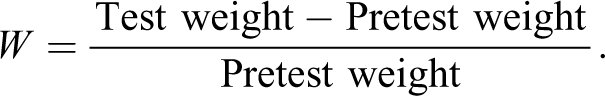Abstract
A novel epoxy resin (EP)/hollow glass microsphere modified (g-HGM) composite was successfully prepared. Studies showed that the water absorption rate of the g-HGMs/EP composite is lower than pure HGMs/EP and HGMs-KH550/EP composites, while the compressive strength of g-HGMs/EP composites could be increased. The enhanced interfacial adhesion between EP and g-HGMs played an important role to improve the compatibility of the two components. The g-HGMs show little effect on density (relative to HGMs) on the g-HGMs/EP composites, which can perform better than the HGMs/EP composites being used in marine environments. It was found that the optimal content of 4,4’-diphenylmethane diisocyanate in the epoxy component was 20 wt%.
Introduction
Hollow glass microspheres (HGMs) are composed of an external rigid glass layer and an internal gas phase, which endowed the composite materials with many unique properties such as lightweight, low thermal conductivity, and low dielectric constants as fillers. HGMs have larger surface area compared to similar solid particles and can be well dispersed in various matrixes. 1 –4 Based on these properties, HGMs are particularly attractive fillers in rubbers, plastics, and coatings. Hollow glass particles filled with polymer composites, also called synthetic foams, have been widely used in aeronautical and marine materials because of their high porosity areas surrounded by thin and hard shells of hollow particles, resulting in a higher mechanical strength and modulus compared to the other types of foams. 5 –11 As early as 2002, China listed the development of deep-sea manned submersible as the National High-Tech Development Plan and began to independently develop the Jiaolong, whose maximum diving depth is 7062 m. These unique closed porous structures endow the synthetic foams with low moisture absorption and high thermal stability. Accordingly, these lightweight composites are considered promising materials for weight-sensitive components of aeronautical or marine equipment.
Epoxy resins (EPs) are widely used in various industrial fields such as adhesives, paints and potting materials, laminates, encapsulants for semiconductors, and insulating materials for electrical devices. 12 –16 EPs have shown many advantages such as good adhesion to many substrates, low shrinkage on curing, solvent/chemical resistance, high tensile strength and modulus, good electrical properties, and excellent dimensional stability, which are important for the above applications. HGMs/EP synthetic foams have been prepared and used in a wide range of marine applications. However, HGMs/EP synthetic foams tend to absorb water. The mechanism of water absorption in HGMs-reinforced composites involves the diffusion of water molecules within the microvoids between polymer chains, capillary transport into voids and defects in the interface between the HGMs and the matrix, and transport of microcracks in the matrix. 17 –22 Water absorption property is also related to the porosity in the resin matrixes, relaxation of the resin upon the moisture and high temperatures, and binding of water molecules by the molecular structure of the resin.
In past studies, most of the studies have been directed to the problem of water adsorption in resins. The EPs used in HGMs/EP synthetic foams generally absorb water. This water absorption can be attributed primarily to the hygroscopic, polar nature of the specific functional groups of the cured EP. The interface between HGMs and EP has been studied only rarely. To obtain good dispersion of the HGMs/EP synthetic foams and to create better compatibility between the HGMs and the EP, it has been recommended to use different coupling agents such as trialkoxysilanes for surface modification on the HGMs. Appropriate surface modification of the HGMs not only results in better dispersion and compatibility of the EP matrix but also results in chemical and physical interactions with the polymer matrix. This ensures a durable chemical bond between the two incompatible phases. 23 –25 In addition, to make the hollow glass microsphere modified (g-HGM) participate in the curing process with EP, we decided to graft the curing agent onto the surface of HGMs. MDI is used to graft the curing agent onto the surface of HGMs, because MDI has symmetrical NCO structure, and it can react with –NH in silane coupling agent and curing agent, respectively, to form stable chemical structure.
