Abstract
A syntactic foam was prepared from an epoxy resin matrix and modified hollow glass microsphere fillers. Modification by silane coupling agents with different molecular structures was analyzed, and the optimal content of the silane coupling agent was determined. The results demonstrated that all silane coupling agents enhanced the adhesion between the hollow glass microspheres and epoxy resin matrix, resulting in enhanced water absorption, compressive performance, tensile performance, and bending performance compared to those prepared using unmodified hollow glass microspheres. Among silane coupling agents with different end groups, the one with a sulfhydryl end group exhibited optimal modification for hollow glass microspheres. Among the silane coupling agents with different backbone structures, the one with silanol groups exhibited the optimal modification of hollow glass microspheres. Additionally, the performance of the syntactic foams was optimal when 6% of the silanol-containing coupling agent was used. The results demonstrated that syntactic foams prepared with hollow glass microspheres modified by silane coupling agents exhibited improvements in water absorption, compressive performance, tensile performance, and bending performance, compared with those prepared using unmodified hollow glass microspheres. Among silane coupling agents with different end structures, the one with a sulfhydryl group as end group showed the best modification effect on hollow glass microspheres. The water absorption was 0.35%, the compressive strength was 62.15 MPa, the tensile strength was 40.15 MPa, and the bending strength was 53.17 MPa. Among silane coupling agents with different backbone structures, the one with silanol groupsbonds showed the best results. Its compressive strength was up to 64.15 MPa, the tensile strength was 35.47 MPa, and the bending strength was 53.99 MPa.
Introduction
Syntactic foams consist of hollow particles and a polymers matrix and are characterized by a low density, high strength, good erosion resistance, excellent machinability, reasonable waterproof performance, and thermal insulation. Hence, syntactic foams have been widely applied in aerospace, marine, shipbuilding, and construction industries.1–9 For syntactic foams, resins, polyurethane, and silicone are common matrix materials, while hollow glass microspheres, hollow carbon microspheres, and hollow ceramic microspheres are common fillers. Epoxy resins are organic polymers containing two or more epoxy groups, which have polar hydroxyl and ether bonds, which allow them to strongly adhere to other substances. Epoxy resins also have excellent mechanical properties, good shape stability thermal stability, etc. and are widely used to synthesize syntactic foams.10–16 Hollow glass microspheres are microscale lightweight materials. Their main component is borosilicate, which is a lightweight material with a large specific surface area, stable chemical properties, high specific strength, and acoustic and thermal insulation.17–20
Hollow glass microspheres were employed as fillers for syntactic foams; however, epoxy resins are organic materials, while hollow glass microspheres are inorganic materials. The significant difference in their polarities leads to poor interfacial bonding, which limits the performance of the resulting syntactic foams. To improve the interfacial bonding between polymers and inorganic fillers, surface modification of the inorganic fillers is required. Among various surfactants, silane coupling agents (Y-R-Si(OR)3) have been widely used that contain silicon functional groups (-Si(OR)3) and organic functional groups (-Y).
Silane coupling agents improve the performance of syntactic foams by a series of reactions. The -Si(OR)3 group in the silane coupling agent undergoes hydrolysis, followed by dehydration and condensation with hydroxyl groups on inorganic fillers. This bonds the silane coupling agent molecules to the inorganic filler surface, so that the surface of inorganic fillers carry -Y groups that can react with polymers. By bonding inorganic fillers with -Y groups on the surface and polymers, the bonding between inorganic fillers and polymers is improved.21–26 Hence, the effect of the silane coupling agent on the treatment of inorganic fillers depends on two factors. The first is the extent of reaction between -Si(OR)3 groups in the silane coupling agent after hydrolysis and inorganic fillers. The second is the extent of reaction between -Y groups in the silane coupling agent with the polymer molecular chain; however, there are few published reports that discuss the effect of different silane coupling agent structures on the treatment of inorganic fillers.
