
Out of 24 studies, 17 were selected for meta-analysis on the basis of completeness of data required to pool the risk with other studies. Seven studies were excluded from meta-analysis because they reported stillbirth and neonatal mortality as a composite outcome or failed to provide numerical data for the exposed and non-exposed (study and control) groups necessary for inclusion for meta-analysis.
All statistical analyses were conducted using R version 4.4.2. using {meta} and {metafor} packages.35–37 The effect measure for dichotomous outcomes was the odds ratio (OR) with 95% confidence intervals (CI), and a p value <0.05 was considered statistically significant. A random-effects model was used throughout the meta-analysis to account for between-study variability and expected clinical and methodological heterogeneity across interventions, settings and study designs.
Heterogeneity across studies was assessed using the Cochran’s Q test, tau-squared (τ2) and quantified using the I2 statistic. Heterogeneity was interpreted as low (<50%), moderate (50%–74%) or high (⩾75%). In this analysis, substantial heterogeneity was observed (I2 > 95%), necessitating further exploration through sensitivity analysis, subgroup analysis and meta-regression. To assess the robustness of the results, a leave-one-out sensitivity analysis was conducted. This method involved sequentially omitting one study at a time and recalculating the pooled OR.
The 17 studies used in meta-analysis differed substantially in study design (case–control, cohort, quasi-experimental, randomized trials) and in the type of intervention evaluated. As a result, not all studies were eligible for every subsequent analysis.
Subgroup analyses were conducted based on the type of intrapartum interventions of the selected studies mentioned below, as well as contextual settings.
- Labour monitoring: Monitoring of foetal heart rate and progress of labour (Type A)
- Capacity building of the staff providing intrapartum care by mentoring /and training programmes (Type B)
- Improvement of standards of clinical care through clinical guidelines and audits (Type C)
- Technology use – software for clinical decision support (Type D)
- Intrapartum referral to higher centres (Type E)
- Maternity waiting homes (MWH) during the intrapartum period (Type F)
This was done to examine whether the effectiveness of intrapartum quality improvement interventions varied by intervention type and contextual setting. These groupings reflect distinct theoretical mechanisms of action and implementation approaches and were selected to explore potential sources of heterogeneity in relation to the study objectives.
To explore potential sources of heterogeneity, a meta-regression was performed using year of publication as a covariate. Publication bias was evaluated using both funnel plot inspection and further validated through Egger’s regression test.
Ethics approval
