Abstract
Background:
Alzheimer’s disease (AD) is a progressive neurological disorder characterized by a wide range of cognitive and non-cognitive impairments. The present study was designed to investigate the potential effects of cacao on cognitive and non-cognitive performance and to identify the role of oxidative stress in an AD animal model induced by unilateral intracerebroventricular (U-ICV) injection of amyloid beta1-42 (Aβ1-42).
Methods:
Oral administration of cacao (0.5 g/kg/day) was performed for 60 consecutive days. Following 60 days, the open-field (OF) test, elevated plus-maze (EPM) test, novel object recognition (NOR) test, Barnes maze (BM) test, and Morris water maze (MWM) test were used to evaluate locomotor activity, anxiety-like behavior, recognition memory, and spatial memory, respectively. Total oxidant status (TOS) and total antioxidant capacity (TAC) in plasma were also examined. Furthermore, the number of healthy cells in the hippocampus’s dentate gyrus (DG), CA1, and CA3 regions were identified using hematoxylin and eosin staining.
Results:
The results indicated that the injection of Aβ1-42 in rats led to recognition memory and spatial memory impairments, as well as increased anxiety. This was accompanied by decreased total antioxidant capacity (TAC), increased total oxidative stress (TOS), and increased neuronal death. Conversely, cacao treatment in AD rats improved memory function, reduced anxiety, modulated oxidative stress balance, and decreased neuronal death.
Conclusion:
The findings suggest that cacao’s ability to improve the balance between oxidants and antioxidants and prevent neuronal loss may be the mechanism underlying its beneficial effect against AD-related cognitive and non-cognitive impairments.
Introduction
Alzheimer’s disease (AD) is a progressive neurological disorder that leads to brain tissue degeneration and ultimately results in cognitive impairment, including memory loss. 1 Individuals with AD frequently suffer from various non-cognitive disturbances, such as anxiety and depression, in addition to the progressive impairment of cognitive function. 2 The pathology of AD is characterized by the accumulation of extracellular amyloid beta (Aβ) plaques and intracellular neurofibrillary tangles, which gradually lead to neurotoxicity and brain dysfunction. 3 The hippocampus plays a crucial role in learning and memory processes 4 and is considered the most vulnerable brain region in AD, being heavily affected by oxidative stress-induced damage. 5 There is substantial evidence suggesting that oxidative stress, plays a key role in the early manifestation of AD symptoms. 6 Studies have also shown that the induction of Aβ plaque toxicity acts as a pro-oxidant, leading to oxidative stress in the brain. 7 Increased levels of reactive oxygen species (ROS) can stimulate pro-inflammatory gene transcription and the release of cytokines. This contributes to chronic neuroinflammation and subsequent neuronal loss. 8 Although none of the existing models of AD completely replicate the human disease, Aβ-induced AD is used as a well-defined model to identify the underlying pathophysiological mechanisms of AD.9 -11
The phytoconstituents, which occur naturally in plants, provide a wide range of bioactive chemical compounds, particularly polyphenols, with different pharmacological potentials for treating and preventing AD. 12 These natural compounds exert their therapeutic effects through multiple mechanisms, including promoting Aβ clearance, preventing Aβ aggregation, oxidative stress control via ROS scavenging, lessening acetylcholinesterase activity, and modulation of the Aβ-induced inflammatory response.13,14 The drugs galantamine and rivastigmine, which the U.S. Food and Drug Administration approves for mild-to-moderate stages of AD, are derived from plant sources. 12 Various phytoconstituents have been utilized in AD therapy such as resveratrol, 15 rosmarinic acid, 16 curcumin, 17 asiaticoside, 18 carnosic acid, 19 ginsenoside, 20 Lavender, 21 ginkgo biloba extract, 22 melissa officinalis phenolic compounds, 23 epigallocatechin-3-gallate, 24 bacopa monnieri. 25 Therefore, the phytoconstituents will undoubtedly contribute to developing novel, safer AD treatments.
Cacao, derived from the plant Theobroma cacao, 26 is a major source of varied polyphenol contents such as quercetin, clovamide, procyanidin, epicatechin, catechin, and also methylxanthines such as theobromine and caffeine. 27 Cacao exhibits a wide spectrum of biological properties, including anti-inflammatory, 28 anti-oxidative, 29 anti-apoptotic, 30 anti-depressant, 31 anxiolytic, 32 neurotrophic,33,34 and cognitive-ameliorating effects. 35 Additionally, cacao has neuroprotective properties in neurodegenerative diseases such as AD 36 and Parkinson’s disease.37 -39 Furthermore, dark chocolate as a cacao by-product can prevent the harmful effects of chronic isolation stress on hippocampal synaptic plasticity, learning, and memory. 40 Furthermore, cacao may protect against D-galactose-induced oxidative damage, cholinergic impairment, and apoptosis. This protection might involve activation of the protein kinase B (Akt)-mediated caspase-3 pathway and catalase in the brain, along with inactivation of acetylcholinesterase. 29 However, in the animal model, the effects of cacao on anxiety and learning and memory functions, oxidative status, and histological changes following Aβ1-42 exposure haven’t been fully investigated yet.
Therefore, this study aimed to investigate the effects of chronic oral administration of cacao on cognitive and non-cognitive deficits in an Aβ1-42-infused AD rat model. Additionally, this study aimed to identify the role of oxidative stress as a specific underlying mechanism.
Materials and Methods
Animals and experimental design
Adult male Wistar rats weighing 250 to 270 g were purchased in a quantity of 48 from the animal house of Hamadan University of Medical Sciences (Hamadan, Iran). The rats had free access to standard food (47% carbohydrate, 5% fat, 23% protein, 5% fiber, 20% water, vitamins, and minerals) and water in a standard laboratory cage. The animal room had a controlled temperature of 22 ± 2°C with a relative humidity of 60 ± 5% and a 12-hour light-dark cycle. Animal care and experimental procedures were conducted in accordance with the NIH guidelines and were approved by the ethics committee of Bu-Ali Sina University-Hamadan (ethical code IR.BASU.REC.1398.025). After a 7-day adaptation period to the laboratory environment, the rats were randomly divided into 6 groups of 8 as follows:
I: Control group: Rats received 5 mL/kg/day of 0.9% normal saline via oral gavage (P.O.) for 60 days.