In order to maximize the lightweight performance of composite foam materials, the high volume fraction hollow glass microsphere is generally used to fill, forming a large number of substrates and hollow glass microsphere interface inside the material. The existence of a large number of interfaces provides favorable conditions for the versatility of composite foam materials, and the surface of hollow glass microspheres can serve as a load interface for other functional fillers. However, on the other hand, the weak interface between the substrate and the hollow glass microsphere can easily reduce the material mechanics. Therefore, there are two main research directions for the modification of hollow glass microsphere surface, first, through deposition or graft functional fillers on hollow glass microsphere surface, such as silver, polyphenylamine, nickel, and graphene oxide, to achieve the performance of composite foam material infrared stealth,27–29 electromagnetic shielding,30,31 electromagnetic absorption,32,33 and so on. Second, the binding force with the substrate is enhanced by the surface modification of hollow glass microsphere. At present, the way to enhance the binding force of hollow glass microspheres with substrates is: First, the hollow glass microspheres are treated directly with acid and heat, to control the content of hydroxyl and metal oxides on the surface of the microspheres, which enhanced the binding force between the microspheres and the substrate. 34 Second, by using silane coupling agent modified hollow glass microspheres, the binding force between the substrate and the hollow glass microspheres is enhanced.35,36 Third, after grafting silane coupling agent on the surface of hollow glass microspheres, the active group that can react with the substrate is connected, and the compatibility and binding force of hollow glass microspheres and substrates are enhanced. 25 Among them, silane coupling agent treatment is the most commonly used to enhance the binding force of microspheres and substrates of the modification means, which can enhance the wear resistance of composite foam materials, heat resistance, extreme cold resistance, 37 water resistance, and so on. According to the current literature,23–26,35–37 the main use of 3-Aminopropyltriethoxysilane silane coupling agent modified hollow glass microspheres, for other types of silane coupling agents studied less. Therefore, it is necessary to carry out a study on the effect of different structural silane coupling agents on the performance of composite foams in order to obtain the best hollow glass microsphere treatment effect.
In this study, we synthesized syntactic foams with an epoxy resin matrix and hollow glass microsphere fillers. A silane coupling agent was used to modify the surface of the hollow glass microspheres. To explore the influence of the molecular structure on the modification effect, silane coupling agents with different end groups and different backbone structures were compared, and the content of the silane coupling agent was optimized. The modification effect of different silane coupling agents was evaluated by the microscopic morphology of the syntactic foams, water absorption, mechanical properties, and interfacial bonding of the epoxy resin and hollow glass microspheres.
Experimental
Materials
Information about IM16K hollow glass microspheres.

Sample preparation
Preparation of modified hollow glass microspheres
The flotation of hollow glass microspheres: Glass microspheres were placed in deionized water in a beaker and stirred thoroughly. The mixture was allowed to stand for 2 h. The upper layer of the floating microspheres was extracted and dried. Then, 10 g of the dried hollow glass microspheres was added to a 500 mL three-necked flask, followed by 300 mL of 0.3 M NaOH solution. This mixture was stirred and heated under reflux at 80°C for 1.5 h. It was then washed with distilled water until a pH of 7 was obtained. Water was removed by vacuum filtration, and the pretreated hollow glass microspheres were dried at 100°C for 12 h to obtain hydroxylated hollow glass microspheres. Sample (10 g) was added to a 500 mL flask, followed by 300 mL of ethanol and water mixed in a volume ratio of 1:3. Then, 1 g of silane coupling agent was added. The above mixture was stirred at 80°C for 2 h. After the reaction, the microspheres were washed with ethanol and filtered under reduced pressure to remove the residual coupling agent from the surface of the glass microspheres. After filtration, the hollow glass microspheres were placed in a drying oven at 100°C for 12 h to obtain modified hollow glass microspheres.