In this work, in order to realize an excellent interfacial binding and a good dispersion between HGMs and EP, HGMs grafted with an EP curing agent (g-HGMs) and EP composites containing g-HGMs at various concentrations were prepared. The influence of the incorporation of g-HGMs into EP composites on morphology, mechanical properties, and water absorption was also investigated.
Experiment
Materials
Diglycidyl ether of bisphenol A-based EP E51 with an epoxy value of nearly 0.50–0.54 mol/100 g was purchased from Bluestar New Chemical Materials Co., Ltd (Wuxi, China). Low-molecular-weight polyamide 651 with an amine value of approximately 380–420 mg·KOH−1·g−1 (Chemtura, Shanghai, Co., Ltd, China) was used as the curing agent. HGMs composed of sodium borosilicate (grade HGMs-iM16 K, particle size 10–25 µm, density 0.46 g·cm−3) were obtained from 3M Corporation (Shanghai, China). Sodium hydroxide (NaOH) solution, (3-aminopropyl)triethoxysilane (KH550), n-propylamine (Aldrich), and 4,4’-diphenylmethane diisocyanate (MDI) were purchased from Sigma-Aldrich Co. Ltd. (Beijing, China). Other chemicals and solvents were purchased from Tianjin Chemical Co., Ltd (China).
Preparation of g-HGMs
The HGMs (5.0 g) were firstly washed with ethanol and dried at 80°C in a vacuum oven for 24 h. Then, the HGMs were added to an aqueous solution (250 mL) of NaOH (0.3 mol·L−1), and the obtained solution was stirred at 90°C for 2 h to form hydroxyl groups on the surfaces of the HGMs. The obtained HGMs were washed and filtered several times with water and dried in a vacuum oven at 60°C for 24 h; the obtained HGMs were dispersed in cyclohexane (100 mL), then (3-aminopropyl)triethoxysilane (0.5 g) as a reactant and n-propylamine (0.1 g) as a catalyst were added to the suspension solution, and the HGMs (HGMs-KH550) were terminated with an amine group. The solution was heated at 80°C for 2 h. The resulting solution was filtered, washed in ethanol, and dried in a vacuum oven at 50°C for 12 h. Figure 1 shows the preparation route of g-HGMs. Amine-terminated HGMs (5.0 g) were suspended in dimethylformamide (DMF, 200 mL) and reacted with MDI at 80°C for 2 h. To explore the influence of MDI content on the grafting effect of curing agent, different contents of MDI were selected for comparison. The mass ratio of MDI to hollow glass beads is set as 5%, 10%, 15%, 20%, and 25%, respectively. Then the low-molecular-weight polyamide 651 is added to the reaction mixture for 2 h at 80°C. The product was removed by filtration with a membrane filter, washed several times with DMF to remove residual monomer, and dried in a vacuum oven at 60°C for 24 h.

Schematic preparation for the hollow glass microspheres grafted with the curing agent of epoxy.
Preparation of g-HGMs/EP composites
The EP E51 and polyamide curing agent 651 were uniformly mixed at a mass ratio of 100:43.4. Then, the original or modified HGMs were added to the mixture at a mass ratio of 12, 18, 24, 30, and 36 wt%, and 30% filler was selected to compare different grafting conditions. After mixing uniformly, the mixture was placed in a vacuum drying oven to remove bubbles. Next, the mixture was poured into the mold for water absorption, compressive strength, tensile strength, and bending strength, respectively. The internal dimensions of water absorption molding mold was ϕ40 ×4 mm2. Compression strength molding mold was made according to the standard GB/T 1041-2008 (ISO 604:2002, IDT). Tensile strength molding mold was made according to the standard GB/T 528-2009 (ISO 37:2005, IDT). Bending strength molding mold was made according to the standard GB/T 8812.2-2007 (ISO 1209-2:2004, IDT). Finally, these samples were placed at room temperature for 12 h and then dried in a vacuum oven at 80°C for 4 h.