Ethics approval and participant consent were not applicable to this study since it is a meta-analysis of data from previously published studies available on public information sources. The analysis involved only aggregated data and not any individual-level identifiable data.
Results
Study selection
The initial search yielded a total of 1356 articles from the databases. Of those, 358 duplicates were identified using MS Excel and were excluded. The titles and abstracts of all the 998 studies were reviewed and 817 were excluded. On examining the 181 articles’ full texts (Supplemental Annexure 3), a total of 31 articles that met the eligibility criteria were finally selected for data extraction. Among these, 24 studies specifically reported stillbirth as an outcome, of which 17 were selected for meta-analysis.
Out of the 1356 articles initially selected, 1337 were in English, 8 in Portuguese, 3 Persian, 6 French and 1 each of Spanish and German. In the final list of 24 selected articles with stillbirths as outcome, 23 were in the English language and 1 in French.
The entire selection process is captured in the following figure (Figure 1).
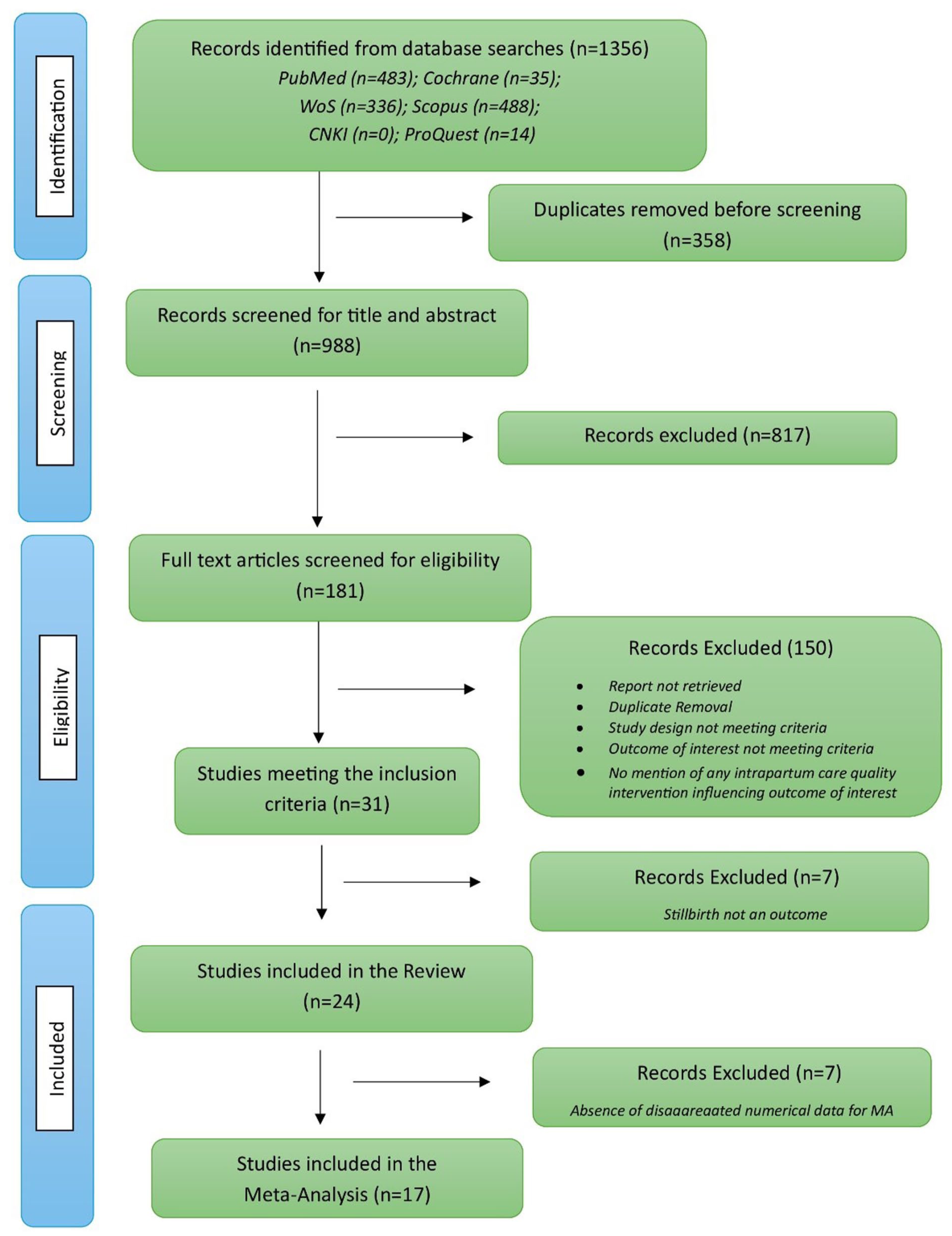
PRISMA diagram showing the process of selection of studies.
Study characteristics
Of the total 24 selected studies with stillbirths as outcome, there were 5 case-control studies, 7 cohort studies and 6 each of quasi-experimental studies and RCTs. Collectively, the 24 studies covered a sample of 4,647,555 in 13 countries; 15 studies were conducted in seven countries of the African region, 2 studies involved two countries from European region, 5 were in two countries of Asian region and 2 studies were in two Western Pacific countries. Three of these countries were high income, two were low income, one high-middle-income and the remaining seven were low-middle income countries.
Study quality
Case-control studies
Out of five case-control studies, two (Kc et al., 52 Gwako et al. 61 ) answered ‘Yes’ to all 11 questions, indicating low risk of bias across design, case-control selection, measurement of exposure and confounder adjustment. One study (Agena and Modiba 41 ) met 10 criteria but reported exposure measurement as a grey area and was placed in low to moderate risk of bias. Two studies, Ouahid et al. 45 and Maaloe et al. 51 , raised concerns about control selection, precision of results and confounder control and ended up as studies of moderate and moderate-high risk, respectively.
The summary of risk-of-bias assessment for case-control studies is placed in Figure 2(a).
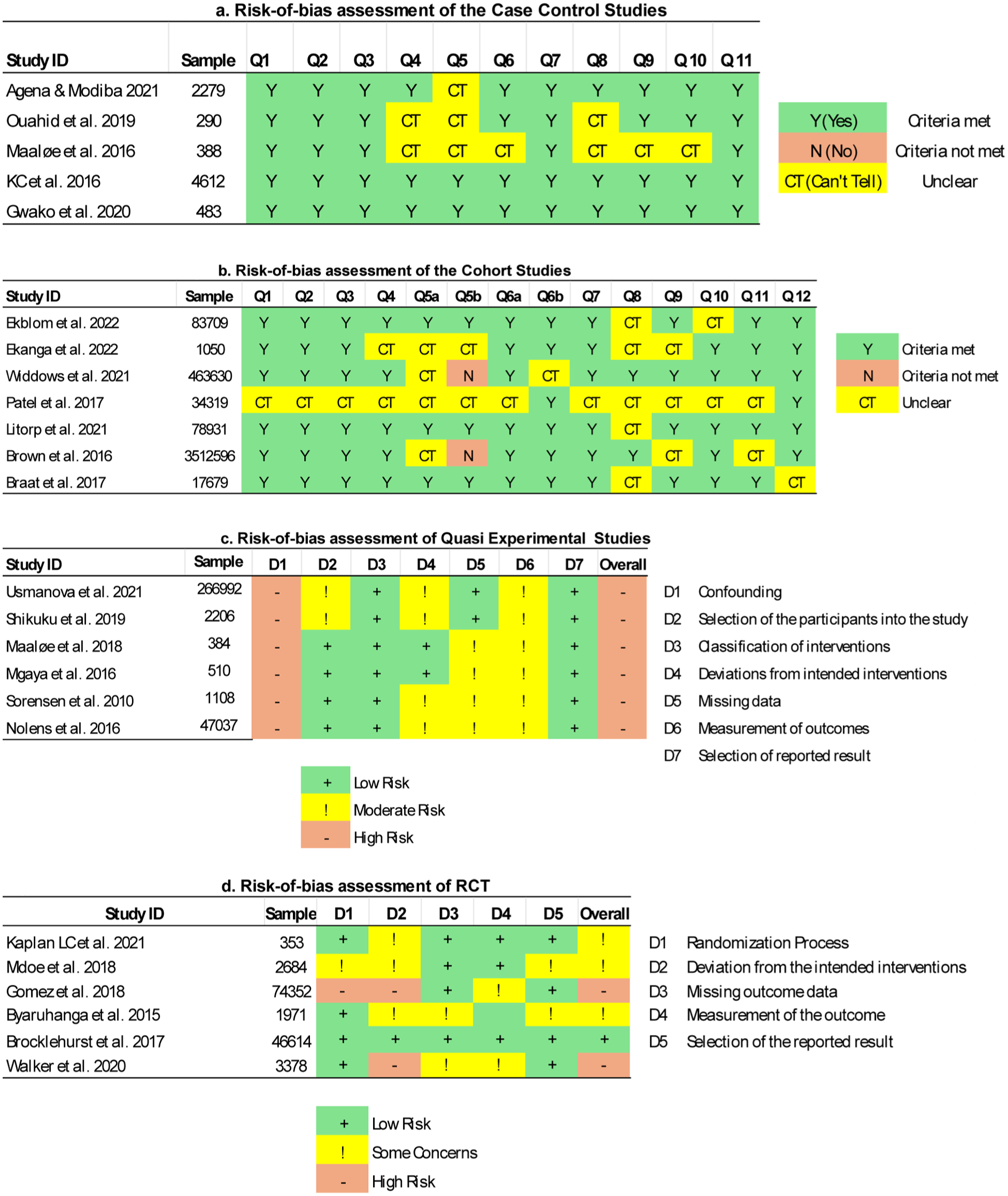
Risk-of-bias assessment of the included studies. (a) Risk-of-bias assessment of the case-control studies; (b) Risk-of-bias assessment of the cohort studies; (c) Risk-of-bias assessment of quasi-experimental studies; (d) Risk-of-bias assessment of RCT.
Cohort studies