Preparation of syntactic foams
In this study, syntactic foams were prepared by mixing bisphenol A epoxy resin E51 (matrix resin) with polyamide curing agent 651 (epoxy curing agent) with hollow glass microspheres modified by silane coupling agents. The manufacturer’s recommended ratio of epoxy resin to curing agent is 100:40–60, the experiment selected 100:43.4, curing conditions selected the previous 25 curing conditions 80°C for 2 h.
Epoxy resin, curing agent, and modified hollow glass microspheres were mixed at a mass ratio of 100: 43.4: 30 and stirred, followed by decompression and defoaming in a vacuum oven. Then, the slurry was poured into a mold for curing at 80°C for 2 h.
Characterization
Modified hollow glass microspheres were evaluated by Fourier-transform infrared (FT-IR, Perkin Elmer Spectrum-680) spectroscopy. Dried IR-grade KBr was used with a sample/KBr ratio of 1: 100; a scanning range of 400–4500 cm−1, eight scans, and a sensitivity of 4 cm−1. The morphologies of hollow glass microspheres and cross-sections of syntactic foams were examined by scanning electron microscopy (FEI-SEM). The accelerating voltage was 15 kV, and gold powder was sprayed on the sample before analysis. Energy-dispersive spectroscopy (EDS) was used to characterize the surface elements and content changes of hollow glass microspheres before and after modification. The magnification was 5000×.
Water absorption
The water absorption of syntactic foams was tested according to GB/T2567-2008 (ISO 2602, IDT). The prepared samples were placed in a blast drying oven and then dried and weighed (M0). Then, the samples were subjected to hydrostatic pressure treatment at 10 MPa. After the reaction, the moisture on the sample surface was immediately wiped off, and the sample was weighed again (M1). The water absorption of syntactic foams was calculated by
Tensile strength
The tensile strength of syntactic foams was tested using an Instron Universal Testing System according to GB/T528-2009 (ISO 37:2005, IDT). The length, width, and thickness of samples were measured with a Vernier caliper. Data were input into the software, the tensile rate was set to 5 mm/min, and the tensile performance was recorded.
Bending strength
Bending strength tests were performed according to GB/T2567-2008 (ISO 2602, IDT). Three-point bending tests were performed on an Instron universal tensile machine with a sample width of 10 mm, a thickness of 2 mm, a span of 60 mm, and a speed of 2 mm/min.
Compressive strength
The uniaxial compressive strength of syntactic foams was measured according to the standard GB/T1041-2008 (ISO 604: 2002, IDT). The sample was cylindrical in shape, with a diameter of 8 mm and a height of 10 mm. Five samples were tested for each condition, and the test rate was 5 mm/min.
Results and discussion
Characterization of modified hollow glass microspheres
To discuss the influence of the silane coupling agent’s molecular structure on the modification effect of syntactic foams, three silane coupling agents with different end groups and three silane coupling agents with different backbone structures were selected in this article. The molecular structures are shown in Figure 1. Chemical structures of the silane coupling agents with different end groups ((a) KH550; (b) KH560; (c) KH591) and different backbone structures ((d) KH553; (e) KH593; (f) KH792).
Figure 2(a) and (b) correspond to the hollow glass microspheres modified by silane coupling agents with different end groups and backbone structures, respectively. There were significant changes in the infrared spectra of hollow glass microspheres modified by silane coupling agents. There were new peaks at 2920 and 2851 cm−1 for modified hollow glass microspheres, which corresponds to overlap of the antisymmetric stretching vibration absorption bands of methyl and methylene groups on the silane coupling agent grafted to the surface of the hollow glass microspheres.
38
The peak at 1496 cm−1 corresponded to the antisymmetric bending vibration of the methylene group and the symmetric bending vibration of the methylene group. The peak at 1638 cm−1 was attributed to the characteristic absorption peak of C=C in KH570. Hence, the silane coupling agent was grafted onto the surface of the hollow glass microspheres. Fourier-transform infrared spectra of hollow glass microspheres modified by silane coupling agents with (a) different end groups (a. blank; b. KH550; c. KH560; d. KH591) and (b) different backbone structures (a. blank; b. KH553; c. KH593; d. KH792).