Characterization
The structure of g-HGMs was confirmed by Fourier transform infrared (FTIR) spectroscopy. FTIR spectra of the product were recorded by Spectrum 100 FT-IR Spectrometer (Perkin-Elmer, Waltham, Massachusetts, USA). IR spectra were collected after 32 scans at 4 cm−1 resolution in the 4000–500 cm−1 region in attenuated total reflectance mode. Thermogravimetric analysis (TGA, TGA-Q50, TA Instruments-Waters China Limited, Hong Kong, China) was performed on the samples to detect the weight fraction of functionalized HGMs. The temperature range is from room temperature to 820°C, and the heating rate is 20°C·min−1. The microscopic morphology of the HGMs was observed by scanning electron microscopy (SEM, XL30 ESEM, Philips, the Netherlands). The density of g-HGMs/EP composites was measured by immersion method.
Water absorption ratio
To confirm the water absorption ratios of the g-HGMs/EP composites, the composites were soaked in a pressure tank (10 MPa) at room temperature for 10 days. In the initial stage, the samples were weighed every day, and in the later stage, the samples were weighed every 2 days after the water absorption data were stable. The test of water absorption was carried out according to GBT 1034-2008 (ISO 62:2008, IDT). The water absorption ratio (W) is defined as:
Compressive strength
Compressive strength testing was carried out using an Instron-3365 universal testing machine (Instron Co. Ltd., Shanghai, China) at room temperature and performed at a crosshead displacement rate of 2 mm·s−1.
Bending strength
The three-point bending test was carried out by Instron universal tensile machine. The sample width was 10 mm, the thickness was 2 mm, the span was 60 mm, and the rate was 2 mm·min−1.
Tensile strength test
The length, width, and thickness of the sample were measured with vernier caliper and then input into the test software. The tensile rate was set as 5 mm·min−1, and the tensile properties of the sample were recorded. Five samples were tested for each data.
Results and discussion
Characteristics of g-HGMs
The formation of g-HGMs was analyzed by FTIR spectroscopy. Figure 2(a) shows the FTIR spectra of g-HGMs, pristine HGMs, and HGMs-KH550. The spectrum of pristine HGMs showed O–H groups at 3400–3500 cm−1. When HGMs were treated with silane coupling agent, the characteristic peaks of –CH2 appeared at 2851 and 2920 cm−1, which indicated that KH550 was successfully grafted onto the surface of HGMs. 26 When HGMs-KH550 was treated with MDI, a characteristic peak of –NCO appeared at 2270 cm−1, which indicated that MDI was successfully modified on the surface of HGMs.

(a) FTIR spectra of HGMs-MDI, pristine HGMs and HGMs-KH550; (b) TGA thermograms of pristine HGMs, HGMs-KH550, and g-HGMs; (c) SEM images of pristine HGMs, HGMs-KH550, HGMs-MDI, and g-HGMs. FTIR: Fourier transform infrared; HGM: hollow glass microsphere; MDI: 4,4’-diphenylmethane diisocyanate; TGA: thermogravimetric analysis; SEM: scanning electron microscopy; g-HGM: hollow glass microsphere modified.
TGA curves for the pristine HGMs, HGMs-KH550, and g-HGMs are shown in Figure 2(b). No weight loss was detected for the original HGMs, the weight loss for the HGMs-KH550 was 3.81%, which is mainly due to the decomposition of KH550 at high temperature, indicating the successful grafting of KH550 onto HGMs. While a weight loss of about 17.82% was observed for the g-HGMs. The increase of weight loss is attributed to the thermal decomposition of MDI and curing agent at high temperature, which indicates the successful grafting of MDI and curing agent.
Figure 2(c) shows SEM images of the virgin HGMs, HGMs-KH550, HGMs-MDI, and g-HGMs. The untreated HGMs had the appearance of a smooth and clean surface. In the formation of aminopropyl-terminated HGMs (HGMs-KH550) and NCO-terminated HGMs (HGMs-MDI), the surfaces of the original HGMs become gradually rough. When HGM-MDI is reacted with the low-molecular-weight polyamide 651, the surface of the HGM is completely encapsulated by the grafted low-molecular-weight polyamide 651 (g-HGM). These results indicated that g-HGMs were successfully prepared.
