Abstract
Increasing evidence documents turnover of the resting-state blood-oxygen-level dependent signal (TBOLD) as a key measure of local cortical brain status. Here we evaluated contemporaneous and lagged associations between TBOLD and cognitive function in 711 participants in the Human Connectome Project on Aging (HCP-A; 316 males and 395 females, age range 36-90 years). We found that TBOLD was negatively associated with Montreal Cognitive Assessment (MoCA) Total scores and with performance on 2 subscales, Delayed Recall and Visuospatial/Executive Function, controlling for sex, age, and handedness. This negative association was largely documented across brain areas and was significantly stronger in the left hemisphere compared to the right. In addition, analyses evaluating forward lagged crosscorrelations between TBOLD and cognitive performance demonstrated that TBOLD predicted decrements in future performance on MoCA Total score, Delayed Recall, and Visuospatial/Executive Function subscales, controlling for sex and handedness. Taken together, we found that increased TBOLD is associated with decreased cognitive performance contemporaneously and in the future. On the hypothesis that increased TBOLD is the outcome of neuroinflammatory processes, these findings provide a mechanism linking neuroinflammation with decreased cognitive performance.
Introduction
Numerous studies have documented the usefulness of the blood-oxygen-level dependent signal (BOLD) as a key measure of cortical brain function. In contrast to changes in task-related BOLD signal, which reflects the coupling of neural events with the hemodynamic response,1,2 resting-state BOLD (BOLD) reflects primarily neurovascular (mainly hemodynamic) factors. 3 The moment-to-moment change of resting-state BOLD, that is, its turnover (TBOLD), possesses several interesting properties, including heritability, 4 increase with age5,6 (particularly in individuals lacking certain immunoprotective human leukocyte antigen alleles), 5 and negative association with brain volume during the lifespan. 6 In a previous study, 6 we attributed the TBOLD increase with age to cumulative insults (ie, reactivation of latent human herpes viruses) causing low-level, chronic neuroinflammation—a hallmark of brain aging. 7 This hypothesis was supported by the finding of a significant negative correlation of TBOLD with brain volume during the lifespan, 6 thus linking the hypothesized chronic neuroinflammation (reflected in increased TBOLD) to decreased brain volume. Since (a) cognitive performance declines with age, 7 (b) chronic neuroinflammation has been postulated to underlie this decline, 8 and (c) TBOLD has been postulated to be a marker of chronic neuroinflammation,5,6 we investigated, in this study, the direct relation between TBOLD and cognitive performance in a large sample of Human Connectome data. In addition to evaluating the contemporaneous association between TBOLD and cognition, we also investigated the possibility that TBOLD would precede cognitive decline in the future. Indeed, various genetic, environmental, and lifestyle factors contribute to neurovascular dysfunction which, in turn, may contribute to cognitive decline and neurodegeneration. 9 Thus, subtle neurovascular alterations captured by TBOLD may reflect the influence of neurovascular insults and may presage cognitive decline; however, the association between TBOLD and cognitive function remains to be investigated. Here, then, we evaluated the association between TBOLD and cognitive function in 711 participants from the Human Connectome Project on Aging (HCP-A; age range 36-90 years). Specifically, we evaluated contemporaneous and lagged associations between TBOLD and performance on the Montreal Cognitive Assessment (MoCA) 10 and 2 of its subscales, controlling for age and sex. The current analyses focused on MoCA subscales reflecting Delayed Recall and Visuospatial/Executive Function as deficits in those 2 MoCA domains are associated with the highest risk for cognitive decline. 11
Materials and Methods
Participants
We analyzed data from the Montreal Cognitive Assessment (MoCA) test and state-of-the-art imaging data of sub-second resting state fMRI (rfMRI) from 711 healthy participants of Human Connectome Project (HCP) on Aging (HCP-A, www.humanconnectome.org/study/hcp-lifespan-aging) 12 lifespan 2.0 data release. The study included 316 males and 395 females younger than 90 (36.0-89.9 years old), representing a sample of the current US population with diverse ethnic, racial, and socio-economic status and with “typical” health for their age. Those with clinical diagnoses of psychiatric and neurological disorders, cognitive impairment, and symptomatic stroke, among other criteria were excluded. All participants provided written informed consent. The Research and Development Committee of the Minneapolis Veterans Affairs Medical Center approved this study.
Cognitive assessment
The total MoCA 10 score and scores of 2 MoCA subscales were used in this study, namely the Visuospatial/Executive Function scale and the Delayed Memory scale. The visuospatial/Executive function scale includes 3 tasks—Trails, Cube, and Clock Drawing (TCCD) that generally capture visuospatial and executive functioning. The Trails involves drawing lines to connect circled numbers and letters in alternating number and alphabet sequence, the Cube involves copying a Necker cube, and the Clock Drawing involves drawing a clock face with the contour, numbers, and time correctly shown. The Delayed Memory scale involves 2 learning trials of a list of 5 words followed by recall of those words 5 minutes later. Points for Uncued Recall (PUR) is determined by the number of words correctly recalled prior to category or multiple choice cue prompting.
Resting state functional MRI data
The 32-channel head coil Siemens 3T Prisma scanner, which enables high acceleration factors via multi-slice acquisitions of MRI, was used. 13 Participants viewed a small white fixation crosshair on a black background during rfMRI scanning. They were instructed to remain awake, still, and blink normally. RfMRI scans were acquired in pairs of 2 runs, with opposite phase encoding polarity so that the fMRI data in aggregate is not biased toward a particular phase encoding polarity. RfMRI data were acquired for 26 minutes of resting state scanning in 4 runs of 6.5 minutes each. Details of acquisition parameter are given elsewhere. 13
rfMRI data extraction and processing
The Desikan-Killiany atlas parcellation, consisting of 68 cortical areas (34 per hemisphere, Table 1), were used. 14 Data of 2 rfMRI acquisition sessions, the first session with one run AP and second run PA and the second session with one run AP and second run PA, were combined. For each participant and brain area, the blood-oxygen-level dependent (BOLD) time series were extracted from recommended minimally preprocessed rfMRI data 15 (N = 1912 BOLD values per vertex).
The 68 areas analyzed (34 for the left and the same 34 for the right hemisphere).