Figure 3 shows the SEM morphology of hollow glass microspheres before and after modification by silane coupling agents with different end groups and silane coupling agents with different backbone structures. Figure 3(a) shows the untreated hollow glass microspheres, which had a smooth, flat surface. After being treated with NaOH, the surface of hollow glass microspheres became very rough because the microspheres were corroded by lye during surface hydroxylation. According to Figure 3(c)–(h), modification by silane coupling agents reduced the surface roughness because they were grafted to the glass microsphere surface. This also demonstrated the silane coupling agents modified the hollow glass microspheres. Additionally, the morphologies of modified hollow glass microspheres depended on the silane coupling agent, indicating that the grafting degree of the different silane coupling agents was different. This may affect subsequent mechanical performance and water absorption. Scanning electron microscopy images of hollow glass microspheres modified by silane coupling agents with different end groups ((a) Hollow glass microspheres IM16K; (b) IM16K-NaOH; (c) IM16K-KH550; (d) IM16K-KH560; and (e) IM16K-KH591) hollow glass microspheres modified by silane coupling agents with different backbone structures ((f) IM16K-KH553; (g) IM16K-KH593; and (h) IM16K-KH792).
Figure 4 shows surface element distributions of hollow glass microspheres modified by silane coupling agents with different end groups and different backbone structures, respectively. The results demonstrated that the main elements on the surface of unmodified hollow glass microspheres IM16K were O, Na, Si, and Ca. The O content increased significantly after surface hydroxylation. Meanwhile, after the hollow glass microspheres (IM16K) were modified with a silane coupling agent, C or N were observed, indicating that organic substances were present on the surface of the glass microspheres. The atomic ratio of O of unmodified hollow glass microspheres was 49.94%, while that of the treated glass microspheres dropped to 36%. This might be due to the reaction between silanol bonds (formed after hydrolysis of one end of the silane coupling agent) and hydroxyl groups on hollow glass microspheres. This also proved that the silane coupling agent was grafted onto the surface of the hollow glass microspheres. Figure 4(e) did not show an S peak, mainly because the S content in KH591 was low, and this peak overlapped with other strong peaks in the EDS spectrum. Energy-dispersive spectroscopy analysis of hollow glass microspheres modified by silane coupling agents with different end groups ((a) IM16 K; (b) IM16K-NaOH; (c) IM16K-KH550; (d) IM16K-KH560; and (e) IM16K-KH591) and different backbone structures ((f) IM16K-KH553; (g) IM16K-KH593; and (h) IM16K-KH792).
Water absorption properties of the syntactic foams
One of the applications of syntactic foams is to provide buoyancy to a variety of underwater equipment. The water absorption at a given hydrostatic pressure is an important indicator of the stability of a material, as the water pressure increases by 1 MPa for every 100 m depth. Figure 5(a) and (b) show the changes in the water absorption of syntactic foams modified by silane coupling agents with different end groups and silane coupling agents with different backbone structures at 10 MPa hydrostatic pressure, respectively. Upon increasing the holding time, the water absorption of all samples increased, the water absorption was the highest in the first 24 h, followed by a slow absorption until 150 h. After 150 h, water absorption equilibrium was almost reached. This phenomenon was consistent with the water absorption process of cured epoxy resins.