II: PBS group: Rats received a stereotaxic unilateral intracerebroventricular (U-ICV) injection of 5 μL/rat of phosphate-buffered saline (PBS) plus normal saline (5 mL/kg/day; P.O. for 60 days).
III: Cacao per se group: Rats received cacao (0.5 g/kg/day; P.O. for 60 days).
IV: Aβ group: Rats received a stereotaxic U-ICV injection of 5 μL/rat of Aβ1-42 (1 μg/μL) plus normal saline (5 mL/kg/day; P.O. for 60 days).
V: Aβ-Pre group: Rats received cacao (0.5 g/kg/day; P.O. for 60 days) before a stereotaxic U-ICV injection of 5 μL/rat of Aβ1-42 (1 μg/μL).
VI: Aβ-Post group: Rats received cacao (0.5 g/kg/day; P.O. for 60 days) after the stereotaxic injection of 5 μL/rat of Aβ1-42 (1 μg/μL).
Cacao powder (Energy 415 kcal, Energy (kJ) 1736 kJ, Carbohydrates 14 g, Sugar 2 g, Protein 7.4 g, Fat 21.5 g, Fiber 33.5 g, Sodium 0.56 g per 100 g; Cadbury Co., UK) was purchased from Amazon company (ASIN: B0BVFQ64XG). The dose of cacao used in the present study (0.5 g/kg/day) was determined based on previous research demonstrating the protective effects of cacao 41 and its by-products, such as dark chocolate with 70% cacao content, 42 against behavioral, biochemical, and histological changes in various models of neurological disorders. In addition, the dosage was selected based on a pilot study conducted in our laboratory. Figure 1 shows the experimental timeline. The behavioral tests of the rats (n = 8) were evaluated by employing open-field (OF) on the 96th day, novel object recognition (NOR) on the 97th and 98th days, elevated plus-maze (EPM) on the 99th day, Barnes maze (BM) during the 100th to 103rd days, Morris water maze (MWM) during the 104th to 108th days. At the end of the study on the 109th day, the rats were euthanized for biochemical assessments (n = 8) and histological investigations (n = 4).
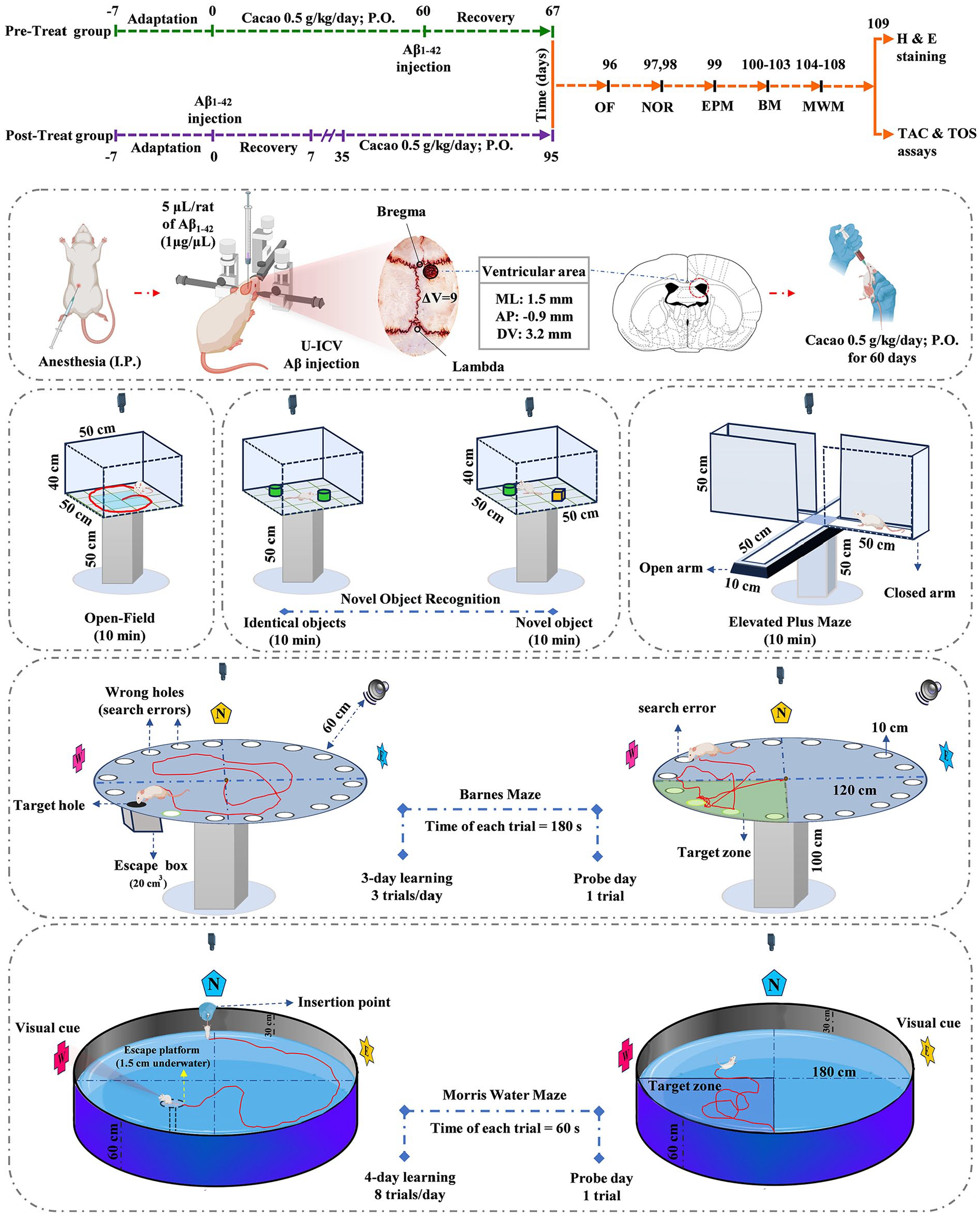
After 1 week of adaptation to the laboratory environment, before (Aβ-Pre group) and after (Aβ-Post group) unilateral intracerebroventricular (U-ICV) injection of a 5 µL solution of Aβ1-42 (1 mg/mL), cacao was administered orally at a dose of 0.5 g/kg daily for 60 consecutive days. Anxiety, recognition memory, and spatial memory tests were performed afterward. At the end of the experiment, plasma biomarkers (TAC and TOS) were measured and hematoxylin and eosin staining was performed on the hippocampal tissue.