Data analysis
TBOLD
A robust, nonparametric estimate of resting-state BOLD turnover was obtained in 3 steps, as described in detail in Ref. 6 Briefly, (i) single BOLD time series for a given kth voxel were first-order differenced; (ii) the median of the absolute values of the differenced time series was computed; and (iii) this median value was divided by 0.8 second (since TR = 0.8 second) to yield the resting-state BOLD turnover per second (TBOLD):

Finally, the mean TBOLD for each area (across M voxels in the area) was computed and used for further analyses:

Handedness
Handedness scores in the databases were integers ranging from −100 and (totally left handed) to +100 (totally right handed), with negative scores indicating left handed, positive scores indicating right handed, and zeros indicating ambidextrous participants. Of the total of 711 participants, 76 (10.7%) were left handed and 635 (89.3%) were right handed; there were no ambidextrous participants. For quantitative analyses, a binary (HAND) variable was created:
Handedness score [−100 to −1] → Left handed (HAND = 0)
Handedness score [1 to 100] → Right handed (HAND = 1)
Statistical analyses
Standard parametric and nonparametric statistical methods were used to analyze the data, including descriptive statistics, and comparisons between means, comparisons between distributions, linear regression analysis, etc. In addition, a crosscorrelation analysis was performed between TBOLD and MoCA, TCCD and PUR scores to find out whether TBOLD is associated with cognitive performance in future years. For that purpose, for each area, the effects of sex and handedness were removed by performing a multiple linear regression of TBOLD, MoCA, TCCD, and PUR against SEX and HAND and retaining the residuals, Next, grand means of these residuals (across areas) were computed for each year-bin, yielding time series of length N = 54 (36-89 annual bins) for each measure. The crosscorrelation function was then calculated between the time series above of TBOLD and those of MoCA, TCCD and PUR scores.