39
Water absorption of syntactic foams prepared using hollow glass microspheres modified by silane coupling agents with (a) different end groups ((a) KH591; (b) resin matrix; (c) KH550; (d) KH560; (e) NaOH; and (f) Unmodified) and (b) different backbone structures ((a) KH553; (b) resin matrix; (c) KH792; (d) KH593; (e) NaOH; and (f) unmodified).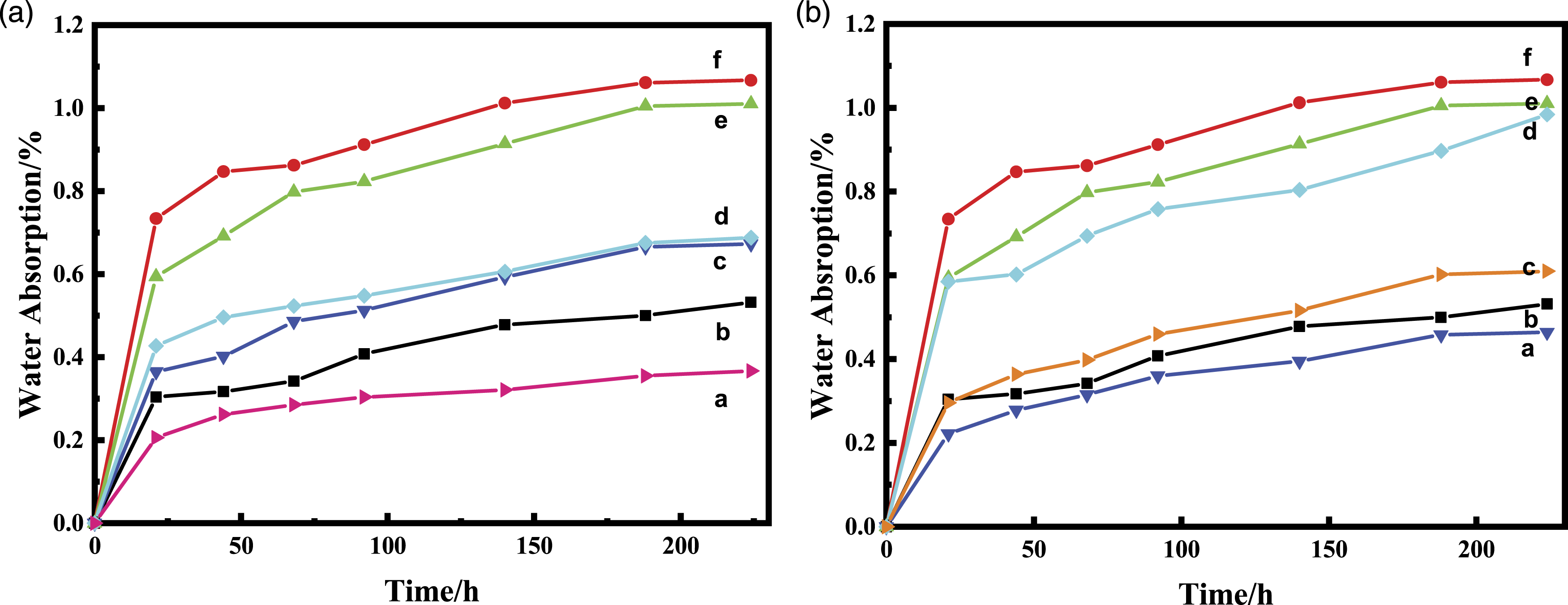
According to the theory, epoxy resins absorb water in three stages: rapid water absorption, slow water absorption, and water absorption equilibrium. The reason for this phenomenon is that at the initial stage of water absorption, water molecules enter the low-crosslink-density region of an epoxy resin, where the free volume is large, allowing water molecules to easily enter and reach equilibrium. This occurs over a relatively short period of time. Subsequently, water molecules continue to penetrate into the highly cross-linked region of the resin, where the free volume is small. This process lasts for a longer time until water absorption reaches equilibrium. As observed, the highest water absorption was obtained for syntactic foams prepared using unmodified hollow glass microspheres (equilibrium water absorption >1%), while the water absorption of syntactic foams prepared using hollow glass microspheres modified by silane coupling agents decreased drastically. The reason is that some hydroxyl groups on the hollow glass microspheres underwent a dehydration condensation reaction with the silanol groups in the silane coupling agent formed after hydrolysis, which reduced the chances of contact between hydroxyl groups on the microspheres and water molecules.