AD induction
A 5-µL solution of Aβ1-42 (1 mg/mL) was injected unilaterally into the ICV region to induce AD in rats.9 -11 Therefore, the Aβ peptide rat (product No/SKU SCP0038-1MG, Sigma Aldrich, USA) was dissolved in PBS solution. Before U-ICV injection, the Aβ was incubated at 37°C for 4 days. This process leads to the production of amyloid fibrils, which are neurotoxic.9,11 Previous studies have reported cognitive and non-cognitive impairments 28 and 35 days after the ICV injection of Aβ1-42. These impairments have been associated with increased amyloid beta plaque accumulation, increased neuronal death, an imbalance in the oxidant-antioxidant system, and impaired synaptic plasticity.9 -11 For AD induction, each rat was anesthetized by intraperitoneally (I.P.) injection of a mixture of ketamine (100 mg/kg) and xylazine (10 mg/kg) and stereotaxic (Dual Lab Standard Stereotaxic apparatus; Stoelting Co., Wood Dale, IL, USA) surgery was performed. The head was shaved and a midline sagittal incision was made in the scalp. A tiny hole was drilled carefully up to the level of the dura mater in the skull over the ventricular area (coordinates relative to bregma: medial-lateral (M/L) = 1.5 mm and anterior-posterior (A/P) = −0.9 mm). Hamilton syringe needle was slowly directed down to beneath the surface of the cortex for the U-ICV injections, into the right lateral ventricle (coordinates relative to the skull: dorsal-ventral (D/V) = 3.2 mm). 43 Bilateral intracerebroventricular injection involves injecting Aβ into both lateral ventricles of the brain. Both unilateral and bilateral injections can induce widespread neurobehavioral alterations resembling AD.44 -46 A 5 μL Aβ solution was administered at a 0.5 μL/min rate. The PBS group received 5 μL of PBS (10 mM). After the surgery, the rats were individually placed in their cages and allowed to undergo a 7-day recovery period with special care. 9
Open-field (OF) test
The open-field test chamber is a 50 cm × 50 cm × 40 cm box positioned 50 cm above the floor. The walls of the box were made opaque, isolating the interior chamber from the external environment and minimizing the impact of environmental factors. The video tracking software (Borj Sanat Co., Tehran-Iran) divided the movement field of the rat into a 16-square grid. Four central squares were designated as the central zone among these squares. Each rat was placed in the chamber for 10 minutes of free exploration. After each trial, the inner walls of the chamber were cleaned and sterilized with 70% ethanol to eliminate any olfactory cues from previous rats. The software automatically recorded the locomotor activity and the time spent in the central zone.11,47
Novel object recognition (NOR) test
The NOR test is a model used to evaluate rodents’ recognition memory and exploratory behavior. 48 The test involved three 10-minute phases conducted in a box measuring 50 cm × 50 cm × 40 cm. These phases included: 1. Habituation to an empty chamber, 2. Familiarization with 2 identical objects (at a 6-hour interval of the first phase), and 3. Identify a novel object (after a 24-hour interval from the second phase). As an indicator of the time spent exploring the novel object, the exploration ratio was calculated by dividing the duration of exploration of the novel object by the total duration of exploration of both old and new objects. Exploration time was defined as sniffing, touching, or directing attention to the object. 49
Elevated plus maze (EPM)
The elevated plus maze (EPM) apparatus is designed based on 2 innate exploratory behaviors of rodents: the tendency to explore and the aversion to open and bright environments. It is used as a tool for evaluating anxiety-like behaviors. 11 The apparatus is placed 50 cm above the floor and resembles a plus sign with 2 open arms and 2 closed arms connected to a central area measuring 10 cm × 10 cm. The open arm is 50 cm long, 10 cm wide, and 1 cm high. The closed arm is 50 cm long, 10 cm wide, and 50 cm high, with opaque walls. For the rats’ safety, a 1 cm high glass border is installed around the open arms to prevent them from falling. Each rat was initially placed in one of the open arms, facing the center of the apparatus. The behavior of the rat was then recorded for 10 minutes using a computerized tracking system. The duration of stay in the open arms was recorded to assess anxiety-like behavior. 50 The ratio of entries into the open arms, determined by dividing the number of entries into the open arms by the total number of entries into both types of arms, was recorded as an indicator of anxiety-like behavior. An entry into the arm was considered when all 4 paws of the rat were positioned within that specific arm. Following each rat’s experiment, the apparatus underwent cleaning and sterilization using 70% ethanol. Anxious animals prefer to stay more in closed arms and spend less time exploring open arms. An increase in the time spent in the open arms or the percentage of entries into the open arms, along with no change in locomotor activity, indicates anxiolytic activity; and a decrease in these 2 parameters is considered anxiety-like behavior. 51
Barnes maze (BM) test