The IBM-SPSS statistical package (version 29) was used to analyze the data. All reported P values are 2-sided.
Results
Age
The frequency distribution of age is shown in Figure 1A. Mean age did not differ significantly between men (mean = 60.2 years, SD = 15.2, N = 316) and women (mean = 59.0, SD = 14.6, N = 395) (P = .268, independent samples t-test). In addition, a comparison of the age distributions of the 2 sexes using the Kolmogorov-Smirnov test was not significant (P = .291), indicating that men’s and women’s age distributions come from the same parent distribution.
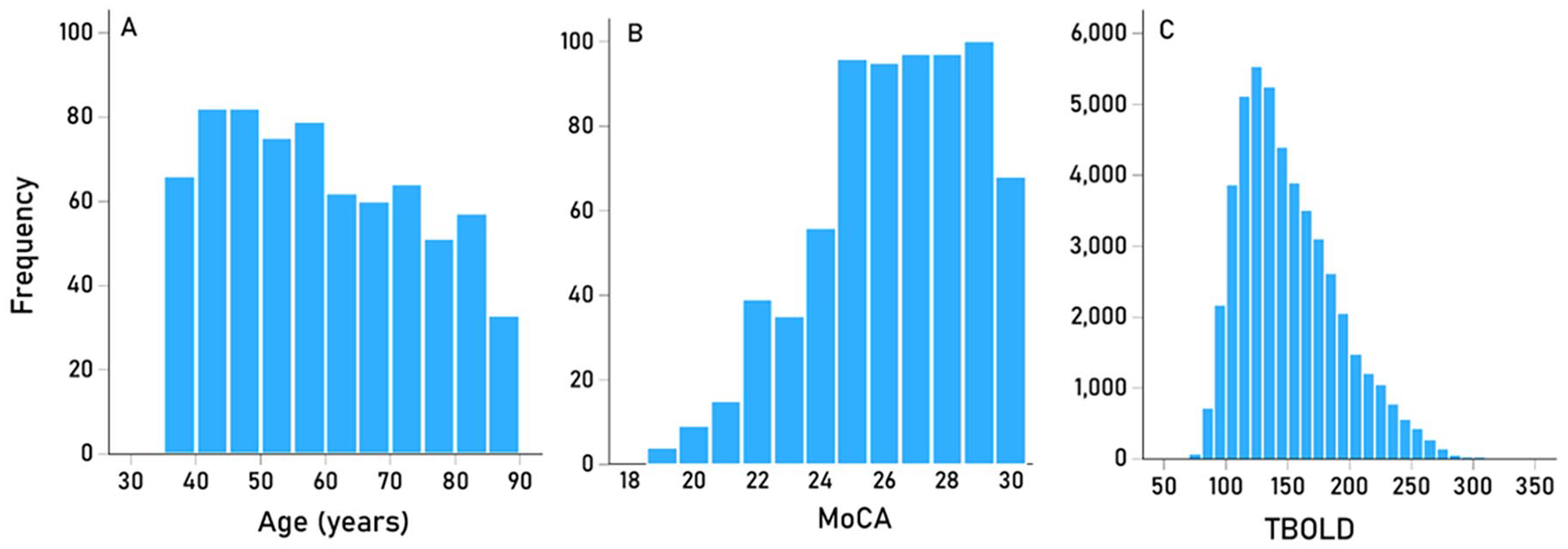
(A) frequency distribution of age of participants (N = 711). (B) frequency distribution of MoCA scores of participants (N = 711). (C) frequency distribution of TBOLD (N = 711 brains × 68 areas = 48 348).
Effect of sex and handedness on cognitive performance
The frequency distribution of MoCA is shown in Figure 1B, and the frequencies of TCCD and PUR scores are given in Tables 2 and 3, respectively. The effects of sex and handedness on performance of MoCA, TCCD and PUR scores was evaluated using an ANCOVA where the cognitive score was the dependent variable, SEX and HAND were fixed factors, and age was a covariate. We found the following. (a) with respect to MoCA scores, women performed significantly better than men (F[1,706] = 6.180, P = .013), whereas neither the HAND nor the SEX × HAND interaction were significant (P = .794 and P = .324, respectively). (b) With respect to the TCCD task, no statistically significant effects were found (P = .155 for SEX, P = .774 for HAND, and P = .229 for SEX × HAND interaction; F-test). Finally, with respect to the PUR task, women performed significantly better than men (F[1,706] = 3.920, P = .048) and right handed participants performed better than left handed participants (F[1,706] = 4.545, P = .033); the interaction term SEX × HAND did not have a significant effect (F[1,706] = 0.124, P = .725).
Performance in the Trails, Cube and Clock Drawing (TCCD) task.