Among silane coupling agents with different end groups, amino groups, sulfhydryl groups, and epoxy groups of the silane coupling agents can react with epoxy or amino groups in the matrix resin, resulting in enhanced interfacial forces between the resin and microspheres, which reduced the water absorption. 40 The KH591 (end group = sulfhydryl group)-modified hollow glass microspheres had the optimal effect, with a water absorption of 0.35%, which is even lower than that of the pure epoxy. The second was the KH550 (end group = amino group)-modified sample, and the worst was the KH560 (end group = epoxy group)-modified sample.
Among silane coupling agents with different backbone structures, KH553 exhibited the optimal modification. Indeed, the water absorption of samples prepared using KH553-modified microspheres was even lower than that of the pure epoxy resin. The main reason is that KH553 contains silanol groups, while the other silane coupling agents have silicon functional groups with methoxy or ethoxy groups at the end. These groups must undergo hydrolysis before they can condense with hydroxyl groups on hollow glass microspheres or form hydrogen bonds. The degree of hydrolysis will limit the final effect of the coupling agent, while KH553 can react directly without hydrolysis; thus, it has the best modification effect.
In summary, the water absorption test results of pure resin and composite foam materials are consistent with the trend of water absorption rate of pure resin, 41 fiber-reinforced composite materials, 42 and composite foam materials 43 reported in literature, and are all rapid water absorption in the early stage and rising slowly in the later stage.
Mechanical performance of syntactic foams
Figure 6 shows the mechanical strength of different syntactic foams in this study. When unmodified hollow glass microspheres were added to the epoxy resin, the compressive strength, tensile strength, and bending strength of materials decreased. This is because the hollow glass microspheres reduced the homogeneity of the resin matrix and the bonding force between them was weak, which decreased the strength of the resulting syntactic foams.
44
After NaOH treatment, the strength of the syntactic foam also decreased because grooves were etched out, even though these grooves increased the contact area between the matrix resin and glass microspheres and increased the number of interaction points; however, the strength of the etched layer itself was very low, which decreased the overall material’s strength. (a) Compressive strengths, (c) tensile strengths, and (e) bending strengths of syntactic foams modified by silane coupling agents with different end groups ((a) pure epoxy; (b) unmodified; (c) NaOH; (d) KH550; (e) KH560; and (f) KH591). (b) Compressive strengths, (d) tensile strengths, and (f) bending strengths of syntactic foams modified by silane coupling agents with different backbone structures ((a) pure epoxy; (b) unmodified; (c) NaOH; (d) KH553; (e) KH593; and (f) KH792).
After modification by a silane coupling agent, the strength of the syntactic foams significantly improved because the silane coupling agents repaired the corrosion layer on the surface of the glass microspheres and enhanced the interfacial bonding between the resin matrix and hollow glass microspheres. This increased the syntactic foam’s strength. 45 As shown in Figure 6(a), (c), and (e), among the silane coupling agents with different end groups, KH591 was the most effective. The maximum compressive strength, tensile strength, and bending strength of the syntactic foams were obtained when using the KH591-modified hollow glass microspheres. Its compressive strength reached 62.15 MPa, its tensile strength reached 40.15 MPa, and its bending strength reached 53.17 MPa. This is because the sulfhydryl groups participated in the curing reactions with epoxy groups, and are more reactive than amino and epoxy groups.
The curing mechanism is: when sulfhydryl and epoxy groups are used alone, the reaction activity is very low, and it is difficult for the reaction to proceed; however, when tertiary amines are present in the curing system, the sulfhydryl groups first reacts with tertiary amines to generate thiol ions, which then react with epoxy groups. Their reactivity is several times that of polyamines. 46 Although tertiary amines are not present in the polyamide curing agent, the primary and secondary amines reacted with the epoxy resin to generate tertiary amines, which play a catalytic role. Hence, glass microspheres modified by the sulfhydryl group of silane coupling agents reacted more thoroughly with the epoxy resin, and their interaction was stronger, which improved the performance.