The BM test is similar to the Morris water maze (MWM) test and allows for the measurement of spatial memory. However, the BM test has less stress for rodents than the water maze, which involves swimming. 52 The Barnes maze is elevated 100 cm above the ground. It consists of a gray circular platform with a diameter of 120 cm divided into 4 equal hypothetical quadrants (northeast, northwest, southeast, and southwest). Eighteen holes with a diameter of 10 cm are located around the platform. An escape box with a volume of 20 cm3 is hidden under one of the holes, called the target hole. In the testing room, various spatial cues are visible to the animal in 4 directions. The goal of this test is to measure the ability of the animal to learn and remember the location of the target hole and escape box using these cues. The rat is initially placed in the center of the platform so that the movement of all rats is recorded equally from the center of the platform. A loud noise at 80 dB, emitted by a concealed sound device located 60 cm from the circular platform, serves as a stimulus for the animal to escape. This loud noise stops when the rat finds the target hole and enters the escape box, encouraging the animal to enter the box. If the rat moves away from the target hole, the bell sound is activated again. After the rat spends 20 seconds in the escape box, it is taken out of the Barnes maze. This learning phase of the test was repeated for 3 consecutive days (3 trials per day, with a 60-minute interval between each repetition, each trial for 180 seconds). During these phases, the learning progress of the animal is measured based on the time and distance traveled to reach the escape box in each trial and the number of search errors (checking non-target holes). On the fourth day, designated as the Probe day, the escape box was removed to conduct a memory test. The rat was placed in the maze for 180 seconds during this phase. Throughout the entire duration of the test, the noise was continuously emitted without any interruptions. The time spent in the target zone and search errors were recorded using a camera and video tracking software.44,45,53
Morris water maze (MWM) test
The MWM is a tank with a black interior covering with a diameter of 180 and a depth of 60 cm, filled with water at approximately 20°C. The maze is divided into 4 hypothetical equal sections (northeast, northwest, southeast, and southwest). A 10 cm diameter circular escape platform is positioned 1.5 cm below the water surface, centered in the southwest zone. This hidden platform is the only means of escape for the animal to prevent drowning. In the testing room, various spatial cues are visible to the animal in 4 directions. A camera installed above the tank tracks and detects the animal’s movement. To acclimate the rats to the MWM test, they swim in a tank without a platform for 1 minute 1 day before training. During the 4 days of learning, the animal was released at the location determined by the system (E, W, N, and S) while facing the spatial cue. The maximum time allowed to find the platform was 60 seconds. If the animal fails to find the platform within this time, it is guided toward the platform to rest on it for 30 s. During these 30 seconds, the rat memorizes its position based on the location of the platform and the cues on the walls. If the animal finds the platform, it is given 30 seconds to rest and memorize the cues. The learning phase of the MWM test was repeated for 4 consecutive days (8 trials per day, with a 5-minute interval between the first and second 4 trials). During these phases, the learning progress of the animal is measured based on the time and distance traveled to find the platform in each trial. 11 On the fifth day (probe day), a memory recall test was conducted by removing the hidden platform and releasing the rats into the water from the south starting point. This phase lasts for 60 seconds for each animal. The time spent in the target zone (southwest zone) was measured as an indicator of spatial memory. 54 At the end of the behavioral test, animal vision was also evaluated. In this stage, a platform was placed above the water level and its surface was made visible by white polystyrene. Then, the animal was randomly released into the water and had 60 seconds to find the platform. If there was a visual impairment, it was excluded from the statistical population.55,56
Biochemical assay
Blood samples from the hepatic portal vein after anesthesia with a mixture of ketamine and xylazine were collected. Each sample was centrifuged at 3500 rpm for 20 minutes, then the clear plasma was divided into 100 μL aliquots and stored at −80°C until use. Kits for measuring total oxidant status (TOS) and total antioxidant capacity (TAC; Kiazist Life Sciences, Iran) were used according to the manufacturer’s protocols to calculate the values of oxidative and antioxidant biomarkers, respectively. 45
Histology analysis
Rats from each group were perfused with 0.9% normal saline and 10% formalin solution. After removing the brain for fixation in 10% formalin solution for 72 hours, a 21-hour protocol was performed in a tissue processor and the brains were embedded in paraffin. Sections were cut to a thickness of 5 µm. After deparaffinization and rehydration, sections were washed in distilled water. Hematoxylin-eosin staining was performed according to standard methods: staining with hematoxylin (8 minutes), rinsing with tap water, immersion in 1% HCL and lithium carbonate (each for 30 seconds), staining with eosin (2 minutes), rinsing with tap water, graded alcohol rinses, and finally clearing in xylene. The number of healthy pyramidal cells in the hippocampus’s CA1, CA3, and dentate gyrus (DG) regions was counted.44,54,57
Statistical analysis
The data were analyzed and plotted using GraphPad Prism software, version 9 (GraphPad Software, San Diego, California, USA). The normality of the data was tested using the Shapiro-Wilk test. Analysis of variance (ANOVA) was performed, and if significant, post hoc analysis was conducted based on equal variances of groups as assessed by Bartlett’s test. Data from the BM test (distance traveled, escape latency, and search errors in learning days) and MWM test (swimming distance, escape latency) related to the treatment (normal saline or Cacao) and exposure (Aβ or non-Aβ) were compared and analyzed by Repeated-measures two-way ANOVA (two-way RM ANOVA) followed by Tukey’s post_hoc test. Results of the time spent in the target zone on probe day of BM and MWM, oxidative stress, and neuron counting data were compared using one-way ANOVA followed by Tukey’s post hoc test. The results were presented as mean ± standard deviation (mean ± SD). A significance level of P < .05 was considered in all statistical analyses.