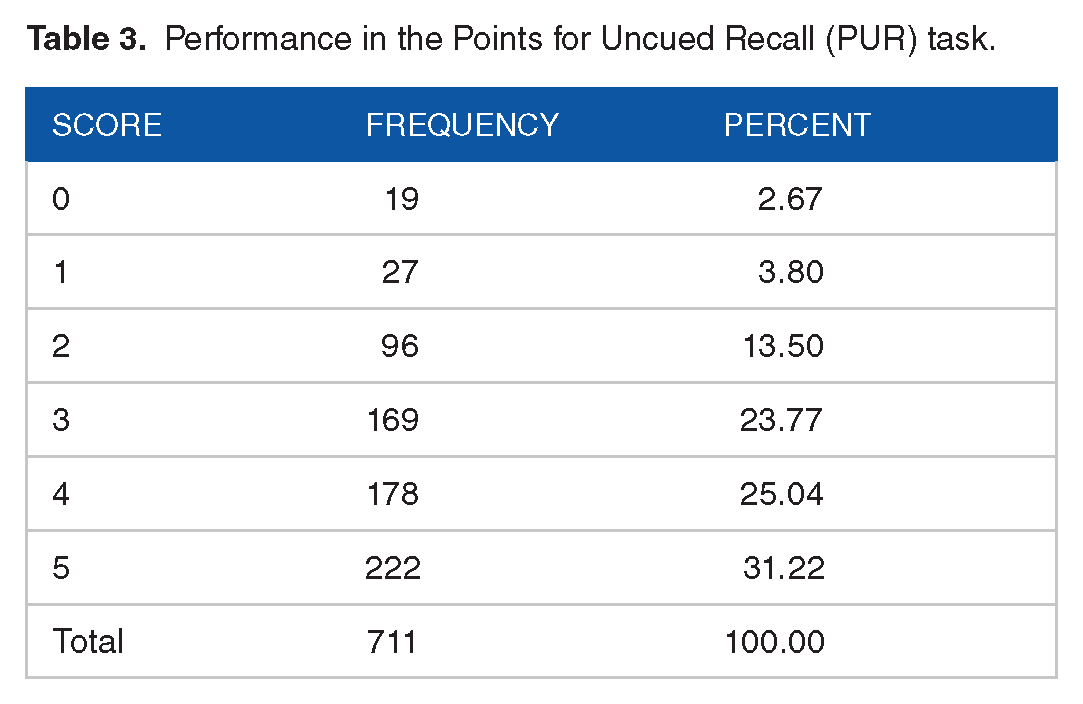
Performance in the Points for Uncued Recall (PUR) task.
Association between TBOLD and cognitive performance
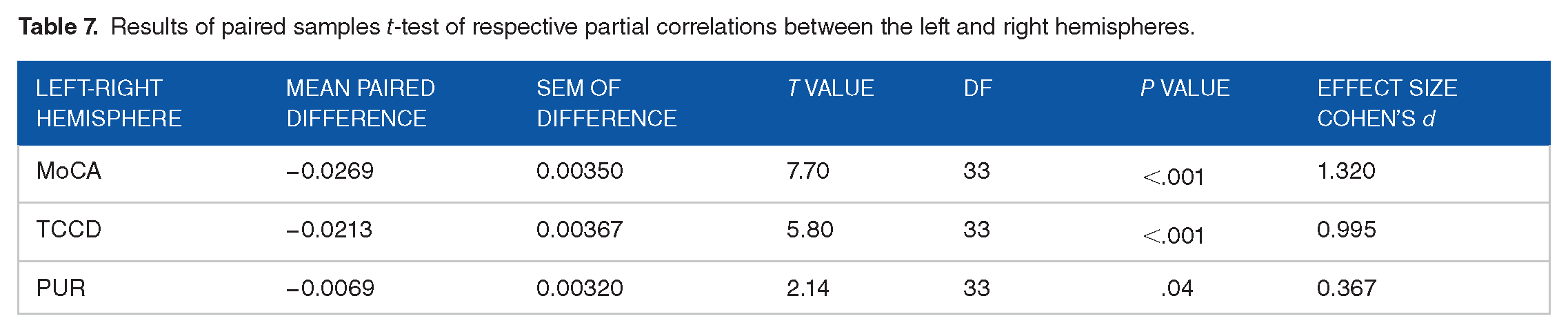
The frequency distribution of TBOLD is shown in Figure 1C. The association of TBOLD with cognitive performance was assessed by computing, for each one of the 68 cortical areas, the partial correlation between TBOLD and MoCA, TCCD and PUR scores, controlling for age, sex, and handedness. The results are shown in Tables 4 and 5. We found the following. With respect to the TBOLD versus MoCA, 32/34 (94.1%; P < .001, Wilson test for binomial proportion against the null hypothesis of 50%) partial correlations in the left and 29/34 (85.3%; P < .001, Wilson test) in the right hemispheres were negative; overall, the partial correlations were highly correlated between the 2 hemispheres (Figure 2A; rp = 0.821, P < .001, N = 34 areas). With respect to TBOLD versus TCCD, 29/34 (85.3%; P < .001, Wilson test) partial correlations in the left and 25/34 (73.5%; P = .006, Wilson test) in the right hemispheres were negative; overall, the partial correlations were highly correlated between the 2 hemispheres (Figure 2B; rp = 0.855, P < .001, N = 34). With respect to TBOLD versus PUR, 32/34 (94.1%; P < .001, Wilson test) correlations in both the left and right hemispheres were negative; partial correlations were highly correlated between the 2 hemispheres (Figure 2C; rp = 0.888, P < .001, N = 34). Finally, with respect to hemisphere, partial correlations were more negative in the left than the right hemisphere for all 3 cognitive measures (Table 6). These differences were highly statistically significant (Table 7).
Partial correlations between MoCA scores and TBOLD, controlling for age, sex, and handedness (N = 711).