Among the silane coupling agents with different backbones, the most effective modification of the hollow glass microsphere was achieved by KH553, which has silanol groups at the end. The syntactic foams prepared from KH553-modified hollow glass microspheres showed the highest strength—its compressive strength was 64.15 MPa, its tensile strength was 35.47 MPa, and its bending strength was 51.99 MPa. This is because the hydroxyl groups of KH553 can react directly with hydroxyl groups on hollow glass microspheres, while the other three silane coupling agents must be hydrolyzed before forming silanol bonds. The degree of hydrolysis will limit the effect of the silane coupling agent.
Adhesion of hollow glass microspheres and epoxy resin
To discuss the degree of adhesion between the resin matrix and hollow glass microspheres in syntactic foams, the Turcsanyi equation was used, and the results are shown in Figure 7. The Turcsanyi equation
47
is a semi-empirical equation to obtain the compressive strengths of syntactic foams using the strength of the resin matrix and microspheres: Interfacial adhesion coefficients of different syntactic foams (a = unmodified; b = NaOH; c = KH550; d = KH560; e = KH591; f = KH553; g = KH593; and h = KH792).

As shown in Figure 7, modification by silane coupling agents increased the interfacial adhesion coefficients of the syntactic foams (>3 in most cases), demonstrating the positive effects of silane coupling agents on the interfacial bonding between hollow glass microspheres and epoxy resin. Additionally, the KH591-modified and KH553-modified syntactic foams exhibited maximum interfacial adhesion coefficients, indicating optimal modification by KH591 and KH553.
According to the literature, 10 the β value of composite foam materials made of hollow glass microspheres that have not been surface modified is less than 3. In this article, there are four kinds of silane coupling agent modified hollow glass microspheres made of composite foam materials β value is greater than 3, the modified hollow glass microspheres have a good enhancement effect on composite foam materials, of which KH553 and KH591 compared to the most commonly used KH550 (3-Aminopropyltriethoxysilane) has a better enhancement effect.
K591 and KH553 have the best mechanics and water resistance by comparing the properties of composite foam materials made of six different structural silane coupling agent modified hollow glass microspheres. This is because sulfhydryl group in KH591 has the highest reactive activity compared to other active groups, while the silicon hydroxyl in KH553 does not need hydrolysis and is directly dehydrated from hydroxyl on the surface of the hollow glass microsphere Reaction, with hollow glass microsphere reaction has the highest activity, which shows that silane coupling effect depends on the reaction activity of the active group at both ends of its molecules, in the selection of silane coupling agent needs to consider the silane coupling agent and the base and filler reaction activity. When the activity of the silane coupling agent and the substrate and filler reaction activity increases, the binding force of the substrate and filler interface is enhanced, which enhances the material mechanics. For composite foam materials, a stronger internal binding interface can also effectively prevent the water molecule extension interface from transmitting inward.
Effects of the silane coupling agent content on the performance of syntactic foams
The above discussion indicated that different silane coupling agents have different effects on the modification of hollow glass microspheres. To systematically investigate the effect of the silane coupling agents on the modification of hollow glass microspheres, the content of the silane coupling agent KH553 was investigated in this section. The mass ratio of KH553 to hollow glass microspheres was 4%, 6%, 8%, 10%, and 12%.
As shown in Figure 8(a), the water absorption of the syntactic foams prepared from 6%, 8%, and 12% KH553 treated hollow glass microspheres was lower than that of the pure epoxy, indicating that modification by KH553 using these three contents was effective. According to Figure 8, the compatibility of hollow glass microspheres and the matrix resin was optimal using 6% KH553. The reason is that the surface of the hollow glass microspheres grafted with a silane coupling agent has a critical point, before which the interactions between microspheres and matrix resin increased with the grafting rate. When the critical point is exceeded, the grafting rate of the silane coupling agent on the microsphere surface increases, that is, the number of layers coated by the silane coupling agent on the microsphere surface increases. This decreases the interfacial compactness and interfacial forces between the microspheres and matrix resin. Effect of KH553 content on (a) water absorption ((a) 6% KH553; (b) 12% KH553; (c) 8% KH553; (d) pure epoxy; (e) 10% KH553; (f) 4% KH553; (g) NaOH; and (h) unmodified), (b) compressive strength, (c) tensile strength, and (d) bending strength ((a) pure epoxy; (b) unmodified; (c) NaOH; (d) 4% KH553; (e) 6% KH553; (f) 8% KH553; (g) 10% KH553; and (h) 12% KH553) of syntactic foams.