Results
The effects of cacao on the OF test in AD rats
Samples of recorded activities in the OF test are shown in Figure 2a. The one-way ANOVA did not show a significant difference in the distance traveled between groups [F(5, 42) = 1.85; P = .124, Figure 2b]. A one-way ANOVA showed a significant difference in the time spent in the central zone among the groups [F(5, 42) = 4.24; P = .003, Figure 2c]. According to the post hoc Tukey’s test, the Aβ group exhibited a significant decrease in time spent in the central zone compared to the control group (P < .05). Additionally, the Aβ-Pre group showed a significant increase in the time spent in the central zone compared to the Aβ group (P < .05). The Aβ-Post group also showed a significant increase compared to the Aβ group (P < .05).

Samples of recorded activities of rats in the OF test (a). The effects of cacao administration on the locomotor activity (b), time spent in the central square (c) in the open-field test, and discrimination index of the novel objective recognition (d). Data is presented as means ± SD of 8 animals per group (one-way ANOVA, Tukey’s post-hoc test).
The effects of cacao on the NOR test in AD rats
A one-way ANOVA revealed a significant difference in the discrimination index among the groups [F(5, 42) = 6.27, P < .001, Figure 2d]. According to the post hoc Tukey’s test, there was a significant decrease in the exploration time of the novel object in the Aβ group compared to the control group (P < .01). Additionally, the Aβ-Pre group demonstrated a significant increase in the exploration time of the novel object compared to the Aβ group (P < .05). The Aβ-Post group also showed a significant increase compared to the Aβ group (P < .05).
The effects of cacao on the EPM test in AD rats
Samples of recorded activities in the EPM test are shown in Figure 3a. One-way analysis of variance revealed a significant difference in the percentage of entries into the open arms among the groups [F(5, 42) = 9.70, P < .001, Figure 3b]. According to the post hoc Tukey’s test, there was a significant decrease in the Aβ’s group compared to the control group (P < .001). Additionally, the Aβ-Pre group exhibited a significant increase in the percentage of entries into the open arms compared to the Aβ group (P < .05). The Aβ-Post group also showed a significant increase compared to the Aβ group (P < .05).

Samples of recorded activities of rats in the EPM (a). The effects of cacao administration on the open arms entries (b), time spent in open arms (c) of the elevated plus maze Test. Data is presented as means ± SD of 8 animals per group (one-way ANOVA, Tukey’s post-hoc test).
A one-way ANOVA revealed a significant difference in the time spent in open arms among the groups [F(5, 42) = 6.93, P < .001, Figure 3c]. According to the post hoc Tukey’s test, there was a significant decrease in the Aβ group compared to the control group (P < .001). Additionally, the Aβ-Pre group displayed a significant increase in the time spent in open arms compared to the Aβ group (P < .01). The Aβ-Post group also exhibited a significant increase compared to the Aβ group (P < .01).
The effects of cacao on the BM test in AD rats
A sample of activities recorded in the BM test is shown in Figure 4a. A two-way RM ANOVA was conducted and revealed a significant effect of time [F(2, 21) = 202.1, P < .001] and treatment [F(5, 105) = 6.66, P < .001], and time × treatment interaction effect [F(10, 105) = 2.80, P = .004] in the distance traveled to reach the escape box during the 3 days of training (Figure 4b). Tukey’s post hoc analysis indicated a significant increase in the Aβ group compared to the control group on the second day (P < .01) and the third day (P < .001) of training. Additionally, the Aβ-Pre group demonstrated a significant decrease in the distance traveled to reach the escape box compared to the Aβ group on the second day (P < .05) and the third day (P < .01) of training. Likewise, the Aβ-Post group exhibited a significant decrease compared to the Aβ group on the second day (P < .05) and the third day (P < .01) of training.

Samples of recorded activities of rats in the BM test (a). The effects of cacao administration on the distance traveled (b), escape latency (c), search errors of the training days (d), time spent in the target zone (e), and search errors of the probe day (f) in the Barnes maze test. Data is presented as means ± SD of 8 animals per group (two and one-way ANOVA, Tukey’s post-hoc test).
Two-way RM ANOVA showed a significant effect of time [F(2, 21) = 83.11, P < .001] and treatment [F(5, 105) = 9.20, P < .001] in the time to reach the escape box during 3 days of training. However, there was no significant time × treatment interaction effect [F(10, 105) = 0.68, P = .73] (Figure 4c). Tukey’s post hoc analysis indicated a significant increase in the Aβ group compared to the control group on the second day (P < .05), and the third day (P < .01) of the training days. Additionally, the Aβ-Pre group demonstrated a significant decrease in the time to reach the escape box compared to the Aβ group on the second day (P < .05) and the third day (P < .05) of training. Likewise, the Aβ-Post group exhibited a significant decrease compared to the Aβ group on the second day (P < .05) and the third day (P < .01) of training.
Two-way RM ANOVA revealed a significant effect of time [F(2, 21) = 252.5, P < .001] and treatment [F(5, 105) = 8.58, P < .001] in search errors during 3 days of training. However, there was no significant time × treatment interaction effect [F(10, 105) = 0.37, P = .956] (Figure 4d). Tukey’s post hoc analysis indicated a significant increase in the Aβ group compared to the control group on the second day (P < .01) and the third day (P < .01) of training. Additionally, the Aβ-Pre group demonstrated a significant decrease in search errors compared to the Aβ group on the second day (P < .05) and the third day (P < .05) of training. Likewise, the Aβ-Post group exhibited a significant decrease compared to the Aβ group on the second day (P < .05) and the third day (P < .05) of training.
The one-way ANOVA results indicated a significant difference between the groups [F(5, 42) = 6.17, P < .001] in the time spent in the target zone on the probe day. According to the post hoc Tukey’s test, there was a significant decrease in the Aβ group compared to the control group (P < .01). Additionally, the Aβ-Pre group displayed a significant increase in the time spent in the target zone on the probe day compared to the Aβ group (P < .05). The Aβ-Post group also exhibited a significant increase compared to the Aβ group (P < .05; Figure 4e).