The overall risk of bias of seven cohort studies ranged from low to moderate. Three studies (Ekblom et al. 38 , Litorp et al., 55 Braat et al. 59 ) scored positively on all but one criterion; they measured exposure and outcome reliably, controlled confounders and used precise outcome definitions and rigorous statistical assessments. Identifying and adjusting confounders or presenting results with precision and statistical power, or both, were challenging criteria for most of the teams except for the studies mentioned above.
The summary of risk-of-bias assessment for cohort studies is placed in Figure 2(b).
Quasi-experimental studies
None of the six studies could be classified as of low risk and could be equated with a RCT and were judged to have a serious overall risk of bias. Confounding was a challenge, except Shikuku et al., who used a comparison facility. Selection bias, measurement bias, intervention fidelity were all concerns for multiple studies; however, reporting bias was low for all.
The summary of risk-of-bias assessment for quasi-experimental studies is placed in Figure 2(c).
Randomized controlled trials
Out of six RCTs, only Brocklehurst et al. 57 reported a low risk of bias, whereas Kaplan et al., 40 Mdoe et al., 47 Byaruhanga et al. 53 reported some concerns. Gomez et al. 48 and Walker et al. 60 were found to have a high risk. Most of the trials have good randomization processes, minimal missing data and acceptable measurement of outcome. However, deviation from intended intervention was a potential source of bias for all trials except the Brocklehurst et al. The summary of risk-of-bias assessment for quasi-experimental studies is placed in Figure 2(d).
Findings from the systematic review
Labour and FHRM, maternal uterine contractions and vitals were found to be potentially beneficial interventions to reduce stillbirths (Ekblom et al., Gwako et al., Maaloe et al., Kc et al., Byaruhanga et al., Agena and Modiba). Quality improvement collaboratives with training and mentoring of providers on intrapartum care and protocols also were seen in reducing stillbirths across different settings (Walker et al., Ekanga et al., Shikuku et al., Maaloe et al., Gomez et al., Brown et al.). Another intervention that was seen to be pointing towards reducing stillbirths was improvement of intrapartum clinical practice and standard of care through criteria-based audits or locally tailored clinical guidelines (Nolens et al., Mgaya et al.). The use of computerized interpretation of foetal heart rate during labour with clinical decision support or software in clinical decision making during the intrapartum period provided equivocal results or a non-significant reduction of stillbirths (Brocklehurst et al,. Usmanova et al.). Interventions such as oxytocin use and clinical referral during labour were seen to increase the risk of stillbirths (Litorp et al., Patel et al.). The overall characteristics of the included studies are captured in Table 1.
Characteristics of the included studies.