Figure 3(a) shows the TGA curves of the HGMs/EP and g-HGMs/EP composites. The weight loss of HGMs/EP was 88.3%, which is due to residues in the HGMs/EP, such as minerals. The weight loss of g-HGMs/EP was 80.4%, which was attributed to the incorporation of g-HGMs into the matrix. This result demonstrates that the introduction of g-HGMs can greatly improve the thermal stability of the obtained g-HGMs/EP composites. Therefore, the thermal weight loss rate of the composite decreased. 27

(a) TGA thermograms of the g-HGMs/EP and HGMs/EP (20% MDI). (b) Changes in density of HGMs/EP composites containing various loadings of HGMs. TGA: thermogravimetric analysis; EP: epoxy resin; HGM: hollow glass microsphere; MDI: 4,4’-diphenylmethane diisocyanate; g-HGM: hollow glass microsphere modified.
An important property of HGMs/EP synthetic foams is the low density. As shown in Figure 3(b), the density of the HGMs/EP complexes decreases with increasing concentration of HGMs. The density of the g-HGMs/EP complex is not affected by the g-HGMs relative to the HGMs/EP complex, which is beneficial to the application of g-HGMs/EP in marine environment.
It is well-known that the properties of composite materials strongly depend on the interaction between filler and resin matrixes. To investigate this interaction, the microstructure of the composite material, including the dispersion of g-HGMs and the state of the interface between g-HGMs/EP, was observed by SEM. Figure 4 shows the distribution of HGMs in the composite. The pristine HGMs are unevenly distributed in the EP with little interfacial interaction with the matrix (Figure 4(a), (c), and (e)), while the g-HGMs are uniformly dispersed in the EP and sufficiently covered with the matrix (Figure 4(b), (d), and (f)). These results indicate that the g-HGMs have higher interfacial adhesion to the substrate than the original HGMs. 28,29 This is because the amino groups on the surface of g-HGMs and the epoxy groups in EP had a curing cross-linking reaction, which improved the adhesion between the resin and glass beads and there are also hydrogen bonds interaction between them.

Cross-sectional SEM observation of pristine HGMs/EP (a, c, and e) and g-HGMs/EP composites (b, d, and f) (20% MDI). SEM: scanning electron microscopy; HGM: hollow glass microsphere; EP: epoxy resin; MDI: 4,4’-diphenylmethane diisocyanate; g-HGM: hollow glass microsphere modified.
When submerged in a marine environment, all EPs will absorb some water, but it is important that the synthetic foam material has low water absorption in water to protect the material and avoid significant deterioration of its properties. In general, it has been found that the presence of HGM reduces the water absorption of the modified composite compared to pure EPs. This phenomenon can be attributed to the excellent barrier properties of the filler. 30 The presence of the high aspect ratio filler may create tortuous pathways for water molecules to diffuse into the composite. Figure 5(a) shows that the water absorption of the g-HGMs/EP composite is lower than that of the pure HGMs/EP and HGMs-KH550/EP composites. Since the pristine HGMs have weak adhesion to the matrix, a void is formed between the pristine HGMs and the EP in the HGMs/EP composite, therefore the water absorbing medium penetrates into the void in the HGMs/EP composite. On the contrary, the void in the g-HGMs/EP composite greatly decreases due to the higher compatibility between g-HGMs and EP matrixes. Figure 5(b) shows the water absorption of g-HGMs/EP composites with various MDI contents as a function of immersion time in water. It can be seen that all g-HGMs/EP composites exhibit the typical water absorption behavior of polymers following Fick’s law. 31,32 It can be seen that the water absorption of the g-HGMs/EP composite increases with time and reaches a stable value. It is estimated that the water absorption reaches equilibrium regardless of the g-HGMs content when the water absorption does not further change with the increase of the immersion time. The water absorption of the g-HGMs/EP composite decreases and then increases with increasing MDI content in the composites. This phenomenon can be attributed to the high affinity between the matrix and the filler and to the increase in the volume ratio of g-HGMs per unit volume of the matrix. The water absorption is lowest when the MDI concentration is 20%. The adhesion between g-HGMs and EP is very stable due to hydrogen bonds and covalent bonding between amino groups on the surface of g-HGMs and epoxy groups in EP.