A one-way ANOVA results indicated a significant difference between the groups [F(5, 42) = 6.81, P < .001] in search errors in the probe day. There was a significant increase in the Aβ group compared to the control group (P < .01). Additionally, the Aβ-Pre group displayed a significant decrease in search errors on the probe day compared to the Aβ group (P < .05). The Aβ-Post group also exhibited a significant increase compared to the Aβ group (P < .05; Figure 4f).
The effects of cacao on the MWM test in AD rats
A sample of activities recorded in the MWM test is shown in Figure 5a. Two-way RM ANOVA revealed a significant effect of the time [F(3, 28) = 281.8, P < .001] and treatment [F(5, 140) = 9.09, P < .001] on swimming distance to reach the escape platform during the 4 days of training. However, there was no significant time × treatment interaction effect [F(15, 140) = 1.25, P = .238] (Figure 5b). Tukey’s post hoc analysis indicated a significant increase in the Aβ group compared to the control group on the third day (P < .05), and the fourth day (P < .001) of training. Additionally, the Aβ-Pre group demonstrated a significant decrease in the swimming distance to reach the escape platform compared to the Aβ group on the third day (P < .05) and the fourth day (P < .05) of training. Likewise, the Aβ-Post group exhibited a significant decrease compared to the Aβ group on the third day (P < .05) and the fourth day (P < .01) of training.

Samples of recorded activities of rats in the MWM test (a). The effects of cacao administration on the swimming distance (b), escape latency to the hidden platform in the training trials (c), and time spent in the target zone (d) in the Morris water maze Test. Data is presented as means ± SD of 8 animals per group (two and one-way ANOVA, Tukey’s post-hoc test).
Two-way RM ANOVA revealed a significant effect of time [F(2, 21) = 83.11, P < .001] and treatment [F(5, 105) = 9.20, P < .001] on the time to reach the escape platform during 4 days of training. However, there was no significant time × treatment interaction effect [F(15, 140) = 1.004, P = .454] (Figure 5c). Tukey’s post hoc analysis indicated a significant increase in the Aβ group compared to the control group on the third day (P < .01), and the fourth day (P < .001) of training. Additionally, the Aβ-Pre group demonstrated a significant decrease in the time to reach the escape platform compared to the Aβ group on the third day (P < .05) and the fourth day (P < .05) of training. Likewise, the Aβ-Post group exhibited a significant decrease compared to the Aβ group on the third day (P < .01) and the fourth day (P < .01) of training.
The one-way ANOVA results indicated a significant difference between the groups [F(5, 42) = 5.66, P < .001] in the time spent in the target zone on probe day. According to the post hoc Tukey’s test, there was a significant decrease in the Aβ group compared to the control group (P < .01). Additionally, the Aβ-Pre group demonstrated a significant increase in the time spent in the target zone on the probe day compared to the Aβ group (P < .05). The Aβ-Post group also exhibited a significant increase compared to the Aβ group (P < .01; Figure 5d).
Effect of cacao and Aβ on TAC and TOS
Significant differences were observed in TOS concentration among the different groups [F(5, 42) = 12.37, P < .001]. Tukey’s test for TOS concentration showed a significant increase in the Aβ group compared to the control group (P < .001). Additionally, the Aβ-Pre group displayed a significant decrease in the TOS concentration compared to the Aβ group (P < .01). The Aβ-Post group also exhibited a significant decline compared to the Aβ group (P < .01; Figure 6a).
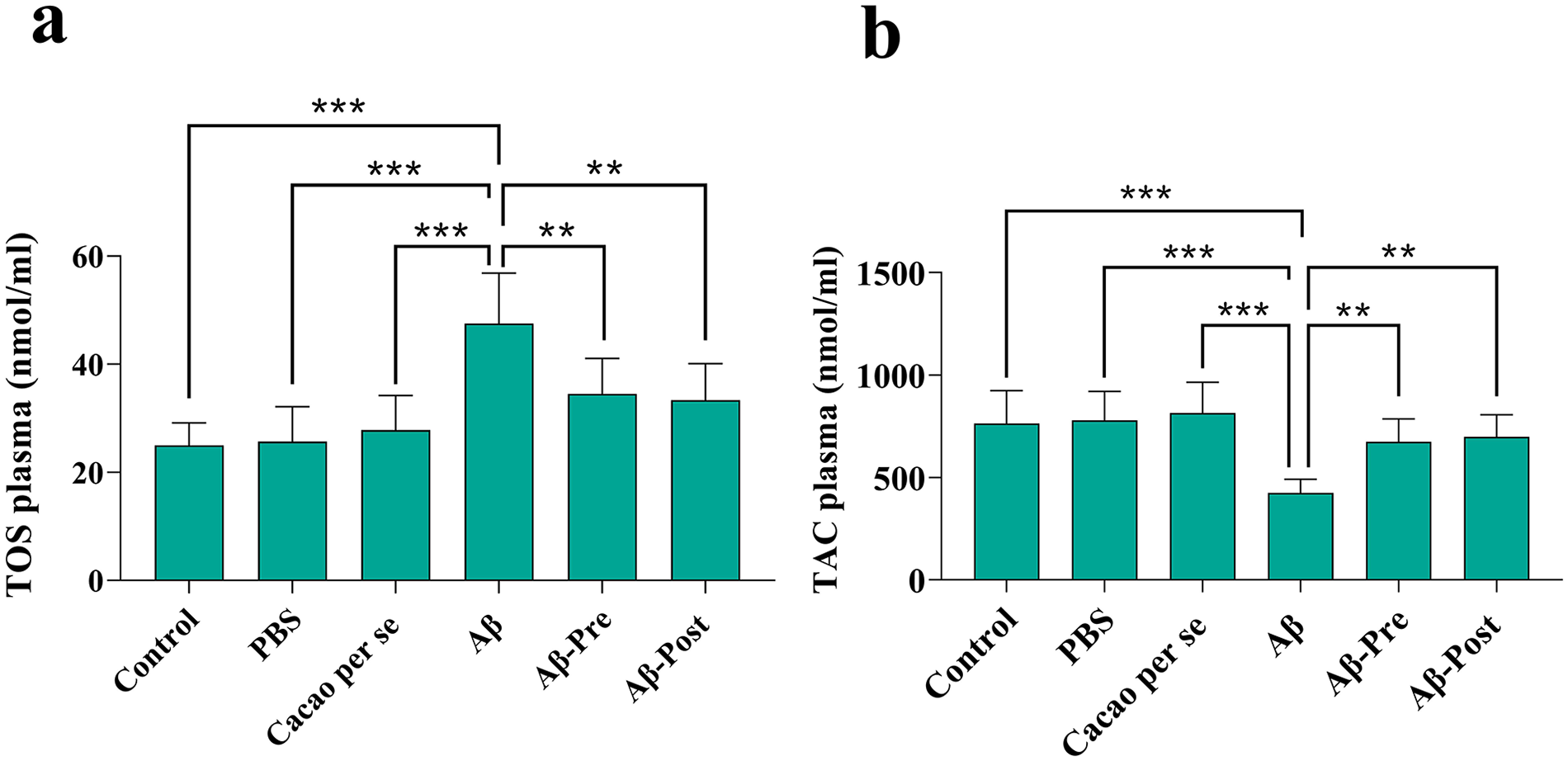
The effects of cacao administration on the plasma parameters of total oxidant status (TOS) (a) and total antioxidant capacity (TAC) (b) of rats using assay kits. Data is presented as means ± SD of 8 animals per group (one-way ANOVA, Tukey’s post-hoc test.