Abbreviations:
Partial correlations between TCCD and PUR scores and TBOLD, controlling for age, sex, and handedness (N = 711). Conventions as in Table 4.


(A) MoCA partial correlations (
Descriptive statistics of partial correlations (

Results of paired samples t-test of respective partial correlations between the left and right hemispheres.
Crosscorrelations
Given the pervasive negative effect of TBOLD on cognitive measures of performance, we further explored, for all 3 measures (MoCA, TCCD, PUR), the hypothesis that an increase in TBOLD would precede a decrease in cognitive performance, in addition to their contemporaneous negative association documented above. For that purpose, we computed the crosscorrelation function between the time series of TBOLD and MoCA, TCCD, and PUR, after removing the effect of SEX and HAND (see Methods). We found the following. (a) The autocorrelation and partial autocorrelation functions (ACF and PACF, respectively) of the TBOLD residuals time series did not show any statistically significant value across all 15 year-lags evaluated (Figure 3), approximating white noise. (b) The crosscorrelation function (CCF) analyses revealed a pervasive negative effect of TBOLD on: MoCA (Figure 4), TCCD (Figure 5) and PUR (Figure 6) across the 15 forward lags tested, namely 13/15 (86.7%) negative CCF values for MoCA CCF, 15/15 (100%) for TCCD CCF, and 11/15 (73.3%) for PUR CCF. (c) CCF values were highly correlated among cognitive measures (MoCA CCF vs TCCD CCF: r = .817, P < .001, N = 15; MoCA CCF vs PUR CCF: r = .859, P < .001; TCCD CCF vs PUR CCF: r = .716, P = .002) and did not differ significantly (P > .5 for all 3 comparisons).

(A) Autocorrelation and (B) partial autocorrelation functions (ACF and PACF, respectively) of the TBOLD time series used for crosscorrelation analyses. Lines indicate 95% confidence intervals.

Crosscorrelation function (CCF) between TBOLD (independent variable) and MoCA (dependent variable). For CCF at lag 2, P = .022, and at lag 3, P = .018.

CCF between TBOLD and TCCD. For CCF at lag 3, P = .037, and at lag 7, P = .049.