(a) Water absorption curves of HGMs/EP composites with different HGMs. (b) The g-HGMs/EP composites with difference of concentrations of MDI. (c) Compressive strength of the HGMs/EP and g-HGMs/EP composites containing various contents of HGMs and g-HGMS, respectively. (d) Compressive strength of the g-HGMs/EP composites containing various concentrations of MDI. (e) Tensile strength of the g-HGMs/EP composites containing various concentrations of MDI. (f) Flexural strength of the g-HGMs/EP composites containing various concentrations of MDI. HGM: hollow glass microsphere; EP: epoxy resin; MDI: 4,4’-diphenylmethane diisocyanate; g-HGM: hollow glass microsphere modified.
Figure 5(c) shows the compressive strength of HGMs/EP and g-HGMs/EP composites with different filler contents. It is clear that the compressive strength decreases with increasing content of HGMs. The results show that the compressive strength of the HGMs is lower than that of EP. However, the g-HGMs/EP composite material tends to have a low compressive strength. This is because the interfacial adhesion between the matrix and the HGMs is strong. The g-HGMs have good compatibility with EP. Figure 5(d), (e), and (f) illustrates the mechanical properties of g-HGMs/EP composites, including their compressive strength, tensile strength, and flexural strength in terms of MDI content. The mechanical properties of the g-HGMs/EP composites were also investigated. This indicates that MDI can effectively graft the curing agent onto HGM surface, and with the increase of MDI, more curing agents are grafted onto the HGMs surface, so that g-HGMs can participate in the curing reaction with EP and improve the binding force between them. This improvement in the mechanical properties of the g-HGMs/EP composite in terms of compressive strength can be attributed to the payload of the g-HGMs and the strong interfacial binding between the g-HGMs and the EP matrix. 33 However, when the MDI content is increased to above 25 wt%, the compressive strength is reduced. The reduction in compressive strength for high concentrations, such as 25 wt%, can be attributed to incomplete grafting of MDI onto the surfaces of the HGMs. Because the content of MDI has exceeded the limit of grafting on HGMs, 20 wt% is the best content for this experiment. The tensile strength and bending strength of g-HGMs also compound this result. As mentioned above, strong interfacial adhesion can be attributed to hydrogen bonding and covalent bonding between g-HGMs and EP. Another reason is the strong chain entanglement resulting from the increased surface area of the HGMs grafted by the low-molecular-weight polyamide 651. As the volume fraction of modified HGMs increases, the g-HGMs/EP composite has a higher surface area of the modified HGMs particles and therefore has better interfacial adhesion to the matrix.
Conclusions
A novel EP/g-HGMs composite material is successfully prepared. The chemical and physical properties of the novel g-HGMs/EP composite were investigated to evaluate their suitability for use as synthetic foams for subsea installations. The preparation of g-HGMs was confirmed by FTIR and TGA. The manufacturing steps of the modified HGMs and the dispersion of g-HGMs in the matrix were confirmed by SEM. The water absorption behavior of g-HGMs/EP composites with different concentrations of MDI in water reaches equilibrium when the volume does not change further with the immersion time. The water absorption of g-HGMs/EP compounds is generally lower than that of HGMs/EP compounds and is lowest at MDI concentrations of 20%. The compressive strength of the g-HGMs/EP composite is much higher than that of the HGMs/EP composite. The compressive strength increases with increasing MDI concentration, reaching a maximum at an MDI content of 20 wt%. This is because of the high interfacial adhesion between the filler and the matrix, that is, hydrogen bonding of HGMs-KH550 and g-HGMs grafted with low-molecular-weight polyamides 651, which can react with EP. The g-HGMs do not increase the density of the g-HGMs/EP composite, which performs better than HGMs/EP composites used in marine environments.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (21875054) and the Fundamental Research Funds of the Central University (3072020CF1005).