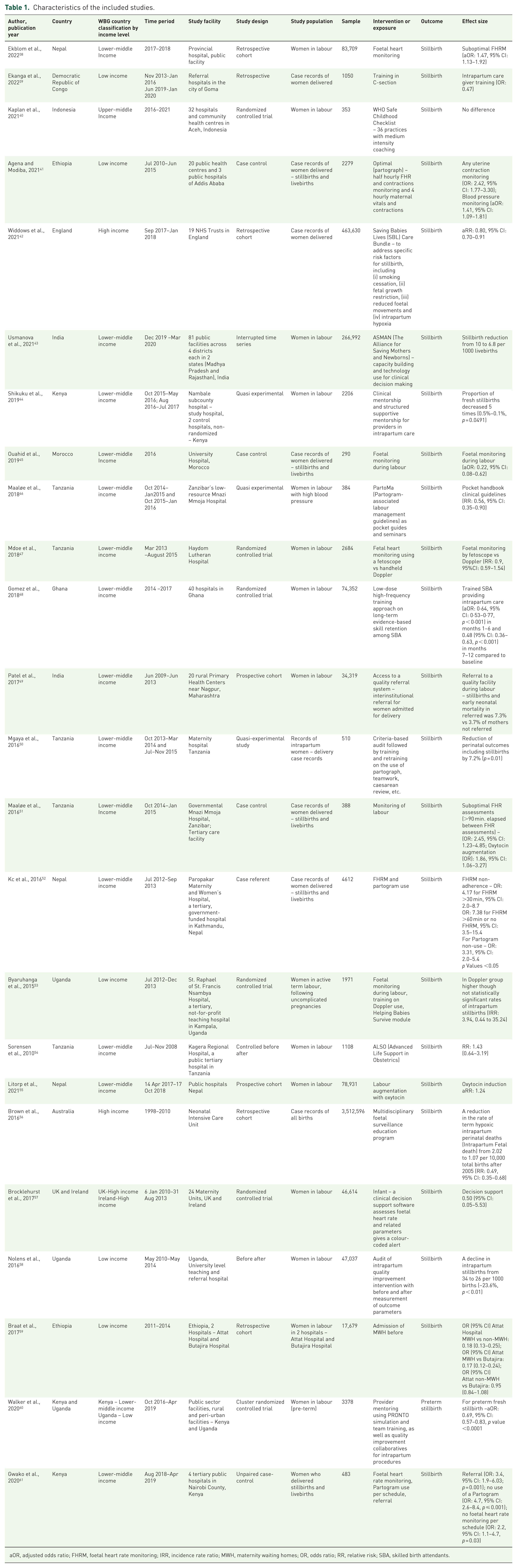
aOR, adjusted odds ratio; FHRM, foetal heart rate monitoring; IRR, incidence rate ratio; MWH, maternity waiting homes; OR, odds ratio; RR, relative risk; SBA, skilled birth attendants.
Meta-analysis findings
A meta-analysis was carried out with 17 of the 24 studies to unfold the individual and pooled estimates of OR of stillbirths for women receiving any quality intrapartum care intervention. The overall risk of bias of the 17 studies was variable; 9 studies were at high risk of bias, 3 at moderate risk and 5 at low risk.
Forest plots
The forest plot below presents meta-analysis of 17 studies (Figure 3) evaluating the effect of various intrapartum interventions on the risk of stillbirth, with results expressed as OR and 95% CIs. The pooled estimate yielded an OR of 0.25 (95% CI: 0.06–1.06), suggesting a trend towards reduced odds of stillbirth with intrapartum interventions; however, the CI encapsulates the line of no effect, indicating the finding is not statistically significant.

Forest plot displaying odds of stillbirth for women who received intrapartum quality improvement interventions.
Most individual studies showed ORs less than 1, pointing towards a decreased risk of stillbirth in the intervention/exposed groups. For example, Agena and Modiba 41 (OR = 0.41 (0.36–0.47)), Maaløe et al. 46 (OR = 0.13 (0.09–0.20)) and Gwako et al. 61 (OR = 0.02 (0.01–0.03)) show strong and statistically significant protective effects, implying that labour monitoring or FHRM or similar interventions reduced stillbirths substantially.
Conversely, Byaruhanga et al. 53 (OR = 3.96) assessing FHRM with Doppler and Patel et al. 49 (OR = 2.07) assessing intrapartum referral to higher centres reported increased odds of stillbirth, although these estimates had wide CIs, reflecting greater uncertainty.
Gomez et al. 48 reported a significant protective effect with an OR of 0.65 (0.55–0.76), and Ekblom et al. 38 demonstrated an OR of 0.75 (0.65–0.86), also indicating significant benefit. Strong protective effects were also observed in Braat et al. 59 (OR = 0.17 (0.12–0.24)) and Walker et al. 60 (OR = 0.58 (0.45–0.74)).
The meta-analysis showed substantial heterogeneity, with a Q statistic (χ2 = 1052.98, p < 0.001) and an I2 of 98.5%, indicating that nearly all variability between studies was due to true differences in study effects rather than chance. This heterogeneity likely arose from differences in study design (case control vs prospective), populations, intervention types and settings. The extremely high I2 values indicate substantial between-study variability and that pooled estimates should therefore be interpreted as an average effect across diverse contexts, rather than a precise summary effect.