CCF between TBOLD and PUR. For CCF at lag 0, P = .023, and at lag 3 P = .005.
Discussion
Here we evaluated the association between TBOLD, that is, the moment-to-moment turnover in the task-free resting-state BOLD signal, and cognitive performance in a large sample of adult participants of the Human Connectome Project on Aging. The findings documented that increasing TBOLD was associated with decreased performance on MoCA total score as well as delayed recall and visuospatial/executive function tasks. Those negative correlations were documented contemporaneously and prospectively, suggesting that increases in resting-state TBOLD are not only associated with but also predict subsequent decline in cognitive performance. On the hypothesis that neuroinflammatory processes underlie increased TBOLD and associated atrophy, 6 the present findings provide a mechanism linking neuroinflammation with decreased cognitive performance.
Previous studies using different measures of BOLD variability have documented that variability in the resting-state BOLD correlates with age 16 and cognitive performance,17,18 although, the direction of those effects has been shown to vary over the lifespan in specific brain regions 19 and across cognitive domains. 20 In the present study we first controlled for age, permitting evaluation of fundamental associations between TBOLD and cognitive performance without potentially conflating age effects. In doing so, we found that increased TBOLD was associated with decrements in cognitive performance, independent of age, and across each of the cognitive domains evaluated. Furthermore, the vast majority of the correlations between TBOLD and cognitive performance were negative across brain regions, albeit more strongly in the left hemisphere, suggesting a pervasive negative influence of TBOLD on cognition. Given these robust associations, we then evaluated the influence of TBOLD on cognitive performance across forward lags and found that TBOLD predicted decrements in future cognitive performance. These findings are in keeping with the postulated TBOLD as dynamic marker for chronic neuroinflammation and, furthermore, provide an estimate of forward negative effects on cognition, ranging from 2 to 3 years, and up to 7 years (Figures 4-6). However, it should be noted that these results are based on cross-sectional data that warrants replication in a longitudinal study.
To date, we have documented that resting-state TBOLD is heritable, 4 varies across brain areas, 5 is negatively associated with brain volume 6 and cognitive performance (current study), and increases with age but only in individuals lacking a specific neuroprotective human leukocyte antigen (HLA-DRB1*13:02).5,6 Taken together, these findings highlight TBOLD as a fundamental indicator of brain activity and show that increases in TBOLD reflect abnormal brain function. In light of the role of HLA in immune protection against foreign antigens such as viruses, we have surmised that increases in TBOLD and associated brain volume loss and cognitive decline may result from chronic low-grade neuroinflammation associated with exposure to common pathogens in the absence of immune protection against them.5,6 Neuroinflammation induces vasodilation via production of nitric oxide 21 mediated by activation of microglia22,23 which upregulate the inducible nitric oxide synthase isoform (iNOS). 24 In addition, astrocytes have also been implicated in neuroinflammation25,26 and associated vasodilation mediated by D-serine and endothelial nitric oxide synthase. 27 TBOLD, which reflects primarily neurovascular activity, 3 is likely to be particularly sensitive to effects of vasodilation. To that end, recent studies have documented that increased BOLD variability is higher in individuals with cerebral small vessel disease 28 and in hypertensive elderly patients. 29 The current finding that TBOLD is negatively associated with cognitive performance contemporaneously and prospectively in healthy adults suggests that subtle neurovascular changes precede cognitive decline and likely reflect the effects of brain insults in the absence of immune-neuroprotection against them.
Study limitations
It should be noted that this study was focused on the overall, “bird’s eye view” association of neurovascular coupling with basic, well established assessments of cognitive function, such as the overall MoCA score and the scores of 2 of its domains (visuospatial [TCCD]) and delayed recall [PUR] tasks) over the lifespan. This design provides a framework for exploring associations between neurovascular coupling at the task-free, resting state (TBOLD) with performance scores of other tasks. By contrast, the study of the neural mechanisms underlying task performance requires the use of fMRI recorded during task performance, a condition where synaptic neural activity contributes substantially to the BOLD signal, in contrast to the task-free, resting-state fMRI where the contribution of synaptic activity is small (~10%). 3
Footnotes
Acknowledgements
Research reported in this publication was supported by the National Institute On Aging of the National Institutes of Health under Award Number U01AG052564 and by funds provided by the McDonnell Center for Systems Neuroscience at Washington University in St. Louis. The HCP-Aging 2.0 Release data used in this report came from DOI:10.15154/1520707.
Author Contributions
APG, PC: conceived the research; PC, APG: analyzed the data; LMJ, PC, APG: wrote the paper; all authors edited and approved the final version of the paper.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial funding for this study was provided by the University of Minnesota (the Kunin Chair in Women’s Healthy Brain Aging, the Brain and Genomics Fund, the McKnight Presidential Chair of Cognitive Neuroscience, and the American Legion Brain Sciences Chair) and the U.S. Department of Veterans Affairs. The sponsors had no role in the current study design, analysis or interpretation, or in the writing of this paper. The contents do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
Study Approval
This study was approved by the Minneapolis VAHCS Research and Development Committee (#1594569).