As indicated by one-way ANOVA, there is a significant difference in the plasma concentrations of TAC among the different groups [F(5, 42) = 9.94, P < .001]. The TAC concentration was significantly lower in the Aβ group compared to the control group (P < .001). The TAC concentration was significantly increased in the Aβ-Pre group compared to the Aβ group (P < .01). The Aβ-Post group also exhibited a significant increase compared to the Aβ group (P < .01; Figure 6b).
The effect of cacao and Aβ on the histological changes in the hippocampus
Hematoxylin & Eosin staining was conducted to confirm the histological changes in the rats’ hippocampal CA1, CA3, and DG regions. As illustrated in Figure 7a, a significant difference was found in the number of intact neurons in the hippocampal CA1 [F(5, 18) = 33.23; P < .001]; CA3 [F(5, 18) = 9.27; P < .001] and DG [F(5, 18) = 45.54; P < .001] regions across the groups. There was a significant decrease in the Aβ group compared to the control group in the hippocampal CA1 (P < .001), CA3 (P < .001), and DG (P < .001) regions. Additionally, the Aβ-Pre group demonstrated a significant increase in the number of intact neurons compared to the Aβ group in the hippocampal CA1 (P < .001), CA3 (P < .05), and DG (P < .001) regions. Similarly, the Aβ-Post group exhibited a significant increase compared to the Aβ group in the hippocampal CA1 (P < .001), CA3 (P < .05), and DG (P < .001) regions (Figure 7b).

Effects of cacao and Aβ on histological changes in the hippocampal CA1, CA3, and DG regions of rats. (a) figure illustrates intact neurons (identified by black arrows) and dark neurons (identified by blue arrows, H & E stain, ×400 magnification). (b) represents the quantitative data of the number of intact neurons. Data is presented as means ± SD of 4 animals per group (one-way ANOVA, Tukey’s post-hoc test).
Discussion
This study’s key findings are: (1) cacao modulated AD rats’ oxidant/antioxidant status. (2) cacao prevented neuronal loss in the hippocampus of AD rats. (3) cacao reduced anxiety-like behavior in the EPM test. (4) cacao improved recognition memory in the NOR test. (5) cacao promoted spatial learning and memory in the BM and MWM tests.
In the present study, the injection of Aβ1-42 did not affect the locomotor activity in the OF test, which is in congruence with the previous study. 49 Also, consistent with the previous study, 32 the administration of cacao showed no significant effect on the locomotor activity of rats. On the other hand, the anxiety-like behavior of rats was evaluated based on their locomotor activity and exploratory behavior in the OF test and EPM test, which showed that injection of Aβ1-42 significantly increased anxiety-like activity in the Aβ group. Studies using an AD rat model have demonstrated that Aβ injections trigger a cascade of inflammation, oxidative stress, and neuronal death, ultimately resulting in anxiety-like behaviors in the animals.58,59 In our work, administration of cacao significantly reduced anxiety-like activity in the Aβ-Pre and the Aβ-Post groups. Similarly, it has been reported that oral consumption of cacao has anxiolytic effects in rats. 32 In humans, flavanol-rich cacao drinks reduced anxiety during highly effortful cognitive tasks. 60 Previous studies have documented the anxiolytic properties of polyphenols.61,62 Polyphenols exert their anxiolytic effects by eliciting biochemical and structural changes in the hippocampus, a crucial brain region for mood and cognition.63,64 Although we did not evaluate the specific polyphenolic compounds in the cacao powder used in the study (and this is a limitation of our work), studies have shown that flavanols found in natural products such as cacao induce mood-related advantages through the modulation of dopaminergic or serotonergic systems, which are targeted by anxiolytic drugs. 64 The association between reduced brain-derived neurotrophic factor (BDNF) and adult depression suggests that flavanol-mediated increases in neurotrophin expression could contribute to their mood-improving effects.36,65 -67
Research suggests a strong association between rising Aβ1-42 levels, progressive loss of synapses, and neuronal death, ultimately leading to cognitive decline.9,11,68,69 U-ICV injection of Aβ1-42 in rats leads to learning and memory deficits, as evidenced by performance in NOR, BM, and MWM tasks.53,68,70 The results of this study revealed that injection of Aβ1-42 induced cognitive impairments in the Aβ group. However, oral consumption of cacao improved these impairments in the Aβ-Pre and the Aβ-Post groups. Also, in line with the results of the present study, it has been reported that the administration of cacao seed extract protected against aluminum-induced recognition memory impairment in rats. 41 A similar finding reports that dietary supplementation with LMN, a cacao-rich diet supplement, significantly improves spatial decline associated with aging. 71 Additionally, it has been shown that Acticoa powder, a cacao polyphenol extract, improves age-related cognitive impairment in rats. 35 The protective effects of cacao and its by-products can be explained, at least in part, by several mechanisms detailed below: (1) improving cerebral blood flow72,73; (2) potentiating neurotrophic factors 34 ; (3) strengthening neurogenesis in the hippocampus 71 ; (4) enhancing the cholinergic neurotransmission in the hippocampus 42 ; (5) modulating BDNF signaling pathway 36 ; (6) activating nuclear factor-erythroid-2-related factor 2 (Nrf2) and an increase in the neuroprotective heme oxygenase-1 (HO-1) enzyme 74 ; (7) up-regulating cyclic adenosine monophosphate (cAMP)/cAMP-response element binding protein (CREB)/BDNF pathway 75 ; and (8) preventing mammalian target of rapamycin (mTOR) signaling cascade. 76