To summarize, while the pooled estimate suggested a possible benefit of interventions in reducing stillbirth, the moderate to high risk of bias of the majority of studies, the high heterogeneity and wide CIs necessitated cautious interpretation. Further analyses, including subgroup, sensitivity analyses and meta-regression, were done to explore sources of variability and strengthen conclusions. Accordingly, the validity of pooling under such heterogeneity is limited, and results should be interpreted in conjunction with subgroup, sensitivity and meta-regression analyses.
Sensitivity analysis
The sensitivity analysis (Figure 4) indicates that no single study has a dominant influence on the overall pooled estimate or heterogeneity. The overall OR remains stable (~0.25) regardless of which study is omitted, except for the exclusion of Kc et al. 52 and all CIs include 1, indicating a lack of statistical significance. High heterogeneity persists across all leave-one-out scenarios (I2 ≈ 98%), suggesting that variability among studies is widespread and not driven by any one study. These findings suggest the overall result is robust but inconclusive; therefore, we further explored the analysis through meta-regression and subgroup analysis, which is needed to understand the sources of heterogeneity.

Sensitivity analysis.
Subgroup analysis
To address the objective of identifying sources of heterogeneity and differential effectiveness, subgroup analyses were conducted based on intervention type, choosing studies from each type of intervention. The subgroup analysis for type of intervention (Figure 5) revealed notable variation in the effectiveness of different intrapartum interventions in reducing stillbirths. Labour/foetal heart monitoring interventions (Type A) showed a statistically significant reduction in risk with a pooled OR of 0.75 (0.66–0.86) and low heterogeneity (I2 = 9.6%), indicating consistent benefit across the three studies, which had only low to some concerns of risk of bias. MWH, where women in late pregnancy have early labour before being transported to a health facility for delivery or in case of complications (Type F), also demonstrated a strong protective effect (OR = 0.17 (0.12–0.24)) by Braat et al., which also had a low risk of bias methodologically. In contrast, capacity building interventions (Type B) had a pooled OR of 0.67 (0.28–1.59) but with very high heterogeneity (I2 = 94%) and high risk of bias in the studies, suggesting inconsistent effects across studies. Interventions focused on improving standards of care (Type C) showed a trend towards benefit (OR = 0.64 (0.40–1.01)), though not statistically significant; the studies fell in the high risk of bias category but displayed a relatively less heterogeneity of 65%. The intrapartum referral intervention (Type E) was associated with a significantly increased risk (OR = 2.07 (1.69–2.54)), likely reflecting the inclusion of high-risk cases; however, the study had a very high risk of bias. Technology-based intervention (Type D) yielded an inconclusive result due to limited data. Overall, the test for subgroup differences was highly significant (p < 0.0001), confirming that the type of intervention substantially influences outcomes.
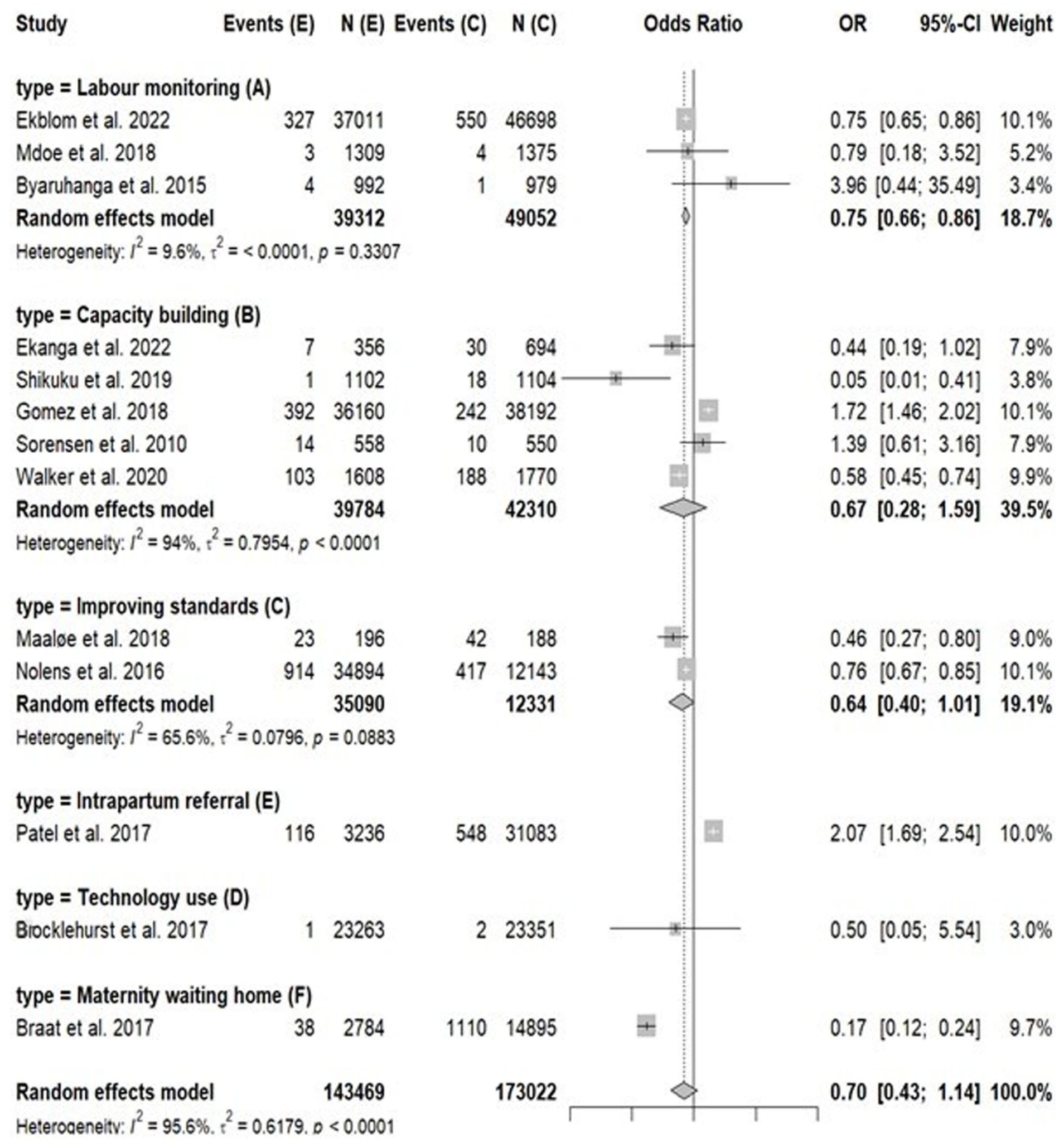
Subgroup analysis for the type of intervention.
The subgroup analysis (Figure 6) based on country income classification (World Bank 2024) revealed differential effects of interventions across income groups

Subgroup analysis for study settings (income of countries).
In low-income countries, the pooled OR indicated a protective but statistically non-significant effect (OR = 0.52, 95% CI: 0.19–1.43), accompanied by high heterogeneity (I2 = 95.9%). In contrast, lower-middle-income countries demonstrated a modest but non-significant reduction in stillbirths (OR = 0.84, 95% CI: 0.54–1.29, I2 = 94.2%). The high-income subgroup had only a single study (Brocklehurst et al. 57 ), which showed a wide CI (OR = 0.50, 95% CI: 0.05–5.54), limiting interpretation. The test for subgroup differences was not statistically significant (χ2 = 0.85, df = 2, p = 0.6550), suggesting that the intervention effect did not significantly vary across income categories. Overall, while some country groups showed trends towards benefit, the high heterogeneity and overlapping CIs indicate caution in attributing differential effectiveness solely to income classification.