Figure 8(b)–(d) are the compressive strengths, tensile strengths, and bending strengths of syntactic foams prepared using microspheres modified by different KH553 contents, respectively. The results demonstrated that the maximum compressive strength, tensile strength, and bending strength of the syntactic foams were obtained using 6% KH553. The compressive strength was 64.76 MPa, and the tensile strength was 45.05 MPa. The reason is similar to the water absorption in Figure 8(a). When the content of KH553 was 6%, its interfacial force with matrix resin was the strongest, and the strength of the material was also higher—a grafting rate that was too low or too high deteriorated the material’s properties.
Figure 9 shows the cross-sectional SEM images of syntactic foams prepared using unmodified and modified hollow glass microspheres. The hollow glass microspheres before and after modification showed greatly different interfaces after filling and preparing syntactic foams. There were obvious gaps between the unmodified glass microspheres and the resin matrix. This is because the physical and chemical properties of the untreated hollow glass microspheres and matrix resin are very different, and they are simply physical blends without chemical bonds. Thus, the compatibility is poor, which is also the reason for the high water absorption and poor mechanical performance of the syntactic foams. After filling the glass microspheres modified by silane coupling agents with epoxy resin, the interfacial gap disappeared. The compatibility between the microspheres and resin was significantly improved. The microspheres and resin were no longer simply a physical mixture, and contained chemical bonds, which made the internal structure of the material denser and the interactions stronger; thus, the water absorption of syntactic foams and the mechanical performance were improved. Scanning electron microscopy images of cross-sections of the syntactic foams ((a) blank-1000×; (b) blank-2000×; (c) blank-5000×; (d) KH553-1000×; (e) KH553-2000×; and (f) KH553-5000×).
Conclusions
In this article, the mechanical properties and water resistance of six different molecular structure silane coupling agent modified hollow glass microsphere preparation composite foam materials are compared, and it is found that KH550, KH560, KH553, and KH591 can effectively enhance the binding force of the substrate with the hollow glass microsphere, and improve the mechanical properties and water resistance of the composite foam material. Among them, KH553 and KH591 have better surface modification effect than the KH550, which is often used in hollow glass microsphere surface modification in literature, and the content of silane coupling agents was also optimized.
The results demonstrated that syntactic foams prepared with hollow glass microspheres modified by silane coupling agents showed improved water absorption, compressive performance, tensile performance, and bending performance compared with those prepared with unmodified hollow glass microspheres. Among the silane coupling agents with different end groups, KH591 showed the best modification effect on hollow glass microspheres. The water absorption of syntactic foams prepared by KH591 was only 0.35%, which was lower than the equilibrium water absorption of a pure epoxy resin. The compressive strength was 62.15 MPa, which was 47.6% higher than that of unmodified syntactic foams. The tensile strength was 40.15 MPa, which was 60% higher. The bending strength was 53.17 MPa, which was 40% higher. Among the silane coupling agents with different backbone structures, KH553 showed the best results, and its water absorption was lower than the equilibrium water absorption of a pure epoxy resin. Its compressive strength reached 64.15 MPa, its tensile strength was 35.47 MPa, and its bending strength was 53.99 MPa. When the content of KH553 was 6%, the corresponding syntactic foam had the best performance, with compressive and tensile strengths of 64.76% and 45.05 MPa, respectively.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article is supported by National Natural Science Foundation of China (21875054).