Oxidative stress plays a fundamental role in the development of various neurological and cardiovascular diseases, and it is considered a core mechanism underlying AD.77,78 Aβ peptide acts as an oxidative toxicant, causing an imbalance of oxidative-antioxidative status. 79 The present study examined the effects of cacao on peripheral TAC and TOS levels, not on cerebral or hippocampal oxidant/antioxidant status, which is one of the limitations of this study. Nevertheless, we aimed to determine if Aβ-induced AD in rats resulted in increased peripheral oxidative stress. In this study, injection of Aβ1-42 into the right lateral ventricle showed an imbalance of oxidative-antioxidative status in the plasma of the rats which was indicated by a decrease in the concentration of TAC and an increase in TOS. Current evidence suggests that oxidative stress in neurodegenerative disorders, like AD, is not limited to the central nervous system. 80 In this regard, increased peripheral oxidative stress in AD has been revealed in prior studies.49,81 However, oral consumption of cacao in the Aβ-Pre and the Aβ-Post groups improved oxidative/antioxidative status by decreasing the TOS level and increasing the TAC concentration, representing its antioxidant activity. Consistent with these results, the antioxidant properties of cacao have been previously reported in different neurodegenerative diseases such as AD 36 and Parkinson’s disease. 82 Flavonoids have been shown to neutralize free radicals, chelate metal ions such as copper and iron, inhibit enzymes that produce ROS, and enhance antioxidant defense systems. 83 Specifically, epicatechin and catechin, the main flavonoids found in cacao, are effective in scavenging ROS and inhibiting lipid peroxidation.84,85 Nevertheless, to the best of the authors’ knowledge, this is the first animal study reporting oxidative stress as a specific underlying mechanism for cacao’s effectiveness against Aβ1-42-induced anxiety-like behavior and cognitive deficits.
Neuronal loss is an outcome of neurodegenerative diseases such as AD. 86 Some studies have shown that neuronal loss primarily occurs in areas where Aβ deposits are present, indicating a link between Aβ deposition and neuronal loss. 87 Additionally, neuronal loss associated with Aβ can lead to behavioral disturbances in AD. 88 Furthermore, it has been demonstrated that local oxidative stress around plaques contributes to long-term toxicity and selective neuronal death in AD. 86 Consistent with previous studies, 53 Aβ injection in rats resulted in increased neuronal death in various hippocampus regions stained with hematoxylin-eosin. Interestingly, treatment with cacao successfully prevented neuronal death in the hippocampus of Aβ-injected rats. Consistent with these results, it has been reported that cacao procyanidin fraction protects against 4-Hydroxynonenal-induced apoptosis by blocking mitogen-activated protein kinase kinase 4 activity as well as ROS accumulation. 30 Therefore, cacao’s ability to inhibit neuronal death may explain its anxiolytic and protective effects against Aβ-induced cognitive and non-cognitive deficits. This suggests that cacao’s anxiolytic and protective effects against cognitive decline in the Aβ1-42-injected AD rat model may be, at least partly, due to its modulation of oxidative stress and inhibition of neuronal death.
Conclusions
Overall, the findings of the present investigation showed that chronic oral administration of cacao improved cognitive and non-cognitive deficits in an Aβ1-42-infused AD rat model, possibly through modulation of oxidative-antioxidative status and alleviating neuronal loss in the hippocampal regions. This work suggests that cacao could be a promising therapeutic agent for improving cognitive and non-cognitive deficits in AD. However, further research is needed to evaluate the mechanisms underlying cacao’s protective effects against AD-induced cognitive decline, particularly the mechanisms involved in its antioxidant properties.
Footnotes
Acknowledgements
This paper was extracted as a part of Hamid Shokati Basir’s M.Sc. thesis.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Approval for Animal Experiments
Experimental methods and animal care were in accordance with the National Institutes of Health (NIH) and ARRIVE Guidelines and were approved by Bu Ali Sina University-Hamedan’s Ethics Committee (Ethic code: IR.BASU.REC.1398.025).
Author Contributions
HSB: writing–review and editing, writing–original draft, visualization, methodology, software, formal analysis, and data curation. NM: writing–review and editing, writing–original draft, validation, supervision, software, resources, project administration, methodology, funding acquisition, and conceptualization. AK: writing–review and editing, writing–original draft, visualization, validation, supervision, resources, project administration, methodology, funding acquisition, and conceptualization. MR: writing–review and editing, writing–original draft, validation, methodology, software, formal analysis, and data curation. AH: writing–review and editing, writing–original draft, validation, software, formal analysis, and data curation.
Availability of Data and Materials
All data generated and analyzed during the current study are available with the corresponding author upon reasonable request.
Consent to Participate/Consent to Publish
Not applicable