Meta-regression
Meta-regression (Figure 7) using year of publication as a covariate suggests a mild decreasing trend in the odds of stillbirths over time. However, this relationship is not statistically significant (p = 0.305), and only 1.85% of the between-study heterogeneity is explained by year. Substantial residual heterogeneity (I2 = 96.65%) remains unexplained, indicating that other covariates or study-level factors (e.g. type of intervention, setting, population) may be more relevant drivers of heterogeneity.

Meta-regression for year of publication.
The evaluation of publication bias in this meta-analysis for 12 studies was conducted using both a funnel plot (Figure 8) and Egger’s regression test. The funnel plot appears visually symmetrical, suggesting no evident publication bias. This visual impression is supported by the results of Egger’s regression test for funnel plot asymmetry, which yielded a non-significant result (t = −0.33, df = 10, p = 0.7503). The bias estimate was −0.6459 (SE = 1.9741), indicating that there was not statistically significant small-study effect. Additionally, the multiplicative residual heterogeneity variance was relatively high (τ2 = 18.8391), which reflected underlying between-study variability. Taken together, both the funnel plot and Egger’s regression test suggested a low likelihood of publication bias influencing the meta-analysis results.

Funnel plot of effect estimates for stillbirths and intrapartum interventions.
However, the very high between-study heterogeneity (τ2 = 18.84, I2 > 95%) limits the reliability of funnel plot symmetry and Egger’s regression in this context. Therefore, despite the absence of statistically significant small-study effects, the assessment of publication bias should be interpreted with caution.
Discussion
This systematic review and meta-analysis assessed the impact of intrapartum quality improvement packages and related facility-based clinical, technological and health system interventions at maternity units of the health facilities on stillbirths. The findings indicated that intrapartum quality improvement interventions could be beneficial in reducing stillbirths. Among the various approaches assessed, comprehensive QI programmes encompassing fetal and labour monitoring, coupled with structured training and ongoing support for healthcare providers and birth attendants, appeared to be yielding the most substantial benefits. In addition, improving clinical practice and standards of care, and the use of technology for clinical decision making, seemed to have had an impact on stillbirth reduction. This highlights the critical role of multifaceted interventions that not only improve clinical monitoring and practice but also strengthen the capacity and responsiveness of healthcare teams during labour and delivery. Our meta-analysis demonstrated significant gains with FHRM, which many studies have echoed.62–64 However, efficient FHRM did not always translate to reduced stillbirths. In the study by Byraruhanga et al., 53 it was seen that FHRM by handheld Doppler led to increased identification of foetal heart rate abnormalities but reported high SBRs in comparison to the standard partograph plus Pinard stethoscope method. Many factors, including insufficient sample size, could have led to this observation as explained by the authors, but it is certain that FHRM had to be backed up by a responsive action, including delivery by caesarean section, to have an impact on stillbirth reduction. A study also reported that in women receiving continuous FHRM, stillbirth risk did not vary between those who received software-based decision support in analysing cardiotocographs and who did not receive technology-based decision support (Brocklehurst et al. 57 ).
Capacity building of healthcare providers in intrapartum care using a mix of training, mentorship and monitoring through quality improvement initiatives has worked in many settings, especially in African countries. A capacity building and clinical mentorship model is a low-dose high-frequency approach, which includes on-site trainings of providers in short sessions followed by repetitive practice under supervision of peer coordinators, and on-site mentoring with telephonic follow-ups by trainers. The studies in our meta-analysis strongly endorsed the role of skilled and mentored intrapartum caregivers in the significant reduction of stillbirth (Ekanga et al., 39 Shikuku et al., 44 Gomez et al., 48 Walker et al. 60 ). Although the findings find reflections in literature about the role of capacity building of providers, in achieving positive outcomes,65–67 the methodological limitations of our studies weaken the strength and certainty of the evidence. Another systematic review investigating the impact of training on cardiotocography on neonatal outcomes reported that the evidence was limited, inconsistent and of low quality. 68
The initial step of a QI process is an audit, which identifies opportunities of improvement, especially where compliance with identified standards is a prerequisite for high quality care. 69 Clinical audit of intrapartum care quality indicators is a retrospective analysis of routinely documented medical records to detect areas of poor quality and to institute quality improvement measures that will prevent further harm. 65 Audits identify systemic gaps in adherence to standard protocols, offering guidance for improvement of clinical practice and standards. 67 Our systematic review and meta-analysis indicated that improvement of clinical practice and standards may play a role in reducing stillbirths (Maaloe et al., 46 Nolens et al. 58 ); however, the high risk of bias of the two studies makes the conclusion guarded.
Among the multiple modifiable health system risk factors, having to wait more than 10 min to receive care after reaching a facility was found to be associated with higher odds of stillbirth. 70 Hence, we included a study which evaluated the role of MWH, which were made available to antenatal women in resource constricted areas to stay and wait for labour. MWH is not essentially an intrapartum intervention, but we decided to include the study (Braat et al. 59 ) since the MWH of the study was located in the hospital where the women delivered. The study was strongly in favour of MWH for stillbirth reduction, but in current evidence, despite there being a general agreement of the beneficial impact of MWH in areas of poor access and other challenges, they have not been incorporated as a routine strategy for want of quality evidence.71,72
WHO underlines that ‘timely identification, appropriate management and referral of women and newborns with complications during labour and childbirth are essential to prevent stillbirths and neonatal deaths’. 21 The study we included supported the necessity of timely referral and indicated that referral during labour would be associated with a higher risk of stillbirth and thus might end up as a risk factor (Patel et al. 49 ). The study had severe methodological limitations, and the finding was not statistically significant, the indication towards wrong timing of intrapartum referrals cannot be ignored.
Our review included studies across a wide range of healthcare settings and populations enhancing the generalizability of these findings and underscoring the universal relevance of QI initiatives. The diversity of settings – from low resource to more developed healthcare systems – demonstrated that QI efforts can be adapted successfully to different contexts, reinforcing their potential as key strategies to improve maternal and neonatal outcomes globally. The interventions seemed to help in the reduction of stillbirths in lower and lower-middle income countries, more markedly in low-income countries, but the non-significant findings, heterogeneity and moderate to high risk of bias of studies limited any sound conclusions.
The heterogeneity observed across included studies – in terms of study design, quality, intervention components and contextual factors – pointed to some limitations and areas for future research. Variability in methodological rigour and reporting standards introduced uncertainty into the pooled estimates and may have affected the comparability of results. Furthermore, contextual factors may play a role in the success of QI interventions. Contextual factors may include not only the organizational factors such as size, structure or performance but also those in the external political, regulatory, socio-cultural-professional and technological environments. 73
We acknowledge the importance of designing and tailoring QI strategies to the unique needs and resources of specific healthcare environments. Contextual adaptation not only has the potential to enhance the effectiveness of interventions but also promotes their sustainability and scalability. Future research should focus on identifying the core components of successful QI programs, understanding implementation barriers and evaluating long-term outcomes across diverse settings. Additionally, rigorous, well-designed studies with standardized outcome measures are needed to strengthen the evidence base and guide policymakers and practitioners in optimizing intrapartum care.
Strengths
Our key strength was the comprehensive search strategy involving literature search in multiple databases and studies published between 2004 and 2025, without geographic or language restrictions. Our final list of 17 studies in meta-analysis also included an article in the French language. Another strength was that the sample size was substantial (4,647,555 participants). The comprehensive inclusion of a diverse range of interventions in the intrapartum period provides a valuable, holistic overview of the field, guiding future research to focus more precisely on targeted interventions. A risk-of-bias assessment was conducted for all studies included in the systematic review and meta-analysis. Moreover, sensitivity analyses and subgroup analyses were performed to investigate potential sources of heterogeneity, thereby enhancing the robustness and rigour of the findings.
Limitations
The review also has several limitations which should be considered while interpreting the findings. The inclusion of multiple, heterogenous interventions such as foetal monitoring, mentoring, referral, technology for clinical decision support, audits, etc. has introduced a lot of complexity in synthesis of findings as the variations posed difficulties to isolate and clearly identify the most effective interventions for stillbirth reduction. Additionally, the heterogeneity induced by the included study designs (RCTs, quasi, cohort and case-control studies) also made direct comparisons and interpretations difficult. We also recognized that the variability of methodological rigour among the selected studies may have compromised the reliability of the overall findings. Almost three quarters of the studies had moderate to serious risk of bias which made interpretations and conclusions guarded. Fidelity of interventions was inconsistently reported and hence it was difficult to assess whether suboptimal delivery of interventions played a role. Also, the selected studies were conducted in diverse settings (countries and populations) which may limit the generalizability of findings to specific populations or regions. Additionally, some studies might have been missed, for example, our final list did not have a single study from the American continents, despite following all the standard procedures for conducting a comprehensive systematic review and meta-analysis.
Conclusion
This systematic review and meta-analysis assessed the impact of intrapartum quality improvement interventions on the reduction of stillbirths. A comprehensive search was conducted across multiple credible databases to identify studies examining QI strategies implemented during labour and delivery. The selected studies assessed strategies that encompassed fetal heart rate and labour monitoring, capacity building of healthcare providers, implementation of clinical care protocols and audit systems, integration of technological tools, referrals during labour and interventions of housing late pregnancy and early labouring women in MWH.
The findings indicated that certain intrapartum QI interventions could have a beneficial role in reducing stillbirths. Notably, comprehensive QI programs – characterized by fetal and labour monitoring, alongside the training and support of healthcare providers and birth attendants – demonstrated the most beneficial effects. The conclusion on the effectiveness of these quality improvement interventions has to be guarded because of the high heterogeneity in study design, methodological quality of the included studies and a poor understanding of the contextual factors of different settings.
Nevertheless, the inclusion of studies from diverse healthcare settings highlights the broad applicability and importance of QI efforts across varying contexts. The limitations notwithstanding, our review has brought out a comprehensive synthesis of a heterogenous group of intrapartum interventions for stillbirth outcomes and brings to sharp focus the critical methodological and implementation barriers which could be addressed in future research.
Supplemental Material
sj-docx-1-reh-10.1177_26334941261431235 – Supplemental material for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis
Supplemental material, sj-docx-1-reh-10.1177_26334941261431235 for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis by Preetha Gopinathan, Neha Lakshman, Himanshu Tolani, Vinay Tripathi, Sumant Swain, Eshika Bindal, Manish Prajapat and Jagdish Prasad in Therapeutic Advances in Reproductive Health
Supplemental Material
sj-docx-5-reh-10.1177_26334941261431235 – Supplemental material for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis
Supplemental material, sj-docx-5-reh-10.1177_26334941261431235 for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis by Preetha Gopinathan, Neha Lakshman, Himanshu Tolani, Vinay Tripathi, Sumant Swain, Eshika Bindal, Manish Prajapat and Jagdish Prasad in Therapeutic Advances in Reproductive Health
Supplemental Material
sj-xls-2-reh-10.1177_26334941261431235 – Supplemental material for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis
Supplemental material, sj-xls-2-reh-10.1177_26334941261431235 for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis by Preetha Gopinathan, Neha Lakshman, Himanshu Tolani, Vinay Tripathi, Sumant Swain, Eshika Bindal, Manish Prajapat and Jagdish Prasad in Therapeutic Advances in Reproductive Health
Supplemental Material
sj-xls-3-reh-10.1177_26334941261431235 – Supplemental material for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis
Supplemental material, sj-xls-3-reh-10.1177_26334941261431235 for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis by Preetha Gopinathan, Neha Lakshman, Himanshu Tolani, Vinay Tripathi, Sumant Swain, Eshika Bindal, Manish Prajapat and Jagdish Prasad in Therapeutic Advances in Reproductive Health
Supplemental Material
sj-xls-4-reh-10.1177_26334941261431235 – Supplemental material for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis
Supplemental material, sj-xls-4-reh-10.1177_26334941261431235 for Impact of intrapartum quality improvement interventions on stillbirths: a systematic review and meta-analysis by Preetha Gopinathan, Neha Lakshman, Himanshu Tolani, Vinay Tripathi, Sumant Swain, Eshika Bindal, Manish Prajapat and Jagdish Prasad in Therapeutic Advances in Reproductive Health
Footnotes
Acknowledgements
We express our gratitude to the staff of the library of the International Institute of Health Management Research, Delhi, for their assistance in developing the search strategy.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
